Abstract
Spinal cord injury (SCI) is a devastating disease, with a high rate of disability. In this meta-analysis, we aimed to comprehensively assess the efficacy and safety of mesenchymal stem cells (MSCs) in treating clinical SCI patients. We systematically searched the PUBMED, EMBASE, Chinese Biomedical (CBM), Web of Science and Cochrane databases using the strategy of combination of free-text words and MeSH terms. The indicators of the American Spinal Injury Association (ASIA) impairment scale (AIS)-grading improvement rate and adverse effects were displayed with an overall relative risk (RR). For the continuous variables of the ASIA motor score, light-touch score, pinprick score, activities of daily living (ADL) score, and residual urine volume, we used odds ratio (OR) to analyze the data. Eleven studies comprising 499 patients meeting all inclusion and exclusion criteria were included. No serious heterogeneity or publication bias was observed across each study. The results showed that significant improvements of total AIS grade (RR: 3.70; P < 0.001), AIS grade A (RR: 3.57; P < 0.001), ASIA sensory score (OR: 8.63; P < 0.001) and reduction of residual urine volume (OR: −36.37; P = 0.03) were observed in experimental group compared with control group. However, no significant differences of motor score (OR: 1.37, P = 0.19) and ADL score (OR: 2.61, P = 0.27) were observed between experimental and control groups. In addition, there were no serious and permanent adverse effects after cell transplantation. Cell transplantation with MSCs is effective and safe in improving the sensory and bladder functions of SCI patients.
Introduction
Spinal cord injury (SCI) is a devastating disease, with a high rate of disability. Patients with SCI always suffer from paralysis, locomotor and sensory dysfunction, urinary incontinence or gastrointestinal dysfunction 1,2 . The incidence of SCI is 27–83 per million in the US and 10–30 per million in Europe 3,4 , which poses a great burden on society. Therefore, there is an urgency to develop an effective therapy for cure of these patients.
The underlying mechanisms of SCI include direct mechanical damages and secondary injuries. Direct mechanical damages involve compression and contusion from the fractured and dislocated bone fragments and discs around the spinal cord 5 . Secondary injuries include neural apoptosis, spinal cord swelling, inflammatory response, oxidative stress and electrolyte disturbance 6 –8 . Both primary and secondary injuries can cause devastating tissue damage, axonotmesis, demyelination, Wallerian degeneration, syringomyelia, and glial scar formation 9 –11 . Many methods have been explored to treat SCI, including surgery, drugs, and rehabilitation, however, no treatment with good efficacy has been reported.
Recently, stem cell transplantation has attracted attention and is reported to be an effective treatment in treating SCI in animal models 12,13 . A variety of stem cell types have shown their potential for transplantation, such as neural stem cells 14 , mesenchymal stem cells (MSCs) 4 , Schwann cells 15 , embryonic stem cells 16 , and induced pluripotent stem cells 17,18 . Among these, MSCs have played a pivotal role in repairing the damaged spinal cord. MSCs can not only differentiate and replace the damaged cells, but also secrete neuroprotective cytokines, including vascular endothelial growth factor (VEGF), glial-cell-line-derived neurotrophic factor (GDNF) and brain-derived neurotrophic factor (BDNF), which all increase neural regeneration, strengthen axon growth, and revive damaged neurons 19,20 . Currently, the efficacy and safety of cell transplantation with MSCs have been verified in the animal SCI models. However, it is unclear whether MSCs have the same efficacy in treating SCI in humans as in animals. No meta-analysis has extensively evaluated the efficacy and safety of MSCs in treating patients with SCI.
Therefore, we conducted this meta-analysis to comprehensively assess the efficacy and safety of MSCs in treating clinical patients by evaluating outcomes including the American Spinal Injury Association (ASIA) motor score, ASIA sensory score (including light-touch and pinprick scores), ASIA Impairment Scale (AIS)-grading improvement rate, activities of daily living (ADL) score, residual urine volume, and adverse events.
Materials and Methods
Protocol
We conducted this meta-analysis based on the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) 21 (Supplemental Table 1).
Search Strategy
The databases of PUBMED, EMBASE, Chinese Biomedical (CBM), Web of Science and Cochrane were systematically searched for eligible studies (up to 25 July 2018). The search strategy consisted of free-words and MeSH terms as follows: (a) “mesenchymal stromal cells” or “MSCs,” and (b) “spinal cord injury” or “SCI,” (c) patient. In addition, other potential eligible studies were identified manually from references of included studies or other reviews pertaining to this topic.
This selection process of eligible articles was conducted by two authors independently (P Xu and X Yang).
Selection Criteria
Data Extraction and Quality Assessment
We extracted the following data from included studies. (a) Identity: authors, years. (b) Patients included in each study: age, duration of injury, size of each group. (c) Treatments: treatment strategy, transplantation methods, cell sources, cell number, follow-up period. (d) Outcomes: AIS grading, ASIA motor score, ASIA sensory score (including light-touch and pinprick scores), ADL, residual urine volume, incidence of adverse effects.
The data of interest from included articles were extracted and processed by two authors, independently (Xu PF and Yang XL). Any disagreement was settled by discussion.
The risks of bias within the included studies were evaluated with the domain-based Cochrane Collaboration tool 22 . Detailed content of this assessment tool included random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other bias. Any dispute was resolved by discussion.
Types of Outcome Measures
The following indicators were applied to assess the efficacy and safety of treatment with MSCs for SCI patients: AIS grading improvement rate, ASIA motor score, ASIA sensory score (including light-touch and pinprick scores), ADL score, residual urine volume, and adverse effects.
Statistical Analysis
This meta-analysis was completed with Stata 14 and Review Manager version 5.0 software (Cochrane Collaboration, software update, Oxford, UK), which was provided by the Cochrane Collaboration. The indicators of AIS grading improvement rate and adverse effects were displayed with an overall relative risk (RR), with corresponding 95% confidential interval (CI). For the continuous variables of ASIA motor score, light-touch score, pinprick score, ADL score, and residual urine volume, we used odds ratio (OR), with corresponding 95% CI to analyze the data.
We used the chi-squared value test and inconsistency index (I2) to assess the heterogeneity across each study. A value of P < 0.1 or I2 > 50% was deemed to have significant heterogeneity, a random-effect model was then used to analyze the data. Otherwise, the fixed-effect model was used. Subgroup analysis was used to find potential source of heterogeneity 23,24 . We adopted the Egger funnel plot and Egger’s test to test publication bias with Stata14.0 (StataCorp LP, College Station, TX, US) 25,26 , with P < 0.05 indicating significant asymmetry 27 .
Results
Study Screen
Searches of the PUBMED, EMBASE, Chinese Biomedical (CBM), Web of Science and Cochrane databases identified 566, 153, 369, 215 and 19 studies, respectively. After removing duplicated studies, 987 studies remained for screening by title and abstract. Subsequently, 59 records with full text were assessed. Finally, 11 studies 28 –37 containing a total number of 499 cases were included in this meta-analysis (Fig 1).

Flow Diagram of the Study Selection Process.
Study Characteristics
The basic characteristics of all 11 studies are summarized in Table 1. The sample sizes ranged from 20 to 96. Nine studies were conducted in China 28,29,31 –38 , one study was conducted in Iran 30 , and another was performed in Egypt 31 . Four studies were reported in English 28 –31 , and the other seven were in Chinese 32 –38 . All studies chose rehabilitation therapy as the control. The MSCs originated from umbilical cord in five studies 29,32,34,37,38 and bone marrow in six studies 28,30,31,33,35,36 . Six studies adopted subarachnoid injection as the process used for cell transplantation 28 –33,38 ; two studies 35,37 used intravenous injection and the other two studies reported both methods 34,36 . The course of SCI ranged from 1 week to 9 years and the follow-up period ranged from 3 to 23.4 months. For reporting the outcomes, five studies reported AIS grading improvement rate 28,30,31,34,36 , ten studies reported ASIA motor score 28,29,31 –38 , five studies reported ASIA light-touch and pinprick scores 28,31,33,34,38 , seven studies reported ADL score 28,29,32 - 34,36,37 , three studies reported residual urine volume 28,29,36 , and 11 studies reported some mild adverse effects 28 –38 .
Characteristics of Included Studies.

M: male; F: female; R: rehabilitation; SCT: stem cell transplantation; BMMSCs: bone marrow mesenchymal stem cells; UCMSCs: umbilical cord mesenchymal stem cells; ASIA: American Spinal Injury Association; AIS: ASIA impairment scale; ADL: activities of daily living; MCS: motor nerve conduction studies; SEP: sensory-evoked potential; SCV: sensory nerve conduction studies; EMG: electromyography; FAC: functional ambulation categories ; SSEP: somatosensory evoked potential; BI: barthel index; PSSEP: paraspinal somatosensory evoked potential.
Methodological Quality of Included Studies
We used the standard Cochrane Collaboration tool to evaluate the risks of bias within included studies, and the results of methodological quality of each study are shown in Fig 2. Ten studies reported random sequence generation and one study did not mention it. Additionally, three studies had the details of blinding of outcome assessment, one study lacked blinding of outcome assessment and the others did not state the blinding of outcome assessment. Overall, the methodological quality of included studies was acceptable.

Risk of Bias Percentile Chart. (2A) Risk of bias graph: A plot of the distribution of review authors’ judgements across studies for each risk of bias item; (2B) Risk of bias summary: A summary table of review authors’ judgements for each risk of bias item for each study.
Efficacy of Treatment
American Spinal Injury Association Impairment Scale Grading Improvement Rate
Five studies containing 189 cases reported AIS grading improvement rate, low heterogeneity was observed across each trial (P = 0.25, I2 = 26%, Fig 3A). A fixed-effects model was applied to evaluate the AIS improvement rate. The results indicated significant improvements of total AIS grade and AIS grade A in the experiment group compared with control group (total AIS grade: RR: 3.70; 95% CI 2.63–6.25; P < 0.001; AIS grade A: RR: 3.57; 95% CI 2.5–6.67; P < 0.001, respectively; Fig 3A and 3B), however, no significant difference was found in AIS grading B/C/D (RR: 4.46; 95% CI 0.42–47.34; P = 0.22; Fig 3C).

Forest Plot of Comparison: Cell Transplantation Group Versus Control Group.
American Spinal Injury Association Sensory Score
Ten studies containing 468 cases reported ASIA sensory score (including ASIA light-touch and pinprick scores); low heterogeneity was observed among included studies (P = 0.16, I2 = 31%). A fixed-effects model was applied to evaluate the ASIA sensory score. The results indicated significant improvements of general ASIA sensory score, ASIA light-touch score and ASIA pinprick score in the experiment group, compared with the control group (general ASIA sensory score: OR: 8.63; 95% CI 4.84–12.41, P < 0.001; ASIA light-touch score: OR: 4.57; 95% CI 0.74–8.4, P = 0.02; ASIA pinprick score: OR: 4.92; 95% CI 1.59–8.25, P = 0.004; Fig 3D, 3E and 3F).
American Spinal Injury Association Motor Score
Ten studies containing 468 cases reported ASIA motor score; low heterogeneity was observed across each trial (P = 0.43, I2 = 1%). A fixed-effects model was applied to evaluate the ASIA motor score. The results showed that no significant improvements of ASIA motor score were observed in the experiment group compared with the control group (OR: 1.37; 95% CI 0.70–3.44; P = 0.19; Fig 3G).
Activities-of-Daily-Living Score
Seven studies containing 278 cases reported ADL score; moderate heterogeneity was observed (P = 0.02, I2 = 59%). Therefore, the random-effects model was applied to evaluate the ADL score. The results showed that no significant improvements of ADL score were observed in the experiment group compared with the control group (OR: 2.61; 95% CI 2.04–7.26; P = 0.27; Fig 3H).
Residual Urine Volume
Three studies containing 84 cases reported residual urine volume; moderate heterogeneity was observed among included trials (P = 0.02, I2 = 73%). A random-effects model was applied to evaluate the residual urine volume. The results showed that significant reduction of residual urine volume was observed in the experiment group compared with the control group (OR: 36.37; 95% CI 68.45–4.29; P = 0.03; Fig 3I).
Safety
Eleven studies containing 499 cases reported adverse effects; low heterogeneity was observed across each trial (P = 0.11, I2 = 38%). The fixed-effects model was then employed in the RR pooled analyses. The results showed that patients receiving cell transplantation of MSCs experience more toxicity than that of the control group (RR: 20.34; 95% CI 8.09–51.18, P < 0.001; Fig 3J). Common adverse effects include fever, headache, backache, numbness, and abdominal distension, which were alleviated spontaneously or following treatment intervention. However, no serious or permanent adverse effects, such as death, tumor, or immune reaction, were observed during follow up.
Subgroup Analysis
We performed subgroup analysis based on cell sources and follow-up period. For analyzing the effects of cell transplantation on motor function and self-care ability, no significant differences were observed between experiment and control groups with different cell sources and follow-up periods (P > 0.05; Fig 4A–4G). However, regarding the adverse effects of cell transplantation, the experiment group showed a higher rate of adverse effects than that in the control group, no matter which different cell sources or follow-up periods (P < 0.05; Fig 4H–4K).

Forest Plot of Subgroup Analysis Based on Cell Source and Follow-Up Periods.
Publication Bias
The Egger’s funnel plot and rank correlation test showed no significant publication bias across each study regarding sensory score, motor function, and adverse effects (P = 0.387, P = 0.091 and P = 0.07, respectively; Fig 5A–5C). For other indicators, the assessment of publication bias cannot be conducted due to insufficient number of studies (n < 10).
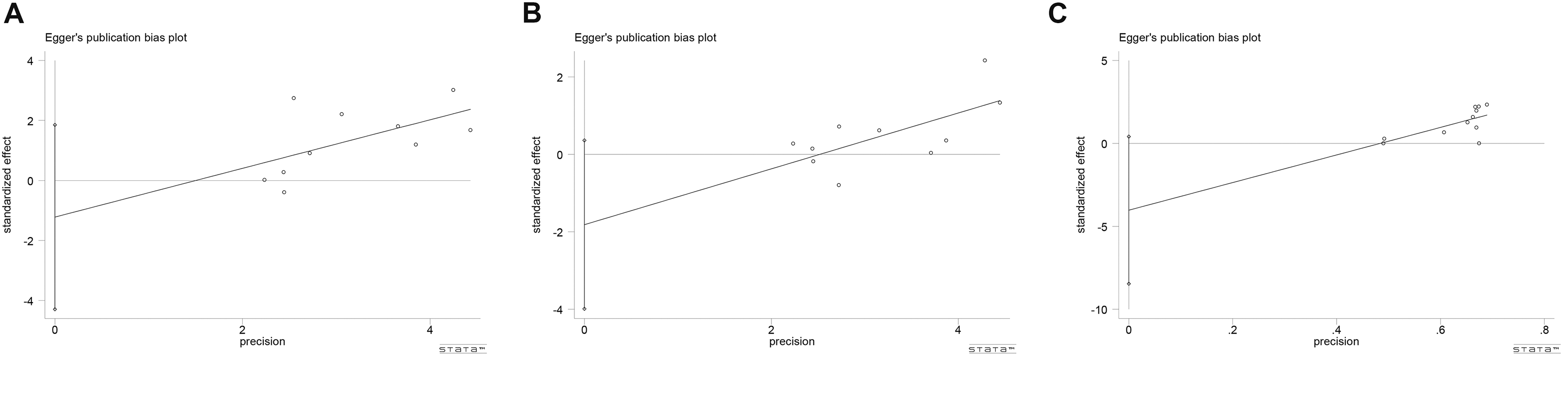
Funnel Plot of Publication Bias.
Discussion
This meta-analysis included 11 studies and comprehensively evaluated the safety and efficacy of MSC transplantation for treating patients with SCI. Although the efficacy of this method for treating SCI patients remains unclear, this study showed that when compared with rehabilitation therapy, MSC transplantation significantly improved the neurological functions, including ASIA light touch, pinprick, ADL, and bladder function. However, this meta-analysis found that no significant difference was observed for the improvement of motor score, and the patients who received MSC transplantation displayed some mild and temporary side effects.
MSCs could be extracted from autologous bone marrow, umbilical cord or adipose tissue 39,40 . In the animal studies, MSCs have displayed many advantages in the treatment of SCI. Some studies showed that MSCs could survive in the site of injury and differentiate into different types of cells (neurons, oligodendrocytes, vascular endothelial and astrocytes) 41 . In addition, MSCs can also secrete neuroprotective cytokines, including VEGF, GDNF, and BDNF, which support neural regeneration, promote axon growth, and activate damaged neurons 19,20 . Furthermore, MSCs are reported to inhibit glial scar formation, alleviate scar obstructions, and activate endogenous neural stem cells 42,43 .
Although some studies demonstrated no advantages with stem cell transplantation in the treatment of SCI 30 , most of the studies included in this meta-analysis indicated that MSC transplantations could significantly improve the sensory functions, including light touch and pinprick. Their results showed that sensory functions were improved in two respects. On the one hand, the improvement was reflected on the downward level of damaged spinal cord with loss of sensation. On the other hand, the sensory functions were improved from insensitivity or weak sensation to strong sensation. In addition, regarding AIS grading, cell transplantation could significantly improve AIS grading in all patients and in patients with AIS grade A before cell transplantation. However, for the patients with AIS grade B or lower before cell transplantation, no advantage was gained from cell transplantation. Furthermore, some studies reported that cell transplantation could significantly improve motor functions 32,33 , but the results of the present study demonstrated that no significant difference was observed between patients receiving cell transplantation and those in the rehabilitation group. Seven studies reported self-care ability. The results of this study showed that no significant improvement of self-care ability was observed in both short-term and long-term follow up after cell transplantation. Regarding bladder function, urine residual volume was significantly reduced after cell transplantation. Cheng et al. also performed urodynamic examination. Their results showed that maximum bladder capacity increases, residual urine volume decreases, maximum detrusor pressure decreases, and maximum urinary flow rate increases 29 .
Although the results showed some adverse effects in the present study, all were only displayed as temporary and light side effects, including fever, headache, backache, numbness, and abdominal distension, primarily caused by spinal puncture. No devastating and long-term adverse effects, such as wound infection, cerebrospinal fluid leakage from incision, or intracranial infection were observed in patients receiving stem cell transplantation, indicating the safety of cell transplantation.
Limitations
Some limitations cannot be ignored in this study. First, only 11 studies were included in this study. Some important subgroup analyses, such as different courses of disease and number of cells given to the patients, could not be performed. Second, we only included the papers published in English and Chinese in this meta-analysis, which may leave out other eligible studies that were reported in other languages. In addition, most of these studies included were conducted in China. There may be some differences of the effects and safety in other populations. Therefore, the results should be interpreted cautiously due to the limited data, although the results of this meta-analysis are robust. Third, no significant publication bias in the overall analysis was observed in this study, but many factors, including stem cell preparation and identification, social economic level, or nursing care could have impacts on the outcomes of stem cell transplantation for SCI. Therefore, multi-centric and large-sample randomized controlled studies with reasonable random sequence generation, adequate allocation concealment, and low risk of reporting bias are required to provide more medical evidence base.
Conclusions
Cell transplantation with MSCs is effective and safe in improving the sensory and bladder functions in SCI patients; however, its effect on motor function is unclear.
Supplemental Material
Supplemental_materials - The Efficacy and Safety of Mesenchymal Stem Cell Transplantation for Spinal Cord Injury Patients: A Meta-Analysis and Systematic Review
Supplemental_materials for The Efficacy and Safety of Mesenchymal Stem Cell Transplantation for Spinal Cord Injury Patients: A Meta-Analysis and Systematic Review by Panfeng Xu, and Xianliang Yang in Cell Transplantation
Footnotes
Ethical Approval
Ethical Approval is not applicable to this study.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
