Abstract
Poor graft function (PGF) is a refractory complication that occurs after allogeneic hematopoietic stem cell transplantation (allo-HSCT). In the present study, we prospectively evaluated the efficacy and safety of mesenchymal stem cells (MSCs) expanded from the bone marrow of a third-party donor to patients with PGF after allo-HSCT. Twenty patients with PGF (7 with primary and 13 with secondary PGF) received MSCs (1 × 106/kg) one to three times at 28-day intervals. Seventeen patients were responsive to MSCs, whereas three were not. Within the first 100 days after MSC treatment, 13 patients developed 20 episodes of infection. Moreover, five patients experienced cytomegalovirus-DNA viremia, and seven experienced Epstein—Barr virus (EBV)—DNA viremia within the first 100 days after MSC treatment; three of the latter developed EBV-associated posttransplant lymphoproliferative disorders (PTLD) within the follow-up period. Grade II acute graft-versus-host disease (GVHD) occurred in one patient, and local chronic GVHD occurred in two patients after receiving MSC treatment, including one acute GVHD and one chronic GVHD, respectively, after accepting donor lymphocyte infusions due to PTLD. After a follow-up period of an average of 508 days (range 166-904 days) posttransplantation, 11 patients died. No short-term toxic side effects were observed after MSC treatment. Two patients experienced leukemic relapse. With the exception of three patients with PTLD, no secondary tumors occurred. These results indicate that MSCs derived from the bone marrow of a third-party donor are beneficial in the treatment of both primary and secondary PGF that develops after allo-HSCT. However, additional studies will be needed to determine whether such treatment might increase the risk of EBV infection and reactivation or the development of EBV-associated PTLD.
Keywords
Introduction
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) can cure many hematologic diseases. Although significant progress has been made in the prevention and treatment of side effects associated with transplantation, poor graft function (PGF) remains an important complication that occurs in 5-27% of patients and is associated with considerable morbidity and mortality related to infections or hemorrhagic complications (7,20,31). Graft function may be poor as a result of slow or incomplete reconstitution of blood counts or decreasing blood counts after successful and prompt hematopoietic reconstitution in the presence of complete donor chimerism and in the absence of severe graft-versus-host disease (GVHD) and relapse (16). PGF treatment usually involves the prescription of hematopoietic growth factors, such as granulocyte colony-stimulating factor (G-CSF) or repeat transplantation. However, these methods are associated with short-term effects and a significant risk of GVHD development, respectively (3-5,15,16,27).
Mesenchymal stem cells (MSCs) are a form of multipotent adult stem cells that are isolated from bone marrow (BM), adipose tissue, and cord blood. Such cells possess the capacity to differentiate into several types of mesenchymal tissues, suppress immunological responses, and support hematopoiesis (18). Clinical applications of human MSCs are evolving rapidly with goals of improving hematopoietic engraftment, preventing and treating GVHD after allo-HSCT, etc. (1,2,9,10,19,21,24,26,28,33, 39). Clinical studies typically use ex vivo-expanded MSCs cotransplanted with hematopoietic stem cells (HSCs), MSCs from the BM of second-party donors (HSC donors), or MSCs originating from the umbilical cord blood of third-party donors (unrelated donors) (1,2,8-10,19,21,24,28). The efficacy of treatment of PGF that develops after allo-HSCT using expanded BM-derived MSCs from a third-party donor is rarely reported (26,33). If such treatment was effective and safe, then BM-derived MSCs could be used as universal donor material. This would have a major impact because the generation of donor-specific MSCs is time consuming, costly, and often impractical if the clinical status of a patient is urgent.
Clinical studies have shown that MSC recipients do not experience immediate or long-term toxic effects associated with MSC treatment originating from either a second-party or third-party donor, regardless of whether the medium contains serum; however, the controversy remains as to whether MSCs increase the incidence of infection, leukemia relapse, and secondary tumor formation in allo-HSCT due to MSC immunosuppressive properties. Some studies have suggested that MSCs do not increase the incidence of infection and leukemia relapse after allo-HSCT. However, this effect is controversial (12-14,17,25,30,32,35-38,42). Therefore, the safety of MSC treatment needs further investigation.
In the present study, we prospectively evaluated the utility of treating patients experiencing PGF after allo-HSCT with ex vivo-expanded BM-derived MSCs from third-party donors. Our objectives were to investigate the safety of MSC treatment on PGF and to evaluate its effect.
Materials and Methods
Patient Eligibility and PGF Diagnosis
Criteria used to determine patient eligibility were the presence of primary or secondary PGF development after allo-HSCT (see Transplantation section) and a patient age of 14-60 years. Fulfillment of any of the following criteria caused a patient to be excluded: any abnormality in a vital sign (e.g., heart rate, respiratory rate, or blood pressure) or the presence of an active visceral hemorrhage. This study was performed in accordance with the tenets of the modified Helsinki Declaration, and the protocol was approved by the relevant Ethics Review Board for each location prior to initiation. All recipients and/or guardians and all donors provided written informed consent.
Diagnosis of PGF (both primary and secondary) was based on criteria in the literature (3,16). The criteria were the presence of 2-3 cytopenic counts [absolute neutrophil cell (NEU) counts ≤ 0.5 × 109/L and platelet (PLT) counts ≤ 20 × 109/L, regardless if the hemoglobin (Hb) levels were ≤70 g/L] for at least 30 consecutive days; the presence of hypoplastic—aplastic BM when donor chimerism was complete; and the absence of GVHD, active infection, administration of a myelotoxic agent, and relapse. The blood values were determined by a hematology analyzer (Beckman Coulter, Brea, CA, USA). Primary PGF was defined as a slow or incomplete recovery of blood cell counts (NEU ≤0.5 × 109/L and PLT ≤20 × 109/L) at least 30 days after allo-HSCT. Secondary PGF was defined as a fall in blood cell counts to levels fulfilling the diagnostic criteria for PGF after successful and prompt hematopoietic engraftment.
Patients
Twenty patients were enrolled in our prospective multicenter trial at Nanfang Hospital, Southern Medical University and Guangzhou General Hospital of Guangzhou Command, and Zhujiang Hospital, Southern Medical University between July 2009 and July 2011. Six patients were female, and 14 were male. The median patient age was 26 years (range 14-46 years). Primary diseases included 18 cases with leukemia (5 were in complete remission and 13 were not in remission at the time of transplantation) and two cases with severe aplastic anemia (sAA) (sAA-I and sAA-II). Of these 20 patients, 7 displayed primary PGF, and 13 displayed secondary PGF. Patient characteristics are shown in Table 1.
Clinical Transplant Characteristics and Outcomes of Patients With Poor Graft Function (PGF)

CML, chronic myeloid leukemia; BC, blast crisis; MDS, myelodysplastic syndrome; AML, acute myeloid leukemia; AA, aplastic anemia; ALL, acute lymphoblastic leukemia; CP, chronic phase; NK-T; natural killer T-cells; AUL, acute undifferentiated leukemia; RD, related donor; URD, unrelated donor; BM, bone marrow; PBSC, peripheral blood stem cell.
In the Disease/Status column: NR, no remission; CR, complete remission. In the Effect column: PR, partial response; NR, no response; CR, complete response.
Did not improve after treatment;
x 106 cells/kg body weight;
patients died at PR status before the second round of MSC treatment.
Transplantation
Of the 20 patients, 11 transplantations were related donors, and 9 were unrelated donors. Four patients were human leukocyte antigen (HLA) locus-matched, and 16 were mismatched. Twelve patients received just peripheral blood stem cells (PBSCs), whereas eight received both PBSCs and a BM transplant. The median number of transfused cluster of differentiation 34-positive (CD34+) cells/kg was 5.51 × 106 (range 3.07-8.45 × 106). CD34+ cell selection was performed by immunomagnetic separation using the CliniMACS Device (Miltenyi Biotec GmbH, Gladbach, Germany). The conditioning regimens for the 18 patients with leukemia were as follows: four patients underwent total body irradiation (TBI; 4.5 Gy/day of TBI on days -5 and -4 administered from linear accelerators; Elekta AB, Stockholm, Sweden) + cyclophosphamide (CY; 60 mg/kg/day on days -3 and -2; ASTA Medica AG, Dresden, Germany); four were treated with busulfan (Bu; 3.2 mg/kg/day on days -7 to -4; Heumann Pharma GmbH, Nurember, Germany) + CY (60 mg/kg/day on days -3 and -2); 10 were treated with fludarabine (Flu; 30 mg/m2/day intravenously on days -10 to -6; Bayer, Leverkusen, Germany) and cytarabine (Ara-C; 2.0 g/m2/day, intravenously on days —10 to —6; Pfizer, New York, NY, USA) in addition to TBI + CY. CY (50 mg/kg/day on days -5 to -2) and anti-human thymocyte globulin (ATG; Genzyme, Cambridge, MA, USA; 3.0 mg/kg/day on days -5 to -1) were given to patients with sAA. Cyclosporine A (CsA; Novartis, Basel, Switzerland), in combination with methotrexate (MTX; Pfizer) (on days +1 and +3) was administered in leukemia patients undergoing HLA-matched sibling donor transplants for GVHD prophylaxis. CsA + MTX + ATG (total ATG dose of 6-10 mg/kg, on days -3 to -1 or -4 to 0) and/or mycophenolate (MMF, 0.5 g/day, b.i.d., on days 0 to +28; Roche, Basel, Switzerland) were given to patients undergoing HLA-mismatched related or unrelated donor transplantations. G-CSF (Amoytop Biotech, Fujian, China) was administered at 5 μg/kg/day commencing 3 days after the day in which the NEU was > 0.5 × 109/L for 2 consecutive days. Patients received red blood cells or PLT transfusions if Hb levels fell to ≤70 g/L or if the PLT counts reached ≤20 × 109/L. The characteristics of all transplant patients are shown in Table 1. The diagnosis, clinical grading, and treatment of acute GVHD (aGVHD) were performed according to established criteria for nonmyeloablative hematopoietic cell transplantation (NMA HCT). Diagnosis and grading of chronic GVHD (cGVHD) were performed using the National Institutes of Health (NIH) consensus criteria. Treatment was given for grades II—IV aGVHD and for extensive cGVHD. Initial treatment usually consisted of methylprednisolone (MP; Pharmacia Italia S.p.A, P, Milan, Italy) 0.8-1.6 mg/kg/day, with tapering doses initiated within 14 days. Steroid-refractory aGVHD was treated as per available investigational protocols or standard practice.
Infection Prophylaxis
Sulfamethoxazole (Xinhua Pharmaceutical Co. Ltd., Shandong, China) and norfloxacin (Xinhua Pharmaceutical Co. Ltd.) were given to all patients orally. Acyclovir (Qidu Pharmaceutical Co. Ltd., Shandong, China) was given daily from the beginning of conditioning to engraftment and then daily for 7 days every 2 weeks for up to 1 year after transplantation. Ganciclovir (Luoxin Pharmaceutical Co. Ltd., Shandong, China) was given for 2 weeks prior to transplantation for prophylaxis of cytomegalovirus (CMV) infections and then again during periods of CMV viremia within 1 year of transplantation. An antifungal agent (fluconazole or voriconazole; Pfizer) was given as an antifungal prophylaxis from the beginning of the conditioning until day +30 to +90 after transplantation.
Isolation and Ex Vivo Expansion of MSCs
Bone marrow-derived MSCs were obtained from five healthy HLA-unrelated third-party donors (aged 18 to 35; three males and two females) after obtaining written informed consent. Each 40 ml of BM aspirate was diluted 1:1 with human MSC growth medium [low-glucose Dulbecco's modified Eagle's Medium (L-DMEM; Hyclone, Logan, UT, USA)] with 10% (v/v) fetal bovine serum (FBS; Hyclone) and layered over a Ficoll-Paque solution (1077 mg/ml; Amersham Biosciences, Uppsala, Sweden). After centrifugation, mononuclear cells were collected from the interface, washed, and suspended in human MSC growth medium at a density of 5,000/cm2. After 3 days, the medium was replaced and nonadherent cells were removed. Adherent cells were further cultured, and medium was changed every 3 days. When monolay-ers attained 70-80% confluence, cells were detached using a trypsin—EDTA solution (Gibco, Grand Island, NY, USA) and passaged at a ratio of 1:3. Cells at passage 1 (and thereafter) were plated at a mean density of 5 × 103/cm2 in T175 cell culture flasks (Greiner, Frickenhausen, Germany). Cells were harvested after four to five passages, and MSCs were detached, washed in CliniMACS PBS—EDTA buffer (AmCell; Miltenyi Biotec GmbH) three times, and resuspended in 0.9% NaCl (w/v; Qilu, Shandong, China) for clinical use. Flow cytometric analysis was performed using a FACSort (Becton Dickinson, San Jose, CA, USA), and data were analyzed using CellQuest software (Becton Dickinson). Standard staining and analysis protocols were applied using commercial fluorescein isothiocyanate (FITC)- or phycoerythrin (PE)-conjugated antibodies against CD11a, CD29, CD34, CD44, CD45, CD73, CD90, CD105, and CD166 (Becton Dickinson). The potential for osteogenic and adipogenic differentiation was proven by culturing in standard differentiation media (Miltenyi Biotec). (Fig. 1A, B). The expanded cells expressed CD29, CD44, CD73, CD90, CD105, and CD166, but not CD11a, CD34, or CD45 (Fig. 1C). All MSCs were screened (negative) for pathogens and contaminants, including bacteria, fungi, viruses, mycoplasma, and endotoxin before use.

Characterization of MSCs. The quality control of mesenchymal stem cells (MSCs) for clinical use was confirmed by their multilineage potential, such as adipogenic and osteogenic differentiation (A, B), as well as the surface markers (C). Scale bar: 50 μm.
MSC Treatment and Response Criteria
MSCs (1 × 106 cells/kg) were given at 4-week intervals and were intravenously infused via a central venous catheter (Smiths Medical Deutschland GmbH, Grasbrunn, Germany) over 15 min. If the NEU and PLT levels did not attain the complete response (CR) standards within 4 weeks, a second round of MSC treatment was given. Vital signs of all patients were closely monitored for 24 h after MSC administration. During MSC treatment, hepatic and renal function [alanine aminotransferase (ALT), aspartate aminotransferase (AST), γ-glutamyl transpeptidase (GGT), alkaline phosphatase (ALP), albumin (Alb), total bilirubin (TBIL), direct (conjugated) bilirubin (DBIL), indirect (unconjugated) bilirubin (IBIL), serum creatinine (SCr), uric acid (UA), and blood urea nitrogen (BUN)] and myocardial enzyme levels [creatine kinase (CK), creatine kinase-muscle 7 brain (CK-MB), lactate dehydrogenase (LDH), and AST] of serum were monitored weekly by fully automatic biochemical analyzer (Beckman Coulter), whereas electrocardiography (ECG-1350P, Nihon Kohden, Tokyo, Japan) was performed once every 2 weeks. Blood counts were performed twice weekly until the NEU counts reached > 0.5 × 109/L, and the PLT counts reached > 20 × 109/L; the counts were then performed once weekly for 12 weeks. Bone marrow aspiration and biopsies were performed at 28 days and 56 days post-MSC treatment, respectively. T-, B-, and natural killer (NK) cells in peripheral blood were enumerated 28 days before MSC treatment and 56 days after MSC treatment. Lymphocyte subsets were enumerated by using three-color flow cytometry (Becton Dickinson). Briefly, blood mononuclear cells (MNCs) were stained with mouse monoclonal antibodies conjugated to fluoro-chromes [FITC, PE, and PE plus cyanine 5(PE/Cy5); BD Biosciences, San Jose, CA, USA]. Flow cytometry data were acquired by using a FACSCalibur flow cytometer (BD Biosciences) and were analyzed with Winlist software (Verity, Topsham, ME, USA). T-cells were defined as CD3+ MNCs. B-cells were defined as MNCs expressing CD19 or CD20 and not brightly expressing CD3, CD10, CD13, CD14, CD16, CD34, or CD56. NK cells were defined as MNCs expressing CD16 or CD56 and not expressing CD3 or CD14.
The response criteria were defined as follows: a CR was defined as an NEU > 1.5 × 109/L and PLT > 50 × 109/L for 3 consecutive days after MSC treatment. A partial response (PR) was defined as an NEU > 0.5 × 109/L and PLT > 20 × 109/L for 3 consecutive days after MSC treatment, thus not attaining the CR standard. No response (NR) was defined as persistent PGF for more than 28 days after MSC treatment.
Additional Treatments Prescribed During PGF
G-CSF was given at 5 μg/kg/day, commencing on the day after MSC treatment and continuing to the time at which NEU reached > 0.5 × 109/L for 2 consecutive days. Patients received red blood cells and PLT transfusions if Hb levels were ≤ 70 g/L and PLT levels were ≤ 20.0 × 109/L.
Monitoring of EBV and CMV DNA Blood Loads
Molecular studies were done for Epstein—Barr virus (EBV) and CMV by in situ hybridization for EBV- and CMV-encoded RNA. Generally, EBV—DNA and CMV—DNA copy numbers in plasma were detected weekly within 3 months posttransplantation with real-time quantitative polymerase chain reaction (RQ-PCR; ABI, Foster City, CA, USA). The mixture of plasma (50 μl) with nucleic acid extract (50 μl) was heated at 99°C for 10 min, followed by centrifugation at 11,340 × g for 10 min. The supernatant was mixed with the primers at the concentrations specified by the manufacturer (Shanghai ZJ Bio-Tech Co., Ltd., Shanghai, China) and PCR was performed under the described conditions. CMV detection was performed as follows: 1 cycle at 50°C for 2 min, 95°C for 10 min, and 45 cycles at 95°C for 15 s and 60°C for 1 min. The CMV forward primer was 5′-GAAG GTGCAGGTGCCCTG-3′, CMV reverse primer was 5′-GT GTCGACGAACGACGTACG-3′, and the probe primer was 6-carboxyfluorescein (FAM)-ACGGTGCTGTAGAC CCGCATACAAA- tetramethylrhodamine (TAMRA). EBV detection was performed as follows: 37°C for 2 min, and 94°C for 2 min followed by 40 cycles at 93°C for 15 s and 60°C for 1 min. The sequences of the TaqMan probes and primers for EBV were as follows: EBV forward primer was 5′-CCAGTGCTGTGATCGAGCATCT-3′, EBV reverse primer was 5′-CTGCTGACAAACTGCTGCATTC-3′, and EBV TaqMan probe was (FAM)-TCTGCTGTTGTTTCT GTCTCACCTACCGG-(TAMRA). The normal threshold for CMV-DNA and EBV-DNA copies in plasma was less than 500 copies/ml according to the manufacturer. Therefore, CMV-DNA or EBV-DNA was considered positive when the copies exceeded 500 copies/ml (40).
During months 4-9, EBV—DNA and CMV—DNA loads were detected once every 2 weeks. From months 10-24 posttransplantation, EBV—DNA and CMV—DNA loads were detected once a month. If EBV—DNA and/or CMV—DNA were positive, then EBV—DNA and/or CMV—DNA were monitored twice per week. Patients with PGF who accepted MSC treatment were monitored for EBV—DNA and CMV—DNA plasma loads once weekly for 6 months.
Evaluation Points and Definitions
In the present study, we focused on infections, leukemic relapse, any toxic side effects, and the effect of MSC treatment on hematopoietic recovery. Hematopoietic reconstitution posttransplantation was defined as reconstitution of both neutrophil and PLT numbers. Neutrophil reconstitution was defined as occurring during the first 3 consecutive days with an NEU > 0.5 × 109/L, and PLT reconstitution was defined as the first time levels reached > 20 × 109/L for 3 consecutive days. Donor chimerism in peripheral blood and BM was analyzed using fluorescence in situ hybridization (FISH) in gender-mismatched transplantations and by short tandem repeat (STR) analysis via PCR-based technology on peripheral blood in gender matched. FISH was performed using a chromosome in situ hybridization kit as recommended by the manufacturer (Oncor, Gaithersburg, MD, USA), and STR analysis was performed with the AmpFlSTR Blue PCR amplification kit (PE Applied Biosystems, Foster City, CA, USA). Complete chimerism was defined as > 95% donor derived and mixed chimerism was defined as present when 5-95% of cells were donor derived (22). Infections mainly occurred within the first 100 days after MSC treatment. Toxic side effects caused by the treatment included acute toxicity and late side effects. Acute toxicity principally involved the heart, liver, and kidney. Late toxic side effects primarily involved the development of secondary tumors and primary disease relapse. Molecular and cryptogenic relapse was assessed based on chimerism status and the presence of tumor markers, defined as a 0.5% decrease in donor chimerism status or the reappearance of tumor gene markers (22). Hematologic relapse was defined as the reappearance of blasts in the peripheral blood, by any manifestation of leukemia outside of the hematopoietic system, or by the presence of > 5% blasts in a BM smear (23).
Statistical Analysis
The SPSS software package (SPSS, Chicago, IL, USA) was used to analyze all data. Differences among lymphocyte subsets pre- and posttreatment were assessed using Student's t test, and differences between the effects on primary and secondary PGF patients were explored using the chi-squared test. A value of p < 0.05 was considered to indicate statistical significance.
Results
Patient Clinical Characteristics Prior to MSC Treatment
All patients received sporadic PLT transfusion and repeat red blood cell transfusions prior to MSC treatment and displayed complete chimerism at day +28 posttransplantation and at the time of MSC treatment. The median neutrophil and PLT counts and Hb levels were 0.35 × 109/L (0.31 - 0.47 × 109/L), 11.0 × 109/L (9.0 - 17.0 × 109/L), and 86.9 g/L (72.3 - 99.6 g/l), respectively, at the time of MSC treatment. Eight patients, including five with primary PGF and three with secondary PGF, had bacterial and/or fungal infections (bacterial septicemia in one patient, bacterial pneumonia in one patient, fungal pneumonia in three patients, and mixed bacterial and fungal pneumonia in three patients) at the time of MSC treatment. Three patients exhibited EBV—DNA viremia, whereas five exhibited CMV—DNA viremia before MSC treatment. However, the peripheral blood of all patients was EBV—DNA and CMV—DNA negative at the time of MSC treatment.
Of the 20 patients, 11 (one with primary PGF and 10 with secondary PGF) received MP during PGF periods. The characteristics of these patients are outlined below. Four experienced a fever for more than 1 week during PGF but showed no evidence of infection; such patients were considered to exhibit GVHD and were treated with MP, following which their body temperature returned to normal, but PGF was still evident. Two patients with extensive chronic GVHD who were treated with combinations of various immunosuppressant agents, including MP, developed secondary PGF when the chronic GVHD improved. Five patients with acute GVHD were treated with MP plus CsA, but developed secondary PGF when GVHD was in complete remission. MP had been given at a median dosage of 12 mg/day (range 8-24 mg/day) for a median of 23 days (range 5-109 days) before MSC treatment.
PGF Response to MSC Treatment
Seven patients with primary PGF received MSCs at a median time of 33 days (range 31-35 days) after allo-HSCT and 13 patients with secondary PGF received MSCs at a median of 35 days (range 34-41 days) after PGF occurred. Seventeen patients were responsive, and three (two with primary PGF and one with secondary PGF) were not responsive to MSC treatment (one to three times) at 28-day intervals. Five patients (two with primary PGF and three with secondary PGF) achieved CR, and 12 achieved PR; two unresponsive patients died of septicemia and graft rejection at 18 days and 20 days, respectively, after MSC treatment, and two PR patients died of septicemia and severe pneumonia at 19 days and 23 days, respectively, after MSC treatment. Of the 11 patients, including 10 in PR and one in NR, 10 patients (one with primary PGF and nine with secondary PGF) achieved CR after the first round of MSC treatment, and the patient who was unresponsive to MSC treatment remained NR after the second and third rounds of MSC treatment and died due to hemorrhagic cystitis 93 days after the first round of treatment. Of the 17 patients who were responsive to MSC treatment, the median time to PR and CR (the two patients with PR that died before the second round were not analyzed for CR), as assessed using neutrophil counts, were 16 days (range 11-37 days) and 39 days (range 23-55 days), respectively, after MSC treatment. The median time to PR and CR, as assessed using PLT counts, was 24 days (range 17-31 days) and 42 days (range 26-55 days), respectively, after MSC treatment. Recovery trends of NEU and PLT counts of all patients after MSC treatment are shown in Figure 2. No significant difference was evident when primary and secondary PGF patients were compared in terms of total recovery or the proportion of patients exhibiting CR or PR after each round (values were p = 0.344, p = 0.06, and p = 0.9, respectively). Of the 15 responsive patients who survived, BM smears (Fig. 3, BM smears of patient No. 4) and biopsy data (Fig. 4, BM biopsy of patient No. 4) revealed improvements in the extent of cell proliferation, cellularity, and the proportion of hematopoietic tissue after one to three MSC treatments.

Recovery trends of neutrophil (NEU) (A) and platelet (PLT) (B) counts of all patients after MSC treatment.

Bone marrow smear of patient No. 4 before and after MSC treatment. Bone marrow smear the closest day before MSC treatment (A), 28 days (B), and 56 days (C) after treatment (magnification: 40×). Bone marrow (BM) smears revealed improvements in the extent of cell proliferation (compared by the M:E ratio, the ratio of all granulocytic plus monocytic cells to all erythroblasts) and cellularity. Scale bar: 50 μm.
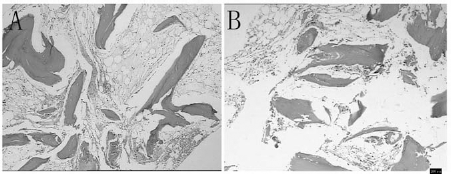
Histology of BM biopsy of patient No. 4 before and after MSC treatment. Photomicrographs of hematoxylin and eosin-stained BM biopsy of patient during poor graft function (PGF) periods (A) and after the second round of MSC treatment (B). During diagnosis, only 5% of hematopoietic tissue was detected, whereas 25 days after the second treatment, recovery of the hematopoietic system was observed up to 35%. The percentage of cells was quantified by the percentage of hematopoietic tissue area to the whole area of the biopsy. Scale bar: 200 μm.
Infections
Within the first 100 days after MSC treatment, 13 patients developed 20 episodes of infection (CMV—DNA and EBV—DNA viremia were not considered an event of infection). Seven patients had a bacterial infection, two had an invasive fungal infection (IFI), three had a viral infection, four had a mixed infection (three bacterial and fungal; one bacterial and viral), and four had infections of unknown etiology. Of the eight patients who suffered from infections at the time of MSC treatment, three infections (two fungal and one bacterial) were under control, whereas five infections were progressing. In addition, within the first 100 days after MSC treatment, five patients (three with primary PGF and two with secondary PGF) experienced CMV—DNA viremia, whereas seven (two with primary PGF and five with secondary PGF) experienced EBV—DNA viremia. Within a median follow-up period of 125 days (range 11-579 days) after MSC treatment, six patients (three with primary PGF and three with secondary PGF) experienced CMV—DNA viremia, whereas eight (two with primary PGF and six with secondary PGF) developed EBV—DNA viremia, including three which obtained EBV-associated posttransplant lymphoproliferative disorders (PTLD) at 7, 9, and 15 days after commencement of EBV—DNA viremia. Two patients with PTLD attained complete remission, but one patient died due to PTLD after treatment with rituximab and donor lymphocyte infusion (DLI). Blood samples became (and remained) negative for EBV—DNA during follow-up of other patients with EBV—DNA viremia with a gradual decrement of immunosuppressant agents, antiviral therapy, rituximab, etc. Six patients with CMV—DNA viremia did not develop any CMV-associated disease within a median follow-up time of 508 days (range 166-904 days) posttransplantation. Seven patients died from infections, including three with mixed infections and three with bacterial infections within 100 days post-MSC treatment and one with viral myocarditis (a Coxsackievirus infection) at 173 days after MSC treatment.
GVHD After MSC Treatment
One of the five patients with acute GVHD who had achieved complete remission at the time of MSC treatment developed local chronic GVHD after such treatment. Two patients who accepted DLI for PTLD developed grade II acute GVHD and local chronic GVHD on days +24 and +89 after DLI, respectively, which was on days +107 and +147 after MSC treatment, respectively. Two patients who exhibited extensive chronic GVHD at the time of MSC treatment improved after such treatment.
Lymphocyte Subsets in Peripheral Blood
Flow cytometric analysis was used to examine the lymphocyte subsets in peripheral blood pre- and post-MSC treatment. In 15 patients who were responsive to such treatment, a significant difference was not evident between patients with primary and secondary PGF in terms of the proportions of CD3+CD4+ and CD3+CD8+ T-cells, CD19+ B-cells, NK cells, or CD4+CD25+ FoxP3 regulatory T-cells prior to MSC treatment; the proportion of CD3+ T-cells and CD3+CD4+ T-cells following 56 days after MSC treatment was higher than those prior to MSC treatment (88.05 ± 1.26% vs. 79.05 ± 3.23%, p = 0.015; and 24.09 ± 1.95% vs. 17.40 ± 1.57%, p = 0.012, respectively) (Fig. 5). The proportion of CD3+CD8+ T-cells present following 56 days after MSC treatment was lower than that prior to treatment with MSCs (51.91 ± 2.86% vs. 61.39 ± 3.43%, p = 0.043) (Fig. 5), and the ratio of CD3+CD4+ to CD3+CD8+ T-cells was higher (0.31 ± 0.04 vs. 0.51 ± 0.06, p = 0.01) (Fig. 5). The proportion of CD4+CD25+ FoxP3 regulatory T-cells, CD19+ B-cells, and NK cells did not change significantly after MSC transfusion (values were p = 0.231, p = 0.493, and p = 0.327, respectively). Statistical analysis could not be conducted on data from the three patients who did not respond to MSC treatment because the data were too few.

Lymphocyte subsets of all patients before and 56 days after MSC treatment. The proportion of CD3+ T-cells and CD3+CD4+ T-cells following 56 days after MSC treatment was higher than those prior to MSC treatment (88.05 ± 1.26% vs. 79.05 ± 3.23%, p = 0.015; and 24.09 ± 1.95% vs. 17.40 ± 1.57%, p = 0.012, respectively) (A), and the ratio of CD3+CD4+ to CD3+CD8+ T-cells was higher (0.31 ± 0.04 vs. 0.51 ± 0.06, p = 0.01) (B).
Toxic Side Effects
No patient experienced any acute toxicity (a significant problem in heart, lung, liver, or kidney function during the first 24 h following MSC treatment). Apart from three instances of EBV-associated PTLD, no other secondary tumor was observed over a median follow-up time of 508 days (range 166-904 days) posttransplantation.
Survival and Relapse
At a median follow-up time of 508 days (range 166-904 days) posttransplantation, nine patients survived, whereas 11 died (six with primary PGF and five with secondary PGF). The causes of death included infection alone (n = 6), graft rejection plus infection (n = 1), hemorrhagic cystitis (n = 1), PTLD (n = 1), and leukemia relapse (n = 2). The two patients that experienced leukemic relapse did so on days +193 and +273 posttransplantation (114 and 180 days after MSC treatment, respectively).
Discussion
PGF is a life-threatening side effect that occurs after allo-HSCT. The pathogenesis of PGF remains poorly understood. Many factors may influence the development of primary or secondary PGF, including prior alloimmunization, intensity of the conditioning regimen, hematopoietic stem cell dose transplanted, HLA matching, donor type, GVHD, and infection (7,29). In the present study, all patients showed at least two of the above risk factors for PGF (prior alloimmunization, HLA mismatching, unrelated donor, and GVHD). Additionally, an immunologic risk factor was evident for each patient (Table 1). Moreover, the ratio of CD3+CD4+ to CD3+CD8+ T-cells was inverted at the time of MSC treatment and improved after MSC treatment in 15 patients who were responsive to MSCs. This is reminiscent of a manifestation of AA, which is considered an immunologic disease (41). Thus, we considered the possibility that an immunologic factor might play a role in pathogenesis of PGF in this group. However, the pathogenesis of PGF could not be fully explained by the immunologic risk factors evidenced by our patients. We concluded that PGF developed in five patients with acute GVHD and two with chronic GVHD, even after such patients achieved either CR or improved GVHD; however, the PGF did not improve when MP and/or other immunosuppressive agents were given. Therefore, additional studies will be required to determine whether PGF is associated with immunologic factors.
Data from several reports show that MSC infusion was effective for the treatment of both primary and secondary PGF (21,26,28,33). Meuleman et al. reported that two primary PGF patients experienced hematopoietic recovery, whereas four showed no response upon treatment with ex vivo-expanded MSCs derived from the BM of a third-party donor (26). Additionally, Sanchez-Guijo et al. described four patients with secondary PGF with MSCs from a third-party donor that obtained hematopoietic recovery over a mean follow-up period of 12.5 months (33). In the reports aforementioned, MSCs were infused solely, but in our patients, MSC infusion was used simultaneously with G-CSF. Seventeen of 20 patients with PGF experienced hematopoietic recovery after MSC treatment. Our small sample also indicated that BM-derived MSCs from a third-party donor were effective for both primary and secondary PGF. However, it is necessary to clarify the role of G-CSF in PGF. Owing to the limitation of sample resources, we did not design a control group in our prospective trial. There was no significant improvement of PGF with G-CSF treatment alone for a median of 27 days (range 23-30 days) before MSC treatment, but PGF did improve after adding MSCs. It is necessary to determine whether MSCs combined with G-CSF can lead to a better prognosis of PGF than MSCs alone. MSCs used to treat PGF might secrete hematopoietic cytokines and/or reconstitute the BM stroma. Our T-cell subset data described above might indicate that MSCs counter PGF by regulating the immune response by appropriately balancing the ratio of CD3+CD4+ T-cells to CD3+CD8+ T-cells.
It has been debated as to whether MSCs with immunosuppressive properties can increase the risk of infection or tumor relapse (12-14,17,25,30,32,35-38,42). Potian et al. found that third-party MSCs did not suppress the proliferation induced by recall antigens, such as Candida albicans and tetanus toxin, during a study on infections (32). Moreover, Karlsson et al. reported that MSCs had little effect on the T-cell response to EBV or CMV (12). In contrast, Maitra et al. observed that MSCs suppressed interferon-γ (IFN-γ) production, as shown by the Elispot assay, in response to a purified derivative of tuberculin (25). In our present work, we showed that seven patients (35%) experienced EBV—DNA viremia within the first 100 days after MSC treatment, and three of these (15%) developed EBV-associated PTLD. This proportion was higher than that seen in patients who did not receive MSC treatment, as evidenced both in the present study and previous reports (6,11,22). However, we could not exclude the possible presence of other risk factors that might cause EBV—DNA viremia and development of EBV-associated PTLD. Such factors include the use of ATG, HLA mismatching, and unrelated donor transplantation. Of our 20 patients, 14 accepted ATG for GVHD prophylaxis, and 19 were unrelated or HLA mismatch-related transplantations (Table 1). The use of ATG, unrelated, and HLA mismatching-related transplantations are major risk factors for EBV infection and reactivation and for the development of EBV-associated PTLD (5,6,11,22). In addition, other factors, for example, prolonged neutropenia or long-term corticosteroid treatment, may increase the susceptibility to infection. Therefore, additional studies will be needed to determine whether MSCs increase the risk of EBV reactivation or the occurrence of EBV-associated PTLD. Although six of 20 patients died from infections within the first 100 days after MSC transplantation, we could not safely conclude that MSCs increased the incidence of infections other than EBV. Other factors, for example, the presence of a severe infection at the time of MSC treatment, prolonged neutropenia, and/or long-term treatment with immunosuppressant agents, might affect susceptibility to infection.
In an open-label randomized clinical trial, HLA-identical sibling-matched HSCs were transplanted or cotransplanted with MSCs in patients with hematologic malignancies. Of 15 patients in the non-MSC group, three (20%) experienced tumor relapse, whereas six out of 10 (60%) experienced tumor relapse in the MSC-treated group. The relapse rate in the MSC group was thus significantly higher than that in the non-MSC group (30). However, another randomized, controlled clinical study showed that cotransplantation of MSCs and HSCs did not increase the recurrence of leukemia (21). In our present prospective study, only two of 20 patients experienced leukemia relapse after MSC treatment. Our series was too small to allow us to draw any firm conclusion on the potential increase in relapse rate evident after MSC treatment. In addition, several studies have suggested that MSCs promote secondary tumor growth via enhancement of neovascularization and boosting of metastatic potential (13,34,37). In contrast, MSCs have been reported to inhibit tumor growth in a model of Kaposi's sarcoma (14). We found that three patients developed EBV-associated PTLD, while secondary tumors were not noted.
Conclusions
Our data indicate that BM-derived MSCs from a third-party donor can effectively be used to treat patients who develop primary or secondary PGF after allo-HSCT. Additional studies are required to determine whether MSCs increase the risk of EBV infection and reactivation or the development of EBV-associated PTLD.
Footnotes
Acknowledgments
This project was supported by the National High Technology Research and Development Program of China (863 Program) (2011AA020105), the National Natural Science Foundation of China (30971300, 81000231), the Science and Technology Planning Project of Guangdong Province, China (2009A03020007, 2007A032100003), and the Key Scientific and Technological Program of Guangzhou City (2008A1-E40115, 2010U1-E00551). The authors declare no conflicts of interest.
