Abstract
Obesity is a well-known risk factor for many diseases, but the impact of baseline body mass index (BMI) on the outcomes of allogeneic hematopoietic stem cell transplantation (allo-HSCT) remains controversial. To elucidate the influence of pretransplant BMI on post-allo-HSCT outcomes including graft-versus-host disease (GVHD), overall survival (OS), relapse-free survival (RFS), and nonrelapse mortality (NRM), we conducted a retrospective study using registry data which comprised 1092 adult patients who underwent allo-HSCT between 2015 and 2023. Among the 1092 eligible patients (53.2% male), 56.5% were normal-weight; 24.8% were overweight and 9.1% were obese. Multivariable analyses revealed that compared with normal-weight patients, obese individuals had a higher risk of grade II–IV and III–IV acute GVHD (aGVHD), especially in the gastrointestinal system, with aHRs of 2.08 (95% CI, 1.47–2.94), 2.60 (95% CI, 1.52–4.44), and 3.71 (95% CI, 2.00–6.88), respectively. The probability of OS and RFS was significantly lower in overweight (P = 0.034, P = 0.015, respectively) and obese patients (P = 0.033, P = 0.024, respectively) as compared with normal-weight patients, with aHRs increasing by ~38% (aHR, 1.38; 95% CI, 1.03–1.86), ~40% (aHR, 1.40; 95% CI, 1.07–1.83), ~58% (aHR, 1.58; 95% CI, 1.04–2.40), and ~56% (aHR, 1.56; 95% CI, 1.06–2.29), respectively. Furthermore, the NRM of obese patients was statistically higher than normal-weight patients (P = 0.02, sHR, 2.19; 95% CI, 1.12–4.27). A subgroup analysis revealed that the adverse effects of obesity on OS, RFS, and NRM were primarily observed in the subgroup of patients aged < 40 years and patients with acute lymphoblastic leukemia. The increased risk of grade II–IV aGVHD due to obesity was observed across all subgroups. In conclusion, Obesity prior to allo-HSCT increases the risk of aGVHD and NRM, leading to poorer OS. These findings underscore the importance of closely monitoring high-risk patients and offering opportunities for early intervention.
Background
In recent decades, the prevalence of overweight and obesity has shown a sharp upward trend globally, accounting for 60% to 70% of the adult population in developed countries, with more than half of U.S. adults considered overweight or obese1–3. Overweight and obesity serve as prominent risk factors for the onset and mortality of various diseases. Statistically, being overweight is associated with an increased risk of cancer in at least 13 anatomical sites, such as endometrial cancer, pancreatic cancer, and multiple myeloma2,4. Obesity also poses a risk factor for numerous chronic diseases such as hypertension, type 2 diabetes, cardiovascular diseases, and renal diseases3,5, and is associated with increased incidence and mortality in cancer patients. Therefore, overweight and obesity are regarded as adverse prognostic indicators for many human health conditions. However, as a heterogeneous condition, there is data suggesting that some late-stage cancer and chronic disease patients may exhibit a survival advantage if they have a higher body mass index (BMI). This phenomenon, where a higher BMI is associated with an increased risk of morbidity but confers a survival advantage in late-stage disease, is termed the “obesity paradox”5–7, and its specific mechanisms remain unclear. To date, there is currently a lack of definitive data on whether obesity impacts the prognosis of patients with hematologic malignancies, especially in the context of hematopoietic stem cell transplant.
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is widely used as an efficient method for treating hematologic malignancies. It primarily relies on the graft-versus-tumor (GVT) effect mediated by donor T cells to eradicate residual cancer cells and prevent disease recurrence 8 . However, the clinical application of HSCT is limited due to the high incidence of graft-versus-host disease (GVHD), which weakens the GVT effect. GVHD can manifest as acute or chronic processes, each with distinct pathophysiological pathways. Acute GVHD (aGVHD) is characterized primarily by an inflammatory cascade reaction leading to a “cytokine storm,” where pro-inflammatory cytokines attack multiple organs such as the gastrointestinal tract, skin, and liver, causing tissue damage, organ failure, and death. Severe acute intestinal GVHD is a major cause of early onset. Chronic GVHD (cGVHD) occurs later and is dominated by tissue fibrosis, severely impacting patient quality of life8–12. GVHD is extremely challenging to treat, and broad-spectrum immunosuppressive agents are currently used for treatment or prevention, indirectly affecting the GVT effect and leading to increased relapse rates8,10. While comprehensive evaluations of prognostic factors for leukemia patients undergoing HSCT have been conducted, the influence of overweight and obesity remains ambiguous. Some studies propose that obese and overweight patients have longer overall survival (OS) compared with those with normal weight 13 . Conversely, alternative perspectives posit that BMI does not affect transplant outcomes 14 . However, a meta-analysis showed that pretransplant overweight recipients were associated with higher rates of aGVHD and poorer survival rates 15 ; Similarly, in a study from the University of Minnesota, patients with obesity had higher transplant-related mortality, lower survival rates, and more severe GVHD after allo-HSCT 9 . So far, the relationship between BMI and the prognosis of allo-HSCT patients remains inconclusive.
Considering the numerous and changing variables involved in hematopoietic stem cell transplantation, such as human leukocyte antigen (HLA) typing, conditioning regimens, stem cell sources, choice of immunosuppression, posttransplant support, and the presence of comorbidities9,10, the heterogeneity of the results can be partially explained. Besides, it is worth noting that the majority of the aforementioned studies were conducted in Western countries. And Asians typically have higher body fat percentages than Europeans, which may lead to health risks even at relatively lower BMI1,13. The discrepancy might also be attributed to the diversity in race, genetics, lifestyle, and so on. Therefore, the present study aims to explore the impact of BMI on transplant outcomes among Asian recipients.
Materials and methods
Patients
The study reviewed 1113 consecutive adult patients with acute leukemia who underwent allo-HSCT in the First Affiliated Hospital of Zhejiang University School of Medicine from January 2015 to February 2023 (Supplemental Fig. S1). The inclusion criteria were (1) age ≥ 18 years; (2) acute leukemia, including acute myeloid leukemia (AML), acute lymphocytic leukemia (ALL), and mixed phenotype acute leukemia (MPAL); (3) receiving a first allo-HSCT; (4) peripheral blood stem cell transplantation from haploidentical relatives, matched sibling donors or unrelated donors. Patients with primary graft failure or incomplete clinical information were excluded. Finally, a total of 1092 patients who met the criteria were included. The study adhered to the Declaration of Helsinki and was approved by the Ethics Review Committee of the First Affiliated Hospital of Zhejiang University School of Medicine (The ethics approval number: 浙大一院伦审2025研第0516号-快 ([2025B] IIT Ethics Approval No. 0516)). The data cutoff date was December 31, 2023. Follow-up time was calculated from the date of transplantation to the event or the last contact. Additional follow-up data were obtained from the review of the hospital records.
Patient and public involvement
Patients or the public were not involved in the design, conduct, reporting, or dissemination plans of our research.
Transplant regimens
As previously described 16 , all patients received either a myeloablative busulfan/cyclophosphamide-based conditioning regimen or a reduced intensity regimen consisting of fludarabine/busulfan. Anti-thymocyte globulin-Genzyme (Thymoglobulin) or anti-human T-lymphocyte immunoglobulin (Grafalon) was applied as preparation for haplo-HSCT or unrelated donor HSCT. All patients received G-CSF mobilized peripheral blood stem cells and no graft was subjected to ex vivo T-cell depletion. GVHD prophylaxis consisted of cyclosporine, a short course of methotrexate, and mycophenolate.
Evaluation of baseline BMI
To examine the relationship between obesity and study outcomes, patients were stratified into subgroups based on their relative weight recorded before the start of conditioning therapy. The BMI was calculated as weight in kilograms divided by the square of the patient’s height in meters. In accordance with the consensus of National Institute for Health and Care Excellence (NICE) 17 regarding weight designations for the Asian population and Working Group on Obesity in China (WGOC) 18 , the suggested BMI categories are as follows: ≤18.5 kg/m2 (underweight); 18.5–23.9 kg/m2 (normal-weight); 24–27.5 kg/m2 (overweight); and ≥27.5 kg/m2 (obese).
Study endpoint definition
The endpoints of this study included aGVHD, OS, relapse-free survival (RFS), nonrelapse mortality (NRM), relapse incidence, and cGVHD. Overall survival was defined as the time from allo-HSCT until death from any cause or last follow-up, whichever occurred first; patients alive at the last follow-up were censored. Relapse-free survival was calculated as the time from transplantation to the first observation of disease relapse, death from any cause, or last follow-up, whichever occurred first; patients alive without disease at the last follow-up were censored. Nonrelapse mortality was defined as death from any cause unrelated to relapse. Relapse was defined as the time of hematologic relapse or disease progression. aGVHD was graded per Mount Sinai Acute GVHD International Consortium criteria 19 . The censoring date for aGVHD was set at 100 days posttransplantation. cGVHD was scored according to the National Institutes of Health (NIH) Consensus Criteria 20 .
Statistical analysis
Descriptive statistics were employed to characterize patient demographics. Continuous variables were presented as median with interquartile ranges (IQR), while categorical variables were presented as percentages. Chi-square tests were utilized to compare patient characteristics for categorical variables, with multiple comparisons corrected using the Bonferroni correction method. Survival functions were evaluated using the Kaplan–Meier method, with differences analyzed via the log-rank test. Cox proportional hazard regression models were used for univariate and multivariate analyses of GVHD, OS, and RFS. In the multivariate analysis, variables significant at P < 0.1 in univariate analyses, along with clinically relevant variables were included in the Cox model to obtain adjusted hazard ratios (aHRs) with 95% confidence intervals (95% CI). Cumulative incidence and group differences for relapse and NRM were estimated using proportional hazard models for competing risk, yielding subdistribution hazard ratios (sHRs) with 95% CI, and groups comparisons were performed using the Fine-Gray test. The sensitivity analysis used the restricted cubic spline function. Fully adjusted models were adjusted for patient age and sex, diagnosis, disease risk index (DRI), and anti-thymocyte globulin (ATG) use or not. Given the impact of obesity on study outcomes, a subgroup analysis was conducted based on patient age, sex, disease diagnosis, and the refined DRI classification 21 . All analyses were performed using SPSS 19, survival analysis plots were generated using the R 3.2.3 software package, and forest plots were constructed using GraphPad Prism 9.5.
Results
Patient, donor, disease, and transplantation characteristics
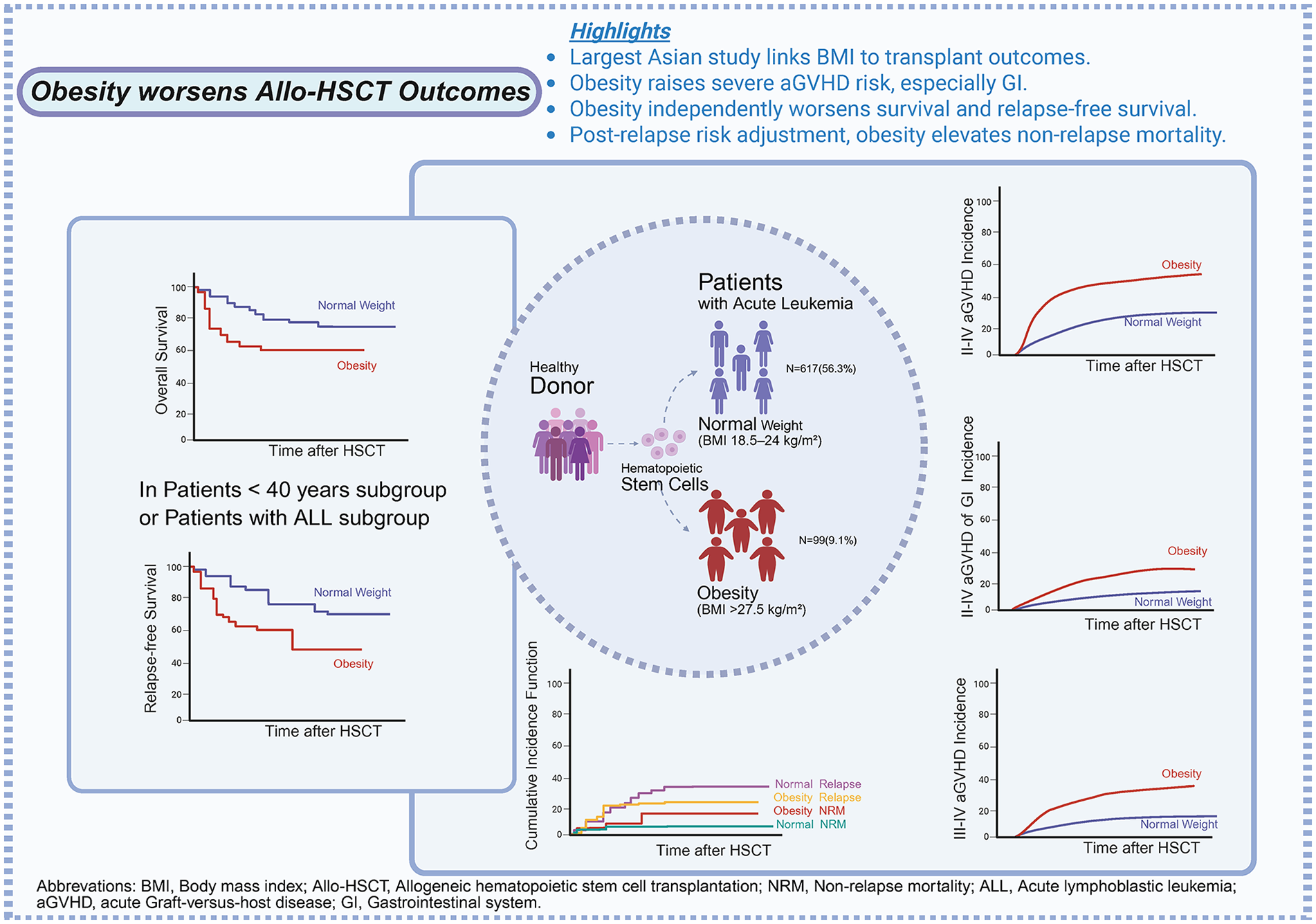
A total of 1092 adult patients were included. Patient-, donor-, disease-, and transplant-related characteristics among the BMI subgroups are summarized in Table 1. The numbers of underweight, normal-weight, overweight, and obese patients were 105 (9.6%), 617 (56.5%), 271 (24.8%), and 99 (9.1%), respectively. AML was the most common diagnosis, accounting for 55.8% of cases, while 42.7% were diagnosed with ALL. 53.2% of patients were male. There were no significant differences among the four subgroups of patients regarding donor/recipient HLA compatibility, donor sex, sex match, DRI stratification, conditioning regimen, and pretransplant disease status.
Baseline characteristics across body mass index groups.

ALL: Acute Lymphoblastic Leukemia; AML: Acute Myeloid Leukemia; MPAL: Mixed Phenotype Acute Leukemia, comprising both Acute Lymphoblastic Leukemia and Acute Myeloid Leukemia; DRI: Disease Risk Index, developed for adult patients with hematologic malignancies undergoing HSCT, commonly used as a tool for predicting transplant outcomes; MAC and RIC: categorized based on conditioning regimen intensity, with MAC referring to myeloablative conditioning and RIC indicating reduced-intensity conditioning; CR1: Complete Remission 1, indicating that a patient achieved complete remission after chemotherapy for the first time; CR2/CRn: Complete Remission 2/n, indicating second or n-th complete remission after disease relapse and subsequent treatment; MRD: Minimal Residual Disease, denoting a small amount of cancer cells remaining in the body after cancer treatment, with positivity indicating detectable residual disease after cancer treatment, while negativity implies the absence of residual disease after cancer treatment; MNC: mononuclear cell. Unless otherwise specified, the meanings of abbreviations are consistent throughout the text.
Data are presented as number (percentage) of participants unless otherwise indicated.
There is a significant difference. When comparing the remaining groups with the normal weight group, multiple comparisons of sample rates were involved in the analysis, which was conducted using the Bonferroni correction method (considering P < 0.017 as indicating significant difference).
Furthermore, there were significant differences in other baseline characteristics among different BMI subgroups. Compared with the normal-weight patients, a larger proportion of patients in the underweight group were aged < 40 years (81.9% vs 55.3%, P < 0.001) and had donors aged ≥ 40 years (57.1% vs 35.2%, P < 0.001), while the patients aged < 40 years in the overweight group were fewer (45.8% vs 55.3%, P = 0.011). No statistically significant difference was observed in the age distribution between obese and normal-weight patients, as well as in the donor ages (both P >0.05). Overweight and obese patients were more likely male (60.1% vs 49.4%, P = 0.004; 63.6% vs 49.4%, P = 0.009, respectively), while the sex distribution was similar between the underweight and normal-weight groups (P > 0.05). No significant differences were observed in the infused CD34+ cell and mononuclear cell (MNC) doses among the different BMI groups (P > 0.05).
aGVHD
The Kaplan–Meier survival curves illustrate the impact of BMI on grades II–IV and III–IV aGVHD, grades II–IV skin or gastrointestinal aGVHD. The cumulative incidences of 100-day grades II–IV (43.4% [95% CI, 33.6%-53.2%] vs. 22.5% [95% CI, 19.2%-25.8%], P < 0.001) and III–IV aGVHD (19.2% [95% CI, 11.4%-27.0%] vs. 7.9% [95% CI, 5.7%-10.1%], P < 0.001) (Fig. 1a, b), as well as grades II–IV acute skin (27.6% [95% CI, 18.0%-37.2%] vs. 19.5% [95% CI, 16.2%-22.8%], P = 0.043) and gastrointestinal system (GI) GVHD (19.5% [95% CI, 10.9%-28.1%] vs. 5.6% [95% CI, 3.6%-7.6%], P < 0.001) (Supplemental Fig. S2 A and B) were higher in the obese group than those in the normal weight group.
We further sought to determine whether the impact of obesity on aGVHD persisted after adjusting for various factors. In the multivariate analyses, obesity was indeed associated with a higher incidence of grades II–IV and III–IV aGVHD posttransplantation compared with normal weight (aHR, 2.08, 95% CI, 1.47–2.94; aHR, 2.60, 95% CI, 1.52–4.44, respectively; both P < 0.001) (Table 2). Especially in the gastrointestinal system, the difference is statistically significant (aHR, 3.71, 95% CI, 2.00–6.88, P < 0.001) (Supplemental Table S1). In addition, other risk factors related to aGVHD after the multivariate Cox regression model included donor/recipient HLA compatibility and/or donor age, which are detailed in Table 2.
Multivariable analysis for risk of aGVHD, OS, RFS, NRM, and Relapse.

aGVHD: acute graft-versus-host disease; OS: Overall Survival; RFS: Relapse-free Survival; NRM: Nonrelapse Mortality; aHR: adjusted hazard ratio; sHR: subdistribution hazard ratio; 95% CI: 95% confidence interval; NR: Non-CR, no complete remission; NA: Not available.
Grade II–IV aGVHD, III–IV aGVHD, OS and RFS were calculated using a multifactor Cox proportional hazards model reported as aHR (95% CI), while NRM and relapse were calculated using a Fine-Gray competitive risk model, reported as sHR (95% CI).
Using the Fine-Gray competing risks model to calculate NRM and relapse risk, only the comparison between the obese group and the normal weight group is presented. Comparisons between the overweight group, underweight group, and normal weight group are provided in eTable 2 in Supplement 1.
When calculating NRM and relapse risk using the Fine-Gray competing risks model, age was treated as a continuous variable.
Disease diagnoses were not included in the hazard ratio estimates.
Sensitivity analysis demonstrated that the risk of aGVHD (both grades II–IV and III–IV) remained relatively stable at lower BMI ranges, whereas it exhibited a progressive increase with higher BMI levels (Supplemental Fig. S3A and B). A similar trend was observed for grade II–IV gastrointestinal aGVHD (Supplemental Fig. S2C). In contrast, the risk of grade II–IV skin aGVHD only reached statistical significance at substantially elevated BMI levels (Supplemental Fig. S2D).
OS and RFS
A Kaplan–Meier survival rate curve was plotted representing the relationship between BMI and both OS and RFS among the four BMI subgroups. With a median follow-up of 31.7 (range 0.7–92.5) months after allo-HSCT, the 5-year OS rates across underweight, normal-weight, overweight, and obese patients were 74.9%, 74.2%, 69.7%, and 68.6%, respectively. The estimated 5-year relapse-free survival rates across four BMI groups were 67.1%, 70.9%, 61.8%, and 59.3%, respectively. As shown in Supplemental Fig. S4, when compared with the normal-weight patients, there were no remarkably significant differences in OS and RFS among the other three groups (all P > 0.05) (Supplemental Fig. S4A and B).
However, the Cox multivariate analysis revealed a statistically significant difference in the impact of BMI on survival outcomes (Table 2). The estimated probabilities of OS and RFS were decreased both in overweight (aHR, 1.38, 95% CI, 1.03–1.86, P = 0.034; aHR, 1.40, 95% CI, 1.07–1.83, P = 0.015) and obese group (aHR, 1.58, 95% CI, 1.04–2.40, P = 0.033; aHR, 1.56, 95% CI, 1.06–2.29, P = 0.024), respectively (Table 2).
NRM, relapse, and cGVHD
To further explore the impact of BMI on NRM and posttransplant leukemia relapse, we conducted a competing risk analysis using the Fine-Gray model, treating relapse as the competing risk for NRM, and vice versa. Patients with obesity were at greater risk of NRM as compared with normal-weight patients (P = 0.042) (Fig. 1c, d), while no statistical difference was observed among other subgroups (Supplemental Fig. S5). There were no significant differences between the four subgroups for either relapse or cGVHD (Supplemental Fig. S6).

Cumulative incidence of aGVHD and NRM according to BMI. (a, b) The obese patients exhibited a significantly higher incidence rate of grade II–IV and III–IV aGVHD than normal-weight individuals (both P < 0.001). (c) Compared with the normal weight group, the obese group demonstrated a higher risk of NRM (P = 0.042). (d) Results of competitive risk model: Relapse was calculated as the target event, with NRM considered as a competing risk event. After adjusting for NRM, there was no significant difference in relapse rates between the obese and normal groups (P = 0.667); however, after adjusting for relapse, the nonrelapse mortality rate was significantly higher in the obese group compared with the normal group (P = 0.042).
In the multivariate analysis, obesity predicted higher NRM (sHR, 2.19, 95% CI, 1.12–4.27, P = 0.022), while not influencing relapse probability (sHR, 0.97, 95% CI, 0.57–1.63, P= 0.900) (Table 2). The sensitivity analysis reveals a U-shaped relationship between BMI and the risk of NRM (Supplemental Fig. S3C). Compared with the normal weight group, there were no significant differences in the probability of NRM and relapse among the remaining groups (Supplemental Table S2).
Subgroup analysis
To further identify specific categories where the impact of obesity was more pronounced, we conducted an analysis across different subgroups. For grade II–IV aGVHD, the adverse effects of obesity were observed in almost all subgroups, regardless of patient age, sex, disease diagnosis, DRI risk stratification, or whether ATG was used or not (Fig. 2a). For OS and RFS, the negative impact of obesity was primarily seen in the subgroup of patients aged <40 years and the ALL subgroup, with less noticeable effects in other subgroups. In detail, among the subgroup of patients aged <40 years, the probability of OS and RFS was lower in obese patients, and the difference was both significant (HR, 1.82, 95% CI, 1.13–2.92; HR, 1.61, 95% CI, 1.03–2.53, respectively) (Fig. 2b, c) (Supplemental Fig. S4C and D), and among the ALL subgroup, obese patients had shorter OS and RFS (HR, 1.78, 95% CI, 1.03–3.05; HR, 1.68, 95% CI, 1.01–2.79, respectively) (Fig. 2b, c) (Supplemental Fig. S4E and F), in comparison with patients with normal weight. Sensitivity analysis in the subgroup of patients aged <40 years revealed a U-shaped association between BMI and both mortality risk and the composite risk of relapse or mortality (Supplemental Fig. S7A and B). A similar pattern was observed in the ALL subgroup (Supplemental Fig. S7C and D).

Subgroup analysis forest plot. (a–d) illustrates the impact of obesity on transplant outcomes across different subgroups.
Regarding NRM, the increased risk associated with obesity was mainly observed in the subgroups of patients aged <40 years, the ALL subgroup, and those with low to medium DRI risk (Fig. 2d).
Discussion
In this retrospective study of acute leukemia patients undergoing HSCT, we observed that obese patients had a higher incidence of grade II–IV and III–IV aGVHD compared with recipients with normal weight. This difference was more pronounced in gastrointestinal aGVHD. More strikingly, obesity has been recognized as an independent risk factor for NRM, contributing to a worsened post-HSCT prognosis in terms of OS and RFS. This highlights the significance of addressing obesity as part of the comprehensive care for individuals undergoing HSCT, emphasizing the need for strategies to manage weight and its associated health implications in this context.
Further research is needed to explain the reasons for the different outcomes in patients with higher and normal body mass indexes. Several hypotheses have been proposed to account for this disparity. Currently, preclinical trials have validated the following mechanisms: obesity leads to systemic immune stimulation, resulting in the release of a plethora of pro-inflammatory cytokines (including tumor necrosis factor (TNF) and Interleukin-6 (IL-6)), increased intestinal permeability, and disrupts the intestinal barrier and integrity, and endotoxin transintestinal transport into the bloodstream causes systemic sepsis, which correlates with increased mortality from acute severe intestinal GVHD 12 . In addition, pretransplant conditioning regimens cause extensive DNA damage and cell apoptosis in the digestive tract, resulting in decreased proliferation of intestinal stem cells; concomitantly, obesity results in decreased intestinal microbiota diversity, with decreased abundance of Clostridia species associated with improved GVHD outcomes, while the relative abundance of Enterococcus species in fecal samples, associated with adverse GVHD prognosis, significantly increases. The use of prophylactic antibiotics can ameliorate these outcomes. This also elucidates why obesity selectively affects the digestive tract in aGVHD9–11. The aforementioned mechanisms have only been validated in preclinical trials, and further research is needed to confirm their clinical implications.
In addition, obese patients may have concomitant liver and kidney dysfunction, leading to increased drug accumulation and the risk of transplant-related toxicity. Glucocorticoids are a common therapeutic approach for posttransplant GVHD, placing patients’ immune systems in a suppressed state and increasing the risk of infection-related complications. Moreover, overweight and obese patients generally have poorer overall health statuses, making them prone to various complications such as cardiovascular diseases and strokes, which can lower posttransplant survival rates and increase the risk of NRM5,10,15,22–24. However, J. Yang et al. argue that obesity also brings benefits to transplant patients on the other hand. They suggested that weight loss and cachexia were definite death factors for cancer patients, therefore, declining nutritional status was a risk factor for adverse outcomes of HSCT 25 . Overweight and obese patients have more nutritional reserves and are better able to tolerate the high stress and metabolic states during transplantation5,13,26,27.
Some scholars also propose that obese patients receive higher chemotherapy doses before transplantation, resulting in greater tissue damage and increased NRM. At the same time, high-dose chemotherapy drugs can trigger cytokine storms, leading to severe aGVHD. Clinically, many drugs in practice are poorly soluble in lipids and distribute inadequately in adipose tissue, altering their pharmacokinetics. Consequently, patients may receive relatively insufficient chemotherapy doses, which could result in poorer chemotherapy efficacy, decreased posttransplant survival rates, and increased mortality. However, some scholars believe that patient nutritional status affects the metabolism of chemotherapy drugs, resulting in higher cumulative drug doses or longer exposure times in obese patients, leading to better outcomes13,28. Prospective pharmacokinetic studies are recommended to better understand the correlation between effective drug doses and therapeutic outcomes4,14,15,22,24,28–31.
The impact of obesity on immune status is complex and multifactorial. Adipose tissue is a highly active endocrine organ capable of synthesizing and secreting various adipokines, such as leptin, causing a chronic inflammatory state that affects the proliferation and function of T regulatory cells, increasing the risk of alloimmune responses posttransplant1,5,22,28. Through endocrine and paracrine effects, adipose tissue can directly promote tumor growth and angiogenesis. Moreover, the anti-apoptotic effect of hyperinsulinemia associated with obesity further promotes tumor progression4,15,24. All these factors contribute to an increased risk of relapse. However, our study failed to demonstrate a significant association between BMI and the risk of relapse posttransplant. This null finding may be potentially attributed to the limited sample size of obese patients in our cohort, which could have resulted in insufficient statistical power to detect a potential effect. Future studies should increase the sample size of obese patients to better elucidate the true relationship between BMI and posttransplant relapse.
This study revealed a clinically intriguing trend wherein underweight patients demonstrated numerically superior outcomes compared with normal-weight counterparts, albeit without reaching statistical significance (P > 0.05). This observation diverges from conventional expectations and may be explained by the following mechanisms: (1) Selection Bias: Institutional protocols typically mandate rigorous pretransplant evaluation for severely underweight candidates. Consequently, enrolled underweight patients likely represent a selectively healthier cohort after excluding those with cachexia or profound malnutrition. (2) The distinct adiposity distribution patterns in Asian populations may be contributory. Normal-weight subjects might include metabolically obese individuals with visceral fat accumulation, whereas underweight patients potentially have lower prevalence of metabolic disorders (e.g., diabetes, hepatic steatosis). The absence of HCT-CI scoring (as noted in limitations) precludes definitive assessment. (3) Weight-based chemotherapy dosing in underweight patients may yield more accurate pharmacokinetic exposure, minimizing toxicity from overdosing or efficacy compromise from underdosing. (4) Limited sample size represents another important factor. The relatively small number of patients in the underweight group (N = 105) may have reduced statistical power and increased the risk of false-positive results.
Limitations of this study should be addressed. Firstly, as a retrospective observational study, this research only included Chinese patients who underwent allo-HSCT at the First Affiliated Hospital of Zhejiang University School of Medicine. The possibility of selection bias cannot be ruled out, and it is uncertain whether similar research results would occur in other countries/regions. Future studies should reassess the results using other databases. Secondly, Pereira AZ et al’s 32 study clearly demonstrated the crucial role of body composition in determining the nutritional status and transplant outcomes of hematopoietic stem cell transplant patients. Despite this study combining the consensus on BMI from NICE and WGOC to distinguish overweight and obesity, BMI itself is not precise in measuring fat content and cannot differentiate between muscle tissue and adipose tissue, thus failing to fully reflect the patient’s nutritional status, which is also an explanation for the obesity paradox. Currently, there is no standardized nutritional screening tool specifically designed for patients undergoing allo-HSCT. Other obesity indices, such as waist-to-hip ratio and skinfold thickness, can be used to assess the risk of adverse health outcomes. Anthropometric measurements such as bioelectrical impedance analysis (BIA) and dual-energy X-ray absorptiometry can also accurately reflect the patient’s body composition, which is crucial for predicting outcomes such as mortality and complications in HSCT5–7,13,23,27,29,31–33. Regarding the limitations of this study, it is important to emphasize that our analysis did not thoroughly investigate the impact of comorbidities, an issue particularly prominent in the obese patient group, which may introduce confounding effects to the interpretation of our results. As indicated by the research of Emily Gallagher et al 34 , the coexistence of obesity and type 2 diabetes significantly elevates the mortality risk among patients. Besides, the absence of Hematopoietic Cell Transplantation-Specific Comorbidity Index (HCT-CI) scores limits our ability to assess whether comorbidity burden mediates the observed BMI-outcome associations. Therefore, future studies should prioritize the inclusion of comorbidity-related data, particularly information on metabolic disorders such as diabetes, to more precisely elucidate the true influence of obesity on posttransplant survival outcomes in patients with acute leukemia.
Conclusions
In conclusion, our study indicates that a high body mass index prior to allogeneic hematopoietic stem cell transplantation was associated with a worse prognosis, including increased risks of aGVHD and NRM, and inferior OS. Future prospective studies are needed to validate our findings and provide a better understanding of the underlying pathophysiological mechanisms. Based on the current results, we recommend integrating comprehensive pretransplant weight management strategies into clinical practice, particularly for obese patients. In addition, transplant teams should enhance monitoring of obese patients, especially during the acute posttransplant period, thereby optimizing prognostic outcomes through timely and targeted interventions.
Supplemental Material
sj-docx-1-cll-10.1177_09636897251349377 – Supplemental material for Prognostic effect of body mass index in patients with acute leukemia undergoing allogeneic hematopoietic stem cell transplantation: A retrospective cohort study
Supplemental material, sj-docx-1-cll-10.1177_09636897251349377 for Prognostic effect of body mass index in patients with acute leukemia undergoing allogeneic hematopoietic stem cell transplantation: A retrospective cohort study by Jing Yu, Zhuoyue Shi, Hengwei Wu, Yi Luo, Jian Yu, Yamin Tan, Xiaoyu Lai, Lizhen Liu, Huarui Fu, Yishan Ye, Luxin Yang, Congxiao Zhang, He Huang, Jimin Shi and Yanmin Zhao in Cell Transplantation
Footnotes
Acknowledgements
We sincerely thank the patients who participated in this study.
Abbreviations
BMI: body mass index; allo-HSCT: Allogeneic hematopoietic stem cell transplantation; GVT: graft-versus-tumor; GVHD: graft-versus-host disease; aGVHD: acute graft-versus-host disease; cGVHD: chronic graft-versus-host disease; OS: overall survival; HLA: human leukocyte antigen; AML: acute myeloid leukemia; ALL: acute lymphocytic leukemia; MPAL: mixed phenotype acute leukemia; NICE: National Institute for Health and Care Excellence; WGOC: Working Group on Obesity in China; RFS: relapse-free survival; NRM: non-relapse mortality; NIH: National Institutes of Health; IQR: interquartile ranges; aHR: adjusted hazard ratio; 95% CI: 95% confidence interval; sHR: subdistribution hazard ratio; ATG: Anti-thymocyte Globulin; DRI: disease risk index; MNC: mononuclear cell; GI: gastrointestinal system; MRD: measurable residual disease; CR: complete remission; NR: no complete remission; TNF: tumor necrosis factor; IL-6: Interleukin-6; BIA: bioelectrical impedance analysis; HCT-CI: Hematopoietic Cell Transplantation-Specific Comorbidity Index.
Ethical considerations
Ethical approval for this study was approved by the Ethics Review Committee of the First Affiliated Hospital of Zhejiang University School of Medicine.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
There are no human subjects in this article and informed consent is not applicable.
Author contributions
JY and ZYS contributed equally to this work, performed the study and wrote the manuscript. HWW, FRF, YSY, LXY, and CXZ were responsible for data collection. YMT, XYL, LZL, YL, and JY provided administrative, technical, or material support for this study. HH, JMS, and YMZ as the guarantors of this study contributed to conception and design of the study. All authors read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Key Research and Development Program of China (grant no. 2022YFA1103500) and the National Natural Science Foundation of China (grant no. 82170210).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
