Abstract
Interruption of spinal cord (SC) continuity leads to functional loss below the lesion level. The purpose of this study was to evaluate the safety and efficacy of bone marrow nucleated cell (BMNC) and multiple mesenchymal stem cell (MSC) transplantations in spinal cord injury (SCI). A patient with total SC interruption at the Th2-3 level underwent experimental therapy with BMNC and MSC transplantations followed with intensive neurorehabilitation treatment. At admission, 6 h after SCI, the patient was scored ASIA A, had a Th1 sensation level, paraplegia with sphincter palsy, and was without the ability to control trunk movement. Neurophysiology examination showed bilateral axonal damage to the motor and sensory neural fibers with no motor unit potentials or peripheral motor nerve conduction in the lower extremities. The standard therapy had been applied and had not produced any positive results. The patient was treated with autologous BMNCs injected intravenously (3.2 × 109) and intrathecally (0.5 × 109) 10 weeks after the SCI and with five rounds of MSCs every 3-4 months (1.3-3.65 × 107) administered via lumbar puncture. Total number of transplanted MSC cells during the course of treatment was 1.54 × 108. There were no complications related to transplantations and no side effects related to the therapy during 2 years of treatment. The ASIA score improved from A to C/D (from 112 to 231 points). The sensation level expanded from Th1 to L3-4, and the patient's ability to control the body trunk was fully restored. Bladder filling sensation, bladder control, and anal sensation were also restored. Muscle strength in the left lower extremities improved from plegia to deep paresis (1 on the Lovett scale). The patient's ability to move lower extremities against gravity supported by the movements in quadriceps was restored. The patient gained the ability to stand in a standing frame and was able to walk with the support of hip and knee ortheses. Magnetic resonance imaging (MRI) revealed that at the Th2/Th3 level, where the hemorrhagic necrosis was initially observed, small tissue structures appeared. Our results suggest that repeated intrathecal infusions of MSCs might have the potential to produce clinically meaningful improvements for SCI patients.
Introduction
Spinal cord injury (SCI) results in an irreversible loss of neuronal junctions leading to SC dysfunction, profound disability that significantly impacts the quality of life, and even death (18, 48). Every year, approximately 130,000 people worldwide, predominantly young males (82%), sustain new SCIs. Progress in emergency medicine, nursing care, and long-term care of SCI patients have resulted in increased survival. However, survival decreases with age, and it is dependent on the injury level (15).
Standard management after a SCI includes stabilization of the injured region and subsequent long-term conservative rehabilitation treatment (67). Patients with SCI develop several long-term complications, such as pneumonia, atelectasis, deep venous thrombosis (DVT), pressure ulcers, fracture spasticity (up to 70%), and neuropathic pain (40-50%) (38, 44, 54). The complications are a frequent cause of morbidity and mortality and lead to an increased rate of rehospitalizations (38). Thus, modern conservative treatment involves prevention of post-traumatic complications, analgesic treatment, as well as rehabilitation. Nevertheless, we still lack standard treatment methods to achieve regeneration of sensory and motor neurons, which would enable total or partial functional recovery of the patient.
Experimental approaches of SCI treatment using cellular delivery of growth factors demonstrated that IGF-1 supports corticospinal axon survival (23); NT-3 causes partial functional recovery (20); and placement of molecular, cellular, or “synthetic ” bridges in the lesion cavity supports axonal regeneration across the lesion site (45). Sciatic nerve conditioning lesion resulted in regeneration of the injured SC (43), and stimulating cAMP signaling increased the intrinsic growth capacity of injured sensory axons (42) overcoming myelin inhibition (52).
Sensory and functional recovery was also demonstrated using the strategy of glial scar inhibition using chondroitinase ABC (ChABC) (7). Neutralization of an important neurite outgrowth inhibitor, Nogo-A was reported to stimulate axonal sprouting (17).
Bone marrow (BM) was demonstrated to be a source of different stem and progenitor cells, including hematopoietic, endothelial, epithelial (13, 32), and mesenchymal (6), and also to contain neural tissue-committed stem cells, which can be mobilized into a peripheral blood during injury, such as cerebral stroke (30). Moreover, BM cells were shown to produce numerous trophic growth factors (36). Intravenous administration of bone marrow CD34+ cells was demonstrated to promote neovascularization of ischemic brain, enabling neuronal regeneration (66). Intracerebral CD34+ and/or CD133+ cell transplantations improved neurological functions in animal models (61). Bone marrow MSCs (BMMSCs) administered intracerebrally (70) or intracaratoidally (60) into ischemic rats' brains caused significant improvement of the functional performance. Remyelination of the SC axons following intravenous delivery of BM cells (31, 58), and functional recovery after intraspinal administration of BM stromal cells was also demonstrated (12).
Since 2005, numerous clinical trials with over 400 patients evaluated safety and efficacy of autologous BM cell transplantations alone or in combination with GM-CSF for acute (4, 14, 18, 48, 63, 69), subacute (63, 69), and chronic (8, 10, 18, 33, 63, 69) adult SCI. Safety and feasibility of single intravenous and intraspinal single implantations were demonstrated by all groups with only minor adverse events in some patients, like fever, increased leukocytosis, skin rash, dizziness, itching, headache, rigidity, or abdominal discomfort (33, 48, 69). These studies provide evidence of the possibility of neurological, sensory, and functional improvement, better quality of life, and partial restoration of the neural connections in acute, subacute, and chronic SC patients. However, more significant improvements were seen in acute patients, and the time interval from the injury to cell therapy negatively correlated with the outcome (18, 63). Three reports demonstrated the recovery in chronic patients transplanted with BM cells with intrathecal administration (10, 18, 33), while one study revealed no change (69). Regardless of the time from injury, intramedullary rather than intravenous administration was more likely to induce neurological improvement (63). Cell number and dosing seemed to influence the appearance and the degree of the improvement (4). Application of a single implantation of BMMSCs caused moderate improvement in acute (57), subacute, and chronic SCI patients (47, 49) although considerable improvement in subacute patients was also reported (29). Allogeneic umbilical cord MSCs were demonstrated to cause moderate recovery in incomplete SCI (35) and cord blood MSCs in complete chronic SCI (28). Considerable improvement was reported in incomplete SCI after multiple transplantations of cord blood CD34+ in combination with umbilical cord-derived MSCs (25). No improvement and some side effects were also reported after multiple MSC transplantations in chronic patients (30).
Single use of olfactory ensheathing cells (OECs) demonstrated moderate (24) and multiple use of OEC considerable improvement (53) in chronic complete SCI patients. Transplantation of olfactory mucosa in complete chronic SCI patients caused modest (11) or considerable improvement in some patients (34). Recently, olfactory bulb cells transplanted intrathecally in multiple sites were reported to bring considerable recovery in a chronic patient (65). Administration of Schwann cells was demonstrated to be safe and cause moderate patient improvement (56, 71). Similarly, correction in sensation was seen in some patients after single administration of a combination of MSCs and Schwann cells (68). Considerable improvement for chronic patients was obtained using combination of MSCs with patient's autoimmune T-cells (39). The use of neuronal stem/progenitor cells was also shown to be very promising in brain regeneration (21, 59).
Based on our previous experience of multiple bone marrow nucleated cell (BMNC) transplantations (27) and building suggestions and reports from animal and human studies that MSCs have potential to support regeneration after SCI, we set up this study to assess the safety and efficacy of combined intravenous/intrathecal BMNCs and multiple lumbar puncture MSC administrations to treat subacute complete SCI.
Materials and Methods
Patient
We present a case report of a 15-year-old girl with complete SCI. The patient had been involved in a moving vehicle accident (MVA) resulting in a vertebral column fracture at the Th2-Th3 level. The patient did not receive methylprednisolone treatment in the first 8 h after MVA. Standard therapy, including anti-inflammatory therapy, stabilization of the destroyed level, and neurorehabilitation (including kinezy therapy; manual therapy: stretching, massage, hands-on strengthening exercises; ESTIM: functional electrical stimulation of lower limb muscle groups and use of Lokomat Gait Orthosis; 5 days a week, 6 h per day), which started 21 days after injury, did not produce any improvement. However, the patient did not develop any signs of autonomic dysreflexia.
At admission, 3 months after SCI, the patient was paraplegic (0 on the Lovett scale) with the Th1 sensory level. She was able to breathe independently and eat using both hands but was unable to control the body trunk. The patient couldn't control her bladder and bowels.
The study was performed at University Children's Hospital (UCH) of Cracow, Poland, with the approval of the Jagiellonian University Medical College Bioethical Committee (KBET/31/L/2012). The girl's parents, as well as the girl herself, provided written informed consent.
Bone Marrow Aspiration
Autologous BM was harvested from the posterior superior iliac crest under general anesthesia using aspiration from multiple punctures of the iliac ala. BM was collected in a medium consisting of 0.9% NaCl (B. Braun, Melsungen, Germany), heparin (Polfa Warsaw, Warsaw, Poland), and gentamicin (PAA Laboratories, GmbH, Goetzis, Austria).
Bone Marrow Nuclear Cell Isolation, Evaluation, and Preparation for Transplantation
Isolation of BMNCs from the BM suspension was performed in the Department of Transplantation of Jagiellonian University Medical College using 10% hydroxyethyl starch solution (HES) (B. Braun). HES had been added to BM until final 3% concentration was reached. Afterward, the obtained solution was mixed and left in a sterile bottle for 40-50 min for distribution. After all erythrocytes had sedimented at the bottom of the bottle, upper cell suspension was collected to 50-ml tubes and centrifuged. The supernatant was discarded and the cell suspension was washed with phosphate-buffered saline (PBS) solution (PAA Laboratories) and suspended in PBS. Cell number was evaluated using a hematocytometer, and cells were divided into two parts.
Viability of the cells was checked in trypan blue exclusion assay (0.4% solution; Sigma Chemicals, Seelze, Germany). Percentage of CD34+ and CD133+ cells was assessed according to ISHAGE protocol (19) using 20 μl of monoclonal anti-CD34-PE and anti-CD133-PE antibodies per 100 μl of the cell suspension and cytofluorymetric evaluation on a FACSCalibur (Becton Dickinson Immunocytometry Systems, San Jose, CA, USA). Sterility of the material was confirmed via microbiological evaluation.
The first part of BMNCs, comprising 0.5 × 109 cells, suspended in 5 ml of 5% glucose solution (FreseniusKabi, Kutno, Poland) was injected intrathecally. The second part, comprising the rest of the cells, was prepared in 30 ml of NaCl (B. Braun) for intravenous injection.
Isolation and Culture of the Mesenchymal Stem Cells
MSCs were isolated and cultured according to the protocol established by our group previously (26) with a slight improvement. Mononuclear cells (MNCs) were isolated from the BM sample using density-gradient centrifugation. The CD271+ population was isolated according to the manufacturer's instructions (Stem Cell Technologies, Vancouver, Canada). MNCs were incubated with antihuman CD32 blocker (25 μl per 1 ml of cells) and anti-CD271 monoclonal antibodies (50 μl per 1 ml of cells) labeling the LNGF receptor (or p75 NTR) (Stem Cell Technologies) for 15 min at room temperature; then nanoparticles (50 μl per 1 ml of cells) were added and incubated for another 15 min. Isolation was performed using a magnet. Isolated cells were plated into 75-cm2 tissue culture flasks (Sarstedt Stare Babice, Poland) in Dulbecco's modified Eagle's medium (StemCell Technologies) supplemented with 10% fetal bovine serum (FBS) (Stem Cell Technologies), platelet-derived growth factor (PDGF) (PeproTech Inc., Rocky Hill, NJ, USA), and epidermal growth factor (EGF) (PeproTech). Flasks were incubated at 37°C in a humidified atmosphere containing 5% CO2, and after 7 days, the medium was changed. At confluence, the adherent cells were detached using 0.25% trypsin (PAA Laboratories) and reseeded at 7.5 × 104 cells/75 cm2 flask and incubated again until confluence with a weekly medium change.
Phenotypic Evaluation of Cultured Mesenchymal Stem Cells
The MSC phenotype was analyzed with 20 μl of antibodies specific for MSCs (CD73, CD90, and CD105) and hematopoietic cells (CD45) (all antibodies from BD Biosciences, San Jose, CA, USA) per 100 μl of cell suspension, using a FACSCalibur cytometer with Cellquest software (BD Biosciences). Expanded MSCs were used for transplantation only if they were ≥95% positive for CD73, CD90, and CD105, and <3% positive for CD45.
Evaluation of the Cytogenetic Stability of Cultured Mesenchymal Stem Cells
Cytogenetic stability of cultured MSCs was confirmed via GTG banding. Metaphases were analyzed under an Olympus BX51 microscope with a camera (Olympus Corporation, Tokyo, Japan) to document photomicrographs. A Cytovision program (Leica Microsystem, Inc., Buffalo Grove, IL, USA) was used to arrange chromosomes into a karyogram.
Surgical Procedure and Cell Transplantation Procedure
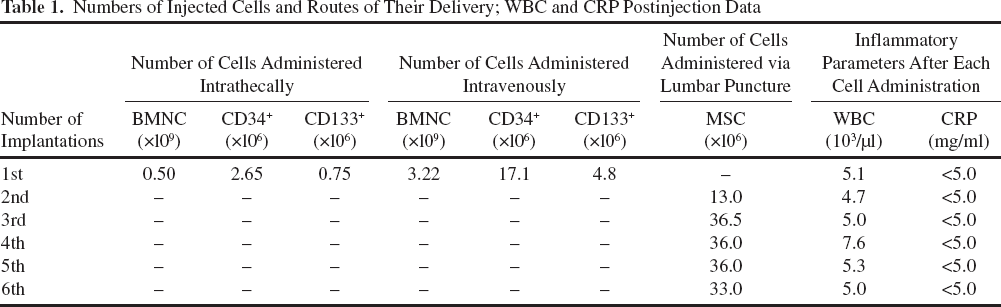
The surgical procedure was performed by the team of specialists from the Department of Pediatric Neurosurgery, University Children's Hospital of Cracow. During the surgical procedure, the SC was exposed using a laminectomy, the injury was verified under a microscope, and the necrotic tissue was resected. BMNCs (0.5 × 109) were administered to the SCI cavity and intravenously (3.22 × 109) via cubital vein over 10 min using a drip infusion set (Table 1). The wound was closed in a typical manner, the meninx was closed with a watertight suture, and the resected lamella of the vertebral arch was restored. The patient was subsequently transplanted with five rounds of cultured MSCs (dose range: 13.0-36.5 × 106) every 3-1 months via lumbar puncture (LP) into cerebral-spinal fluid (CSF) (Table 1). The patient underwent intensive neurorehabilitation after the BMNC transplantations.
Numbers of Injected Cells and Routes of Their Delivery; WBC and CRP Postinjection Data
Pretreatment and Follow-up Efficacy Assessment
Pretreatment and follow-up neurological examinations were performed according to the guidelines for the conduct of clinical trials for SCI developed by the ICCP panel (62). They included American Spinal Injury Association (ASIA) impairment scale and the Frankel grading system; sensory deficits were evaluated using dermatome sensory maps. Posttraumatic spasticity was assessed using the Ashworth scale, and bladder function evaluation was based on the patient's reports. Muscle strength was evaluated based on the Lovett scale (0-6 terms). Quality of live evaluation was assessed based on the functional recovery estimated using the SCIM (Spinal Cord Independence Measure) method. When the first improvement appeared, additional objective tests were included in the evaluation. EMG and ENG (Neurowerk Sigma MedizinTechnik, Gelenau, Germany) were used to test peripheral conductivity, and the motor-evoked potentials (EP) method was used to test the conductivity from the cerebral cortex and spine to the motor units and back. MRI (Siemens Healthcare Poland, Warszawa, Poland) was used to assess the structural changes in the SC.
Safety Evaluation Criteria
The safety criteria of the transplantation procedure included the following: infection, fever, headache, pain, increased level of C-reactive protein (CRP), increased leukocytosis, allergic reaction (presenting as shock), and perioperative complications (anesthesia and analgesia complications, infections of the wound). The safety criteria of the therapy included cancer development, deterioration of the neurological status, and development of neuropathic pain, secondary infections, urinary tract infections, and pressure ulcers.
Results
MSCs used for transplantations had fibroblast-like morphology (Fig. 1A) and expressed key MSC markers, according to the International Society for Cellular Therapy (16), CD73, CD90, and CD105 (Fig. 1B). They did not express hematopoietic marker CD45 (Fig. 1B). The cytogenetic analysis did not show any chromosomal aberrations (Fig. 1C).

Characteristics and quality control of transplanted MSC cells. (A) Phase-contrast morphology. (B) Surface markers. (C) Karyotype.
There were neither early adverse events like infection, fever, pain, headache, increased level of CRP, increased leukocytosis, allergic reaction shock. nor other perioperative complications that could be related to the transplantation procedures (Tables 1 and 2). We did not observe any late adverse events related to therapy like aggravation of neurological status or cancer development observed during 2 years of treatment (Table 2).
Summary of Early and Late Complications Over 2-Year Follow-Up

Throughout the treatment, the patient demonstrated continuous improvement (Tables 3-5). Among the first signs of improvement, occurring 2-3 weeks after the first MSC administration, was a decrease in sensation level to Th5-6 and partial restoration of bladder sensation. Hip muscle strength slightly increased (1 to 1+), and quality of life increased to 50 on the SCIM scale. In addition, the patient gained the ability to control her trunk. The next improvement, which occurred after the second MSC administration, included an additional sensation level decrease to Th8, the ability to stand in a standing frame, further hip muscle strength increased (1+ to 3), and a quality of life improvement to 54. The ASIA score advanced from A to B/C. After the third MSC implantation, the sensation level decreased to L1, bladder control was partly restored, further hip muscle strength increased (2 to 3), and the quality of life increased to 61. The ASIA score changed to C, and the plegia improved to deep paresis. The patient gained the ability to walk supported by a hip orthesis. The fourth MSC administration produced a further sensation improvement to L2/3, bladder control was fully restored, and anal sensation developed. The patient's quality of life increased to 67. The fifth MSC administration resulted in further sensation improvement to L3/4 and a further hip muscle strength increase. The patient gained the ability to walk supported by a knee orthesis. The changes in ASIA, Frankel, sensation level, functional improvement, and conductivity are summarized in Table 3. The muscle strength improvement results are presented in Table 4, and life quality improvement changes are summarized in Table 5.
Change in ASIA, Frankel, Sensation Level, Functional Improvement, and Conductivity

Evaluation was performed 3 months after each implantation. nd, not done.
Changes in Muscle Strength After MSC Implantations
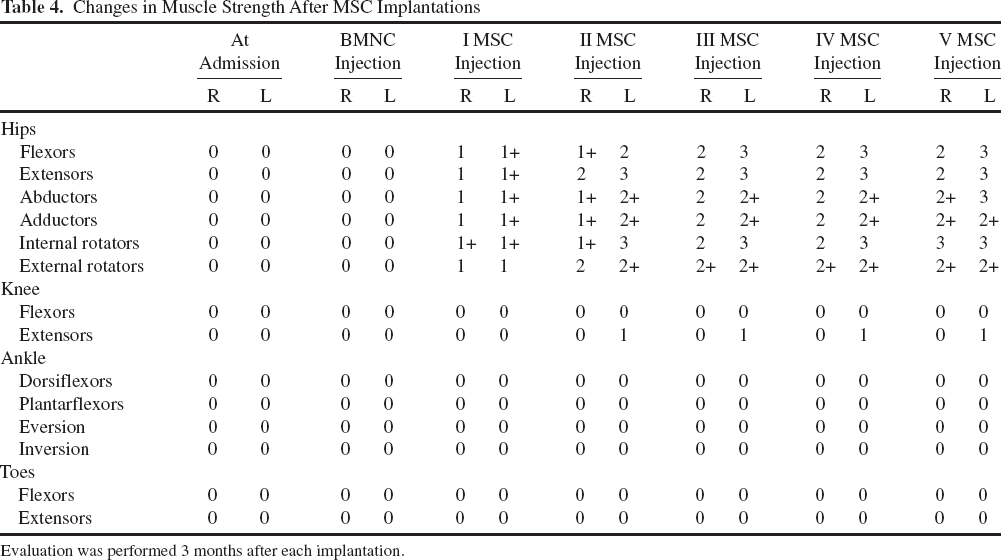
Evaluation was performed 3 months after each implantation.
Quality of Life Improvement Evaluated Using the SCIM Scale
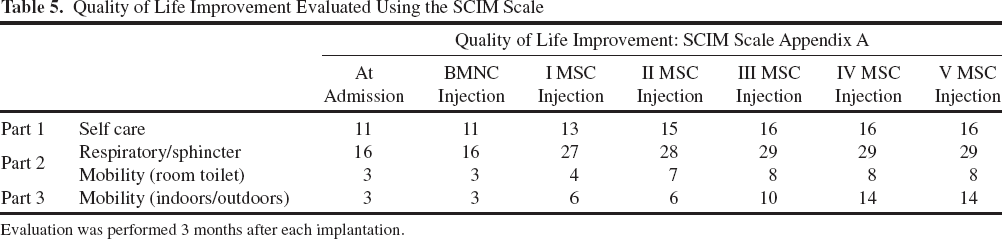
Evaluation was performed 3 months after each implantation.
MRI revealed that in the place of destroyed SC structures at level Th2/Th3, where the hemorrhagic necrosis was initially observed, small tissue structures appeared, which may represent a zone of regenerated neural tissue (Fig. 2). Additionally, near-normal appearance of neural tissue surrounding the degeneration zone at Th3/Th4 was detected by MRI (Fig. 2). The MRI signal in the injured area of the spine improved. EP evaluation of the superficial sensations demonstrated restoration of normal potentials down to Th10 and restoration of normal abdominal muscle activity, improved activity of the lower extremities, and restoration of normal conductivity of the tibialis anterior and peroneus longus nerves. Efferent conductivity from the cortex to the abdominal and lower extremity muscles was restored. EMG, ENG, and EP evaluations objectively confirmed trend of improvement.

MRI evaluation of the SC structure. (A) At admission (coordinates ET: 21, TR: 3128, TE: 60, DFOV: 26 × 26cm, W: 2348, L: 1350). (B) After 2 years of treatment (coordinates ET: 27, TR: 2980, TE: 112.8, DOV: 36 × 36cm, W: 2582, L: 1290).
Discussion
In the past two decades, reports have demonstrated the safety and potential efficacy of cell therapy using cells from various adult tissues for SCI adult patients, including BM cells (4, 8, 10, 14, 18, 27, 33, 48, 63, 69), MSCs (3, 25, 28, 29, 35, 47, 49, 57), OECs (11, 24), olfactory mucosa (11, 34), olfactory bulb cells (65), Schwann cells (56, 71), or cell combinations (2, 39). More considerable improvements have been observed for acute patients, with multiple transplantations, or when treatment strategies combined different cell types.
Our therapy approach for a 15-year-old girl with complete SCI (verified by MRI and clinical examination) with vertebral column fracture at the level Th2/Th3 included single intravenous administration of BMNCs and multiple intrathecal MSC implantations.
The transplanted BMNCs and MSCs were obtained according to established protocols (26). Quality of MSC preparations was verified according to the International Society for Cellular Therapy (16). Microbiological and cytogenetic safety was always checked. The transplantation regimen was developed according to our previous studies (27). Safety and efficacy evaluation was performed following ASIA scale and the Frankel grading system, Ashworth scale and Lovett scale, SCIM method, EMG, ENG, MEP, SSEP, and MRI (2, 59).
Within 2 years of treatment, the ASIA score improved gradually from A to C/D (from 112 to 231 points), and it reflected the sensation level change from Th1 to L3-4 and the patient's ability to control the body trunk. The patient regained bladder control, and the muscle strength in lower extremities improved from plegia to deep paresis. Most importantly, the patient recovered some of the movement activities in lower parts of the body and gained the ability to stand in a standing frame and was able to walk with the support of hip and knee ortheses. Regeneration of neural tissue was demonstrated in macroscale by the only possible way: MRI evaluation. Although histological confirmation could not be performed, the electrophysiological results and clinical improvement stands for the MRI results.
The safety and feasibility of cell therapies have already been demonstrated through intrathecal and LP implantations in adult patients (8, 10, 11, 14, 18, 24, 25, 28, 29, 33-35, 39, 47-49, 53, 56, 57, 63, 65, 68, 69, 71) and multiple BMNC implantations in pediatric patients (26), with only minor side effects observed in some patients (30, 33, 48, 69). Given that our patient showed neither early nor late complications, our results confirm and expand existing data demonstrating the safety and feasibility of multiple, intrathecal MSC implantations for pediatric SCI patients.
In case of CNS injury, cells can be delivered intravenously or intrathecally. The intravenous delivery is easier and was used in one of the first clinical trials but resulted in inferior improvement compared to intrathecal delivery (63). The intravenous delivery limitation is necessity to migrate through the BBB to get to the CNS and possibility to get entrapped into a pulmonary network and lost in the lungs (9, 37, 46). The intrathecal delivery, technically more difficult, allows introducing cells directly into the cerebral fluid where they exert their therapeutic function and can actively migrate toward the inflammation side, further increasing the chance of positive influence. For the above reasons, most clinical studies conducted so far used intrathecal delivery. We assume that the use of the intrathecal route of delivery in our study likely contributed to an improved overall therapeutic effect.
Most published clinical studies used a single injection of cells either intravenously or intrathecally (4, 8, 10, 11, 14, 24, 28, 33-35, 47-49, 56, 57, 63, 68, 69, 71). Our approach included single BMNC and multiple MSC implantations every 3-4 months. This approach resulted in considerable, continuous neurological and quality of life improvement confirmed by objective MRI, EP, EMG, and ENG assessments. Interestingly, the recovery of bladder sensation and control indicates sequential regeneration of the ortho- and antidromic conductivity of the nerve pathways responsible for bladder innervation. Antidromic regeneration was observed first, and orthodromic regeneration was observed 3 months later. Our data may suggest that the denervation process of the lower extremities was stopped and reversed.
Optimally scheduled multiple injections could provide sustained delivery of pro-neurotrophic factors to the place of injury providing continuous stimulation and regeneration of the damaged tissue. The improvement after using the multiple injection therapy approach seems to be higher than in case of single injections for SCI treatment (18, 29, 39, 53, 65) and also for other diseases (22, 55).
However, contradictory results were also published from animal (3) and human (30) studies showing that multiple injections do not provide additional benefit over single implantation or can even worsen the clinical status of the patients (30). The differences observed might be due to the different cell types, transplantation schedule, and number of cells used in our study versus the other.
The multiple intrathecal injections could also lead to the activation of the immune system (5, 51, 52). However, we did not observe any immunological responses likely due to the use of autologous MSCs, which have well-established immunomodulatory potential (1, 40, 50) (Table 1).
According to our experience (we implanted cells intrathecally to the cerebrospinal fluid at the L4 level, low lumbar spine), there is also a minimal possibility of reinjuring the cord during MSC administration. No change to the inflammatory status of the patient after each injection, as measured by CRP level (Table 1) and lack of early side effects (Table 2), confirms the safety of our approach.
The use of MSCs in cell therapy in pediatric patients has never been reported. The obtained improvement is better than the best improvement that has been previously reported for freshly isolated BM cell-based (14, 18, 33, 48, 69) and MSC-based (6, 10) strategies for adults with complete SCI. Considerable improvement (ASIA A to D) was demonstrated using multiple implantations of cord blood CD34+ cells combined with Wharton's Jelly MSCs, but only for a patient with incomplete injury (25), and no improvement was reported for patients with complete injury (35). Therapeutic outcomes of our strategy have clear advantage over OEC (53, 64), Schwann cell (71), and olfactory mucosa autografts applied in adult patients (31) or recently published olfactory bulb cells (65). It cannot be ruled out that the younger age of the patient has favored better improvement. Some studies actually suggest that a younger age might be beneficial for the cell-based therapy of SCI (41). However, in our previous study when chronic pediatric SCI patients were transplanted with BMNCs, the younger age of the patient did not seem to correspond with better results (27). Thus, to address the age factor in SCI therapy, large clinical studies that directly compare younger and older patients are necessary.
Interestingly, our report, and most others, showed that the most effective treatments for patients with complete SCI are stem cell therapy strategy combined with either intense rehabilitation/neurorehabilitation alone (14, 18, 39, 53, 65) or combined with electrostimulation (29). Additionally, most of these strategies employed multiple implantations or multiple sites of implantation (18, 29, 39, 53, 65).
Thus, available data, including this report, seem to indicate the usefulness of multiple implantations combined with intense rehabilitation/neurorehabilitation to maximize the cell therapy treatment efficacy in SCI patients. We are aware, however, that our case study observations must be confirmed in a larger group of patients. Based on our experience, the future studies should also include more detailed observations of recovery using more sophisticated imaging systems, combined with a cell tracking. Moreover, cell dose escalation, not feasible in a single patient, should also be considered in the future studies. Nevertheless, we believe the multiple implantation approach is worth considering when designing various cell therapy treatment protocols for patients with complete SCI.
Conclusions
We report the safety and feasibility of multiple BMNC and MSC implantations and of the treatment strategy introduced herein in a pediatric patient. Our preliminary results demonstrate the possibility of obtaining considerable, continuous neurological and quality of life improvement in patients with subacute complete SCI. Despite these very promising results, we acknowledge that case reports have to be confirmed on a larger group of patients in randomized clinical trials.
Footnotes
Acknowledgments
This work was partially financed by Jagiellonian University Medical College Grant K/ZDS/004044, by Leading National Research Center (KNOW) scheme supported by the Ministry of Science and Higher Education and by the “Dar Nadziei ” Foundation. The authors declare no conflict of interest.
