Abstract
The number of HLA-haploidentical allogeneic hematopoietic stem-cell transplantation (Haplo-HSCT) is increasing. Comparative studies about Haplo-HSCT versus allo-HSCT with HLA-matched sibling donors (MSD-HSCT) have been tried in leukemias and B-cell lymphomas. Few studies were reported in Peripheral T-cell lymphomas (PTCLs). We performed a multicenter retrospective study about 52 patients with PTCLs undergoing Haplo-HSCT (n = 20) or MSD-HSCT (n = 32). All Haplo-HSCT recipients received antithymocyte globulin (ATG) based graft versus host disease (GVHD) prophylaxis. The median follow-up for all survivors was 38 months. The 100-day cumulative incidence of grade II to IV acute GVHD was similar (19% in the MSD-HSCT group versus 28% in the Haplo-HSCT group, P = 0.52). The 2-year cumulative incidence of chronic GVHD (limited and extensive) after Haplo-HSCT (30%) was also similar with that in the MSD-HSCT group (50%, P = 0.15). The 3-year relapse rates (33% vs 27%, P = 0.84) and non-relapse mortality (21% vs 22%, P = 0.78) did not differ between these two groups. There were also no differences in 3-year overall survival (OS) (48% vs 50%, P = 0.78) and progression-free survival (47% vs 51%, P = 0.95) between these two groups. On multivariate analysis, prognostic index for T-cell lymphoma (PIT) score (higher than 1: hazard ratio [HR], 4.0; P = 0.003) and disease status (stable or progression disease before HSCT: HR, 2.8; P = 0.03) were independent variables associated with worse OS. We concluded that ATG-based haplo-HSCT platform could work as an alternative to MSD-HSCT for patients with PTCLs.
Keywords
Introduction
Peripheral T-cell lymphomas (PTCLs) are a group of malignancies with great heterogeneity in morphology, pathophysiology, immunophenotypes, molecular and clinical characteristics, and geographic distribution. PTCLs account for less than 15% of all non-Hodgkin’s lymphomas (NHL) in Western countries 1 . However, the incidence is approximately 25%–30% in East Asia, where EBV-associated NK/T-cell lymphomas are more frequent 2,3 . In east Asia, more than 90% of PTCLs are peripheral T-cell lymphoma not otherwise specified (PTCL-NOS), anaplastic lymphoma kinase (ALK)-positive or negative anaplastic large cell lymphoma (ALCL), angioimmunoblastic T-cell lymphoma (AITL), and NK/T-cell lymphoma (NK/TCL). Less frequent subtypes include hepatosplenic γ/δ lymphoma (HSL), enteropathy-type T-cell lymphoma, and subcutaneous-like T-cell lymphoma.
Except for patients with ALK-positive ALCL, the prognosis of PTCLs patients was generally poor with standard first-line treatment using CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone) or CHOP-like regimens 1,4,5 . With the rapid growth of knowledge about (epi)genetic changes in PTCLs, more drugs have been investigated and approved for clinical use 6 . With the exception of brentuximab vedotin (BV) showing great efficacy in CD30-expressing PTCLs 7 , few of these novel agents were reported to significantly improved the survival of PTCLs patients 8,9 . Autologous hematopoietic stem cell transplantation (auto-HSCT) was still recommended as the consolidation treatment for most histological subtypes 9,10 . As a valuable treatment option, allo-HSCT has been investigated for patients with refractory or relapsed PTCLs over decades. About one half of the patients with relapsed or refractory PTCLs can obtain long-term survive by allo-HSCT 6,9 . Progress in supportive care and transplant techniques has greatly decreased transplantation-related complications and expanded the eligibility of allo-HSCT. There has been convincing evidence that Haplo-HSCT can achieve comparable outcomes with MSD-HSCT for lymphomas 11,12 . However, there is yet no comparative study about haplo-HSCT versus MSD-HSCT in PTCLs. Herein, in this study, we conducted a multicenter retrospective analysis of 52 patients with PTCLs who underwent haplo-HSCT or MSD-HSCT in 8 hospitals across China between July, 2007 and June, 2017.
Materials and Methods
Study Design and Population
Consecutive patients with PTCLs who received their allo-HSCT between July 2007 and June 2017 at 8 tertiary hospitals across China were analyzed. All diagnosis of these patients was initially confirmed by hematopathologists at each hospital, and then was centrally reviewed again according to the 2016 edition of the WHO classification of lymphoid neoplasms. Exclusions were patients received (1) allo-HSCT other than Haplo-HSCT or MSD-HSCT; (2) non-myeloablative conditioning regimens; (3) prior auto or more than one allo-HSCT. All patients signed consent forms for the retrospective collection and use of their data. This study was reviewed and approved by the Medical Ethics Review Boards at each center.
HLA Matching and Stem Cell Collection
All patients and donors were examined with high-resolution DNA-typing techniques. HLA-matched related donors were considered when they were compatible with the recipient for all the 10 allelic levels. All donors were selected based on HLA-matched loci, younger age, male sex and better performance status. In case HLA-matched related or unrelated donors were unavailable, haplo-HSCT was considered as an alternative. All donors were mobilized with recombinant human G-CSF (Filgrastim, Kyowa Kirin, Tokyo, Japan; 5 μg/kg/day) for 5 to 6 consecutive days. Stem cell harvests were collected from the peripheral blood on the fifth day and continued until sufficient number of cells was achieved. The threshold was >5 × 108/kg of recipient weight for mononuclear cells and >2 × 106/kg of recipient weight for CD34+ cells.
Conditioning Regimens and GVHD Prophylaxis
Allo-HSCT that using nonmyeloablative or reduced conditioning regimens was not routinely performed at these 8 medical centers. Auto-HSCT and myeloablative allo-HSCT were their mainly procedures. So, all allo-HSCT included in this study were myeloablative conditioning regimens (MAC). Three main regimens were as follows 13 : (1) modified BuCy regimen: busulfan (9.6 mg/kg, intravenously, days −10 to −8), carmustine, (250 mg/m2, days −5), cytarabine (4 g/m2, days −7 to −6), cyclophosphamide (100 mg/kg, days −4 to −3); (2) modified FB regimen: substitution of cyclophosphamide in BuCy with fludarabine (150 mg/m2, days −7 to −3); (3) TBI+Cy: total body irradiation (TBI, 8–10 Gy, days −7 to −6), cyclophosphamide (100 mg/kg, days −4 to −3). All recipients received cyclosporine A (CsA), short-term methotrexate (MTX) and mycophenolate mofetil (MMF) based regimens. ATG (Thymoglobulin, rabbit; Genzyme Europe B.V., Naarden, the Netherlands, 10mg/kg, days −5 to −2) was additionally used in haplo-HSCT.
Definitions and Statistics
Demographic and transplantation-related characteristics of patients were collected. Neutrophil engraftment, platelet engraftment, aGVHD, and cGVHD (limited or extensive) were evaluated and graded according to international criteria 14,15 . Patients’ responses were basically evaluated with computed tomography (CT) scan, or positron emission tomography (PET), and bone marrow aspiration or biopsy for patients with bone marrow involvement before HSCT. Responses to therapy were evaluated according to the International standards 16 . GVHD-free/relapse-free survival (GRFS) was refined as survival without the following events: grade 3-4 acute GVHD, cGVHD requiring systemic immunosuppressive treatment, disease relapse, or death from any cause after allo-HSCT. 17 Response evaluations were performed before and 3 to 6 months after allo-HSCT, and thereafter every half-year unless clinically indicated.
Descriptive analyses methods were used to show baseline characteristics of patients. Overall survival (OS) and progression free survival (PFS) were primary outcomes. And secondary outcomes were non-relapse mortality (NRM) and relapse (progression). OS and PFS was calculated with the Kaplan-Meier method and compared using the log-rank test. For NRM, relapse, and GVHD, their cumulative incidences were estimated with competing-risk analysis and compared with Gray’s test. The following variables were tested in univariate analysis: age, sex, type of HSCT, bone marrow involvement at diagnosis, prognostic index for T-cell lymphoma (PIT) score, disease stage at diagnosis, cycles of therapy before HSCT, remission status before HSCT, and time from diagnosis to HSCT. Cox multivariate analysis was used to identify any significant associations between variables and the outcomes of interest; Variables with a p value < 0.50 were entered in Cox multivariate analysis, and only those with p value ≤ 0.05 were retained in the final model. All analysis was performed with R software, version 2.12.
Results
Baseline and Treatment-Related Characteristics
A total of 52 consecutive patients with PTCLs were analyzed including MSD-HSCT (n = 32) and ahaplo-HSCT (n = 20). Baseline patient- and transplantation-related characteristics were shown in Table 1. Most patients in the two cohorts were male, were diagnosed with B symptoms or extranodal involvement, and had higher index scores or advanced disease. There were no significant differences in most variables. However, recipients of Haplo-HSCT all received ATG-based GVHD prophylaxis (P = 0.001), and were more likely to be diagnosed with BM involvement before transplantation (P = 0.03).
Baseline and Transplant Characteristics of Patients Receiving According to the Type of Donor.

HSCT, hematopoietic stem cell transplantation; PTCL-NOS, peripheral T-cell lymphoma not otherwise specified; AITL, angioimmunoblastic T-cell lymphoma; AITL, angioimmunoblastic T-cell lymphoma; ALK-pos ALCL, anaplastic lymphoma kinase positive anaplastic large cell lymphoma; ALK-neg ALCL, ALK-negative ALCL; BM, bone marrow; CNS, central nervous system; aaIPI, age adjusted international prognostic index; PIT, prognostic index of T-cell non Hodgkin lymphoma; CR, complete remission; PR, partial remission; SD, stable disease; PD, progression disease; TBI/Cy, total body irradiation, cyclophosphamide; Bu/Cy, busulfan, cyclophosphamide; BEAM, semustine/carmustine, etoposide, cytarabine, melphalan; FB, fludarabine, busulfan; HLA, human leukocyte antigen; GVHD, graft-versus-host disease; CSA, cyclosporine; MTX, methotrexate; ATG, antithymocyte globulin; MMF, mycophenolate mofetil. a Hepatosplenic γ/δ T-cell lymphoma(n = 1), Subcutaneous panniculitis-like T-cell lymphoma(n = 2), primary cutaneous invasive epidermo-tropic CD8 positive cytotoxicity T-cell lymphoma(n = 1). b: Hepatosplenic γ/δ T-cell lymphoma(n = 3), Subcutaneous panniculitis-like T-cell lymphoma(n = 1).
Engraftments and GVHD
The cumulative incidence of neutrophil and platelet engraftment at day 28 were similar between Haplo-HSCT and MSD-HSCT group. Data on neutrophil recovery were missing in 4 patients (1 in the MSD-HSCT group and 3 in the Haplo-HCT group). The median time of neutrophil engraftment in the Haplo-HSCT group was 13 days (range: 10–22 days), which was similar to the 14 days in the MSD-HSCT group (range: 9–27 days, P = 0.61). Data on platelet engraftment was missing in three patients (1 in the MSD-HSCT group and 2 in the Haplo-HCT group). No difference was observed in the median time of platelet engraftment: 13 days (range: 9–35) in the Haplo-HSCT group versus 16 days (10–38 days) in the MSD-HSCT group (P = 0.29). All patients in both groups had full donor chimerism at day 28 evaluation after transplantation.
The 100-day cumulative incidence of Ⅱ-Ⅳ aGVHD for the entire group was 23% (95% confidence interval [CI], 13%–35%) and was similar between these two groups (19% [95% CI, 8%–35%]) in the MSD-HSCT group versus 28% [95% CI, 10%–50%] in the Haplo-HSCT group, P = 0.52, Fig. 1A). The cumulative incidence of limited and extensive chronic GVHD at 2 years was 40% (95% CI, 26%–52%) for the entire group. The 2-year incidence of cGVHD in the Haplo-HSCT group (30% [95% CI, 10%–53%]) was also similar with that in the MSD-HSCT group (50% [95% CI, 31%–67%], P = 0.15, Fig. 1B).
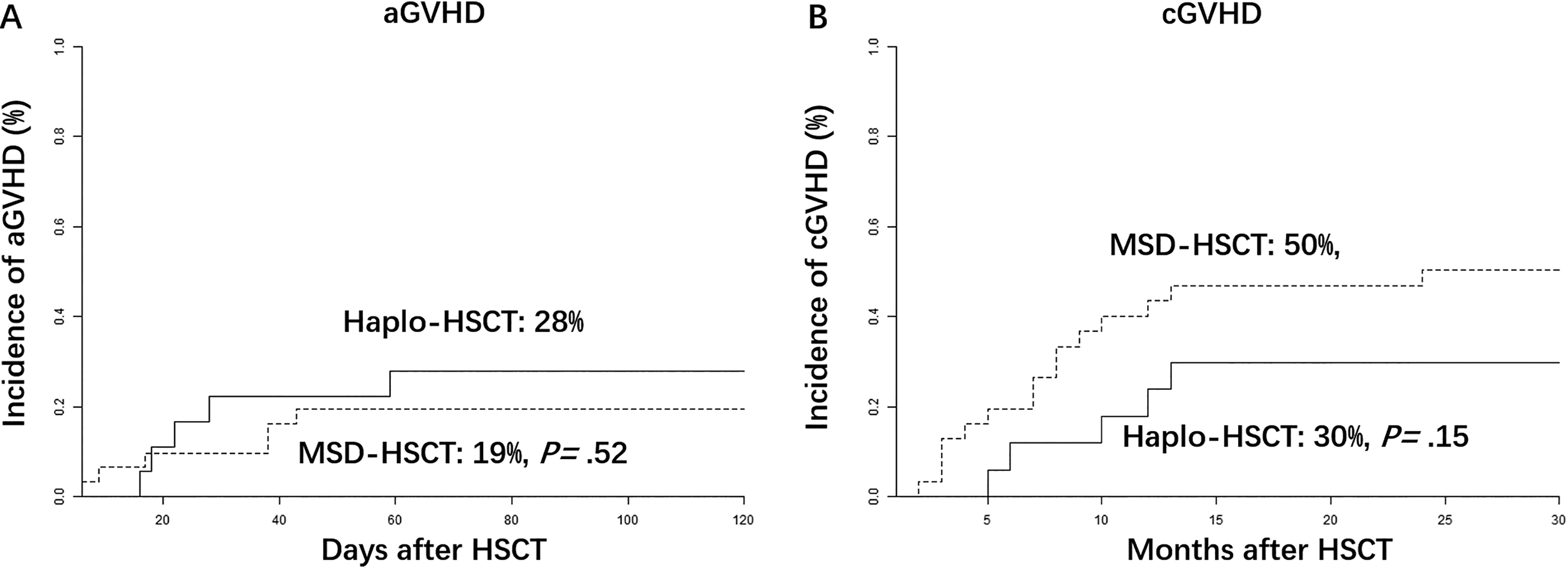
(A) Cumulative incidence of 100-day grade Ⅱ-Ⅳ acute graft versus host disease (aGVHD) after allo-HSCT. (B) Cumulative incidence of 2-year limited and extensive chronic GVHD (cGVHD) after allo-HSCT. The cumulative incidences of both aGVHD and cGVHD were estimated with competing-risk analysis and compared with Gray’s test. Haplo-HSCT, allo-HSCT with HLA-haploidentical donors; MSD-HSCT, allo-HSCT with HLA matched sibling donors.
NRM and Relapse
The 3-year cumulative incidence of NRM was 22% (95% CI, 6%–43%) in the haplo-HSCT group, compared with 21% (95% CI, 8%–37%) in the MSD-HSCT group (P = 0.78; Table 2; Fig. 2A). Further univariate and multivariate analysis were performed for NRM. But none of the variables tested were identified to significantly affect NRM rate in multivariate analysis (Table 2 and Supplemental Table 2).
Univariate Analysis of Clinical Outcomes of Patients with PTCLs After allo-HSCT.
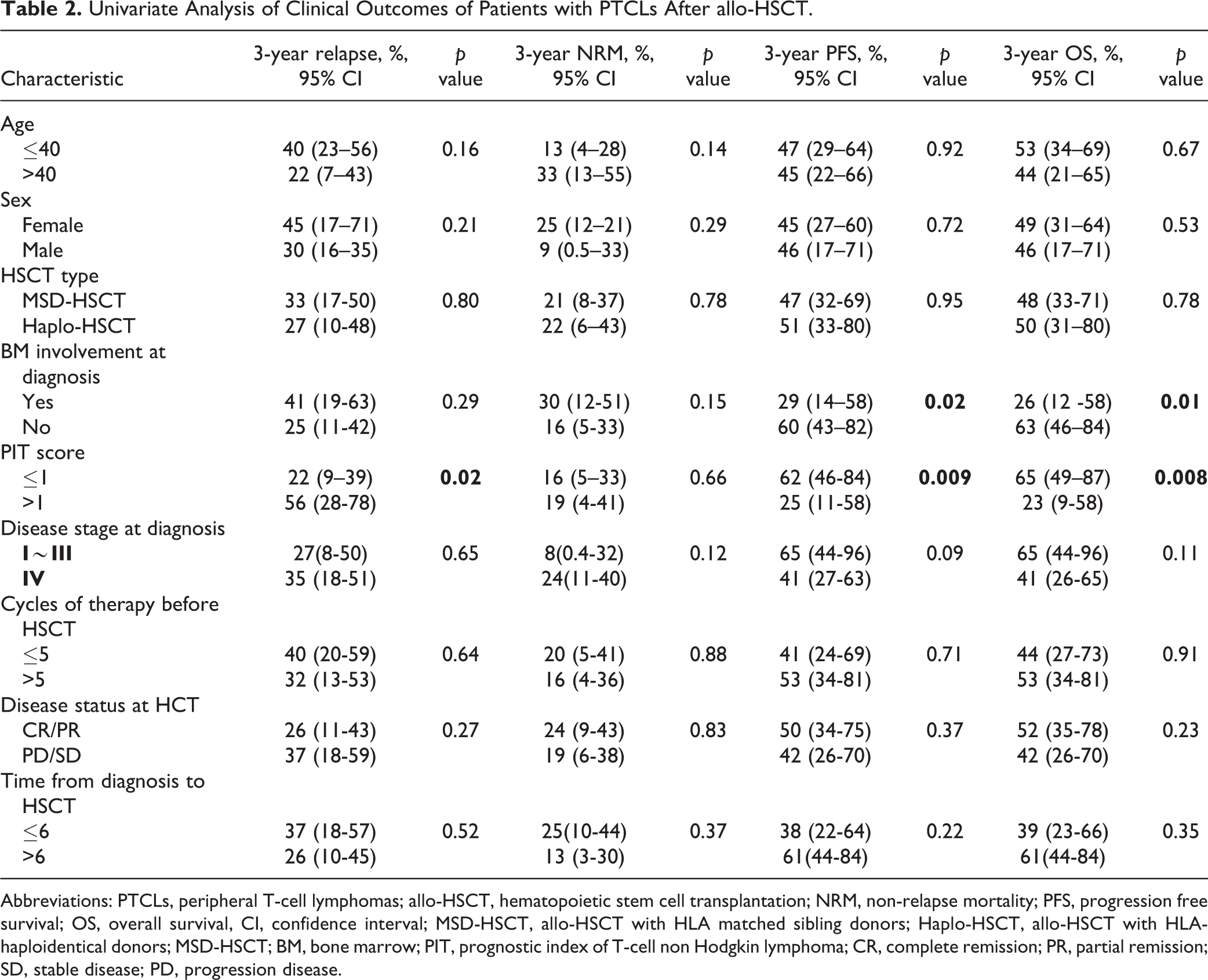
Abbreviations: PTCLs, peripheral T-cell lymphomas; allo-HSCT, hematopoietic stem cell transplantation; NRM, non-relapse mortality; PFS, progression free survival; OS, overall survival, CI, confidence interval; MSD-HSCT, allo-HSCT with HLA matched sibling donors; Haplo-HSCT, allo-HSCT with HLA-haploidentical donors; MSD-HSCT; BM, bone marrow; PIT, prognostic index of T-cell non Hodgkin lymphoma; CR, complete remission; PR, partial remission; SD, stable disease; PD, progression disease.
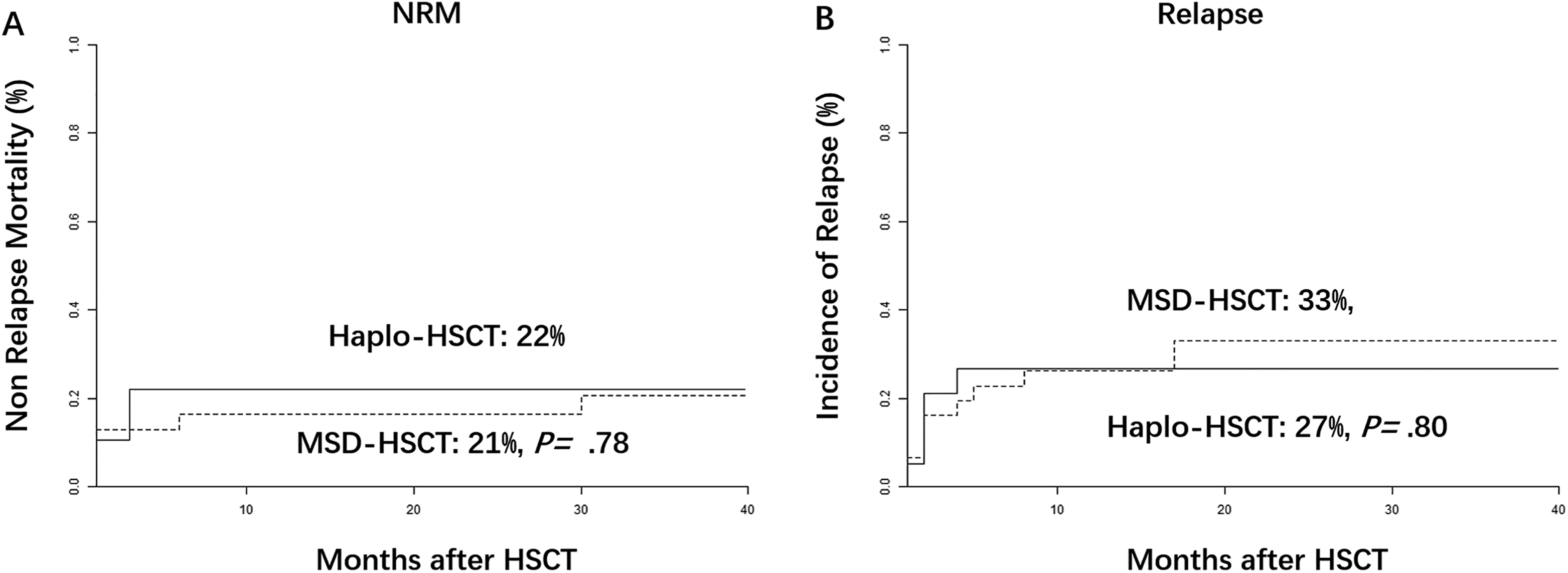
(A) The cumulative incidence of non-relapse mortality (NRM) at 3-year was 22% (95% CI, 6%–43%) in the haplo-HSCT group versus 21% (95% CI, 8%–37%) in the MSD-HSCT group. (B) The 3-year relapse rate was 27% (95% CI, 9%–48%) in the Haplo-HCT group versus 33% (95% CI, 17%–50%) in the MSD group. The cumulative incidences of both NRM and relapse were estimated with competing-risk analysis and compared with Gray’s test. Haplo-HSCT, allo-HSCT with HLA-haploidentical donors; MSD-HSCT, allo-HSCT with HLA matched sibling donors.
The cumulative incidence of relapse/progression at 3 years was 29% (95% CI, 27%–42%) for the entire population. Univariate analysis showed that there was no difference in 3-year relapse rate between these two groups (27% [95% CI, 9%–48%] for Haplo-HCT recipients versus 33% in the MSD group [95% CI, 17%–50%], p= .80, Fig. 2B). PIT score was the only independent variable predicting higher risk of relapse (HR, 6.8; 95% CI, 1.2%–41%; P = 0.03, Table 2 and Supplemental Table 2).
The causes of dead were listed in Supplemental Table 1. Disease progression were the common cause of death in both groups. Other causes in the MSD-HSCT group were hemorrhage (n = 3), multiorgan dysfunction (n = 2), GVHD (n = 1), and infection (n = 1). Five patients in the haplo-HSCT group died of transplantation related complications: multiorgan dysfunction (n = 3), GVHD (n = 1), infection (n = 1).
Survival
With a median follow-up of 38 (range: 2–112) months for all survivors, the 3-year OS for the entire group was 50% (95% CI, 37%–67%). There was no significant difference in 3-year OS between these two groups (49.9% [95% CI, 31%–80%] for Haplo-HSCT versus 48.2% [95% CI, 33%–71%] for MSD-HSCT, P = 0.78, Fig. 3A). Bone marrow involvement and higher PIT score were associated with inferior survival in univariate analysis (Table 1). On Cox multivariate analysis, PIT score (HR, 4.0; 95% CI, 1.6%–9.9%; P = 0.003, Supplemental Table 2) was an independent inferior predictor of inferior 3-year OS. Additionally, remission status (HR, 2.8; 95% CI, 1.1%–%6.9; P = 0.03, Supplemental Table 2) was another independent factor associated with survival.
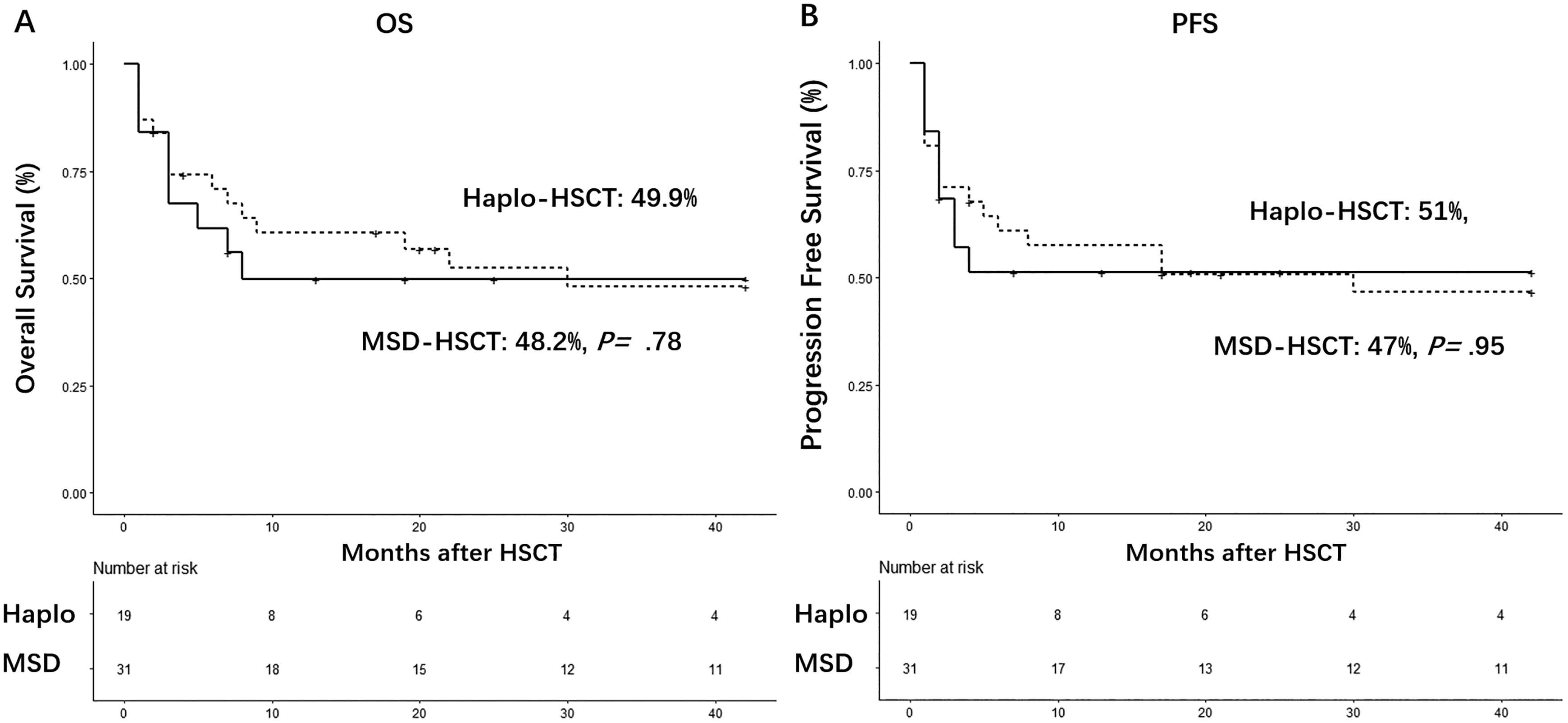
Kaplan-Meier survival analysis. (A) The 3-year overall survival (OS) was 49.9% (95% CI, 31%–80%) in the Haplo-HSCT group versus 48.2% (95% CI, 33%–71%) in the MSD-HSCT group. (B) The 3-year progression free survival (PFS) was 51% (95% CI, 33%–90%) in the Haplo-HSCT groups versus 47% (95% CI, 32%–69%) in the MSD group. Haplo-HSCT, allo-HSCT with HLA-haploidentical donors; MSD-HSCT, allo-HSCT with HLA matched sibling donors.
The 3-year PFS was not significantly different between these two groups (51% [95% CI, 33%–90%] in the Haplo-HSCT groups versus 47% [95% CI, 32%–69%] in the MSD-HSCT group, P = 0.95, Table 2 and Fig. 3B). And this was also confirmed in multivariate analysis (HR, 1.1; 95% CI, 0.40 to 3.3; P = 0.80; Supplemental Table 2). Higher PIT score remained to be the only independent variable associated with worse PFS (p < 0.0001, Supplemental Table 2). There was no statistical difference in the term of GRFS between the 2 groups (34% (95% CI, 14%–56%) in the haplo-HSCD group versus 20% (95% CI, 8%–36%) in the MSD-HSCT group, P = 0.99, Supplemental Fig. 1).
Discussion
The outcome of first-line chemotherapy is far from optimal until today for most PTCLs subtypes. Allo-HSCT remains a valuable treatment to achieve longer survival or cure this disease 9 . Because of the relative rarity of this disease, comparative study about Haplo-HSCT versus MSD-HSCT was rare. To the best of our knowledge, this is the first study that compared outcomes of patients with PTCLs undergoing Haplo-HSCT using ATG-based GVHD prophylaxis with the outcomes of patients received MSD-HSCT in the setting of myeloablative conditioning regimens. Our results suggested that ATG-based Haplo-HSCT can obtain similar outcomes comparable to MSD-HSCT in terms of engraftment, aGVHD, risk of relapse and NRM, and survival.
Although there were no significant differences in most variables in baseline characteristics, more patients seemed to be chemosensitive before transplantation in the haplo-HSCT group and they received haplo-HSCT as a consolidation. Disease status was not statistically different in univariate analysis probably because of the small number of patients included. Most PTCLs were associated with an aggressive nature, somehow like acute leukemia. If auo-HSCT failed, it was more likely that there was no time or chance for allo-HSCT. It was reasonable to receive allo-HSCT as consolidation for patients with high risk disease.
With a median follow-up of 38 months for survivors, the 3-year OS for the whole population was 50% in our study. It was similar to results of large retrospective studies from CIBMTR 18 , Asia 19 , and Europe 20 . In particular, quite a number of allo-HSCT in the above studies received non-myeloablative or reduced conditioning regimens (RIC). All allo-HSCT in this study were MAC. Until now, there were few studies focusing on patients of PTCLs undergoing allo-HSCT in the setting of MAC. The 3-year relapse rate in our study was 29%, similar to the reported 32% in the specific set of MAC subgroup from the CIBMTR study 18 . The 3-year NRM (22%) in this study was lower than that (32%) in the MAC subgroup of the CIBMTR study, somehow reflecting the improvement in allogeneic transplant techniques.
With the advantage that nearly everyone can have a donor from his or her family members, there is a significant growth in the number of Haplo-HSCT both in China and western countries. And also, preparation of an HLA haploidentical donor takes only 2-3 weeks. It’s more meaningful for patients with aggressive diseases like PTCLs. Currently, there are two haplo-HSCT platforms: haplo-HSCT with ATG-based GVHD prophylaxis and the haplo-HSCT platform using post-transplant cyclophosphamide regimen. The relative efficacy of haplo-HSCT versus MSD-HSCT had been compared in both of the two platforms 11,12,21 –26 , but were mainly restricted in leukemias and B-cell lymphomas. Our study provided evidence that haplo-HSCT using myeloablative ATG-based GVHD prophylaxis regimens can also achieve similar outcomes comparable to MSD-HSCT in the specific set of patients with PTCLs.
In our study, there was no significant difference in the incidence of II-IV aGVHD between the haplo-HSCT (28%) and MSD-HSCT group (19%). However, previous studies indicated that the incidence of aGVHD after haplo-HSCT seemed to be higher than that in the MSD-HSCT 22,27,28 . One possible reason is that the above reports are results about patients in leukemia or severe aplastic anemia. Our study was focusing on patients with PTCLs. Secondly, the sample size in our study was smaller than that in the above reports. Although there was no significant difference in the incidence of cGVHD, the cumulative incidence of limited and extensive cGVHD seemed to be a litter lower (30% vs 50%). Unlike in the PT-Cy based Haplo-HSCT platform, where cGVHD was lower than that after MSD-HSCT 29 , the relative rates of cGVHD after ATG-based haplo-HSCT versus MSD-HSCT were not confirmed until now 28 .
There was also no difference in NRM rate (22% vs 21%). The 3-year NRM in the haplo-HSCT group seemed to be a little higher than that with ATG-based Haplo-HSCT regimens in leukemia 30 . But it was acceptable. Because remission status before HSCT for PTCLs patients was worse than that in the above studies for patients with leukemia. In support of the similar efficacy of ATG-based Haplo-HSCT, no significant difference was noted in relapse rates (27% vs33%). Nevertheless, lymphoma relapse or progression was the major cause of death in both groups. Large prospective studies are needed to answer the question whether ATG-based Haplo-HSCT have a stronger GVL effect than MSD-HSCT in PTCLs, which had been implicated in acute leukemia 31 –33 .
The 3-year OS and PFS after haplo-HSCT was similar to that after MSD-HSCT in our study, which were also confirmed in multivariate analysis. And PIT score was identified as the main factor affecting the clinical outcome in relapse, PFS and OS. Together with previous studies 4,34,35 , these results provide evidence about the prognostic value of PIT score in PTCLs. Additionally, Remission status before transplantation was another independent viable affecting OS in our study. Our finding was in accordance with large registry study, supporting supports the importance of better disease control before HSCT 18,19 .
Similar to other retrospective studies, there are limitations in this study. Although we enrolled patients from 8 medical centers across China, the total number of patients was still small. Due to the lower statistical power, it is difficult to find minor differences. Secondly, the comparison in our study was subject to some selection bias because of preferences of each center or physicians due to the complex criteria for allo-HSCT. Finally, although the basalline characteristics were not significantly different between these two groups, there were still some heterogeneity in different subtypes of lymphoma, disease status before HSCT, and conditioning regimens. Our analysis should be interpreted with some caution and need to be confirmed in large prospective studies.
In summary, our analysis suggests that myeloablative ATG-based haplo-HSCT platform could achieve similar survival outcome with that after MSD-HSCT in the specific setting of PTCLs. When an HLA MSD donor was not available, HLA haploidentical donors using ATG based platform can work as an alternative for patients with PTCLs. For patients with aggressive diseases like PTCLs, it may get some benefit from rapid allo-HSCT with ATG-based halplo-HSCT platform. All of these needed to assessed in randomized prospective studies.
Supplemental Material
Supplemental Material, sj-pdf-1-cll-10.1177_0963689721999615 - Myeloablative Haploidentical Transplant as an Alternative to Matched Sibling Transplant for Peripheral T-Cell Lymphomas
Supplemental Material, sj-pdf-1-cll-10.1177_0963689721999615 for Myeloablative Haploidentical Transplant as an Alternative to Matched Sibling Transplant for Peripheral T-Cell Lymphomas by Gu Zhenyang, Li Nainong, Wu Xiaoxiong, Wang Maihong, Fu Xiaorui, Wang Zhao, Ren Hanyun, Li Yuhang, Li Xiaofan, Wu Yamei, Liu Yao, Zhang Mingzhi, Wang Yini, Liu Daihong, Dong Yujun, Hu Liangding and Huang Wenrong in Cell Transplantation
Footnotes
Abbreviations
Acknowledgments
We thank all the faculty members that participated in these studies.
Authors’ Contributions
Yujun Dong, Liangding Hu, Wenrong Huang designed the study and initiated this work; Data were obtained by Zhenyang Gu, Zhao wang, Xiaorui Fu; Nainong Li; Yao Liu; Xiaoxiong Wu; Yini Wang; Yuhang Li; Hanyun Ren; Mingzhi Zhang; Xiaofan Li; Maihong Wang; Yamei Wu; Daihong Liu. All statistical analyses were performed by Zhenyang Gu, Yujun Dong, Xiaorui Fu. Zhenyang Gu wrote the paper; all the authors were involved in the interpretation of the results; read, gave comments, and approved the final version of the manuscript; had full access to the data in the study; and take responsibility for the accuracy of the data analysis.
Gu Zhenyang, Li Nainong and Wu Xiaoxiong are authors contributed equally to this work.
Availability of data and materials
The dataset supporting the conclusions of this article are available in the clinical data (which were sorted from 8 above medical centers) repository of the First Medical Center of Chinese PLA General Hospital, Beijing 100853, China; Tel: +86-010-55499303. The datasets are available from the corresponding author on reasonable request.
Ethical Approval
Ethical approval to report this case series was obtained from the Medical Ethics Review Boards of Chinese PLA General Hospital, the Medical Ethics Review Boards of Fujian Institute of Hematology, the Medical Ethics Review Boards of the Forth Medical Center of PLA General Hospital, the Medical Ethics Review Boards of Xinqiao Hospital, Third Military Medical University, the Medical Ethics Review Boards of the First Affiliated Hospital of Zhengzhou University, the Medical Ethics Review Boards of Beijing Friendship Hospital, Capital Medical University, the Medical Ethics Review Boards of Peking University First Hospital, and the Medical Ethics Review Boards of the Fifth Medical Center of PLA General Hospital.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the by the Medical Ethics Review Boards at the above medical centers.
Statement of Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
