Abstract
Patients with hair-loss experience stress and a loss of confidence. Several factors contribute to hair loss, including aging, androgen hormone effects, genetics, and diseases. Normally, physicians treat hair loss using medications such as minoxidil and finasteride. However, these treatments are associated with side effects and some limitations. Currently, hair transplantation is the most efficient technique; however, it requires the use of numerous hair grafts. Stem cell and hair follicle cell (HFC) treatments have been developed for hair-loss treatment. Previously, stem cell and HFC therapies have been shown to promote hair development, prolong the anagen phase, and increase hair diameter and density. In addition, growth factors and extracellular vesicles (EVs) are important for prolonging the anagen phase, improving hair follicle health, and reducing dihydrotestosterone effects. Furthermore, gene therapy and tissue engineering have also been developed as treatments for hair loss. In this review, we discuss (i) the basic knowledge and molecular signaling relevant to hair regeneration and (ii) advanced technology medical treatments for hair loss (stem cells, HFCs, growth factor, EVs, gene therapy, and tissue engineers).

Introduction
Hair loss, caused by several factors including hormones, genetic and physical factors, medications, and some diseases, affects individuals by reducing confidence and increasing stress 1 . To find a solution, researchers have developed treatments to improve hair growth, including drug treatment2,3, hair transplantation 1 , stem cell treatment 4 , hair follicle cell (HFC) treatment 4 , conditioned medium5,6, gene therapy 7 , and tissue engineering for producing hair follicles in vitro 8 (Fig. 1). Minoxidil and finasteride are Food and Drugs Administration (FDA)-approved hair-loss medications2,3. Minoxidil increases dermal papilla cells (DPCs) proliferation by inducing molecular signaling cascade 9 , vascular smooth muscles relaxation, and opening potassium channels, which affects hair growth and prolongs the anagen phase10,11. Topical minoxidil has been applied at 2% twice daily or 5% once daily. However, the recommended concentration of topical minoxidil for women is 2%. Orally, it is used at 0.25–5 mg/mL for hair-loss treatment11–13. While minoxidil increases blood flow, finasteride is used to treat hair loss in case of hormone effect 14 . Testosterone binds with 5α-reductase to produce dihydrotestosterone (DHT), which harms hair follicles 15 . Finasteride inhibits type II 5α-reductase and increases insulin-like growth factor-I (IGF-I) expression in DPCs10,16 and is administered at 1 mg daily for oral treatment 14 . Topical treatment included doses of 0.005% finasteride twice daily or 0.25% combined with 3% minoxidil twice daily 16 . However, drug treatment has some limitations2,14, and patients are reluctant to take drugs for extended periods 17 .

Hair-loss treatments include (i) medication (minoxidil and finasteride), (ii) hair transplantation (follicular unit transplantation [FUT] and follicular unit excision [FUE]), (iii) stem cell treatment, (iv) hair follicle cells (HFCs) treatments (dermal papilla cells [DPCs], dermal sheath cells [DSCs], and hair follicle stem cells [HFSCs]), (v) condition media (growth factors and extracellular vesicles), (vi) gene therapy, and (vii) tissue engineering. Schematic figures were created using BioRender.com, procreate.com, canva.com, and PowerPoint.
Hair transplantation is a well-known alternative method for hair-loss repair. This technique involves harvesting donor hair grafts from areas with high hair density and transplanting them to bald areas 12 . Autologous hair transplantation is divided into two techniques: follicular unit transplantation (FUT) and follicular unit excision (FUE)1,18. FUT involves harvesting hair grafts in strips, the size of which depends on the number of hair follicles required for transplantation to the recipient area. However, there are linear scars that persist in patients after transplantation. Thus, FUT is unsuitable for short hairstyles1,19. FUE is used to increase the number of hair grafts and reduce the number of scars after transplantation. The harvest donor area for FUE is wider than that for FUT, and it typically results in no visible scarring or only a pinpoint scar 0.75–1.2 mm from the biopsy punch1,18,19. Patients with severe hair loss or those who request hair transplantation more than once often face limitations in the number of available donor hair grafts. Therefore, stem cell and HFC transplantation have been used as alternatives to treat hair loss. Autologous adipose-derived stromal vascular cells (ADSVFs)20,21 and allogeneic mesenchymal stem cells (MSCs) from the umbilical cord22,23 have been reported for human hair-loss treatment. They increase hair diameter, prolong the anagen phase, and induce hair regeneration20–23. HFCs such as DPCs, dermal sheath cells (DSCs), and hair follicle stem cells (HFSCs) are of interest for baldness cell therapy24,25. DSCs have been shown to improve hair density following treatment 26 . Furthermore, growth factors and extracellular vesicles (EVs) are also important for inducing hair regrowth and DHT inhibition5,6. Previous results in animal models show that EVs improve hair growth through injections in the intradermal, subcutaneous, or microneedle patch methods5,6,27. Moreover, gene therapy has also been reported in animal models to solve hair-loss problems 7 . In addition, the idea of producing hair follicles using tissue engineering has also been developing 8 . Basic knowledge and understanding of the signaling molecules that induce hair follicle regeneration are required for successful hair-loss therapy. Therefore, this review aims to present an overview of the current knowledge on hair follicles and signaling molecules that induce hair follicle regeneration. In addition, we highlight the techniques and results of stem cell and HFC transplantation for effective hair-loss treatment, as well as the impact of growth factors and EVs on extending hair follicles and enhancing hair restoration. Update technology such as gene therapy and tissue engineer in hair-loss treatment scope.
Hair shaft structure and hair follicles under the skin
The human body is covered with approximately 5 million hair follicles, including those in the skin, eyebrows, eyelashes, and scalp. The human scalp accounts for 80,000–150,000 of these follicles. Hair follicles are mini-organs comprising two structures: the hair shaft and structures under the skin 28 (Fig. 2). The shaft is divided into three components: cuticle, cortex, and medulla (Fig. 2). The cuticle is the outer layer of the hair shaft and contains an overlapping flat cell layer 29 . This layer is approximately 3–4 μm thick and can reflect light, giving hair a shiny and healthy appearance 15 . The cortex layer contains microfibrils (keratin filaments), remnants, melanin granules, and hair moisture15,29. Melanin, which is important for hair color, is divided into eumelanin and pheomelanin. High eumelanin and low pheomelanin levels result in dark hair formation. Red and blonde hair have low to medium levels of eumelanin and pheomelanin 29 . The center of the hair shaft is the medulla, which contains glycogen and medullary granules 15 .
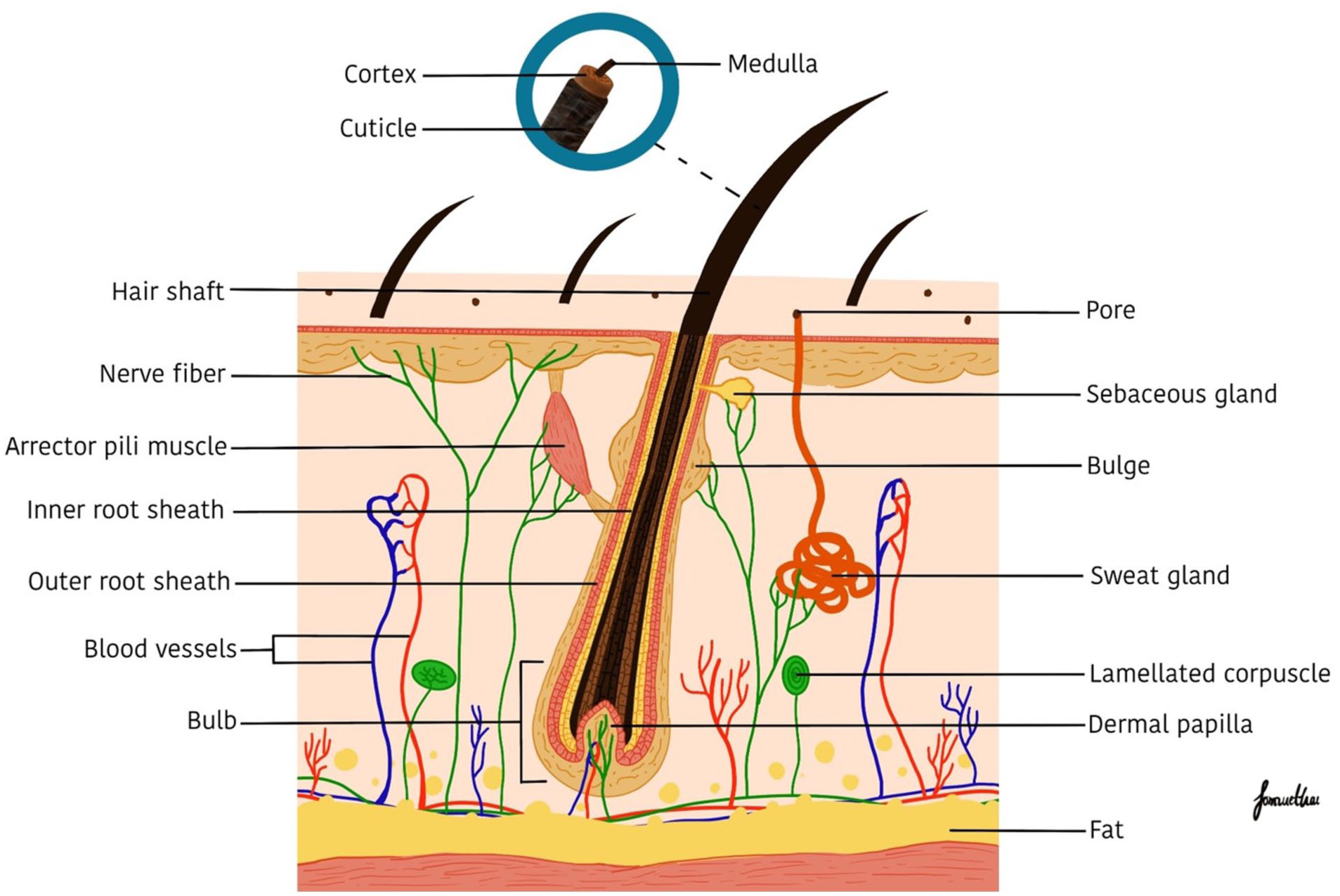
The diagram shows hair shaft and the structures under the skin. The shaft contains the medulla, cortex, and cuticle. The under-skin structures include sebaceous glands, the bulge, inner and outer root sheaths, and the bulb with a dermal papilla in its center. Schematic figures were created using procreate.com.
The hair follicle under the skin (Fig. 2) contains sebaceous glands, bulges, inner (IRS) and outer (ORS) root sheaths, and a bulb. Bulges fully comprise HFSCs, which are important for generating new hair. The IRS is divided into three layers: Henle’s, Huxley’s, and cuticle. Keratinization first occurs in Henle’s layer and then in Huxley’s layer. Finally, keratinized cells are transformed into hair cuticles in the cuticle layer 28 . The ORS contains keratinocytes, melanocytes, Langerhans cells, and Merkel cells, which function as sensory organs and immunological components of the skin 28 . The bulb is the lowermost part of the hair follicle and consists of matrix keratinocytes, follicle pigments, and dermal papilla (DP) 30 .
Hair cycle and molecular signaling
The hair cycle is divided into three phases: anagen (growth phase), catagen (apoptosis phase), and telogen (quiescent phase). Normally, hair on the scalp is in the anagen phase (85%–90%) and maintains this phase for 2–7 years. Hair in the catagen phase comprises only 1%–2% of the scalp and remains in this phase for 2–4 weeks. Approximately 10%–15% of the scalp has hair in the telogen phase, which persists for 3 months15,28 (Fig. 3). During the telogen phase, the DP moves to a location near the bulge and sends molecular signals to the bulge for quiescent HFSC activation. Subsequently, HFSCs migrate to the base of hair follicles in the hair matrix, start proliferating and differentiating for new hair generation, and begin the anagen phase 31 (Fig. 3). It implies that hair loss can be prevented by prolonging the anagen phase and inducing hair regrowth after hair shedding (telogen phase). Several crucial molecular signaling cascades for hair development include the Wnt/β-catenin, bone morphogenetic protein (BMP), Sonic hedgehog (Shh), and Notch pathway.

The hair follicle cycle is divided into three phases: anagen (growth phase), catagen (apoptosis phase), and telogen (quiescence phase). Hair maintains the anagen phase for 2–7 years across 85%–95% of the scalp. In the catagen phase, apoptosis starts in the hair matrix, as well as in the inner and outer root sheaths. Hair shafts start to form hair clubs, and epithelial strands help move the hair shafts up to the bulge. Hair remains in catagen for 2–4 weeks in 1%–2% of the scalp. In the telogen phase, the dermal papilla separates from the club hair and moves up near the bulge. Hair stays in this phase for 3 months in 10%–15% of the scalp. The dermal papilla sends signal molecules to activate hair follicle stem cells (HFSCs) in the bulge to start anagen I, where Wnt levels are high, and BMP levels decrease. Shh induces the dermal papilla to move down from the bulge in anagen II. New hair starts to generate, requiring signal molecules such as BMP, Wnt, Shh, and Notch in anagen III. Schematic figures were created using procreate.com. and BioRender.com.
The Wnt signal binds to its receptors, Frizzle 2/3/7, and lipoprotein receptor-related protein (co-receptor) in the bulge region and activates the component Disheveled (phosphoprotein), resulting in adenomatous polyposis coli, axis inhibition protein, and glycogen synthase kinase-3, which in turn inhibit β-catenin phosphorylation. Stable β-catenin is translocated to the nucleus and binds to transcription factors of the lymphoid enhancer factor/T-cell factor (LEF/TCF) family. This interaction promotes the expression of genes such as axis inhibition protein 2 (AXIN2), LEF1, and leucine-rich repeat-containing G-protein-coupled receptor 5 (LGR5), which in turn activate HFSCs and lead to the generation of hair follicles 32 . The Wnt family proteins, involved in hair development, include Wnt1a, which can induce hair follicle transition of telogen to anagen 33 and stimulate hair follicle formation 34 . Wnt3a and Wnt7b stimulate hair activity, promote hair growth, and induce hair shaft elongation 33 . Furthermore, Wnt3a, Wnt4, and Wnt7b are crucial for enhancing gene expression during the anagen phase 34 . Wnt7b is upregulated to promote early hair follicle formation when BMP is inhibited 35 . However, during the formation of mature hair follicles, Wnt7b is downregulated, leading to a decrease in other Wnt expressions, which affects the transition of hair follicles from anagen to catagen 35 . Thus, Wnt7b can regulate the duration of anagen 35 . Whereas Wnt5 is the target of Shh for early hair follicle formation, and its expression is induced by Shh34,36. However, Wnt5 has been reported to increase the telogen phase and decrease β-catenin. Therefore, Wnt5 inhibits hair follicle formation and effects aging hair follices 33 . On the other hand, Wnt10b is a major component of HFSC activation that promotes hair follicle formation32,34 and induces transition from telogen to anagen causing hair follicle regeneration and hair shaft elongation33,34.
BMP and Wnt/β-catenin pathways are important for the mechanism of activation and quiescence of HFSCs 30 . The transforming growth factor-β (TGF-β) family (cell regulatory protein) contains BMP (growth factor) and TGF-β signal molecules, which are important for hair follicle development but active in different stages of the hair cycle 37 . Quiescent HFSCs have high BMP levels in telogen, which are reduced in the early stage of anagen and subsequently upregulated in the late anagen phase as hair formation progresses 38 . TGF-β1 has been detected in the cuticle area, while TGF-β2 (expressed in ORS) and TGF-β3 have been found in cortex hair follicles at anagen. TGF-β2 is present in hair follicles transitioning from anagen to catagen; therefore, TGF-β2 may induce apoptosis in hair follicles 39 . On the other hand, TGF-β2 reportedly has anti-aging effects on DPCs by affecting collagen expression 40 . BMP and TGF-β have the same receptor, type I and II, on the cell surface but different intracellular signal molecules. BMP phosphorylates Smad1/5/8 (signal transducer), whereas TGF-β phosphorylates Smad2/3, and they use the same co-Smad4 to activate and translocate into the nucleus (Fig. 4)37,39. Moreover, TGF-β and BMP can reduce their effects through binding co-receptor suppression 37 .

The diagram shows TGF-β/BMP signaling pathway. TGF-β binds to receptor types II and I, phosphorating Smad2/3. Then, phosphorylated Smad2/3 activates co-Smad4 to translocate into the nucleus. BMP, binding type II and I receptor, phosphorylates Smad1/5/8. Active Smad1/5/8 activates co-Smad4 for translocation into the nucleus. Schematic figures were created using BioRender.com.
The Shh pathway is a key for activating quiescent HFSCs 32 and induces epithelial cell proliferation 37 . Moreover, it regulates DP function in expanded transient amplifying cells (TACs) 32 . Shh induces the DP to move down from the bulge, thereby increasing BMP levels 38 . Shh is present in the ORS and is important for linear hair formation 30 . The Shh pathway controls both epithelial and mesenchymal cells during hair follicle development 37 .
Notch pathways are activating, proliferating, and differentiating into HFSCs to generate hair follicles 32 . Notch transmembrane protein receptor, involved in epidermal cell 37 and hair shaft development 30 , is bound by ligand Delta-like 1 (Dll1). The Dll1 has been found during early hair follicle development but is unobservable in mature hair follicles 37 . Notch receptors interact with other signaling molecules, such as Shh, to induce the hair cycle30,37.
The interaction between molecular signaling pathways affects hair follicle formation, as shown in Fig. 3. In telogen, BMP2/4 and Wnt levels increase and decrease, respectively, resulting in HFSC quiescence. Contrarily, BMP2/4 decreases while Wnt increases in anagen I. Consequently, β-catenin levels stabilize, leading to the activation and migration of HFSCs down to the bulb30,38,41. Anagen II HFSCs are obtained from TACs when BMP and Wnt levels are low and high, respectively. At this stage, Shh stimulates and maintains Wnt signaling. TACs originate from the hair shaft in anagen III, in which Shh drives the DP lower from the bulge, owing to an increase in BMP. Hair shaft differentiation requires several signal molecules, including Wnt/β-catenin, Shh, Notch, and BMP30,38.
Stem cells for hair-loss treatment
Stem cell treatments for hair-loss using adipose-derived stromal vascular fraction (ADSVFs) and allogeneic MSCs are shown in Table 1. ADSVFs have been approved by the FDA and the European Medicines Agency for use in advanced therapy under doctor’s supervision 42 . ADSVFs include MSCs, hematopoietic stem cells, and endothelial and immune cells21,43. ADSVFs show positivity for markers of CD13, CD14, CD29, CD34, CD117, CD166, HLA-ABC, HLA-DR, CD31, CD45, CD73, CD90, and CD105. ADSVFs have been administered at approximately 0.2 mL per injection spot in a concentration of 1 million cells per 1 mL NaCl20,21,43 and an injection depth of 4 mm, targeting both the dermal21,43 and subcutaneous layers 20 . The results showed that ADSVFs could increase anagen 93.4% and decrease telogen 35.6% after treatment for 6 months 20 . Moreover, hair diameter and density also increased after 3–6 months of treatment 43 . The number of hair falls decreased after hair-pull testing 21 . ADSVFs improve hair-loss by restoring stem cells in damaged hair follicles or by activating quiescent HFSCs. In addition, ADSVFs contain growth factors important for scalp skin repair, vascularization, and hair development, including vascular endothelial growth factor (VEGF), hepatocyte growth factor (HGF), IGF, fibroblast growth factor (FGF), and platelet-derived growth factor (PDGF) which are found in adipose-derived stem cells isolated from ADSVFs21,42,44.
Previous studies on stem cell treatment for generating hair growth.

AA: alopecia areata; ADSVFs: autologous adipose-derived stromal vascular cells; AGA: androgenetic alopecia; AU: alopecia universalis; BMMCs: bone marrow–derived mononuclear cells; FSC: follicular stem cells; GR: glucocorticoid receptor; MSCs: mesenchymal stem cells; SALT: Severity of Alopecia Tool; PDSSCs: proteins derived from stressed mesenchymal stem cells.
Autologous bone marrow stem cell treatment for alopecia areata by injection of 100,000 cells/ml per square centimeter intradermally. The result demonstrates that 50% of cases achieve very good outcomes, with a 45% overall improvement. In cases of androgenetic alopecia (AGA), results show 10% excellent, 50% very good, and 52% improved45,47. In addition, allogeneic MSCs from the umbilical cord have been reported for hair-loss treatment, both with and without prior expansion before treatment22,23. Freeze-thawed MSCs have been injected into the dermis or intradermally22,23. The concentration and volume of the injections differed between the two studies (Table 1)22,23. Fresh MSCs were positive for CD73 (77.48%), CD90 (95.67%), and CD105 (95.40%) 22 . The samples were frozen and thawed before use without further expansion 22 . The treatment results showed that hair growth covered the alopecia area in patients 1 and 2 with no visible alopecia occurring thereafter 22 . However, patient 3 who had alopecia universalis showed improved hair growth after 3 months, but hair regeneration to cover the scalp area was not achieved after treatment for 1 year 22 . Another report on MSC treatment indicated that all patients showed improved hair growth 23 . On average, the Severity of Alopecia Tool (SALT), a standardized method for quantifying scalp hair loss, score decreased by 14.9% after treatment for 12 weeks and 12.2% after treatment for 24 weeks 23 . In addition, MSC protein for topical hair-loss treatment product was developed. MSC was induced stressed by culture in Atalantia monophylla extract (root extract: antioxidant, antifungal, antiviral, anti-inflammatory, immunomodulatory effects and maintain the stemness of MSCs), and then MSC proteins were extracted. The results showed that AGA mice could regenerate hair growth after 24 days of treatment. The immunohistochemistry results showed increased expression of HFSCs markers (K15 and K17). Moreover, the Wnt and BMP signaling pathways also found increased expression of β-catenin, Wnt5a, LEF1, TGF-β, and the glucocorticoid receptor 46 .
Stem cells show effective and safety for hair-loss treatment in some cases22,45. However, the functions of stem cells are not specific to hair follicle development. Whereas DPCs, DSCs, HFSCs, and skin progenitor cells are the focus for restoring hair. DSCs and autologous scalp tissue have been reported for use in human hair-loss treatment48,26.
HFCs for hair restoration
Cultured and expanded HFCs are of interest for hair-loss treatment. DPCs, DSCs, HFSCs, and skin progenitor cells have been reported to use several mechanisms to regenerate and improve hair growth (Table 2).
Previous reports about hair follicle cells for hair-loss treatment.

DPC, dermal papilla cell; DSC, dermal sheath cell; DMEM, Dulbecco’s Modified Eagle Medium; FBS, fetal bovine serum; FGF, fibroblast growth factor; EPC, endothelial progenitor cell; HFSC, hair follicle stem cell; VEGF, vascular endothelial growth fact.
Embryonic hair follicle development begins with the interaction between mesenchymal and epithelial cells. The mesenchymal cells undergo condensation and develop to DP surrounded by the dermal sheath (DS) layer in bulb location 56 . DPCs express ALPL, VCAN, SOX2, β-catenin, and Wnt5a51,57,58 and positive markers for CD133, CD90, and CD10551,59–61. In contrast, DPCs show negative expression in the markers CD34 and CD45 61 . The problem with DPC culture is their loss of ability to induce hair growth after culture at high passages (P); consequently, conditioned media50,49 or 3D culture51,25 has been developed to increase their hair growth induction ability. Wnt conditioned medium has been used to culture DPCs and maintain their hair growth induction ability. Previous studies have shown that DPC transplantation at P2 and P10 with keratinocyte cells at P1 induced 100% hair growth in mice 50 . Moreover, a keratinocyte conditioned medium can maintain the hair growth induction ability of DPCs for more than P90, and implantation induces 93% hair growth 49 . Another method reported to improve the culture of DPCs in a 3D environment involves their aggregation by culturing them in hanging drops to form spheroid DPCs that induce unpigmented hair and lack sebaceous glands 62 . However, encapsulating CD133+ DPCs in hydrogel resulted in increased gene expression and hair growth after grafting mixed CD133+ DPCs and epidermal and dermal cells on the backs of nude mice for 21 days 51 .
HFSCs is another cell type which interests hair-loss treatment, located in the bulge at a quiescent stage. Molecular signaling from the DP is required to activate HFSCs. The markers for identifying HFSCs are CD200, K15, and K1425,63. Previous reports show that HFSCs aggregated in 3D culture using Matrigel showed high gene expression in both mouse and human HFSCs. In addition, transplantation of hair follicle germs (HFGs) (HFSCs with embryonic mesenchymal cells) induces hair growth in mice 25 .
The origins of DS and DP are similar, developing from the condensation of the dermal layer during embryonic development. Therefore, DS can be converted into DP depending on the surrounding microenvironment 64 . However, DS produces smaller and thinner hair follicles than does DP 65 . Moreover, DSCs can induce the ability of aging DPCs as shown in previous report; transplanted DPCs at P60 with DSCs at P1 can generate hair growth 24 .
Clinical trial phase II for human hair-loss treatment involving the injection of DSCs for male pattern hair loss (Norwood grade 3–6) and female pattern hair loss (Shiseido grade 3–6) was conducted 26 . DSCs were injected into the scalp at four spots, comparing high dose (7.5 × 106/ml), medium dose (1.5 × 106/ml), and low dose (3.0 × 105/ml) 26 . The results indicated that the low dose increased hair density, and diameter improved after treatment between 6 and 9 months without serious side effects 26 . They suggest that DSCs can migrate and differentiate into DP, enhancing hair quality 26 . To prove this concept, gene expression analyses were conducted for both the high- and low-efficacy groups 66 . The high-efficacy group upregulated cell proliferation and migration gene expression, whereas the low-efficacy group upregulated basement membrane and vasculature development 66 . In addition, the phase III clinical trial reported an increase in the treatment area to 105 cm2. DSCs (3.6 × 106cells/12 ml) were injected intradermally for treatment at 120 spots, twice every 3 months 54 . The results indicate that hair diameter improves more in the midline area than in the peripheral area. DSC treatment can maintain and extend the anagen phase. This treatment is effective for older patients and females, demonstrating better treatment results than in males 54 . Finally, in 2024, DSC hair treatment received approval for regenerative medicine in Japan 13 . In addition, DSCs and DP spheres also have clinical trial report 55 . The treatment involved injection at 9 spots per 1 cm2, with an injection distance of 0.5 cm and a cell concentration of 1 × 105/spots, and results were obtained after follow-up for 9 months 55 . After treatment at 1 and 3 months, there was an improvement in the proportion of terminal hair and an increase in mean hair diameter. However, the results slightly decreased after 3 months 55 . Furthermore, human micrografts from scalp tissue have been used to treat hair loss, and the results showed an increase in hair density after treatment for 14.5 months 48 . Flow cytometry analysis showed high levels of K15 and CD200 in scalp hair compared with those in the bald scalp, suggesting that scalp tissue contains HFSCs and epithelial cells that induce hair regeneration 67 .
Important considerations for HFC hair-loss treatment
HFCs are used for human hair-loss treatment with safe single-cell injections into the scalp. However, the success rate must concern several factors such as injection position, suitable number and quality of cells, microenvironment, and hormone which effect hair loss.
Injection position for hair-loss treatment
HFC single cells and DP sphere have been utilized for treating human hair loss26,48,55 while aggregates have not been documented for transplantation in humans. Prior studies have demonstrated that single-cell injections can regenerate hair growth, enhance hair diameter and density, and extend the anagen phase26,48,52,62. Therefore, the locations of injection points are crucial for the success rate of transplantation.
Hair regeneration should be injecting HFCs intradermally to promote interactions between the epidermis and dermis, initiating the formation of hair placodes, inducing dermal condensation, and stimulating new hair growth. Key molecular signaling during this phase includes Wnt/β-catenin, Shh, FGF, and TGF-β2, while inhibiting BMP2/430,31. A single HFC contains molecular signals and growth factors that promote hair follicle formation23,48. Furthermore, baldness occurs when hair is shed, and quiescent HFSCs become inactive due to low signaling molecules. Injecting HFCs into the dermal layer near the bulge may enhance molecular signals to activate quiescent HFSCs, prompting them to enter early anagen. Activated HFSCs migrate to the bulb area of the hair follicle for proliferation and differentiate into TACs, which can further develop into melanocyte cells, mesenchymal cells, and epithelial cells, all crucial for hair formation30,31. The important molecular signaling during this period includes elevated levels of Wnt/β-catenin and Shh, along with downregulation of BMP2/430,31,38.
Number and quality of cells for treatment
Optimizing the number of cells and injection distance is essential for effective cell therapy in human hair-loss treatment. Previous phase II clinical trials in humans compared three different concentrations of DSCs injection at four spots (75,000 cells/spot, 375,000 cells/spot and 1,875,000 cells/spot) 26 . The best results were obtained using 75,000 cells/spot, leading to improvements in hair diameter and density after treatment for 6 to 9 months 26 . In addition, a phase III clinical trial reported that DSCs were injected at a rate of 3.6 × 106 cells/12 ml, with a total injection point of 120 spots in 105 cm2 every 3 months for 2 times without serious side effects 54 . However, DSCs and DP spheres were used at 1 × 105 cells per injection spot with an injection distance of 0.5 cm 55 . Moreover, Leng et al. 52 reported the generation of 1 hair using 7,000–8,000 skin progenitor cells after injecting 0.5 to 1 × 106 cells in the dermal layer of a human skin graft at depths of 1 and 2 mm 53 . However, multiple rounds of HFC treatment may be requested depending on hair-loss severity48,54. DPCs, DSCs, and HFSCs can duplicate during culture, eliminating any limitations to their use in treatment. Another important factor is cell quality. Treatment requires high cell viability; therefore, several reports have used fresh cells for transplantation48,55. Even when using freeze-thawed cells, viability control is important for high-efficiency treatments 26
Hair-generation microenvironment
Hair follicles require a suitable microenvironment for generation such as adipose tissue, arrector pili muscle, blood vessels, immune cells, and nerves68,69. Enhanced vascularization and blood supply to the scalp improve transplantation success and maintain hair follicle growth21,68. When transplanted in an unsuitable microenvironment, hair follicles grow poorly or with incomplete parts, such as unpigmented hair and undeveloped sebaceous glands 63 . Transplantation of human progenitor cells into Nude/nude mice resulted only in dark pigment spots, whereas Nude/nude mouse-human skin grafts successfully regenerated hair growth because of a suitable microenvironment. Researchers indicate that the success rate remains low (20%), highlighting the need to improve the microenvironment for better transplantation outcomes 52 . Moreover, both young and aged individuals exhibit differences in hair generation due to aging and blood supply. Young skin demonstrates longer durations of the anagen phase compared to those of aged skin 69 . HFC treatment of bald areas improves the health of scalp skin, which contains cells and growth factors that are important for increasing moisture and blood supply under the skin to support hair growth 48 .
Hormonal effects on hair follicle regeneration
Hormones enhance hair follicle growth while also inducing hair loss. High estrogen levels during pregnancy prolong the anagen phase and increase hair diameter 70 . However, postpartum hair loss occurs 2–4 months after delivery due to changes in hormone levels. This phenomenon affects 90% of women postpartum. Hair loss stops when hormone levels normalize from 6 months to 1 year 71 . Moreover, androgens stimulate hair growth in the beard and axillary regions. DPCs from the beard secrete autocrine growth factors, such as IGF-I, which affect testosterone to increase hair follicle size. On the other hand, androgen stimulates DPCs in the frontal scalp to secrete TGF-β1, which suppresses keratinocytes and leads to the production of miniaturized hair. However, these effects are dependent on genetic factors 72 .
In addition, 5α-reductase converts testosterone to DHT and binds to androgen receptors at the hair follicle surface. DHT has five times more potential to bind androgen receptors than testosterone 72 . Once bound, DHT enters the nucleus and triggers the production of proteins that lead to hair miniaturization 15 . After hair transplantation, hair follicles are also susceptible to hormonal effects; therefore, finasteride and minoxidil are administered to prolong hair follicle lifespan 12 . Minoxidil directly induces the VEGF effect by increasing the DP diameter 41 . Hormonal may have effects on HFCs treatment. Nevertheless, HFCs contain growth factors that protect against these effects. DHT has the effect of decreasing IGF-I, which is important for maintaining the anagen phase, whereas HFCs, conditioned medium, and platelet-rich plasma (PRP) contain several growth factors that have beneficial effects in improving hair quality, such as IGF-I, TGF-β, PDGF, FGF, EGF, and VEGF73,74.
PRP source of growth factor for improving hair growth
PRP are filled with growth factors to enhance hair growth, including IGF-I, PDGF, TGF-β, and VEGF 74 . IGF-I benefits include protecting hair follicles from apoptosis and maintaining the anagen phase 75 . PDGF enhances epithelial cell proliferation 75 . TGF-β is functional in both inhibiting and inducing hair growth effects in aging DPCs39,40, and VEGF increases blood supply, which is essential for improving hair density 13 . PRP treatment is partially effective in promoting hair thickness and density 13 . However, PRP is requested to be used fresh after the process; therefore, microneedle patch techniques were applied to develop PRP methods 76 . Microneedle PRP can sustain growth factor efficiency for 4–6 days, with the needle directed toward the bulge position 76 . PRP microneedles were shown to promote hair growth better than PRP subcutaneous injection in mice after 13 days of treatment without erythema and skin damage 76 .
EVs derived from conditioned media to promote hair growth
EVs for improved hair loss obtained from both stem cells and DPCs condition medium5,61. EVs-MSC from mouse bone marrow have been reported to induce the activation of VEGF and IGF-I in DPCs, thereby increasing their proliferation, migration, and survival rates 5 . EVs derived from adipose mesenchymal stem cells (EVs-ADSC) are highly expressed miR-122-5p, which reduces Smad 3 in the TGF-β pathway and inhibits DHT in DPC culture 6 . Treatment with EVs-ADSC injected subcutaneously into mice resulted in hair follicle growth after 15 days of treatment; the result showed a trend similar to that of minoxidil treatment 6 . EVs-ADSC has been reported to increase LEF1 and Wnt 3a in DPCs while reducing hair inflammatory disorders, as TNF-α levels decrease in DPCs treated with EVs-ADSC 77 . Moreover, a case report from a patient who complete hair loss from chemotherapy shows EV treatment induced hair growth within 3 months by receiving subcutaneous injections of allogenic EVs from placenta at a dose of 140–160 µg (2.5–3.2 × 1010 particles) for three sessions without side effects 78 . They suggest that treating hair loss with EVs enhances DP proliferation and migration 78 . In addition, preclinical studies report cases of trichorrhexis nodosa, a condition affecting the hair shaft that causes breakage and hair loss 79 . The treatment involved injecting 5 ml of EVs derived from stem cells subcutaneously, with injections spaced 0.5–1.0 cm apart every month. The results demonstrated improved hair growth, increased hair thickness, darker hair color, and normalized cuticle layers 79 . Moreover, a human clinical trial has been conducted with EVs-ADSC administered using a microneedle roller at a concentration of 6 × 106 particles/vial for 12 weeks in 39 AGA patients. The results showed that hair density and thickness improved 80 . In addition, a microneedles patch was developed as an alternative method for EV treatment 27 . The results compared between EVs-ADSC microneedles, subcutaneous injection, and tropical 3% minoxidil show hair generation rate of 99%, 86%, and 77%, respectively 27
EVs derived from DPC conditioned medium (EVs-DPCs) have been reported to be injected subcutaneously into mice, resulting in enhanced wound healing 61 . Moreover, they investigate that Krüppel-like factor 4 from EVs-DPC activates the effects of VEGF-A on vascularization during the wound-healing process 61 . Furthermore, EVs-DPC co-cultured with HFSCs contain Wnt3a, LEF1, and IGF-I, which promote the Wnt/β-catenin pathway and activate HFSC proliferation81,82. Therefore, EVs can maintain healthy hair follicles, reduce the harmful effects of hormones, improve scalp skin, and support vascularization.
Gene therapy for hair-loss treatment
Gene therapy is an alternative way to solve the hair-loss problem. CRISPR/Cas9 is a gene-editing technique that uses Cas9 to cut double-stranded DNA. However, a guide RNA was requested in this method to target a specific DNA sequence. After editing the target, the DNA will repair itself, and the genes that cause hair loss will be eliminated 75 . Gene therapy has been reported to be used in animal models such as mice, rabbit, and cashmere goat 83 . Steroid 5 alpha reductase 2 type II (SRD5A2) enzyme was a gene edited by CRISPR/Cas9 in mice. The result showed SRD5A2 expression decreases following gene editing, leading to an increase in hair density and diameter 7 . The FGF5 growth factor functions as a regulator of hair cycle driving to induce anagen to catagen. The result in rabbit editing FGF5 expression led to inhibited BMP2/4 and increased VERSICAN signaling pathway resulting in prolonged anagen 84 . Moreover, cashmere goats were gene edited to increase thymosin beta 4 (Tβ4) a small-molecule protein important for cell proliferation and differentiation, thereby enhancing hair growth. The results show cashmere clone produced a cashmere yield increase of up to 74.5% 85 . Recently, gene therapy has been utilized for the treatment of genetic diseases in humans. A patient with severe carbamoyl-phosphate synthetase 1 deficiency received customized treatment at 7 and 8 months. A patient being enabled to process protein without high blood ammonia level. However, long-term effects still need to be monitored 86 . Conversely, there have been no reports on gene therapy for human hair loss. The research into gene editing of ARD5A2 and FGF5 is promising for potential applications in human hair-loss gene therapy.
Tissue engineers
Preparing tissue-engineered skin substitutes (TESs) for creating skin and hair follicles or producing artificial skin for culturing in vitro hair farms is intriguing for hair-loss therapy. However, methods of producing hair follicles in the laboratory still have problems with incomplete hair function and structure such as sebaceous glands and arrector pili muscles 8 . Epidermal cells from bulge areas are also considered interesting for producing TESs that hair stem cells contain 87 . Moreover, Yao et al. present the idea of generating hair follicles in vitro using 3D scaffold techniques as a structure for skin substitutes. They implant the sphere of neonatal dermal fibroblasts and epidermal cells (ratio = 9:1) in TESs, which consists of dermal and epidermal cell layers. Hair follicles can grow, but the number of hair follicles produced in the laboratory is quite low. TES techniques still need to improve and prepare an optimal microenvironment for hair follicle growth. Hair farms present an interesting approach to producing hair follicles to address hair-loss issues. However, these techniques still require enhancement to generate large number of hair follicles with complete hair structure and function.
Conclusions
Hair loss is a major problem affecting personal life. Drug treatments and hair transplantation have been used to treat hair loss. Furthermore, advanced medical technologies such as stem cell and HFC treatment are entering the clinical trials in humans stage. In addition, gene therapy and tissue engineering are being developed to be used to solve the hair-loss problem. Understanding the hair cycle and molecular signaling is crucial for treatment success.
Hair follicle in the telogen phase, DP move up near the bulge and send signals to the bulge area, thereby activating HFSCs. The pathways that are crucial in this process are Wnt/β-catenin and BMP. When hair follicles initiate growth, Wnt/β-catenin, Shh, and Notch pathways are crucial to generating hair shafts and structures. Stem cells play a key role in repairing the scalp, reducing inflammation, and activating aging hair follicles. Therefore, hair follicles increase in diameter and density after treatment. However, the functions of stem cells are not specific to inducing hair growth. In contrast, HFCs such as DPCs, DSCs, and HFSCs isolated from hair follicles are specific cells that are crucial in promoting hair growth. To achieve success, the injection position for HFC treatment should be carefully considered; injecting near the bulge area may effectively activate the quiescent HFSCs. In addition, quality control and determining the optimal number of cells are crucial for successful treatment. Moreover, microenvironmental factors must be optimized to enhance hair growth and prolong the anagen phase. Molecular signaling and growth factors are important for maintaining a healthy hair microenvironment. Furthermore, the effects of DHT hormones, which seriously cause hair loss, are reduced by using growth factors or EV treatment. Another approach to reducing DHT and extending the anagen phase is gene therapy aimed at decreasing ARD5A2 and FGF5. In cases of severe hair loss, generating hair follicles in vitro through tissue engineering techniques may be a promising avenue. However, these techniques still require further improvement to produce a large number of hair follicles with complete structure and function. In addition, most important of advanced techniques for hair-loss treatment is to indicate both safety and efficacy.
Footnotes
Ethical approval
This study was approved by our institutional review board.
Author Contributions
Kwanrudee Kaewmungkun: Conceptualization, Investigation, Writing—original draft, Writing—review & editing, Visualization, Project administration. Somruethai Kaisang: Visualization. Nattida Yokhaphachon: Review. Rangsun Parnpai: Review & editing.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The references used for the writing of this manuscript are publicly available.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
