Abstract
This study compared the efficacy and safety of the modified BEAM (mitoxantrone hydrochloride liposome replacing melphalan) versus BEAM/BEAC as a conditioning regimen for autologous hematopoietic stem cell transplantation (auto-HSCT) in patients with non-Hodgkin lymphoma (NHL). The data source for the modified BEAM regimen was obtained from a prospective, single-arm study (NCT05681403), while the BEAM/BEAC regimen was retrospectively collected from an electronic health record–derived external control group. Primary endpoint was the cumulative incidence of relapse (CIR). Eighty-three patients were included, with 34 in the modified BEAM group, 23 in the BEAM group, and 26 in the BEAC group. The median times were 10.5, 9.0, and 9.0 days for neutrophil engraftment, and 10.5, 10.0, and 10.0 days for platelet engraftment, respectively. Oral mucositis (14.7%, 21.7%, and 7.7%), nausea (8.8%, 17.4%, and 0.0%), and diarrhea (5.9%, 8.2%, and 0.0%) were the most common grade 3–4 non-hematologic toxicities in three groups. No treatment-related death was reported. Besides, no significant differences were observed in 2-year CIR (9.4%, 26.6%, and 23.6%), progression-free survival (90.6%, 73.4%, and 76.4%), and overall survival (97.1%, 87.0%, and 88.5%) rates among three groups (all P > 0.05). The modified BEAM conditioning regimen may have comparable efficacy and safety with the BEAM/BEAC regimen in treating patients with NHL.

Keywords
Introduction
Non-Hodgkin lymphoma (NHL) is the most common hematologic malignancy, with an estimated 553,010 new cases and 250,475 new deaths worldwide in 2022 1 . The majority of NHLs arise from the clonal expansion of B cells, with a minority originating from T cells or natural killer (NK) cells 2 . Diffuse large B-cell lymphoma (DLBCL) is the most common histologic subtype of NHL representing almost 30% of all cases of NHL 3 . Despite high initial response rates to frontline treatment, many patients with NHL (approximately 40% in DLBCL) may experience relapse or have refractory disease 4 . High-dose chemotherapy followed by autologous hematopoietic stem cell transplantation (auto-HSCT) is one of the recommended treatment options for patients with relapsed/refractory NHL5,6 and consolidation for some subtype of NHL. The conditioning regimen constitutes a pivotal component of the auto-HSCT. Currently, BEAM (carmustine, cytarabine, etoposide, and melphalan) and BEAC (carmustine, cytarabine, etoposide, and cyclophosphamide) are the most widely used conditioning regimens for NHL 7 . However, the application of BEAM and BEAC regimens is limited by dose-limiting toxicities, including myelosuppression, mucositis, gastrointestinal complications with melphalan, and cardiotoxicity with cyclophosphamide8,9. Therefore, it is necessary to explore effective conditioning regimens with low toxicity.
Mitoxantrone hydrochloride liposome (Lipo-MIT) is a pegylated liposomal mitoxantrone formulation with properties of the long circulation time after intravenous infusion and the stable encapsulation of mitoxantrone 10 . The remarkable altered pharmacokinetics of Lipo-MIT compared with mitoxantrone contributed to the improved safety and potential efficacy as shown in a phase I clinical trial 10 . The promising efficacy and acceptable safety of Lipo-MIT have been demonstrated in multiple subtypes of NHL, including peripheral T-cell lymphoma (PTCL), NK/T-cell lymphoma (NKTCL), DLBCL, angioimmunoblastic T-cell lymphoma (AITL), and sézary syndrome11–14(SS). Moreover, it has been approved by the National Medical Products Administration for relapsed/refractory peripheral T-cell lymphoma based on a phase II trial 15 . Importantly, mitoxantrone-based conditioning regimen has proven effective in patients with lymphoma, whereas the role of Lipo-MIT as a conditioning regimen remains unclear16–19. In light of these initial promising findings, we hypothesized that Lipo-MIT-containing regimen may serve as a potential conditioning option for auto-HSCT in patients with NHL. Thus, a modified BEAM regimen with Lipo-MIT replacing melphalan was designed in a prospective, single-arm study. This study indirectly compared the efficacy and safety of the modified BEAM regimen with the BEAM/BEAC regimen from an electronic health record–derived external control group in patients with NHL.
Subjects and methods
Study design and patients
An indirect comparison was conducted in this study to assess the efficacy and safety of the modified BEAM versus BEAM/BEAC as a conditioning regimen for auto-HSCT after first-line treatment in patients with NHL. The data source for the modified BEAM regimen (modified BEAM group) was obtained from a prospective, single-arm, open-label, single-center study, while the BEAM/BEAC regimen was retrospectively collected from an electronic health record–derived external control group (BEAM/BEAC group). The prospective study (NCT05681403) was conducted from March 2022 to December 2023. Between January 2018 and February 2022, patients with NHL who received BEAM/BEAC as a conditioning regimen for auto-HSCT at the same center and with complete clinical and follow-up data were included in the external control group. The study was approved by the ethics committee of The First Affiliated Hospital of Xi’an Jiaotong University (number: XJTU1AF2022LSK-238) and was conducted per the Declaration of Helsinki and Good Clinical Practice Guidelines. Written informed consent was obtained from each patient.
Patients with histologically confirmed diagnosis of NHL and planned to undergo auto-HSCT were eligible for the prospective study. Additional eligibility criteria included 18 to 60 years of age, an Eastern Cooperative Oncology Group (ECOG) performance status of 0-2, and adequate bone marrow and organ function. Key exclusion criteria were any uncontrolled systemic disease or active infection, other malignant tumors, hypersensitivity to any study drug or its components, prior treatment with mitoxantrone or Lipo-MIT, or prior treatment with doxorubicin at a total cumulative dose exceeding 360 mg/m2, or other anthracyclines at equivalent doses.
Conditioning regimens
The modified BEAM regimen consisted of intravenous Lipo-MIT 24 mg/m2 on day –7, carmustine 300 mg/m2 on day –6, and cytarabine 200 mg/m2 q12h and etoposide 200 mg/m2 on days –5 to –2. The BEAM regimen consisted of intravenous carmustine 300 mg/m2 on day –7, cytarabine 200 mg/m2 q12h and etoposide 200 mg/m2 on days –6 to –3, and melphalan 140 mg/m2 on day –2. The BEAC regimen included carmustine (300 mg/m2 on day –6), cytarabine (200 mg/m2 q12h on days –5 to –2), etoposide (200 mg/m2 on days –5 to –2), and cyclophosphamide (35 mg/kg on days –5 to –2). Hematopoietic stem cells were reinfused on day 0.
Outcomes and assessments
The primary endpoint was the cumulative incidence of relapse (CIR), which was defined as the time from transplantation to relapse accounting for non-relapse mortality as a competing event. Secondary endpoints included progression-free survival (PFS), overall survival (OS), times to neutrophil engraftment and platelet engraftment, and non-hematological toxicity. The PFS was defined as the time from auto-HSCT to disease progression, relapse, death, or last follow-up, whichever occurred first. The OS was calculated as the time interval from auto-HSCT until death or last follow-up. The times to neutrophil and platelet engraftment were defined as the first of three consecutive days with neutrophil count ≥ 0.5 × 109/L and platelet count ≥ 20 × 109/L, respectively. Treatment response was evaluated using computed tomography (CT) or positron emission tomography (PET)-CT per the Lugano 2014 classification for NHL 20 . The incidence and severity of non-hematological toxicity were assessed according to the Common Terminology Criteria for Adverse Events version 5.0 (CTCAE v5.0).
Statistical analysis
The sample size calculation of the prospective study was based on the recurrence rate of the study endpoint, with a recurrence rate of 46% at 3 years for NHL patients underwent autologous stem cell transplant (ASCT) reported in the previous literature 21 , an estimated recurrence rate of 33% for the modified BEAM regimen, and a sample size of 31 cases calculated by PASS software(accuracy was 80% and the α was 0.05). Considering that the shedding rate is 10%, a sample size of 34 cases is required. Categorical variables were presented as numbers with percentages, and continuous variables were expressed as median with range (skewed distributed data) or mean with standard deviation (normally distributed data). The Shapiro-Wilk test was used for testing normality. Between-group comparisons of continuous variables were performed utilizing either the t test for normally distributed data or the Mann–Whitney U test for skewed distributed data. Meanwhile, assessments of categorical variable differences across groups were carried out using the chi-square test or Fisher’s exact test. The CIR, PFS, and OS were estimated using the Kaplan–Meier method and compared using the stratified log-rank test. All statistical analyses were conducted using IBM SPSS software (version 25.0 or later, IBM SPSS Institute, Chicago, IL, USA) and GraphPad Prism 8 software (GraphPad Software, San Diego, CA, USA), with statistical significance set at P < 0.05. Independent predictors were determined using univariate and multivariate Cox regression analyses. A post hoc power analysis of actual CIR data was performed with the use of PASS software (version 15.0).
Results
Patient characteristics
The study included data from 34 patients belonging to the prospective cohort (modified BEAM group) and an additional 49 patients drawn from the external control group (BEAM/BEAC group). Table 1 presents the baseline and disease characteristics of patients in three groups. All baseline and disease characteristics were well-balanced among three groups. The median age was 46.0 (range = 18.0–59.0) years in the modified BEAM group, 48.0 (range = 19.0-60.0) years in the BEAM group, and 42.0 (range = 18.0–58.0) years in the BEAC group (P = 0.662). Males constituted 55.9% of the modified BEAM group, 34.8% of the BEAM group, and 61.5% of the BEAC group (P = 0.144). The majority of patients in three groups had an ECOG performance status of 0–1 (100.0%, 91.3%, and 92.3%, P = 0.173), stage III–IV disease (82.3%, 73.9%, and 92.3%, P = 0.572), had achieved complete remission prior to auto-HSCT (94.1%, 91.3%, and 80.8%, P = 0.266), had extranodal involvement (82.4%, 73.9%, and 84.6%, P = 0.605), and had increased lactate dehydrogenase (LDH) at diagnosis (58.8%, 52.2%, and 53.8%, P = 0.868). Histological subtype distribution was similarly distributed among three groups (P = 0.595), with DLBCL being the most prevalent (44.1%, 60.9%, and 50.0%), followed by NKTCL (11.8%, 13.0%, and 23.1%) and anaplastic large cell lymphoma (ALCL; 8.8%, 8.7%, and 7.7%). There were no significant differences in prior lines of chemotherapy, International Prognostic Index (IPI) score, radiotherapy prior to auto-HSCT, and time from diagnosis to auto-HSCT (all P > 0.05).
The baseline and disease characteristics of patients among three groups.

Other IPI included Follicular Lymphoma International Prognostic Index (FLIPI), Mantle Cell Lymphoma International Prognostic Index (MIPI), and prognostic index of natural killer lymphoma (PINK). MYC/BCL2/6 double hit referred to lymphomas with translocations in both MYC and BCL2/BCL6 genes. Modified BEAM, mitoxantrone hydrochloride liposome, carmustine, cytarabine, and etoposide; BEAM, carmustine, cytarabine, etoposide, and melphalan; BEAC, carmustine, cytarabine, etoposide, and cyclophosphamide; IPI, International Prognostic Index; ECOG, Eastern Cooperative Oncology Group; auto-HSCT, autologous hematopoietic stem cell transplantation; LDH, lactate dehydrogenase; CR, complete remission; PR, partial remission; DLBCL, diffuse large B-cell lymphoma; MCL, mantle cell lymphoma; FL, follicular lymphoma; HGBL, high-grade B-cell lymphoma; MZL, marginal zone lymphoma; PMBCL, primary mediastinal large B-cell lymphoma; NKTCL, natural killer/T-cell lymphoma; ALCL, anaplastic large cell lymphoma; AITL, angioimmunoblastic T-cell lymphoma; PTCL, peripheral T-cell lymphoma; MEITL, monomorphic epitheliotropic intestinal T-cell lymphoma.
Hematopoietic engraftment
The median dose of infused CD34-positive cells was 6.2 × 106/kg (range = 2.1–23.4) in modified BEAM group, 5.0 × 106/kg (range = 0.4–30.4) in BEAM group, and 5.5 × 106/kg (range = 1.5–19.2) in BEAC group (P = 0.293), with median dose of infused mononuclear cells of 6.2 × 108/kg (range = 3.2–11.6), 6.0 × 108/kg (range = 4.2–15.3), and 6.7 × 108/kg (range = 3.5–16.3), respectively (P = 0.553). Among three groups, the median times were 10.5 (range = 7.0–15.0) days, 9.0 (range = 7.0–13.0) days, and 9.0 (range = 7.0–13.0) days for neutrophil engraftment (P < 0.001), and 10.5 (range = 7.0-14.0) days, 10.0 (range, 7.0-21.0) days, and 10.0 (8.0-21.0) days for platelet engraftment (P = 0.235), respectively.
The changes in lymphocyte count after transplantation are displayed in Fig. 1. At the first month post-transplantation, the lymphocyte count was significantly lower with modified BEAM than that with BEAM/BEAC (0.4 ± 0.4, 1.3 ± 0.9 vs. 1.2 ± 0.7, P < 0.001), but were similar by months 3 (1.5 ± 0.9 vs. 2.4 ± 2.2, P = 0.379) and 6 (1.3 ± 0.8 vs. 1.2 ± 0.3, P > 0.999).

The changes in lymphocyte count after transplantation. Modified BEAM, mitoxantrone hydrochloride liposome, carmustine, cytarabine, and etoposide; BEAM, carmustine, cytarabine, etoposide, and melphalan; BEAC, carmustine, cytarabine, etoposide, and cyclophosphamide; *P < 0.05.
Non-hematological toxicity
Non-hematological toxicities of any grade occurred in all patients in the modified BEAM and BEAM/BEAC groups (Table 2). The most common non-hematologic toxicities of any grade observed with the modified BEAM and BEAM/BEAC regimens were nausea (88.2%, 95.7%, and 84.6%, P = 0.501), febrile agranulocytosis (79.4%, 87%, and 61.5%, P = 0.095), oral mucositis (70.6%, 73.9%, and 34.6%, P = 0.005), upper respiratory infection (52.9%, 52.2%, and 69.2%, P = 0.364), diarrhea (35.3%, 43.5%, and34.6%, P = 0.774), and vomiting (32.4%, 39.1%, and 57.7%, P = 0.135). Grade 3–4 non-hematologic toxicities were similar between the modified BEAM and BEAM/BEAC groups, including oral mucositis (14.7%, 21.7%, and 7.7%, P = 0.388), nausea (8.8%, 17.4%, and 0%, P = 0.086), and diarrhea (5.9%, 17.4%, and 0%, P = 0.063). Although four (11.8%) patients in the modified BEAM group experienced cutaneous blue pigmentation, it resolved spontaneously within 1 to 3 months. No treatment-related death was reported. Cardiac function was evaluated at baseline and at weeks 1, 2, and 3 after stem cell infusion in modified BEAM group, with no cardiac adverse events observed.
Non-hematological toxicity with modified BEAM or BEAM/BEAC regimen.

Efficacy
At data cutoff, the median follow-up after stem cell infusion was 24.0 (range = 3.0–38.0) months in modified BEAM group and 57.0 (range = 4.0–85.0) months in BEAM/BEAC group (P < 0.001), with three and 15 events of disease progression or death, respectively. There was no significant difference between the 2-year CIR (9.4%, 26.6% and 23.6%, P = 0.155) (Fig 2a), PFS (90.6%, 73.4%, and 76.4%, P = 0.155) (Fig. 2b), and OS (97.1%, 87.0%, and 88.5%, P = 0.239) (Fig. 2c) among three groups.

Kaplan–Meier curves of three groups. (a) Kaplan–Meier curves of CIR. (b) Kaplan–Meier curves of PFS. (c) Kaplan–Meier curves of OS. CIR, cumulative incidence of relapse; PFS, progression-free survival; OS, overall survival; modified BEAM, mitoxantrone hydrochloride liposome, carmustine, cytarabine, and etoposide; BEAM, carmustine, cytarabine, etoposide, and melphalan; BEAC, carmustine, cytarabine, etoposide, and cyclophosphamide.
We screened the prognostic factors of the CIR by univariate and multivariate COX regression analysis, including age, gender, IPI score, ECOG performance status, number of prior lines of chemotherapy, extranodal involvement, LDH levels at diagnosis, double expression, double hit, disease status prior to auto-HSCT, histological subtype, and disease stage. The results showed these variables in the study had no independent and significant effect on cumulative incidence of relapse (CIR).
When outcomes were analyzed by histological subtypes, the 2-year CIRs for B-cell lymphoma were 5.6%, 25.0%, and 24.4% in modified BEAM, BEAM, and BEAC groups (P = 0.118), and 15.4%, 16.7%, and 22.2% for T-cell lymphoma (P = 0.923), respectively (Fig. 3a). For patients with B-cell lymphoma (Fig. 3b, c), the 2-year PFS rate was 94.4% with modified BEAM, 74.5% with BEAM and 75.6% with BEAC (P = 0.118), with 2-year OS rates of 100.0%, 93.8%, and 88.2%, respectively (P = 0.190). In the T-cell lymphoma subgroup (Fig. 3b, c), the 2-year PFS was 84.6% in modified BEAM group, 83.3% in BEAM group, and 77.8% in BEAC group (P = 0.923), and the 2-year OS rates were 92.3%, 83.3% and 88.9%, respectively (P = 0.860).
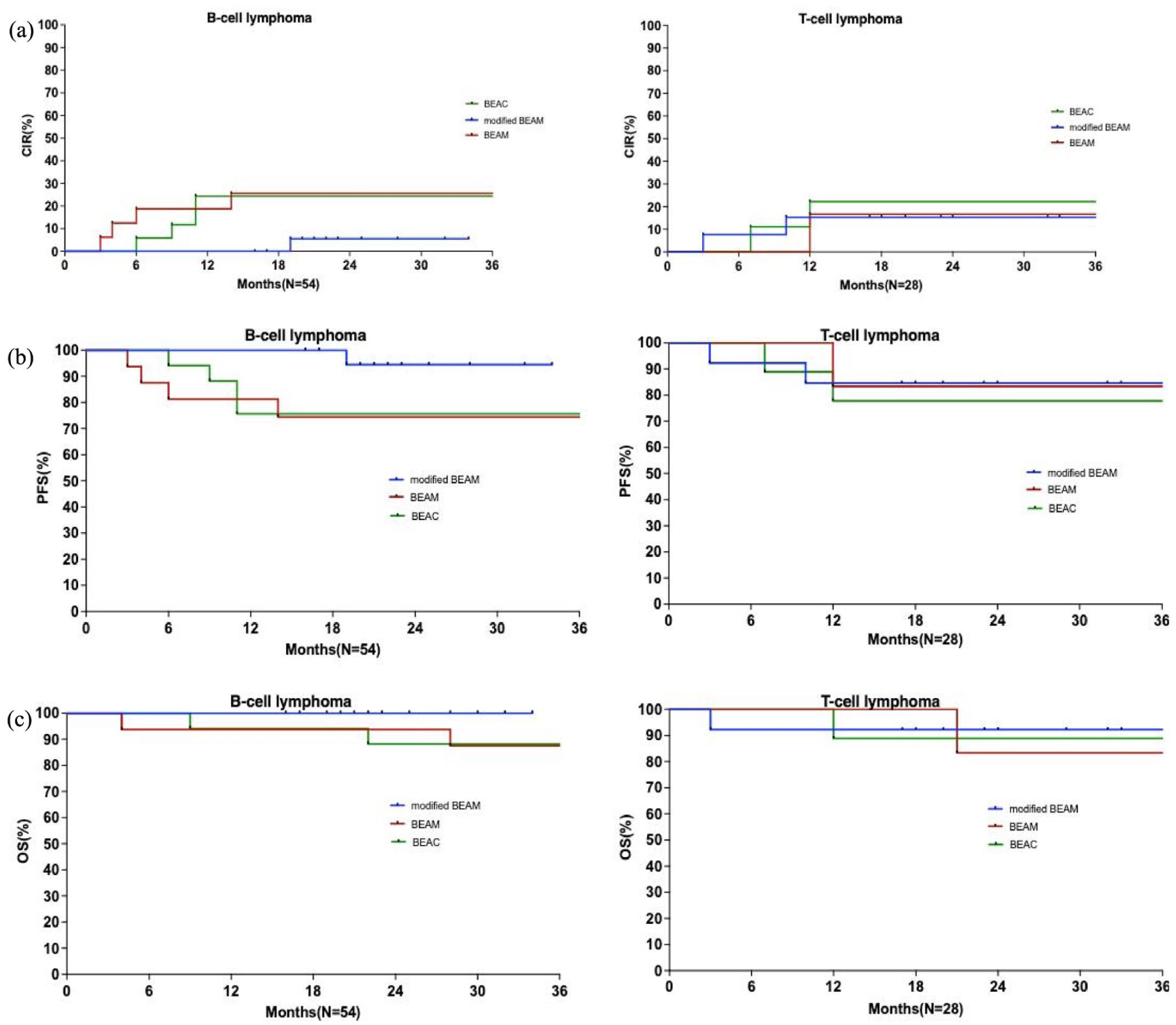
Kaplan–Meier curves according to histological subtypes. (a) Kaplan–Meier curves of CIR for B-cell lymphoma and T-cell lymphoma. (b) Kaplan–Meier curves of PFS for B-cell lymphoma and T-cell lymphoma. (c) Kaplan–Meier curves of OS for B-cell lymphoma and T-cell lymphoma. CIR, cumulative incidence of relapse; PFS, progression-free survival; OS, overall survival; modified BEAM, mitoxantrone hydrochloride liposome, carmustine, cytarabine, and etoposide; BEAM, carmustine, cytarabine, etoposide, and melphalan; BEAC, carmustine, cytarabine, etoposide, and cyclophosphamide.
Table 3-4 presents a comparative analysis of the 2-year CIRs among three groups across various subgroups categorized by histological subtype, disease stage, IPI score, and disease status prior to auto-HSCT. No statistically significant differences were observed among these subgroups (all P > 0.05).
Subgroup analyses for the 2-year CIR between modified BEAM and BEAM group.

Modified BEAM, mitoxantrone hydrochloride liposome, carmustine, cytarabine, and etoposide; BEAM, carmustine, cytarabine, etoposide, and melphalan; CIR, cumulative incidence of relapse; IPI, International Prognostic Index; auto-HSCT, autologous hematopoietic stem cell transplantation; CR, complete remission; PR, partial remission; CI, confidence interval; HR, hazard ratio.
Subgroup analyses for the 2-year CIR between modifed BEAM and BEAC group.

Modified BEAM, mitoxantrone hydrochloride liposome, carmustine, cytarabine, and etoposide; BEAC, carmustine, cytarabine, etoposide, and cyclophosphamide; CIR, cumulative incidence of relapse; IPI, International Prognostic Index; auto-HSCT, autologous hematopoietic stem cell transplantation; CR, complete remission; PR, partial remission; CI, confidence interval; HR, hazard ratio.
Discussion
This study provided an indirect comparison of the efficacy and safety of the modified BEAM conditioning regimen from a prospective study with the BEAM/BEAC regimen from an electronic health record–derived external control group. Our results indicated that patients with NHL receiving the modified BEAM conditioning regimen had a similar non-hematological toxicity profile compared with patients receiving the BEAM/BEAC regimen. Engraftment outcomes were comparable in both groups except for the delayed neutrophil engraftment with the modified BEAM regimen compared with the BEAM/BEAC regimen. It was reported that a day 100 absolute lymphocyte count/monocyte count ratio > 2.1 correlated with lower risk of relapse in patients with relapsed/refractory Hodgkin lymphoma underwent ASCT 22 , and an early recovery of absolute lymphocyte count after ASCT can be regarded as a good prognostic marker in patients with DLBCL who have undergone frontline ASCT 23 . This study showed lower lymphocyte counts at 1 month and recovered similar to other groups at 3 months in the modified BEAM group. It indicated that the immunosuppressive effect of modified BEAM is stronger, and lymphocytes recover more slowly, but it is close to the other group at 3 months post-transplantation, which does not pose a negative impact on the prognosis. In addition, there were no significant differences between the two groups in terms of 2-year CIR, 2-year PFS, and 2-year OS rates, irrespective of the histological subtypes. These findings suggest that modified BEAM represents a valuable alternative option relative to old conditioning regimens, especially when some contra-indications exist such as heart toxicity for cyclophosphamide.
Current National Comprehensive Cancer Network (NCCN) guidelines reserve BEAM/BEAC for transplant-eligible patients with NHL. Previous studies on mitoxantrone liposomes have found that it has good safety and efficacy in the treatment of lymphoma. In this study, mitoxantrone liposome was applied to lymphoma transplantation conditioning for the first time to explore its safety and efficacy24–29. Overall, the modified BEAM conditioning regimen was well tolerated in our patients with NHL, with the majority of adverse events regarded as grade I–II. The incidence and severity of non-hematologic toxicities observed with the modified BEAM regimen were comparable with those described with the BEAM/BEAC regimen in our study. The most frequently observed grade 3–4 non-hematologic toxicities with the modified BEAM regimen were oral mucositis, nausea, and diarrhea, which were also consistent with those previously described with BEAM/BEAC regimen30–32. Compared with the reported incidence of cardiac toxicity ≥grade 3 (6%) 33 and death due to cardiac toxicity (0.26%) in the BEAC regimen and (0.39%) in the BEAM regimen 34 , no cardiac adverse events occurred in the modified BEAM regimen in this study. The results suggest that the modified BEAM regimen has potential cardiac safety benefits, particularly for elderly patients and those with pre-existing cardiac dysfunction. Although neutrophil engraftment was delayed in the modified BEAM group compared with the BEAM/BEAC group, neutrophil (10.5 days vs. 10–12 days) and platelet (10.5 days vs. 11–16 days) recovery in our patients receiving the modified BEAM regimen were in line with previous studies with BEAM/BEAC regimen30,31,35,36. Clinical and preclinical pharmacokinetic studies have shown that Lipo-MIT has a long metabolic time property, which may lead to prolonged bone marrow retention. This may transiently suppress stem cell niches, explaining the engraftment delay despite comparable CD34+ doses10,37–39. Importantly, no treatment-related death was reported in our study. These findings collectively suggest that the modified BEAM regimen with Lipo-MIT replacing melphalan is safe in treating patients with NHL before auto-HSCT.
Over the past few decades, a high recurrence rate has remained a pivotal concern associated with both the BEAM and BEAC regimens in treating patients with NHL30,31,35,40. A retrospective analysis by the EBMT-Lymphoma Working Party revealed that 33% of patients in the BEAM group and 34% of patients in the BEAC group experienced a relapse within 2 year 32 . Similarly, the 2-year CIR was 26.6% and 23.6% for our patients receiving the BEAM and BEAC regimen, respectively. Notably, our study indicated a trend toward better disease control (2-year CIR, 9.4% vs. 26.6% vs. 23.6%) and survival (2-year PFS, 90.6% vs. 73.4% vs.76.4%; 2-year OS, 97.1% vs. 87.0% vs. 88.5%) with modified BEAM regimen, although without statistical significance, potentially due to the short follow-up period and limited sample size. Thus, further updated analyses with extended follow-up periods are necessary to evaluate long-term recurrence and survival outcomes.
There were inherent limitations to this study. First, due to the limited sample size, a propensity score–matched comparison that would reduce the risk of selection bias and uncontrolled confounding was not conducted and the study was not powered to detect subtle differences in these subgroups; only a naive comparison was conducted in our study. However, study populations were well balanced in all baseline and disease characteristics, including age, gender, histological subtype, disease stage, IPI score, ECOG performance status, disease status before auto-HSCT, prior treatment, time from diagnosis to auto-HSCT, extranodal involvement, LDH levels at diagnosis and transplant, double expression, and double hit. Moreover, the efficacy assessment methodologies conducted at the same center were consistent. However, we agree that results may not fully generalize to other settings. Consequently, despite the inherent possibility of bias, it still generates valuable signals and hypotheses for further research. Second, the short follow-up period in our study precluded the comprehensive understanding of the long-term recurrence and survival data. Thus, multicenter, randomized controlled trials with larger sample sizes and long-term follow-up are necessary to further compare the efficacy and safety of the modified BEAM and BEAM/BEAC as conditioning regimens for auto-HSCT in patients with NHL.
Conclusion
In conclusion, our comparative analysis showed that both the modified BEAM and BEAM/BEAC conditioning regimens may have comparable efficacy and safety in treating patients with NHL prior to auto-HSCT. The modified BEAM conditioning regimen with Lipo-MIT replacing melphalan may provide a feasible alternative to the traditional BEAM/BEAC regimen for this disease population. Further prospective studies with larger sample sizes and longer follow-up are needed to validate these findings.
Footnotes
Acknowledgements
Not applicable.
Ethical Considerations and Consent to Participate
The study was approved by the ethics committee of The First Affiliated Hospital of Xi’an Jiaotong University (number: XJTU1AF2022LSK-238) and was conducted per the Declaration of Helsinki and Good Clinical Practice Guidelines. Written informed consent was obtained from each patient.
Author Contributions
Conception and design, P.H. and X.W.; Provision of study materials or patients, J.R. and H.Z.; Collection and assembly of data, Y.W. and R.L.; Data analysis and interpretation, Y.W. and X.W.; Drafting article, X.W.; Administrative support, P.H. All the authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Medical Basic Clinical Integration Innovation Project of Xi’an Jiaotong University (YXJLRH2022007) and the Key Research and National Key R&D Program of China (2022YFC2502700), and was supported by the Clinical Research Project Fund of the First Affiliated Hospital of Xi’an Jiaotong University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
