Abstract
Neural stem cells (NSCs) and mesenchymal stem cells (MSCs) are promising graft materials for cell therapies in spinal cord injury (SCI) models. Previous studies have demonstrated that MSCs can regulate the microenvironment of NSCs and promote their survival rate. Furthermore, several studies indicate that MSCs can reduce stem cell transplantation-linked tumor formation. To our knowledge, no previous studies have determined whether co-transplantation of human umbilical cord mesenchymal stem cells (hUC-MSCs) and human neural stem cells (hNSCs) could improve the outcome in rats with SCI. Therefore, we investigated whether the transplantation of hUC-MSCs combined with hNSCs through an intramedullary injection can improve the outcome of rats with SCI, and explored the underlying mechanisms. In this study, a moderate spinal cord contusion model was established in adult female Wistar rats using an NYU impactor. In total, 108 spinal cord-injured rats were randomly selected and divided into the following five groups: 1) hUC-MSCs group, 2) hNSCs group, 3) hUC-MSCs+hNSCs group, 4) PBS (control) group, and 5) a Sham group. Basso, Beattie and Bresnahan (BBB) behavioral test scores were used to evaluate the motor function of all animals before and after the SCI weekly through the 8th week. Two weeks after transplantation, some rats were sacrificed, immunofluorescence and immunohistochemistry were performed to evaluate the survival and differentiation of the transplanted stem cells, and brain-derived neurotrophic factor (BDNF) was detected by ELISA in the injured spinal cords. At the end of the experiment, we evaluated the remaining myelin sheath and anterior horn neurons in the injured spinal cords using Luxol Fast Blue (LFB) staining. Our results demonstrated that the surviving stem cells in the hUC-MSCs+hNSCs group were significantly increased compared with those in the hUC-MSCs alone and the hNSCs alone groups 2 weeks post-transplantation. Furthermore, the results of the BBB scores and the remaining myelin sheath evaluated via LFB staining in the injured spinal cords demonstrated that the most significantly improved outcome occurred in the hUC-MSCs+hNSCs group. The hUC-MSCs alone and the hNSCs alone groups also had a better outcome compared with that of the PBS-treated group. In conclusion, the present study demonstrates that local intramedullary subacute transplantation of hUC-MSCs, hNSCs, or hUC-MSCs+hNSCs significantly improves the outcome in an in vivo moderate contusion SCI model, and that co-transplantation of hUC-MSCs and hNSCs displayed the best outcome in our experiment.
Keywords
Introduction
Spinal cord injury (SCI) is a devastating injury that can result in permanent motor and sensory function disabilities. However, there presently are no treatments that can cure the neurological deficits. The mechanism underlying acute SCI is a two-step process involving primary and secondary injuries. The post-traumatic environment that leads to secondary damage can be regulated by certain measures, such as stem cell transplantation, drug treatment, and neural tissue engineering approaches. Recent preliminary studies have indicated that cell therapy may be helpful for the effective functional recovery of animals with spinal cord injuries.
Neural stem cells (NSCs) are self-renewing multipotent cells. Because of their ability to differentiate into functional neurons and glial cells after being transplanted into an injured spinal cord, NSCs appear to be a hopeful treatment for spinal cord injuries 1 . Many studies have shown that NSC transplantations can significantly enhance the functional recovery of animals with SCI 2 –6 . Mesenchymal stem cells (MSCs) are multipotent stromal cells with a high capacity for self-renewal and can differentiate into a variety of cell types. MSCs are excellent candidates for cell therapy because they are easily accessible, their isolation is straightforward, they can be bio-preserved with a minimal loss of potency, and they have shown no adverse reactions in allogeneic versus autologous MSC transplants 7 . Many studies have shown that MSCs have a potential ability in the treatment of SCI 8 –13 . Studies have shown that MSCs can regulate the growing microenvironment of NSCs and promote their survival rate 14 . Importantly, MSCs can induce the differentiation of stem cells and reduce stem cell transplantation-linked tumor formation 15 . Although stem cell transplantation therapy has been confirmed as a useful treatment in models of SCI, the outcome is not satisfactory because the functions of the stem cells are limited by the molecular environment in the injured spinal cord tissue, and their survival rate is very low.
Since multiple factors determine the progression of SCI, a combined therapeutic approach is required to more effectively treat SCIs 16 . Thus, we performed an experiment in which SCI animals were treated with human umbilical cord mesenchymal stem cells (hUC-MSCs) combined with human neural stem cells (hNSCs) via a direct intramedullary transplantation during the subacute phase to identify a more valid method for treating spinal cord injuries. Our study provides the first evidence that a combined hUC-MSC and hNSC intramedullary injection improves functional recovery more than that from a hUC-MSC alone or a hNSC alone treatment in a SCI animal model. Furthermore, in this study, the combined hUC-MSC and hNSC treatment was found to result in better transplanted cell survival in the injured site of the spinal cord of rats than that of the treatments with either of the two types of stem cells alone. In general, our data suggested that the combined hUC-MSC and hNSC intramedullary subacute transplantation may be a potentially more promising method for cell treatments in SCI.
Materials and Methods
Preparation of Human Umbilical cord MSCs and Human NSCs
MSC isolation and expansion
We isolated MSCs from the human umbilical cord using the explants culture method. Fresh umbilical cords were obtained from 38- to 40-week-old healthy neonates delivered via Cesarean section in the obstetrical department of Qilu Hospital, Shandong University. Informed consents were signed by the mothers of the neonates who provided the umbilical cords. After removal into a sterile 0.9% sodium chloride solution, the umbilical cords were rinsed with 70% ethanol for 1 min. Then, the umbilical cords were washed with a sodium chloride solution twice for 2 min. After removing the umbilical arteries, veins, and umbilical cord adventitia from the umbilical cord, Wharton’s jelly was obtained. Then, we dissected Wharton’s jelly into small pieces of approximately 1 mm3. Next, the Wharton’s jelly pieces were inserted into T-75 flasks (Corning Inc., Corning, NY, USA) containing 15 ml of complete medium, including Dulbecco’s modified Eagle’s medium(DMEM; Invitrogen, Carlsbad, CA, USA), 10% (v/v) fetal bovine serum (FBS; Invitrogen, USA), 2 mM
HNSC isolation and expansion
Forebrain tissues were obtained from 8- to 10-week-old embryonic fetuses via routine abortions in the obstetrical department of Qilu Hospital, Shandong University. Informed consents were signed by the pregnant women who provided the fetal tissues.
The fetal forebrain tissues were dissected under sterile conditions in a DMEM/F-12 medium (Invitrogen, USA). Then, the forebrain tissues were dissociated mechanically through tissue homogenization. The dissociated cells were collected and resuspended in a neurobasal medium (Invitrogen, USA) containing a 2% B27 supplement (Invitrogen, USA), 20 ng/ml bFGF and 20 ng/ml EGF (R&D Systems, Minneapolis, MN, USA), 2 μg/ml heparin (Sigma, USA), 2 mM
Preparation of stem cells for grafting
On the day of the transplant, the cells were harvested from the culture flasks using 0.25% trypsin/EDTA (hUC-MSCs) or Accutase (NSCs) and subsequently washed and resuspended in PBS. The total concentration of the resuspended cells prepared for each group transplantation was 1 × 105 cells/μl. The hUC-MSCs and hNSCs groups contained hUC-MSCs alone and hNSCs alone, respectively, and the hUC-MSCs+hNSCs group contained both types of cells (1:1). The cells were placed on ice until the transplant. The viability of the cells was assessed using Trypan blue immediately before transplantation. Only cells in populations with >95% viability were used.
Experimental animals
Adult female Wistar rats (weight, 180–210 g) were used in this study. All rats in this study were obtained from the Laboratory Animal Center of Shandong University, kept in standard conditions under a 12-h light/dark cycle, and provided food and water ad libitum. All animal care procedures were conducted based on the guidelines of the Ethical Committee of Qilu Hospital, Shandong University. All animal protocols were approved by the Institutional Animal Care and Use Committee at the School of Shandong University.
SCI model
The rat SCI model was established in our laboratory using a previously described weight dropping method 17 . After the rats were anesthetized with 10% chloralhydrate (0.35 ml/100 g, intraperitoneally), a laminectomy was performed at the T10 spinal cord level with the dura matter intact. A moderate contusion SCI model was created by dropping a 10-g weight rod from a height of 2.5 cm onto the exposed dorsal surface of the dura of the spinal cord with an NYU impactor. The rats in the sham group received a laminectomy only with no SCI. Postoperatively, the animals were treated with anti-inflammatory medication (8 mg/kg gentamycin) and saline (0.9%; 4 ml) via subcutaneous injections to prevent bacterial infections and dehydration. Manual bladder expression was performed three times each day until voluntary urination was established. All animals were tested with the BBB behavioral test 1 day before and after the SCI, and then weekly until the end of the test, and only rats with BBB test scores ≤2 on the 7th day after SCI were included in the study.
Experimental groups
One day after the SCI, 108 spinally injured female Wistar rats were selected and randomly divided into the following five groups: (1) the hUC-MSCs group, in which rats were given an SCI and received a human umbilical cord mesenchymal stem cell transplantation,
Cell transplantation
The transplants were performed using a randomized, blind study design 8 days post-injury. Rats in all groups were anesthetized in the same way as described above, and, using a sterile technique, the spinal dura matter at T10 was exposed. Stem cells were intramedullary injected into the spinal cord using Hamilton needles in manner described previously 17 .
In the experimental hUC-MSCs, hNSCs, and hUC-MSCs+hNSCs groups, 6 μl of isolated stem cells (1 × 105 cells/μl) suspension was injected into the spinal cord (the center and 2 mm rostral and 2 mm caudal to the center of the traumatized area) in three separate injections, and approximately 1.5 mm deep from the dorsal surface of the dura matter. Thus, the total amount was 6 × 105 cells per rat. In the PBS group, the rats were injected with only PBS in the same way. The sham group did not receive any specific treatment, but the spinal dura matter was exposed as performed in the other four groups. All rats received a daily injection of cyclosporine A (10 mg/kg/day; Novartis Pharma AG, Basel, Switzerland) intraperitoneally starting from 1 day before the cell transplantation surgery until the end of the experiment.
Behavioral Tests
To evaluate the locomotor recovery of the SCI animals, we adopted the BBB rating scale method. The BBB scale was developed by Basso, Beattie and Bresnahan and is a reliable and sensitive locomotor rating scale that offers investigators a more discriminating measure of behavioral outcomes to evaluate treatments after SCI 18,19 . In our experiment, two independent examiners who were blinded to the protocol observed all the animals during the tests. Briefly, the rats were placed on a floor within a circular enclosure and assessed for 4 min. Their hindlimb locomotor functions were scored using a 22-point (0–21) scale that evaluates parameters including hindlimb joint movement, paw placement, weight support, forelimb-hindlimb coordination, etc. Animals displaying normal locomotion received a score of 21.
Eight animals were selected randomly from each group and were assessed with the BBB behavioral test 1 day before and after the SCI, and then weekly until the experiment was completed. The scores were obtained by averaging both hindlimb values.
Immunofluorescence and immunohistochemistry
In this experiment, we measured the survival and differentiation of the cells transplanted into the injured spinal cords 2 weeks after transplantation. Six animals were selected randomly from each treated group (the hUC-MSCs group, the hNSCs group, the hUC-MSCs+hNSCs group and the PBS group) and were sacrificed by an intraperitoneal injection of an overdose of chloral hydrate (600 mg/kg) 2 weeks after cell transplantation. The rats were then intracardially perfused with 0.9% sodium chloride and 4% paraformaldehyde in 0.1 M sodium phosphate buffer at pH 7.4. Spinal cord sections surrounding the T10 lesion site (approximately 1 cm long) were carefully extracted from the vertebrae and post-fixed with 4% paraformaldehyde overnight at 4°C. After dehydration in a 25% sucrose solution for 48 h, the specimens were embedded, snap-frozen with a freezing compound (O.C.T. Tissue-Tek, Sakura, Tokyo, Japan) and stored at –80°C until use.
The frozen tissues were longitudinally cryosectioned into a series of slices with 5 μm thickness, and three adjacent 5-μm sagittal slices for every 0.25 mm were selected. Thus, approximately 8–10 sets of triple adjacent sections were obtained for each sample. Then, five sets of triple adjacent slices near the midline of the spinal cord were selected and prepared for the cell survival, cell differentiation, and brain-derived neurotrophic factor (BDNF) expression measurements. After being rehydrated and washed with PBS, the sections were treated with 3% H2O2 for 10 min and then washed with PBS and permeated with 0.2% Triton X-100 for 10 min. After being washed with PBS, the slices were incubated in a blocking buffer (5% goat serum in PBS) for 30 min at 37°C.
To identify the surviving transplanted stem cells in the samples, we incubated the sections with a mouse polyclonal anti-human nuclei (HuNu) antibody (1:100; Millipore, USA) at 4°C overnight (Method (1)). The next day, after washing with PBS and incubating with Alexa Fluor® 594 goat anti-mouse IgG (H+L) (1:200; Invitrogen, USA) for 1 h at room temperature, the slices were washed, and the cell nuclei were stained with DAPI (Vector Laboratories, Burlingame, CA, USA).
To identify differentiation of the hUC-MSCs and hNSCs in the injured spinal cord, the tissues were double stained with a human-specific nuclei marker, mouse polyclonal anti-human nuclei (HuNu) antibody (1:100; Millipore, USA), and a rabbit polyclonal anti-GFAP antibody (1:500; Abcam, Cambridge, UK) or a rabbit anti-β-tubulin III antibody (1:500; Abcam, UK) or a rabbit polyclonal anti-CNP antibody (1:500; Abcam, UK) (Method (2)). We visualized the immunoreactivity with Alexa Fluor® 594 goat anti-mouse IgG (H+L) and Alexa Fluor® 488 goat anti-rabbit IgG (H+L) secondary antibodies (1:200; Invitrogen, USA) and counterstained the nuclei with DAPI (Vector Laboratories, USA). The PBS group was used as the negative control group.
The slides from methods (1) and (2) above were mounted with an antifade mounting medium (Wuhan Boster Biological Technology, Ltd., China).
To identify BDNF expression, immunohistochemistry was performed on the selected frozen slices (Method (3)). The sections were incubated with a rabbit polyclonal anti-BDNF antibody (1:100; Santa Cruz Biotechnology, Santa Cruz, CA, USA) (reactivity: mouse, rat, human) at 4°C overnight. The next day, the slices were washed with PBS and incubated with a goat anti-rabbit IgG and a streptavidin peroxidase (SP) complex for 40 min at 37°C (SP-9001 kit, ZSGB-BIO, Bejing, China). After the sections were washed with PBS, we visualized the peroxidase activity sites with a 3,3’-diaminobenzidine tetrachloride kit (DABkit, ZSGB-BIO, China). The sections were then rinsed with double distilled water and washed with running tap water. The slides were stained with hematoxylin for counterstaining the nuclei for 2 min. Next, the slides were dehydrated in graded ethanol and cleared in xylene. Finally, the slides were mounted with a neutral balsam medium, covered with Leica CV5030 coverslips and dried at room temperature. The negative control tissue sections (from the hUC-MSCs group) were processed similarly but the primary antibody was omitted.
Finally, we obtained immunofluorescent or immunohistochemistry images under a fluorescence microscope (BX53 Microscope, Olympus, Tokyo, Japan) using software (CellSens Standard, Olympus, Japan).
The Alexa Fluor® 594 stained the nuclei of the surviving implanted stem cells red, and the nuclei were measured using Image-Pro Plus 6.0 software (Media Cybernetics, Rockville, MD, USA). The spinal cord tissues on the slides were divided into three regions equally according to the sites of cell transplantation. The number of the stem cells in each region was evaluated in a randomly selected 200x microscopic field. The average number of the counted stem cells in each specimen (per slide) was used for the statistical analysis. The BDNF expression measurements were performed using Image-Pro Plus 6.0 software. The brown particles in the picture were regarded as positive BDNF expression. The integrated optical density (I.O.D.) of the BDNF immunostaining was evaluated in four 400x randomly selected microscopic fields at the glial scars around the cavity of the injured spinal cord. The average I.O.D. of the BDNF expression in each sample (per slide) was used for the statistical analysis.
BDNF Enzyme-Linked Immunosorbent Assay
Two weeks after cell transplantation, six rats selected randomly from each treated group (hUC-MSCs group, hNSCs group, hUC-MSCs+hNSCs group, and PBS group) were sacrificed by the methods described above. The rats were deeply anesthetized, and sections of their spinal cords surrounding the injured site (approximately 1 cm long) were removed and placed on ice. Subsequently, each sample was homogenized in cold 0.9% chloride sodium (1:9 weight/volume). Then, we centrifuged the homogenate and collected the supernatant for analysis. BDNF expression was detected using an enzyme-linked immunosorbent assay (ELISA) kit (Abcam, UK) (reactivity: mouse, rat, human) following the manufacturer’s protocol. The supernatant was diluted two-fold before use. Assays of the standards and all samples were performed in triplicate on a single 96-well plate. We read the ELISA plate at 450 nm with an ELISA reader and calculated the BDNF concentration of the samples using standard curves.
LFB Staining
At the completion of the 8-week study, six animals from each group were selected randomly and sacrificed, and the injured spinal cord was resected (approximately 1 cm long) in the same way as described above. The resected spinal cords were post-fixed with 4% paraformaldehyde for 2 days at 4°C. After washing with tap water and processing the tissue, the spinal cords were embedded in paraffin. Then, the tissues were transversely sectioned into a series of slices with 5-μm thickness, and slices for every 2 mm were selected. A total of five slices centered over the injured epicenter of the spinal cord from each sample were obtained and mounted on glass slides.
The sections were deparaffinized and rehydrated in a 95% ethanol solution. Then, the sections were left in LFB solution (0.1%, Sigma, USA) in a 60°C oven overnight. The next day, the excess dye was rinsed out with 95% ethanol followed by washing with distilled water. Then, the slides were differentiated in a lithium carbonate solution (0.05%, Sigma, USA) for 30 s followed by 70% ethyl alcohol for 30 s. After washing with distilled water, the staining was assessed microscopically to determine whether the gray matter was clear and whether the white matter was sharply defined. The differentiation steps were repeated three times in this experiment. After the differentiation procedure, the slides were rinsed with 0.5% eosin, washed with distilled water and stained with Cresyl violet (Sigma, USA) solution for 10 min at 37°C. After being washed with distilled water, the slides were dehydrated in 95% ethanol once and 100% ethyl alcohol twice. After being cleared in xylene twice, the slides were mounted with a neutral balsam medium with Leica CV5030 coverslips and dried at room temperature.
Five slices from each sample, including the lesion center of the spinal cord (spanning 8 mm), were evaluated for LFB-positive areas, and the number of neurons in the anterior horn of the spinal cord was counted. Images of the slices were visualized via an Olympus BX53 microscope using software (CellSens Standard). The I.O.D. of the myelin sheath and the number of neurons in the spinal anterior horn were obtained using Image-Pro Plus 6.0 software.
Statistical Analysis
The data are expressed as the mean±SD, and the variance was analyzed using SPSS version 16.0 statistical software (SPSS, IBM, Armonk, NY, USA). The locomotor function data were statistically analyzed using a two-way ANOVA followed by Fisher’s least significant difference (LSD) post hoc analysis. For the immunofluorescence, immunohistochemistry, and LFB staining data, one-way ANOVAs comparing the groups followed by Tukey-Kramer tests or Dunnett tests were performed. Differences were considered statistically significant at
Results
Isolation and Culture of hUC-MSCs
Primary hUC-MSCs were successfully isolated by the explant culture method from the Wharton’s jelly matrix. After 7 days, we observed that many cells migrated from the Wharton’s jelly matrix mass. After 12 days, 90% confluent cell colonies at high cell densities were observed in the tissue culture flasks (Fig. 1A). The cells were then sub-cultured to passage 3 for later use.

Stem cell culture. (A) On day 12, high densities of spirally growing hUC-MSCs can be observed in the tissue culture flasks. (B)Neural spheres(hNSCs) formed in the suspension culture medium. Magnification: 10X.
Culture of hNSCs
hNSCs were successfully isolated and cultured from the forebrain tissues obtained from 8- to 10-week-old embryonic fetuses via routine abortions. Neural spheres were formed within 7–10 days of the suspension culture of the original neural stem cells (Fig. 1B). In the primary culture of hNSCs, we can obtain 1–2 x 108 cells per fetal brain. The cells at passages 3–5 were obtained and used in our experiment.
Behavioral Tests
The BBB scale test was performed every week to assess the hindlimb functional recovery of the rats. In total, 108 SCI rats with BBB scores of 0–2 were successfully selected and divided into five groups on the 7th day after SCI. On the 8th day after SCI, rats in the four treated groups were injected with stem cells or PBS (the control group). The BBB score was 21 in the sham group from 1 week after the SCI until the end of the experiment. All rats in the SCI groups exhibited a slow improvement in the 1st week after SCI, which was characterized by slight movement of the hind limb but with no weight support. Thereafter, hind limb function recovery speed increased markedly over the following 4 weeks in the SCI rat groups after the transplantation treatment. Particularly, BBB scores in the hUC-MSCs+hNSCs group were enhanced greatly, and showed significant differences compared with the scores in the other three treated groups from 2 weeks after SCI until the end of the experiment. The significant difference between the hNSCs and PBS groups began 4 weeks after SCI and remained until the end of the trial (+P < 0.05). In the 5th week after SCI, the BBB scores of the hUC-MSCs group increased quickly and showed a significant difference compared with those of the PBS group; this trend was maintained until the 8th week (*

BBB locomotor score. Assessment of the mean BBB locomotor score in rats after SCI until week 8. Each data point represents the mean±SD. Significant differences between the hUC-MSCs+hNSCs group and the hUC-MSCs, hNSCs, or PBS groups started at 2 weeks after the SCI (hUC-MSCs, hNSCs, *
Survival Results
The surviving stem cells (per slide) in the four treated groups were counted (Fig. 3). As shown in the figure, the number of surviving transplanted cells in the spinal cord was 343.0 ± 47.2 in the hUC-MSCs+hNSCs group, 251.3 ± 61.3 in the hNSCs group, and 224.4 ± 57.5 in the hUC-MSCs group. The number of surviving stem cells in the hUC-MSCs+hNSCs group was significantly greater than that in the hNSCs group(*
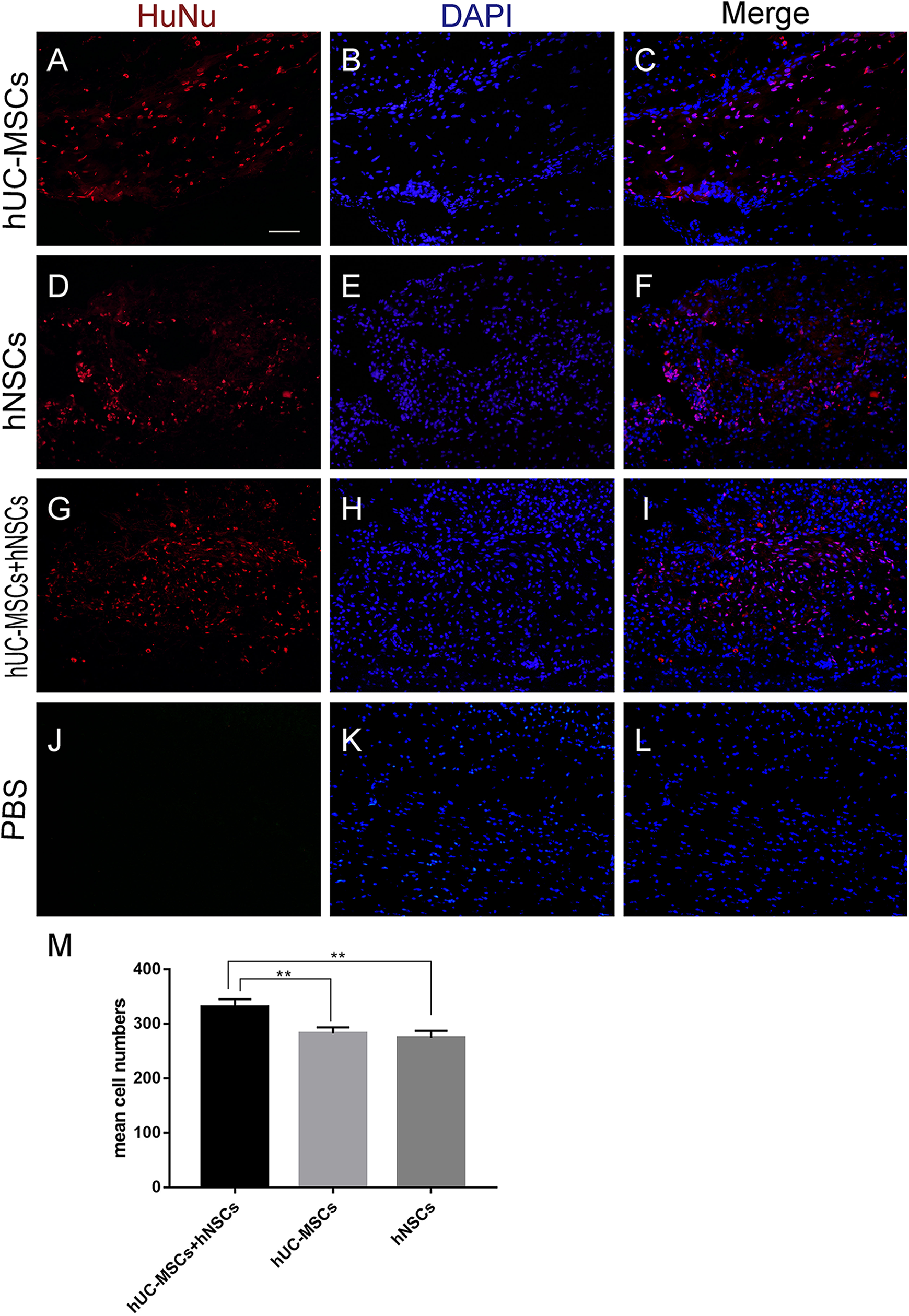
Surviving transplanted stem cells. The numbers of surviving cells in the spinal cord were 343.0±47.2 in the hUC-MSCs+hNSCs group, 251.3±61.3 in the hNSCs group, and 224.4±57.5 in the hUC-MSCs group. No transplanted stem cells were found in the PBS group. The number of surviving transplanted stem cells in the injured spinal cords of rats in the hUC-MSCs+hNSCs group were the highest among the four treated groups and had a significant difference compared with that in the hNSCs group (*
Differentiation of Surviving Stem Cells in Vivo
Differentiation of the transplanted stem cells in the injured spinal cord of the three stem cell transplant groups was identified using cell marker antibodies, and the PBS group was used as the negative control group. In the hUC-MSCs group, cells with HuNu-positive nuclei (red) were observed, and were not double stained with any of the anti-GFAP, anti-β-tubulin III or anti-CNP antibodies (Fig. 4A–C). HuNu-β-tubulin III positive cells were 18.0 ± 5.2 in the hUC-MSCs+hNSCs group and 26.8 ± 7.7 in the hNSCs group. HuNu-GFAP positive cells were 89.6 ± 33.2 in the hUC-MSCs+hNSCs group and 151.4 ± 35.7 in the hNSCs group. HuNu-CNP positive cells were 5.5 ± 1.9 in the hUC-MSCs+hNSCs group and 9.6 ± 3.4 in the hNSCs group (Fig. 4 M). This result indicated that the surviving hUC-MSCs cells in the injured spinal cord did not differentiate into neural cells in our experiment. As shown in Fig. 4D–I, double staining of HuNu and GFAP, HuNu, and β-tubulin III, or HuNu and CNP cells was present in both the hNSCs group and the hUC-MSCs+hNSCs group, indicating that the transplanted cells had differentiated into neurons, astrocytes, or oligodendrocytes. No transplanted stem cells were found in the PBS group. Furthermore, based on the immunofluorescence images, we can observe that the detected hUC-MSCs seem more converged and did not integrate much better with the host, but the transplanted stem cells in the hNSCs group and the hUC-MSCs+hNSCs group dispersed and integrated with the host tissues very well (Fig. 4D–I).

Surviving transplanted stem cells double staining. In the hUC-MSCs group, the transplanted HuNu-positive stem cells (red) can be observed in (A), (B), and (C), and no double stained cells were found. Double staining of HuNu and GFAP, HuNu and β-tubulin III, or HuNu and CNP cells was present in the hNSCs group and the hUC-MSCs+hNSCs group (D–I, white arrow). No positive cells were found in the PBS group. The detected hUC-MSCs seemed more converged and did not integrate well with the host, but the transplanted stem cells in the hNSCs group and the hUC-MSCs+hNSCs group dispersed in the host tissues very well.
BDNF Results
The results of the immunohistochemistry analysis of BDNF expression in the stem cell-treated groups showed higher expression and significant differences compared with the expression in the PBS group (hUC-MSCs+hNSCs, **

Results of the immunohistochemical staining of BDNF. (A) hUC-MSCs+hNSCs group; (B) hUC-MSCs group; (C) hNSCs group; (D) PBS group; (E) negative control images. (F) In the hUC-MSCs+hNSCs group, brown particles were converged around the cavity of the spinal cord. Magnification (A)–(E), 400x; F, 100x. (G) The BDNF I.O.D. were significantly different between the stem cell-treated groups and the PBS group (hUC-MSCs+hNSCs, **
The results of the BDNF expression measurements using a sandwich ELISA were consistent with the immunohistochemistry outcomes. The BDNF levels were significantly increased in the three stem cell-treated groups compared with those in the PBS group (hUC-MSCs+hNSCs, 9.76 ± 1.23, **
LFB Staining
The LFB staining showed large amounts of cell debris, degenerated axons, and cavities in the transverse spinal cord sections of the injured groups (Fig. 6(a) A-D). As shown in Fig. 6(b), the amount of myelin sheath (per slide) evaluated by the I.O.D. in each of the three stem cell-treated groups was larger compared with that in the PBS group (hUC-MSCs+hNSCs, **

The results of the LFB staining at 8 weeks post-SCI. (a, A–E) Histology of the transections near the epicenter of the injured spinal cord. Large amount of cell debris, degenerated axons, and cavities were found at the epicenter of the injured spinal cord in the SCI groups (A–D). (b) The I.O.D. values of the fiber sheaths on the surviving neurons in each of the three stem cell-treated groups were higher compared with those in the PBS group (hUC-MSCs+hNSCs, **
The numbers of cells (per slide) containing Nissl bodies and having typical neuronal morphology were counted as neurons. Fig. 6(a) F–J shows the Cresyl violet-stained neurons in the anterior horn of the transverse spinal cord slices approximately 2 mm caudal from the epicenter of the injured spinal cord. As the statistical results show in Fig. 6(c), the numbers of surviving neurons in the anterior gray horns of the spinal cord in the three stem cell-treated groups were significantly higher than those in the PBS group (hUC-MSCs+hNSCs, hUC-MSCs, **
Discussion
In our study, the locomotor function was measured using the hindlimb BBB scores of the rats in all groups. The results indicated that the functional recovery in the rats in the combined hUC-MSCs+hNSCs group was the best among all the groups, although the hUC-MSCs and hNSCs groups also obtained better functioning than that in the PBS group. The survival of the transplanted cells in the combined hUC-MSCs+hNSCs group also showed the highest numbers of the three stem cell-treated groups. This result indicated that the combined hUC-MSCs and hNSCs treatment can enhance the transplanted cell survival at in the injury site, and the increased number of surviving transplanted cells may have contributed to the achievement of the best functional recovery among the three stem cell-treated groups.
Because we found in the pre-experiment that the neurological recovery of rats with SCI was the fastest within 1–2 weeks after cell transplantation, the main purpose of this study was to find the relationship between the recovery of neurological function and the transplanted cells in the subacute phase of SCI. Therefore, we only observed the survival rate of transplanted cells 2 weeks after cell transplantation. In future work, we need to investigate whether the combined transplantation strategy can improve the long-term survival of transplanted cells and the specific functions of surviving cells in the local transplant microenvironment.
In addition to the pathological damage due directly to the trauma, one of the most important pathological processes in the white matter of injured spinal cords is a chronic and progressive demyelination of spared axons 20 . We evaluated the number of myelinated axons in our trial to determine whether the amount of remaining myelin sheath in the injured spinal cord was consistent with the functional recovery. The amount of myelin sheath measured by LFB staining in the combined hUC-MSCs+hNSCs group was also the highest among all the stem cell-treated groups, which is consistent with the BBB score and surviving cell number data. This result indicated that the greater amount of myelinated nerve fibers remaining in the injured spinal cord may be one of the contributions to the best functional recovery in this histological aspect. The larger number of myelinated nerve fibers may be due to axonal re-myelination, axonal regeneration or the survival of neural fibers with a conserved myelin sheath. The greater number of surviving transplanted stem cells in the combined hUC-MSCs+hNSCs group can possibly better regulate the microenvironment of the injured spinal cord and increase the number of myelinated nerve fibers.
BDNF is a type of neurotrophic factor that can enhance neuronal regeneration, plasticity, re-myelination and cell survival in the nervous system under pathological conditions caused by disease or trauma 21 –23 . Studies have also indicated that NSC grafts exert a neuroprotective effect by upregulating BDNF in injured spinal cords 24 . In our study, the BDNF detection results using immunohistochemistry were consistent with the BDNF ELISA results, and both showed that the amount of BDNF secreted in the three stem cell transplant groups (hUC-MSCs+hNSCs group, hUC-MSCs group, and hNSCs group) was significantly higher than that in the PBS group, and this result indicated that BDNF may be one of the nutritional factors that enhances the functional recovery of rats with SCI. BDNF can be secreted by the stem cells directly or by the host cells that are mediated by the transplanted stem cells. Although the hUC-MSCs+hNSCs group showed the best transplanted stem cell survival, there were no significant differences in terms of BDNF secretion among the three stem cell-treated groups. Therefore, this result implies that BDNF is not mainly secreted by the stem cells, and that host cells, such as glia or neurons, might play a crucial role in BDNF secretion.
Although the immunofluorescence showed that hNSCs can differentiate into neurons, astrocytes and oligodendrocytes, the functional outcome was not different between the hUC-MSCs group and the hNSCs group, and the two stem cell-treated groups both had a better outcome when compared with that of the PBS group. This result indicated that hUC-MSCs and hNSCs promoted the functional recovery in rats with SCI not primarily through cell differentiation and direct replacement of the lost cells, but rather through neuroprotective means, such as providing neural trophic and immunomodulatory factors to enhance axonal growth, modulate the environment, and reduce neuroinflammation 25 . So, many challenges in understanding cell replacement and potential formation of new functional neuronal relays across lesion sites still need to be overcome. The results of our study indicated that the co-transplantation of hNSCs and hUC-MSCs had the best functional recovery. Therefore, a combined hUC-MSC and hNSC therapy may prove to be an ideal method for treating SCI in the future.
Conclusions
The present study demonstrated that intramedullary transplantation of hNSCs, hUC-MSCs, or hNSCs+hUC-MSCs in subacute phase all significantly improved outcomes in an in vivo contusion SCI model. The co-transplantation of hNSCs and hUC -MSCs can increase the survival of transplanted stem cells in vivo and is presented as the best method.
Footnotes
Ethical Approval
Ethical approval to this original research was obtained from the Ethical Committee of Qilu Hospital, Shandong University.
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Institutional Animal Care guidelines of Shandong University, China and approved by Administration Committee of Experimental Animals of Shandong University, Shandong Province, China. All experimental procedures involving humans were approved by the Ethics Committee of Qilu Hospital, Shandong University.
Statement of Informed Consent
Written informed consent was obtained from the patients who provided the tissues used in this experimentation for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: 1) Shandong Cord Blood Bank, Shandong, China; 2) Shandong Disabled Persons’ Federation, Shandong, China; 3) Department of Science & Technology of Shandong Province, China (Project No. 2016ZRA09012).
