Abstract
Transplantation of mesenchymal stem cells (MSCs) is one of the hopeful treatments for spinal cord injury (SCI). Most current studies are in animals, and less in humans, and the optimal transplantation strategy for MSCs is still controversial. In this article, we explore the optimal transplantation strategy of MSCs through a network meta-analysis of the effects of MSCs on SCI in animal models. PubMed, Web of Science, Cochrane Library, Embase, China National Knowledge Infrastructure (CNKI), Wanfang Database, China Science and Technology Journal Database (VIP), and Chinese Biomedical Literature Service System (SinoMed) databases were searched by computer for randomized controlled studies on MSCs for SCI. Two investigators independently completed the literature screening and data extraction based on the inclusion and exclusion criteria. RevMan 5.4 software was used to assess the quality of the included literature. Stata 16.0 software was used for standard meta-analysis and network meta-analysis. Standardized mean difference (SMD) was used for continuous variables to combine the statistics and calculate 95% confidence interval (95% CI). P < 0.05 was considered a statistically significant difference. Cochrane’s Q test and the I2 value were used to indicate the magnitude of heterogeneity. A random-effects model was used if I2 > 50% and P < 0.10 indicated significant heterogeneity between studies, and conversely, a fixed-effects model was used. Evidence network diagrams were drawn based on direct comparisons between various interventions. The surface under the cumulative ranking curve area (SUCRA) was used to predict the ranking of the treatment effects of each intervention. A total of 32 animal studies were included in this article for analysis. The results of the standard meta-analysis showed that MSCs improved motor ability after SCI. The network meta-analysis showed that the best treatment effect was achieved for adipose tissue–derived mesenchymal stromal cells (ADMSCs) in terms of cell source and intrathecal (IT) in terms of transplantation modality. For transplantation timing, the best treatment effect was achieved when transplantation was performed in the subacute phase. The available literature suggests that IT transplantation using ADMSCs in the subacute phase may be the best transplantation strategy to improve functional impairment after SCI. Future high-quality studies are still needed to further validate the results of this study to ensure the reliability of the results.
Introduction
Spinal cord injury (SCI) is a severe injury disorder of the central nervous system. The global incidence of SCI has been reported to range from 12 to over 65 cases per million per year 1 . SCI may be caused by traffic accidents, falls, violence, and others 2 . SCI mainly manifests as sensory, motor, and autonomic dysfunction varying degrees below the plane of injury, seriously affecting patients’ quality of life3,4. The pathological mechanism of SCI is divided into two main stages: primary injury and secondary injury. Primary injury refers to direct damage to the spinal cord tissue by external mechanical forces, accompanied by nerve cell death, vascular damage, oxidative stress, inflammation, and excitotoxicity of nerve cells5,6. Secondary injury occurs hours to weeks after the primary injury. It consists mainly of pathological reactions such as massive neuronal apoptosis, residual nerve fiber demyelination, gilial scar formation, and spinal cord cavitation 7 . Surgery, drugs, hyperbaric oxygen therapy, and physical therapy have been widely used in the clinical treatment of SCI, but with modest efficacy. Therefore, finding effective treatments for SCI has become an urgent problem to be solved.
Mesenchymal stem cells (MSCs) are multifunctional progenitor cells found in tissues such as bone marrow, adipose, and umbilical cord that can self-renew, proliferate, and differentiate into multiple cell lines 8 . MSCs can secrete bioactive molecules such as growth factors and cytokines, inhibit inflammation, reduce glial scarring, and resist apoptosis, thus promoting axonal regeneration, which has essential potential and broad application prospects in SCI treatment 9 . MSCs transplanted into the area of SCI can release trophic factors at the site of injury to maintain the activity of cells in the injured area and release anti-inflammatory factors to further promote vascular regeneration, blood–spinal cord barrier repair, and nerve regeneration10,11. MSCs can also tilt the balance of inflammatory cytokines in an anti-inflammatory direction by regulating the status of macrophages astrocytes and T cells12,13. A study showed that adipose tissue–derived mesenchymal stromal cells (ADMSCs) could activate the transforming growth factor (TGF)-β1/Smad3/procollagenlysine, 2-oxoglutarate 5-dioxygenase 2 (PLOD2) pathway in spinal cord neurons and promote axonal regeneration, enhancing functional recovery after SCI 14 . Zhao et al. 15 found that neural regeneration scaffolds of umbilical cord mesenchymal stem cells (UCMSCs) inhibited glial scar formation, promoted axonal growth, and improved motor and sensory function after SCI. Gu et al. 16 reported that bone marrow mesenchymal stem cells (BMSCs) improve motor function and reduce the expression of the pro-apoptotic transcription factor C/EBP homologous protein (CHOP) and apoptosis after SCI. Although the preliminary results are hopeful, various transplantation strategies exist for MSCs. There is no uniform conclusion on the advantages and disadvantages of different transplantation strategies for SCI treatment.
Therefore, this study used network meta-analysis to comprehensively evaluate the intervention effects of different MSCs transplantation strategies of MSCs on motor function after SCI in animals and to determine the optimal transplantation strategy for clinical decisions.
Materials and Methods
This study was conducted under the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.
Search Strategies
A database search of English and Chinese databases for randomized controlled trials of MSCs for SCI was performed by a computer. English language databases included PubMed, Web of Science, Cochrane Library, and Embase. Chinese databases included China National Knowledge Infrastructure (CNKI), Wanfang Database, China Science and Technology Journal Database (VIP), and Chinese Biomedical Literature Service System (SinoMed). The time frame for the search was from database creation to March 2, 2023 (please see Supplementary Material 1 for specific search strategies).
Study Selection Criteria
Inclusion criteria
Inclusion criteria included (a) randomized controlled trial of MSCs transplantation for SCI in rat models, (b) the experimental group was treated with MSCs on top of the control group, and (c) the outcome indicator was Basso–Beattie–Bresnahan (BBB).
Exclusion criteria
Exclusion criteria included (a) conference papers or duplicate published studies, (b) study data could not be extracted, (c) non-randomized controlled studies, and (d) the low-quality score of the literature.
Data Extraction
Duplicates retrieved were excluded using EndNote 20 and manually. Two investigators (Y.W. and C.G.) independently screened articles based on inclusion and exclusion criteria. When there was disagreement, it was resolved through group discussion. Two investigators (Y.W. and Y.D.) extracted the following information for each of the included animal studies: first author, year in publication, rat species, sample size, gender, age, weight, MSCs source, MSCs transplantation way, the dose of MSCs, transplantation time, final evaluation time, and outcome indicators. In studies that assess outcome indicators at multiple time points, data from the final assessment are extracted for analysis.
Quality Assessment
The risk of literature bias was assessed independently by two investigators (Y.W. and C.G.) using RevMan 5.4 software. Animal studies were assessed using SYRCLE’s risk-of-bias tool for animal studies. The evaluation included 17 (a) Was the allocation sequence adequately generated and applied? (b) Were the groups similar at baseline or were they adjusted for confounders in the analysis? (c) Was the allocation adequately concealed? (d) Were the animals randomly housed during the experiment? (e) Were the caregivers and/or investigators blinded from knowledge which intervention each animal received during the experiment? (f) Were animals selected at random for outcome assessment? (g) Was the outcome assessor blinded? (h) Were incomplete outcome data adequately addressed? (i) Are reports of the study free of selective outcome reporting? (j) Was the study apparently free of other problems that could result in high risk of bias? Low risk of bias, unclear, and high risk of bias were assessed for each entry. Any disagreement in the above quality assessment process was resolved through group discussion.
Statistical Analysis
This study performed the standard meta-analysis and network meta-analysis using Stata 16.0 software (Supplementary Material 2).
Standard meta-analysis
Effect size calculation
Standardized mean difference (SMD) was used for continuous variables to combine the statistics and calculate 95% confidence interval (95% CI). P < 0.05 was considered a statistically significant difference.
Heterogeneity test
Cochrane’s Q test and the I2 value were used to indicate the magnitude of heterogeneity. If I2 > 50% and P < 0.10, it suggested significant heterogeneity among studies, and the random-effects model was used; if I2 ≤ 50% and P ≥ 0.10, it suggested a small heterogeneity among studies, and the fixed-effects model was adopted. The combined results with more significant heterogeneity were searched for sources of heterogeneity by subgroup analysis.
Network meta-analysis
Evidence network diagrams were drawn based on direct comparisons between various interventions. The size of the dots in the evidence network diagram represents the sample size, and the thickness of the lines represents the number of studies 18 . A node-split model was used to test for local inconsistency between direct and indirect comparisons. If P > 0.05, it suggested no significant inconsistency, and the data were analyzed using the consistency model. If P ≤ 0.05, significant inconsistency was suggested, the data were analyzed using the inconsistency model, and the source of inconsistency was explored. The surface under the cumulative ranking curve area (SUCRA) was used to predict the ranking of the treatment effects of each intervention. If SUCRA is close to 1, the intervention is very effective. If SUCRA is close to 0, the intervention is probably ineffective. The symmetry of the two sides of the comparison-corrected funnel plots was used to determine whether there was a publication bias in the analysis results.
Results
Study Selection
A total of 5,605 relevant studies were retrieved. Of total, 2,868 duplicate studies were removed, and 2,563 studies were removed by reading the titles and abstracts. The full text was read carefully, and 142 studies were removed. Finally, 32 studies19 –50 were included, including 1425,28,31,33 –36,40,42,43,46,48 –50 in English and 1819 –24,26,27,29,30,32,37 –39,41,44,45,47 in Chinese (Fig. 1).
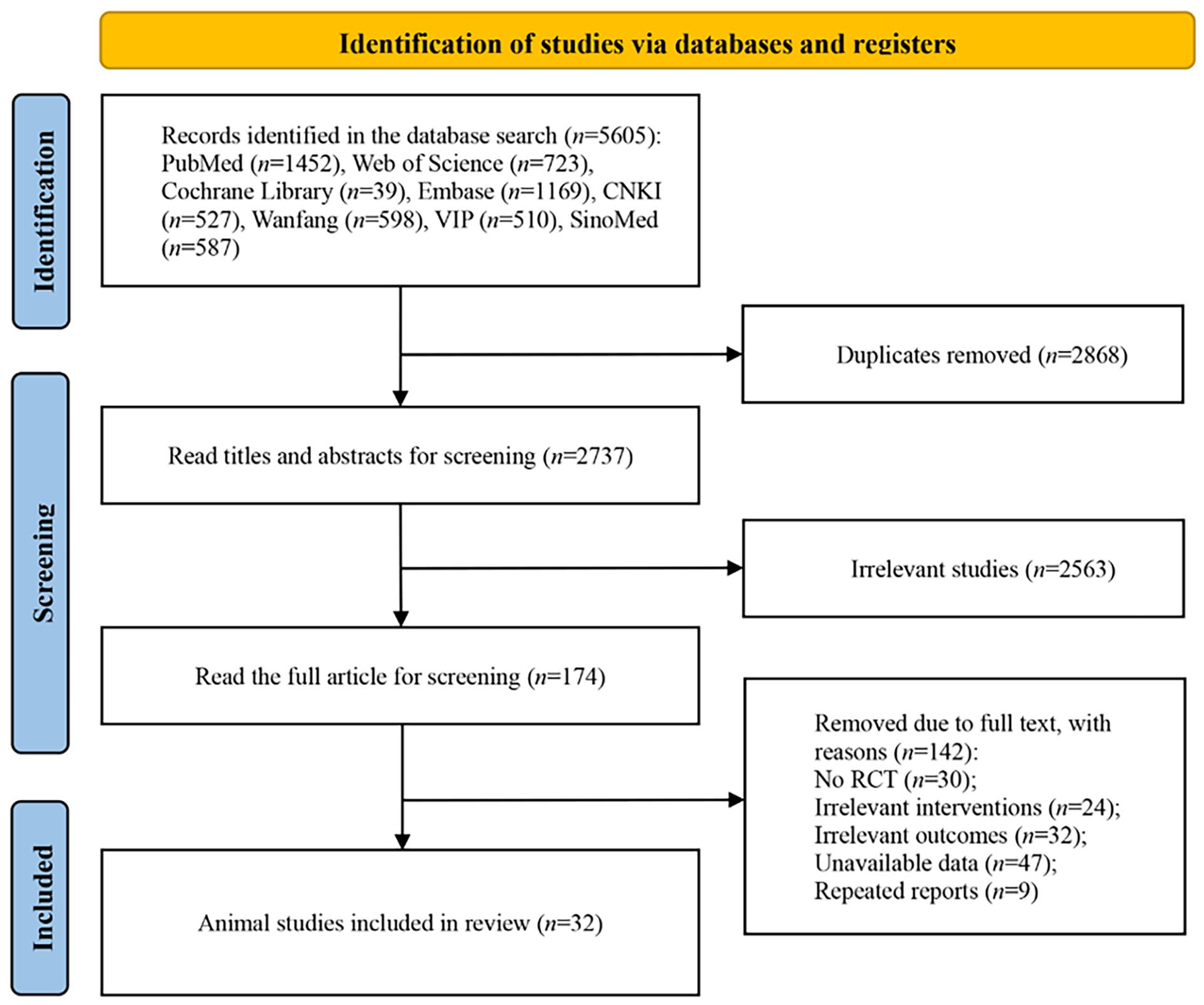
Flow chart of literature screening.
Study Characteristics
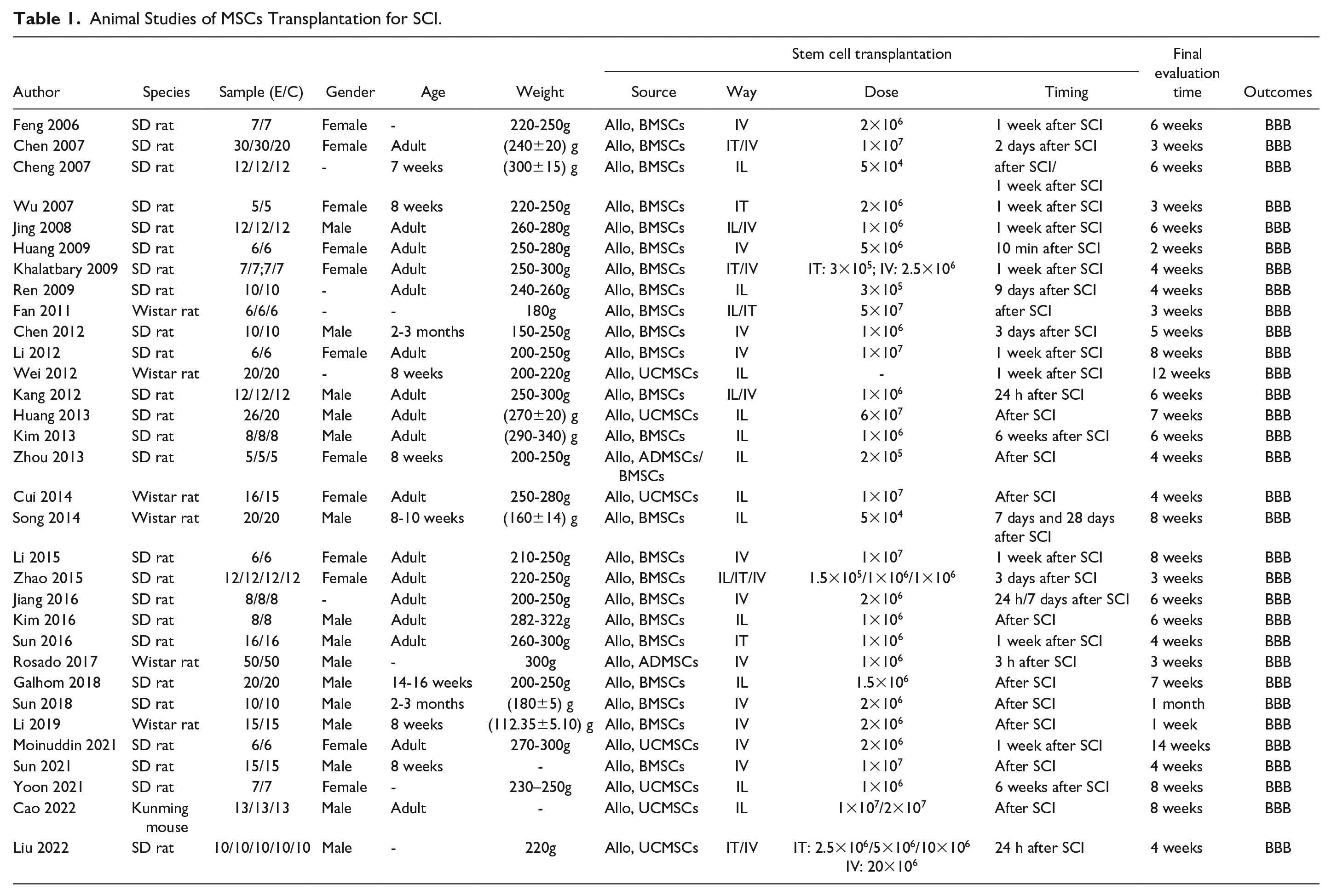
Sources of MSCs included UCMSCs (seven studies), BMSCs (24 studies), and ADMSCs (two studies). One study directly compared ADMSCs and BMSCs. MSCs transplantation route included intrathecal (IT) injection (seven studies), intralesional (IL) injection (16 studies), and intravenous (IV) injection (17 studies). Two studies directly compared IT and IL. Four studies directly compared IT and IV. Three studies directly compared IL and IV. Timing of MSCs transplantation included the acute phase (≤3 days, 19 studies), subacute phase (4–14 days, 13 studies), and chronic phase (≥15 days, 2 studies). 2 study directly compared acute phase transplantation with subacute phase transplantation. The essential characteristics of the animal studies are shown in Table 1.
Animal Studies of MSCs Transplantation for SCI.
Study Quality and Risk of Bias
The risk-of-bias evaluation for animal studies is shown in Fig. 2. Three studies37,41,47 described the generation of allocation sequences using the random number table method. The baseline characteristics of the groups were essentially the same for all studies. All studies did not adequately describe allocation concealment. Seven studies24,31,35,42,45,48,50 described the experimental procedure in which animals were randomly placed. Three studies19,22,40 reported a random selection of animals for measurement during outcome evaluation. A total of 16 studies19,20,24,26,29,30,32 –34,37 –39,42,43,46,48 were blinded to outcome evaluators. All studies were free from missing study data and selective reporting. No other bias was found to exist.
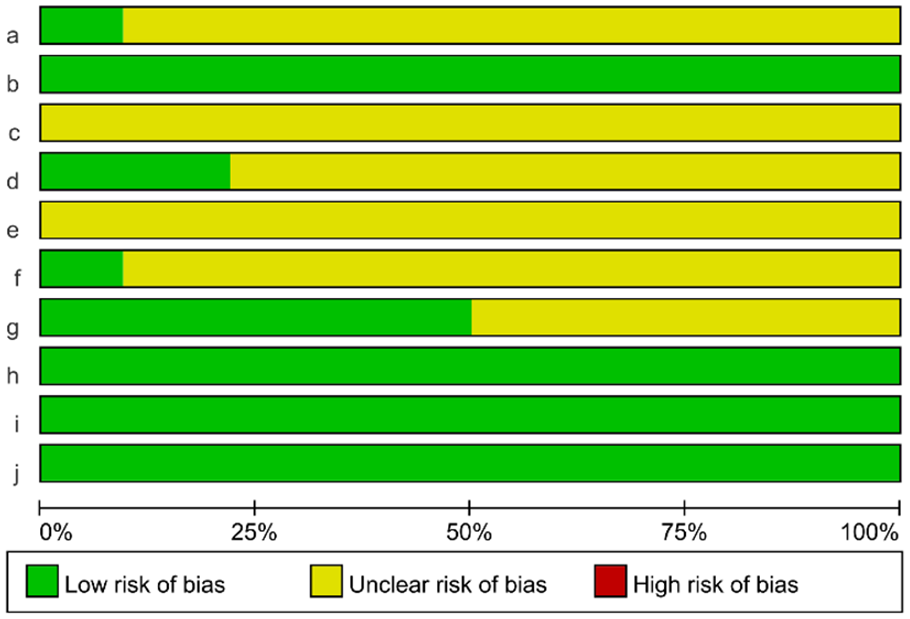
Risk assessment of bias in animal studies.
Standard Meta-Analysis
The acute phase of SCI
A total of 19 articles20,21,24,27,28,31,32,34,35,38 –40,42 –45,47,49,50 were transplanted with MSCs in the acute phase of SCI. Heterogeneity between studies was significant (I2 = 88.1%, P < 0.1) and was analyzed using the random-effects model. Subgroup analysis according to transplantation modality showed that BBB scores were significantly higher in the MSCs group than in the control group (IT: SMD = 3.80, 95% CI = 2.81–4.80, P < 0.05; IV: SMD = 2.27, 95% CI = 1.78–2.77, P < 0.05; IL: SMD = 4.34, 95% CI = 2.87–5.80, P < 0.05) (Fig. 3).
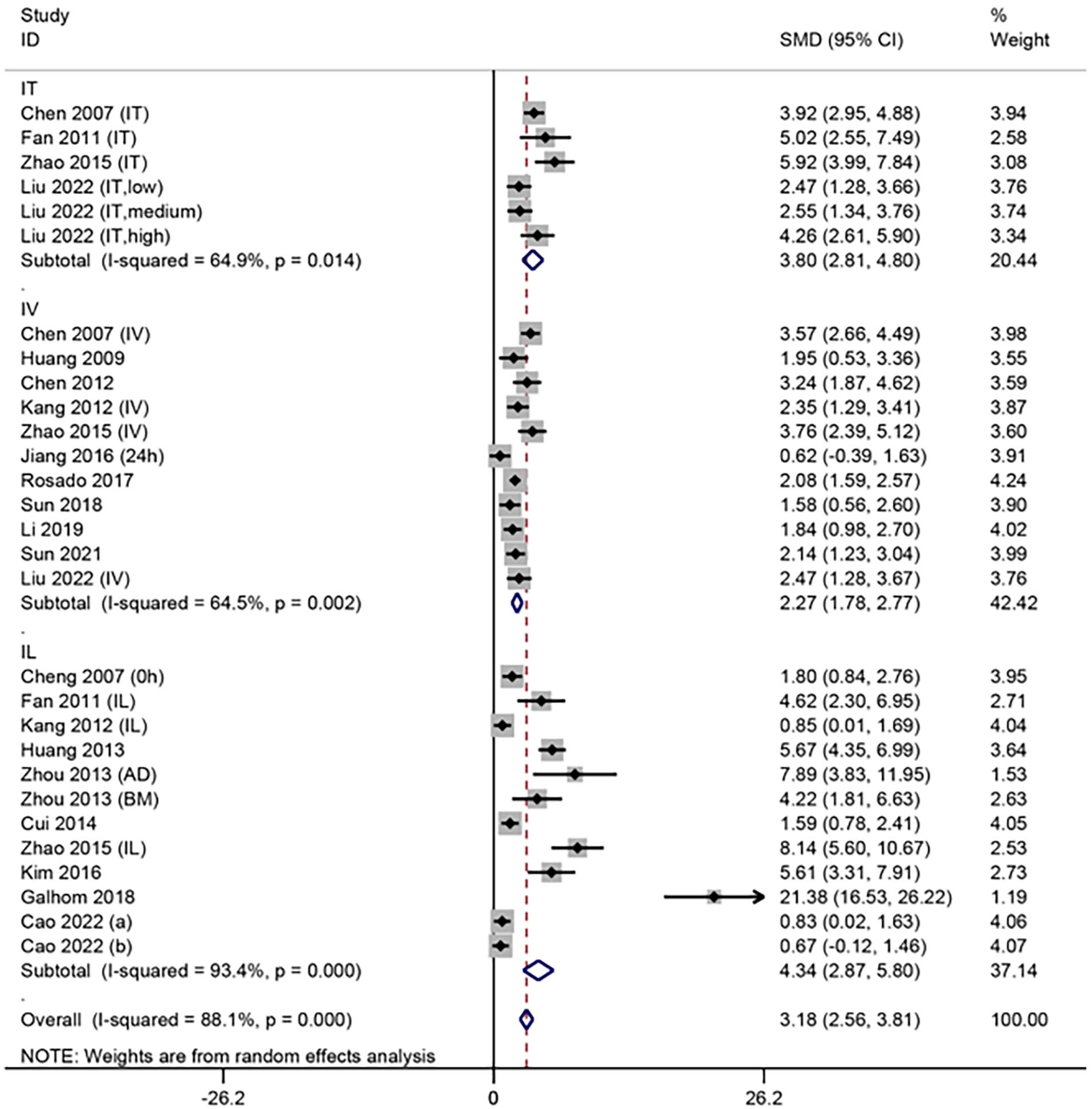
Meta-analysis of MSCs transplantation in the acute phase of SCI.
The subacute phase of SCI
A total of 13 articles19,21 –23,25,26,29,30,36,37,39,41,46 were transplanted with MSCs at the subacute phase of SCI, with significant heterogeneity among studies (I2 = 86.8%, P < 0.1). The random-effects model was used for analysis. Subgroup analysis according to transplantation modality showed that BBB scores were significantly higher in the MSCs group than in the control group (IT: SMD = 6.02, 95% CI = 2.70–9.33, P < 0.05; IV: SMD = 3.96, 95% CI = 2.83–5.10, P < 0.05; IL: SMD = 4.96, 95% CI = 2.51–7.40, P < 0.05) (Fig. 4).
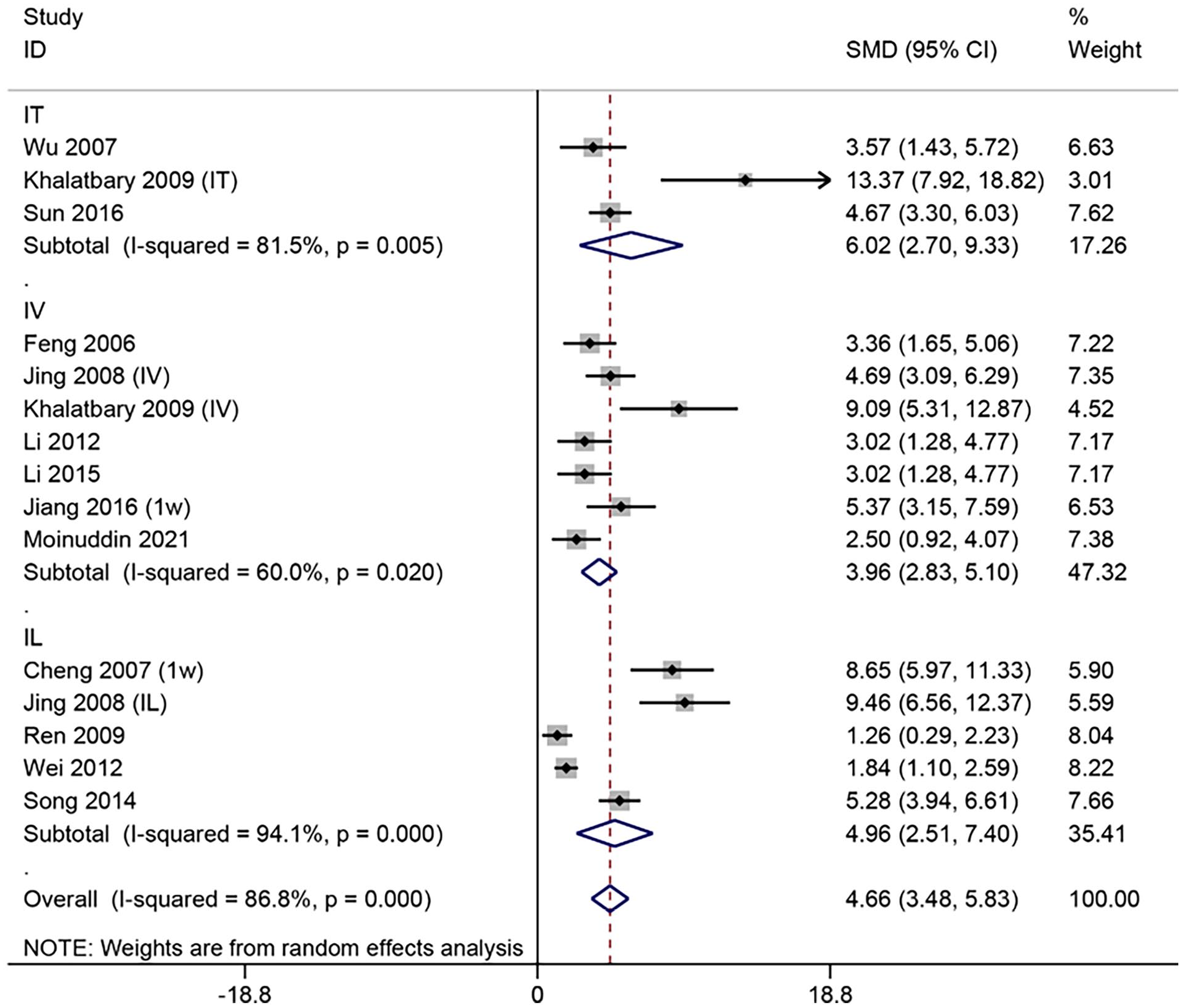
Meta-analysis of MSCs transplantation in the subacute phase of SCI.
The chronic phase of SCI
Two articles33,48 were transplanted in the chronic phase of SCI. Heterogeneity between studies was small (I2 = 0%, P > 0.1) and was analyzed using the fixed-effects model. Subgroup analysis according to transplantation modality showed that BBB scores were significantly higher in the MSCs group than in the control group (IV: SMD = 3.47, 95% CI = 1.86–5.09, P < 0.05; IL: SMD = 4.42, 95% CI = 3.01–5.83, P < 0.05) (Fig. 5).
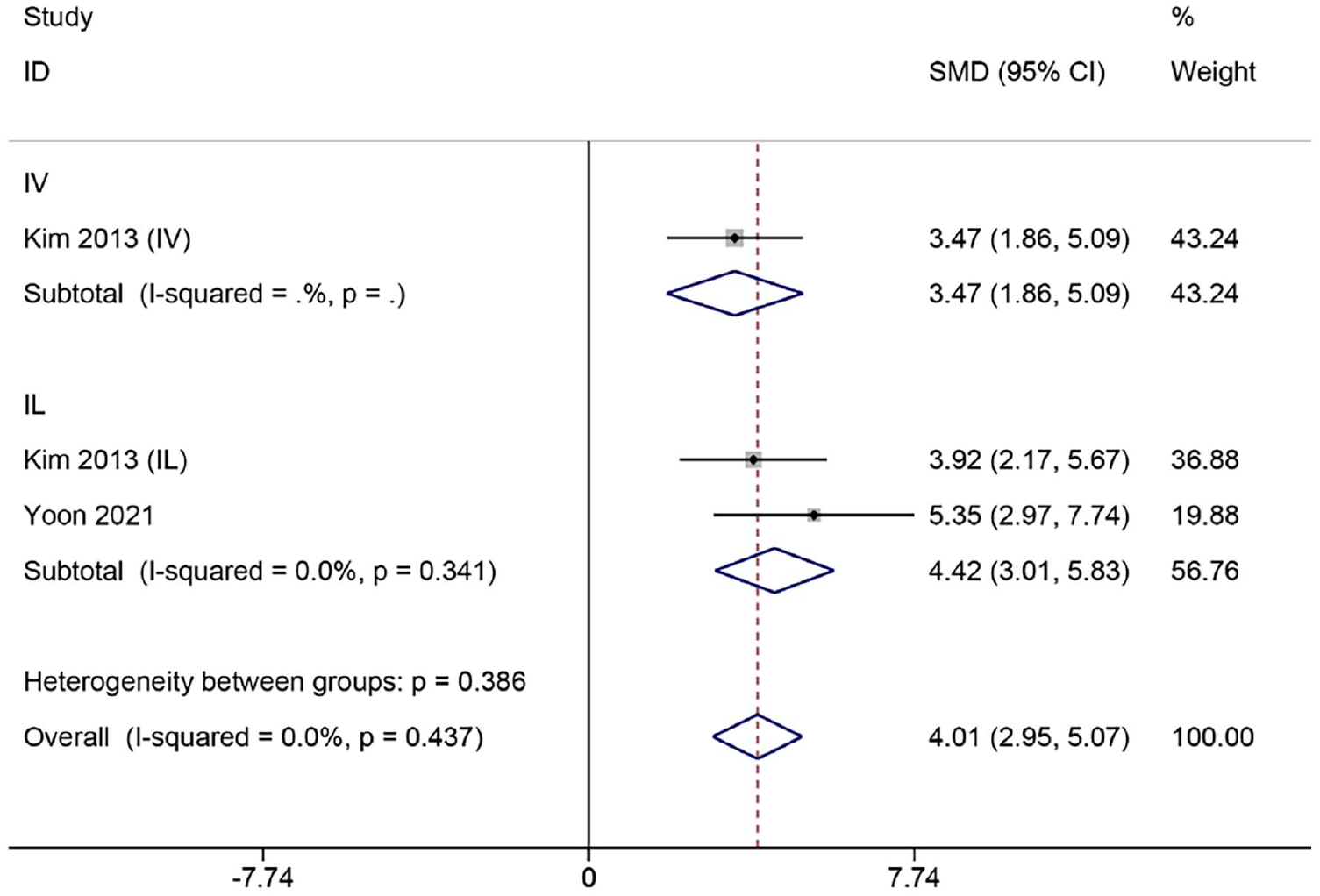
Meta-analysis of MSCs transplantation in the chronic phase of SCI.
Network Meta-Analysis
Network meta-analysis was performed according to different cell sources, transplantation ways, and times using the BBB score as an outcome indicator. The network relationship diagram was sequential (Fig. 6A–C). Local inconsistency tests were performed separately for the closed loops in Fig. 6A–C. The results showed P > 0.05, so the consistency model was used. The SUCRA ranking of different cell sources was AD > BM > UC > CG (83.0% > 78.0% > 39.0% > 0.0%, Fig. 6D). The ranking of SUCRA by different transplantation ways was IT > IL > IV > CG (88.3% > 76.6% > 35.1% > 0.0%, Fig. 6E). The ranking of SUCRA by different transplantation times was subacute > chronic > acute > CG (91.6% > 65.3% > 42.8% > 0.3%, Fig. 6F). The imperfectly symmetrical comparison-corrected funnel plots suggest a possible publication bias and a small sample effect in the above results (Fig. 6G–I).
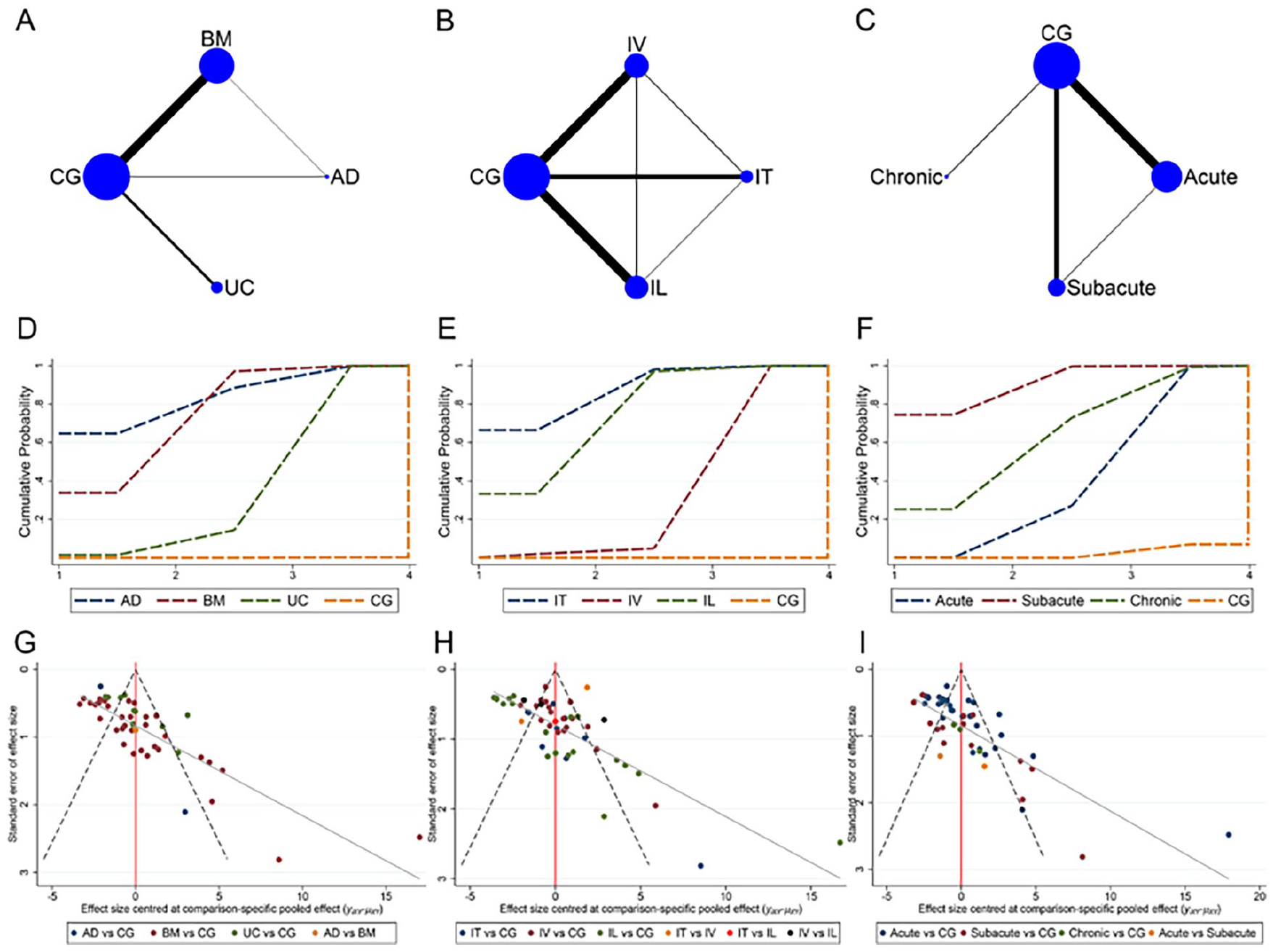
Network meta-analysis of animal studies. A: Network relationship of different cell sources; (B) Network relationship of different transplantation ways; C: Network relationship of different transplantation times; D: SUCRA of different cells sources; (E) SUCRA of different transplantation ways; F: SUCRA of different transplantation times; G: Funnel plot of different cell sources; H: Funnel plot of different transplantation ways; I: Funnel plot of different transplantation times.
Discussion
The standard meta-analysis was performed on 32 animal studies in this study. The results showed that treatment with MSCs was effective in improving motor function after SCI compared with controls. A comprehensive comparison of different MSCs cell sources, transplantation modalities, and transplantation times in animal studies was performed by network meta-analysis using the BBB score as an outcome index. The results showed that ADMSCs were best treated in terms of cell source, IT was best treated in terms of transplantation modality, and transplantation was best treated in terms of transplantation timing when performed in the subacute phase. While ADMSCs demonstrated sound therapeutic effects in animal studies, clinical studies related to ADMSCs are currently less available, and the clinical efficacy in SCI patients needs to be further explored.
ADMSCs can be obtained from fat aspirates obtained by liposuction, are widely available, enable autologous transplantation, reduce immune rejection caused by allografts, have a high ability to differentiate into neurons, and have a few ethical issues 51 . An animal study showed that ADMSCs can reduce Janus family of cytoplasmic tyrosine kinase (JAK)/signal transducer and activator of transcription 3 (STAT3) phosphorylation in astrocytes by inhibiting the Jagged1/Notch signaling pathway to treat SCI rats, which may be a potential mechanism for their improved motor function 52 . UCMSCs can be obtained from waste tissue such as umbilical cord blood and perivascular and subendothelial umbilical veins, and there are no medical ethical issues 53 . UCMSCs are mostly allogeneic and have been gradually applied in basic and clinical research because of their easy access, high value-added rate, low immunogenicity, and non-tumorigenicity 54 . It was shown that pro-inflammatory factors interleukin (IL)-1β, interferon-gamma (IFN-γ), IL-6, and tumor necrosis factor (TNF)-α were elevated in the serum of SCI rats, and there was a tendency to reduce the elevation of IFN-γ, IL-6, and TNF-α after treatment with UCMSCs 50 . BMSCs are stem cells in the bone marrow that have the potential for multidirectional differentiation and contribute to hematopoiesis and bone regeneration 55 . Li et al. 45 found that TLR4 expression was significantly reduced in SCI rats after BMSCs transplantation, suggesting that BMSCs transplantation could significantly inhibit TLR4 expression after SCI, thus reducing the inflammatory response. BMSCs are easy to obtain by autologous isolation and have less immune rejection in SCI patients, so they are primarily used in clinical studies for cell transplantation 56 .
There are some limitations to this study: (a) the included clinical literature was small, and the sample size of individual literature was small; (b) the language type of the included literature includes only English and Chinese, which may have regional and ethnic limitations and is not comprehensive; (c) most of the studies were not allocation concealed, which may produce bias; and (d) the bias analyses both suggested that there may be a small difference and bias.
Conclusion
In conclusion, the currently available evidence in the literature suggests that IT transplantation using ADMSCs in the subacute phase in SCI may be the best transplantation strategy to improve functional impairment after SCI. MSCs transplantation using this strategy could be performed in SCI patients in clinical trials to verify the validity of the findings. Considering the limitations of this study, more large sample sizes, and multicenter, high-quality animal studies are needed in the future to further validate the results of this study to ensure the reliability of the findings. The biological therapeutic mechanisms of MSCs for SCI should also be explored in depth.
Supplemental Material
sj-docx-1-cll-10.1177_09636897241262992 – Supplemental material for Mesenchymal Stem Cells for the Treatment of Spinal Cord Injury in Rat Models: A Systematic Review and Network Meta-Analysis
Supplemental material, sj-docx-1-cll-10.1177_09636897241262992 for Mesenchymal Stem Cells for the Treatment of Spinal Cord Injury in Rat Models: A Systematic Review and Network Meta-Analysis by Yueying Wang, Yi Ding and Chenchen Guo in Cell Transplantation
Footnotes
Author Contributions
Y.W. conceived the theme of the study. Y.W. and C.G. performed the systematic search, reviewed the literature, and extracted the data. Y.W. and Y.D. analyzed data and wrote the first draft of the paper. Y.W. and C.G. checked and modified the manuscript. All authors read and approved the final manuscript.
Availability of Data and Material
The data used to support the findings of this study are available from the corresponding author upon request.
Ethical Approval
Ethical approval is not applicable to this study.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Natural Science Foundation of China (82374556) and the Shandong Traditional Chinese Medicine Science and Technology Development Programs (Q-2023067 and Q-2023069).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
