Abstract
Neuropathic pain is a debilitating complication following spinal cord injury (SCI). Currently, effective treatments for SCI-induced neuropathic pain are highly lacking. This clinical trial aimed to investigate the efficacy of combined intrathecal injection of Schwann cells (SCs) and bone marrow–derived mesenchymal stem cells (BMSCs) in improving SCI-induced neuropathic pain. This study was a parallel-group, randomized, open-label, active-controlled phase II trial with two arms, including treatment and control groups. Patients with complete SCI-induced neuropathic pain in the treatment group received a single combined intrathecal injection of BMSCs and SCs. Study outcome measures were International SCI Pain Basic Data Set (ISCIPBDS) and World Health Organization (WHO) Quality of Life Assessment Instrument (WHOQOL-BREF). A total of 37 (55.2%) and 30 (44.8%) patients in the treatment and control groups were followed up for 6 months, respectively. Significant reductions in mean scores of interference items in the treatment group, including daily activities (P < 0.001), mood (P < 0.001), and sleep (P < 0.001), were found at 6 months after the injection compared with the control one. Similarly, pain frequency (P = 0.002), mean (P = 0.001), and worst (P = 0.001) numeric rating scale (NRS) pain intensity scores showed significant reductions in the treatment group after 6 months compared with the control one. Based on multiple regression analysis controlled for potential confounders, significant associations between changes in all outcome measures over the study period and the treatment group were found. This clinical trial indicated the efficacy of combined cell therapy in improving the neuropathic pain and quality of life in complete SCI patients. Future investigations should evaluate the effects of combination of this strategy with other existing therapies for SCI-induced neuropathic pain. This clinical trial was also registered prospectively at the Iranian Registry of Clinical Trials (IRCT20200502047277N8).

Introduction
Chronic pain is a common condition following traumatic spinal cord injury (SCI) and affects 30%–80% of patients1–4. SCI-related pain is categorized into nociceptive and neuropathic, of which the latter is further divided into above-level, at-level, and below-level pain, which is frequent among patients 3 . Prior reports have shown a significant association between chronic SCI-related neuropathic pain and poorer quality of life (QoL)5,6. Although several treatment strategies have been investigated previously, including both pharmacological and non-pharmacological therapies, their efficacy in reducing neuropathic pain has been limited7–9.
Stem cell therapy is a burgeoning field of treatment for SCI, and the safety and efficacy of various types of cells have been studied in prior investigations. To date, most of these studies have used mesenchymal stem cells (MSCs) derived from various sources, including adipose tissue–derived MSCs (ADSCs), bone marrow–derived MSCs (BMSCs), and umbilical cord–derived MSCs10,11. Given their major characteristics, including immunomodulatory, neuroprotective, and regenerative capacities, the ability to differentiate into nerve cells, and low immunogenicity, MSCs are a potential option for the treatment of SCI12,13. Many prior studies have also suggested that SC transplantation is effective as a therapeutic option for SCI. The SCs are also capable of providing neurotrophic support for axonal regeneration through secretion of glial cell–derived neurotrophic factor (GDNF), ciliary neurotrophic factor, brain-derived neurotrophic factor (BDNF), and neurotrophin 3 14 .
Despite some previous clinical reports on cell therapy with SCs, clinical data concerning the efficacy of combined cell therapy with both BMSCs and SCs in SCI are highly lacking. In addition, there is a lack of clinical evidence concerning the efficacy of stem cell therapy in SCI-related neuropathic pain and patients’ QoL. Thus, this randomized, active-controlled phase II trial was performed to assess the effects of intrathecal combined cell therapy with BMSCs and SCs on SCI-induced neuropathic pain and the QoL of SCI patients.
Methods
Trial Setting and Participants
The present study was a parallel-group, randomized, open-label, active-controlled phase II clinical trial designed according to the Declaration of Helsinki. The ethics committee of the Shahid Beheshti University of Medical Sciences (IR.SBMU.MSP.REC.1401.053) as well as Aja University of Medical Sciences (IR.AJAUMS.REC.1402.215) approved this clinical trial. This study was also registered at the Iranian Registry of Clinical Trials (IRCT20200502047277N8). Patients were provided with written informed consent before performing any procedure in this study, including study intervention, cell culture, and biopsy specimen analysis. Thorough information was given to patients regarding the study intervention and its experimental nature, as well as potential adverse events, such as infection, autonomic dysreflexia, motor or sensory deficit, and spasticity. This study included two groups (treatment and control). Random assignment of participants to treatment and control groups (1:1 ratio) was performed using computer-generated block randomization. A person who was out of the study process performed randomization in this study. Blinding was not performed due to the nature of the study intervention. Similarly, no placebo was used in the control group. Data reporting in this clinical trial were according to the Consolidated Standards of Reporting Trials (CONSORT) guidelines.
In the present study, all SCI patients who presented to the neurosurgery department of Shohada Tajrish Hospital between June 2022 and August 2023 were assessed for eligibility. Inclusion criteria were age between 18 and 65 years, complete SCI [defined as American Spinal Injury Association impairment scale (AIS) grade A], persistent or recurrent pain for at least 3 months (chronic pain) according to US Model SCI systems measures (patient’s score ≥1, using a 0–10 scale for the average pain over the past 3 months)15,16, neuropathic pain category according to the International SCI Pain Basic Data Set (ISCIPBDS) version 2.03, and written informed consent. Exclusion criteria were pre-existing comorbidities, such as diabetes mellitus, cardiovascular diseases (e.g., myocardial infarction or heart failure), neurological and psychological disorders, abnormal hepatic and renal function tests, penetrating spinal cord trauma or transection, active infection such as pneumonia or urinary tract infection, positive test results for hepatitis B virus, hepatitis C virus, human immunodeficiency virus, cytomegalovirus, or venereal disease research laboratory test, and history of malignancy.
Cell Isolation and Characterization
The process of SC harvest was according to our modified protocol. Following the harvesting of sural nerve, under sterile conditions, the specimens were transferred to the laboratory in Dulbecco modified Eagle’s medium (DMEM; Gibco, Grand Island, NY, USA) using a cold and sealed container. At the laboratory, the sural nerves were dissected, adjacent soft tissue was removed, and the nerves were sliced into smaller pieces (1–2 mm). After that, treatment with collagenase (1.4 U/mL; Sigma, St. Louis, MO, USA) and Dispase (2.4 U/mL; Sigma) at 37°C for 3 h was performed. Thereafter, the cells were washed twice with DMEM/F12, and mesh filtering was performed. After that, cells were incubated in DMEM/F12 for 5 days (37°C, 5% CO2) with no fetal bovine serum (FBS). The FBS (Gibco) concentration in the culture medium was then increased up to 10% over 1 week following the fasting period. Isolated cells were characterized and determined using S-100 immunocytochemical staining. Cells were then fixed in 4% paraformaldehyde (Sigma), and for intracellular antigen staining, they were permeabilized with 0.1% Triton X-100 (Sigma) and subsequently blocked with 5% goat serum (Gibco). After that, overnight incubation at 4°C with anti-S100 (Chemicon, Temecula, CA, USA) antibody and then with fluorescein isothiocyanate–conjugated secondary antibody (Abcam, Cold Spring Harbor, NY, USA) at room temperature for 3 h was performed. Finally, fluorescence images of cells were acquired via a fluorescent microscope (TE2000-S; Nikon-Eclipse, Tokyo, Japan) (Fig. 1A).
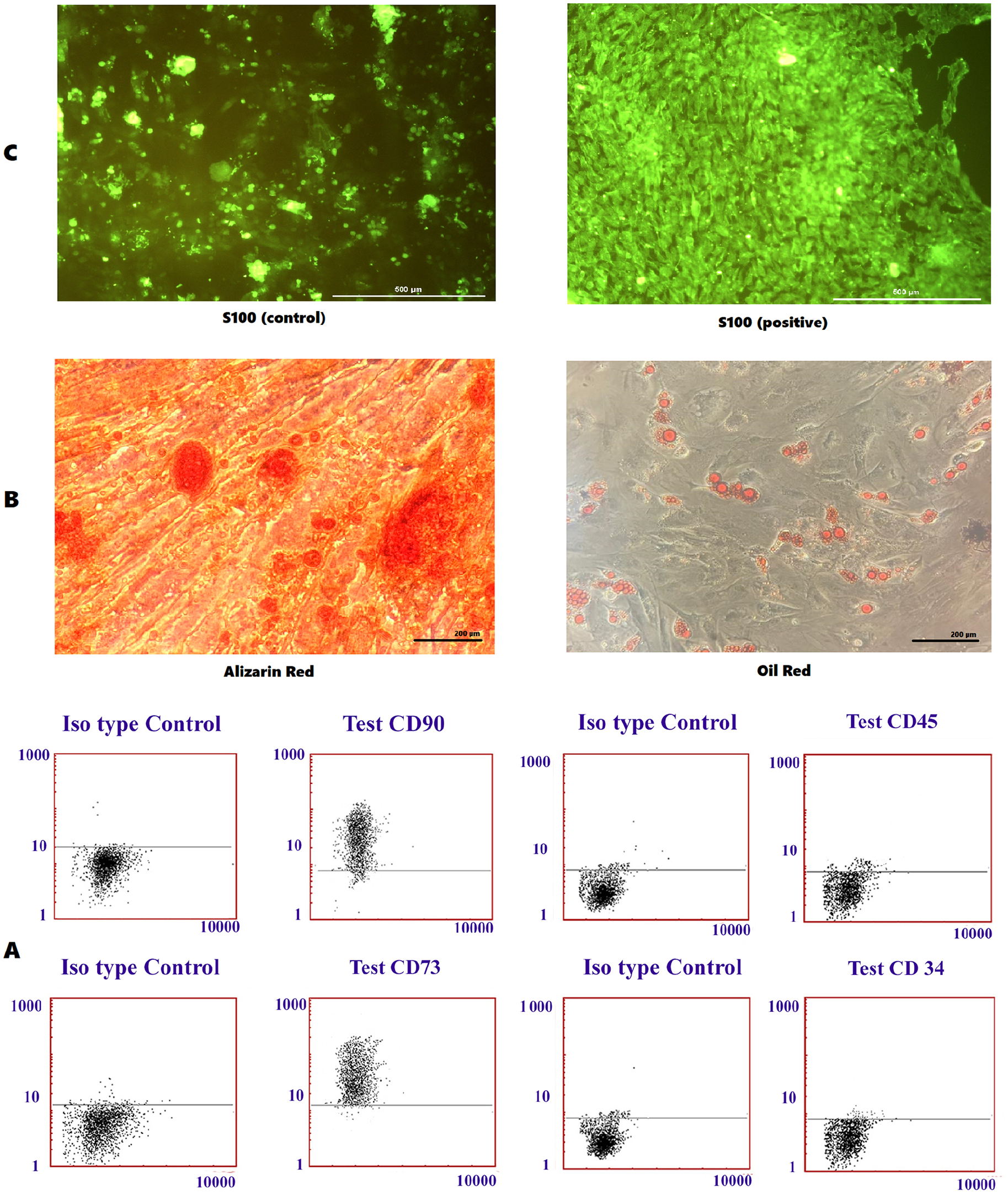
Results of bone marrow MSCs and SCs laboratory assessments. S-100 immunocytochemical staining negative control and S-100 immunocytochemical staining test (A); alizarin red and oil red staining of the MSCs (B); the cell surface markers (CD90, 73, CD45, and CD34) analysis through flow cytometry (C).
For MSC harvest, each participant underwent bone marrow aspiration, and 100–150 mL of bone marrow was aspirated from the iliac crest. The bone marrow was then diluted (1:3) with Hanks balanced salt solution (HBSS; Sigma), and samples then underwent a density gradient (a ratio of 1:3) with Ficoll (1.077 g/L; Sigma). Thereafter, the bone marrow was centrifuged at 400 × g for 40 min, and the mononuclear cell layer was separated and washed with HBSS subsequently. Following the removal of Ficoll, the cells were centrifuged three times to remove platelets and isolate mononuclear cells. To confirm the isolation MSCs, their ability to differentiate into adipogenic and osteogenic cells as well as their surface markers was evaluated. Osteogenic induction was performed by adding 50 mg/mL ascorbic acid 2-phosphate (Sigma), 100 nmol/L dexamethasone (Sigma), and 10 mmol/L glycerol phosphate to the growth medium for 21 days (all from Merck, Rahway, NY, USA). Calcium mineralization was assessed by fixing and staining the samples with Alizarin red (Sigma). Furthermore, adipogenic differentiation was assessed after supplementing the growth medium with 0.5 mmol/L hydrocortisone (Sigma), 0.5 mmol/L isobutyl methylxanthine (Sigma), and 60 mmol/L indomethacin (Gibco) for 3 weeks. The accumulation of the oil droplets was confirmed using the oil red O staining afterward (Fig. 1B). Flow cytometry analysis was then performed using a total of 1 × 105 cells in 100 mL phosphate-buffered saline (PBS; Sigma). Immunostaining with anti-CD45, anti-CD73, anti-CD34, and anti-CD90 (Abcam) human monoclonal antibodies was performed at 4°C for 30 min. After washing the cells twice with PBS, they were fixed in PBS with 1% paraformaldehyde. Flow cytometry was performed on the FACSCalibur (Becton Dickinson, New Jersey, USA) following the fixation. Histograms were created using WinMDI 2.8 software. The flow cytometry analysis indicated that MSCs expressed CD73 and CD90, but did not express CD34 and CD45 (Fig. 1C).
Intervention
Finally, a mixture, including MSCs (5 × 105 cells per mL) and SCs (5 × 105 cells per mL), was suspended in 6 mL of normal saline. Three weeks after the biopsy, patients in the treatment group were hospitalized for intrathecal cell injection. A lumbar puncture was performed at L4/L5 levels via a 24G needle at the operating theater. Following the needle insertion into the subarachnoid space (ascertained by observing the cerebrospinal fluid), the cellular mixture (6 mL) was injected slowly through the lumbar puncture. The needle was kept in place for 1 min to prevent the cerebrospinal fluid leakage. One hour after the procedure, patients were discharged.
Standard Care for Experimental Groups
Patients in both treatment and control groups received standard symptomatic therapy (pharmacological and non-pharmacological) for their neuropathic pain. All participants, whether in the intervention or control group, underwent surgical decompression and vertebral fixation during the acute phase of their injuries. Following the cell injections, no additional surgeries or interventions were performed, ensuring that the effects of the cell therapy could be accurately assessed without interference from other medical treatments. During the subacute and chronic phases, participants received a standardized rehabilitation regimen, comprising both physical and occupational therapy. This rehabilitation was administered uniformly across all participants to maintain consistency and avoid any potential influence on the study’s outcomes.
Outcome Measures
All the study outcome measures, including the ISCIPBDS and World Health Organization (WHO) Quality of Life Assessment Instrument (WHOQOL-BREF), were assessed at baseline and 6 months after the intrathecal injection3,17,18. The ISCIPBDS is a self-reported dataset that records pain intensity according to a 0 to 10 numeric rating scale (NRS), pain frequency, and location, as well as pain interference with sleep, mood, and daily activities over the last week. The pain interference items are scored from 0 (no interference) to 10 (extreme interference) 3 . The extended pain dataset was also evaluated in the present study, which includes pain type and common sensory abnormalities related to neuropathic pain (such as hyperalgesia, dynamic mechanical, and thermal allodynia) 18 . The WHOQOL-BREF is a self-reported questionnaire used to assess the patient’s QoL 17 . This questionnaire consists of 26 questions and four main domains (physical health, psychological health, social relationships, and environment). Besides the aforementioned domains, there are two separate questions concerning the general QoL and health. Each question is scored according to a 5-point Likert scale, and all domain scores are then linearly transformed into a 0–100 scale 19 . To perform this, WHO has provided an SPSS syntax, which was used in the present study. The validity of ISCIPBDS and WHOQOL-BREF in the SCI population has been demonstrated previously3,20–22.
Statistical Analysis
The mean pain intensity according to the 0–10 NRS scale was the primary outcome measure in this study. Based on a pilot population in our institute with a standard deviation (SD) of 1.5 for mean NRS scores, to observe a minimum difference of 1 between the two groups, a sample size of 74 patients (37 in each arm) was required (α = 0.05, power = 0.8). Per-protocol approach was utilized to analyze the outcome measures in this study. Quantitative data were reported as mean ± SD, and qualitative data were expressed as frequency and percentage. The Wilcoxon signed-rank test was performed to compare continuous variables between the baseline and 6 months after the injection. The same was performed for categorical variables using McNemar’s test. Differences in continuous variables between the two study groups, including treatment and control groups, were assessed using the Mann–Whitney U test. The same was performed for categorical variables using chi-square and Fisher’s exact tests. Moreover, subgroup analyses were performed based on the level of the injury and chronicity. The level of the injury included cervical and thoracolumbar SCI subgroups. The chronicity included subacute (<6 months elapsed since the injury) and chronic (≥6 months elapsed since the injury) subgroups. Both between-group (treatment vs control) and within-group changes were assessed in each subgroup. Multiple linear regression analysis was also applied to determine independent predictors of change in study outcome measures after 6 months using a backward stepwise method. Changes in the pain items and WHOQOL-BREF domain scores (6-month value—baseline value) were dependent variables, and independent variables included age, sex, level of the injury (cervical or thoracolumbar), chronicity (subacute or chronic), and study group (treatment or control). The model was also controlled for the effect of the baseline value of outcome measures on their change over time. All statistical analyses were performed using SPSS 24 (SPSS, Inc., Chicago, IL, USA), and P values <0.05 were considered statistically significant.
The graphical abstract was created using BioRender.com.
Results
Patient Characteristics
Between June 2022 and August 2023, a total of 103 patients were screened for eligibility in the present study (Fig. 2). Among them, 20 patients did not meet the study criteria and nine refused to participate. Therefore, 74 patients were included in the present study. A total of seven patients in the control group were lost to follow-up. Thus, 67 patients with SCI-related neuropathic pain were followed up for 6 months after the intrathecal injection, including 37 (55.2%) and 30 (44.8%) in the treatment and control groups, respectively. The mean age of the patients was 30.3 ± 8.5 and 30.0 ± 10.8 years in treatment and control groups, respectively. A total of 12 (32.4%) and 15 (50.0%) patients in treatment and control groups had cervical SCI, respectively (Table 1).

Consolidated Standards of Reporting Trials (CONSORT) 2010 study workflow.
Patients’ Clinical and Demographic Characteristics.

SCI: spinal cord injury.
Indicates statistically significant.
International SCI Pain Basic and Extended Data Set
No significant difference in ISCIPBDS items between the treatment and control groups was found at the baseline (Table 2). The mean scores of all three interference items of the treatment group, including daily activities (3.9 ± 2.4 vs 6.6 ± 1.8, P < 0.001), mood (4.2 ± 2.8 vs 6.4 ± 1.9, P < 0.001), and sleep (4.1 ± 2.8 vs 6.5 ± 2.2, P < 0.001), were significantly lower than the control group, at the 6-month follow-up (Fig. 3). Regarding changes over time, there was a significant decrease in mean interference scores of all three items in the treatment group at the 6-month follow-up (P < 0.05). However, no such change was found in the control group. The pain frequency over the previous week (3.9 ± 2.0 vs 5.5 ± 1.7, P = 0.002), mean (4.9 ± 2.3 vs 6.6 ± 2.0, P = 0.001), and worst (6.2 ± 2.4 vs 8.1 ± 1.8, P = 0.001) NRS pain intensity scores was also significantly lower in the treatment group compared with the control group 6 months after the injection. Furthermore, a significant reduction in the mean (P < 0.001) and worst (P = 0.002) NRS pain intensity scores was observed in the treatment group 6 months after the injection. The pain frequency over the previous week also decreased in the treatment group after 6 months, yet it was not significant (P = 0.062). Changes in different neuropathic pain-associated sensory abnormalities in patients who underwent combined cell therapy were also evaluated. In terms of dynamic light touch, the number of patients with absent sensation at (n = 19 vs n = 13; P = 0.031) and below (n = 22 vs n = 16; P = 0.031) the level of injury significantly reduced at the 6-month follow-up. Moreover, the number of patients with normal dynamic light touch sensation significantly increased (n = 5 vs n = 13; P = 0.008) at the 6-month follow-up. Regarding the pinprick sensation, the number of patients with absent sensation at the level of injury significantly reduced (n = 19 vs n = 12; P = 0.016) 6 months after the cell therapy. No significant change was observed in terms of cold sensation both at and below the level of injury among patients after 6 months. Table 3 demonstrates the details of changes in various neuropathic pain-related sensory abnormalities after 6 months among patients who underwent cell therapy.
Analysis of Changes in ISCIPBDS Items in Treatment and Control Groups.

NRS: numeric pain rating scale.
Significance of difference between the treatment and control groups using Mann–Whitney U test.
Indicates statistically significant.
Significance of changes at the 6-month follow-up compared with the baseline using Wilcoxon signed-rank test.

Changes in ISCIPBDS items, including interference with daily activities (A), mood (B), and sleep (C), as well as pain frequency (D), worst numeric rating scale (NRS) pain intensity score (E), and mean NRS pain intensity score (F) in treatment and control groups with subgroup analysis based on level of injury and chronicity.
Neuropathic Pain-Associated Sensory Abnormalities of Patients in the Treatment Group, According to the Extended Pain Dataset, Before and After the Stem Cell Therapy.

Significance of changes at the 6-month follow-up compared with the baseline using McNemar’s test.
Indicates statistically significant.
Subgroup Analysis of the Association Between Cell Therapy and Level of Injury
The level of injury was thoracolumbar in 25 (67.6%) and 15 (50.0%) patients in treatment and control groups, respectively. The cervical region was involved in other patients. Pain interference items in both thoracolumbar and cervical levels of injury in the treatment group were reduced at the 6-month follow-up, yet this reduction was only significant (P < 0.05) in the cervical group (Table 4). There was also a significant decrease in the mean (P = 0.001) and worst (P = 0.017) NRS pain intensity scores, as well as the pain frequency (P = 0.021) over the previous week in patients with the thoracolumbar level of injury in the treatment group. Patients with cervical injury demonstrated an insignificant decrease in the aforementioned pain items at the 6-month follow-up. No significant change in pain items was found over the study period in both cervical and thoracolumbar categories among patients in the control group (Figure 3).
Subgroup Analysis of Changes in ISCIPBDS Items in Treatment and Control Groups.

NRS: numeric rating scale.
Significance of difference between the treatment and control groups using Mann–Whitney U test (cervical level of injury/subacute injury).
Significance of difference between the treatment and control groups using Mann–Whitney U test (thoracolumbar level of injury/chronic injury).
Indicates statistically significant.
Significance of changes at the 6-month follow-up compared with the baseline using Wilcoxon signed-rank test.
Subgroup Analysis of the Association Between Cell Therapy and Chronicity
There was a significant reduction in pain interference items, including daily activities (P = 0.011), mood (P = 0.021), and sleep (P = 0.012), as well as the worst (P = 0.016) and mean (0.011) NRS pain intensity scores at the 6-month follow-up among patients with subacute injury in the treatment group (Table 4). Except for a significantly reduced mean NRS pain intensity score (P = 0.013) among patients with chronic SCI in the treatment group, no significant change in other items was observed in this group of patients, similar to both subacute and chronic subgroups in the control group. Pain interference items (P < 0.05), mean (P = 0.001), and worst (P = 0.008) NRS pain intensity scores were significantly lower in patients with subacute SCI in the treatment group compared with the control group at the 6-month follow-up (Figure 3). The pain frequency (P = 0.017) over the previous week, worst (P = 0.032), and current (P = 0.011) pain intensity scores of the chronic subgroup in the treatment group were significantly less than the control group at the 6-month follow-up.
QoL
Patients in the treatment group showed significant improvements in all WHOQOL-BREF domains, including the general perception of QoL (P = 0.026) and health (P = 0.019), physical (P = 0.032), psychological (P = 0.002), social (P = 0.034), and environment (P = 0.047) domains at the 6-month follow-up compared with the baseline (Table 5). No significant change was observed in WHOQOL-BREF domains in the control group at the 6-month follow-up. No significant difference was found in the baseline values of WHOQOL-BREF domains between the two study groups. The general perception of QoL or Q1 (P = 0.030), psychological (P = 0.010), social (P = 0.028), and environment (P = 0.025) domains was significantly higher in the treatment group compared with the control one at the 6-month follow-up.
Changes in WHOQOL-BREF Domains of All Patients.

Significance of difference between the treatment and control groups using Mann–Whitney U test.
Indicates statistically significant.
Multiple Regression Analysis
Multiple regression analysis controlled for age, sex, level of injury, chronicity, and baseline value of the outcome showed a significant association between the study group (treatment vs control) and changes (6-month value—baseline value) in all study outcomes, including pain items and WHOQOL-BREF domain scores. The reduction in pain interference scores, including daily activities (B = −1.59, P = 0.001), mood (B = −1.44, P = 0.012), and sleep (B = −1.74, P = 0.004), was significantly associated with the study group. Pain frequency in the previous week (B = −0.85, P = 0.014), mean (B = −1.68, P = 0.002), and worst (B = −0.91, P = 0.038) NRS pain intensity scores were also significantly associated with the study group. Similarly, changes in all WHOQOL-BREF domain scores, including the general perception of QoL (B = −19.53, P = 0.013) and health (B = −16.60, P = 0.040), physical (B = −11.30, P = 0.036), psychological (B = −14.37, P = 0.017), social (B = −12.25, P = 0.041), and environment (B = −9.89, P = 0.041) domains, 6 months after the injection were significantly associated with the study group.
Discussion
The major finding of the present study was the efficacy of a single combined intrathecal injection of autologous BMSC and SCs on both neuropathic pain and QoL improvement over 6 months in patients with SCI. Moreover, subgroup analysis revealed that the subacute injury was associated with improvement in pain-related outcomes. In addition to subgroup analysis, multiple regression showed that the study intervention (treatment group) was the main predictor of improvement in pain items and QoL. Thus, this study supports that intrathecal combined cell therapy would be a viable therapeutic option for SCI-related neuropathic pain.
To date, much attention has been paid to the effects of stem cell therapy on improvements in AIS and sensorimotor outcomes, yet its impact on neuropathic pain has been less investigated11,23. A limited number of reports have evaluated neuropathic pain after stem cell therapy24–26. Mendonça et al 25 assessed the safety and efficacy of direct transplantation of autologous BMSCs into the lesion in a phase I non-controlled clinical trial, including 14 patients with chronic traumatic complete SCI. They found a reduction in visual analog scale (VAS) scores and pain rating index in 67% of patients 6 months postoperatively, yet it was not statistically significant. Vickers et al 26 evaluated the safety and efficacy of local injection of ADSC in 10 patients with neuropathic trigeminal pain. In addition to safety, they found a significant reduction in mean NRS pain scores and the need for medication 6 months after the injection. In the study conducted by Vaquero et al 24 , 10 patients with chronic SCI and neuropathic pain received a total of 300 million autologous MSCs via three intrathecal injections (with a 3-month interval between doses). Similar to our findings, their results demonstrated a significant decrease in the VAS scores at 4, 7, and 10 months after the injection compared with the baseline. Another remarkable finding of the present study was the significant improvement in QoL, which was observed among patients who underwent cell therapy at the 6-month follow-up. This is in line with prior results showing a significant association between chronic pain conditions, in particular, neuropathic pain, and poorer QoL among patients5,6,27,28. Therefore, it is likely that cell therapy’s impact on pain reduction has contributed to the improvement in QoL of patients with neuropathic pain in this study. Although merely a limited number of studies have assessed the effects of stem cell therapy on neuropathic pain, the results are varying due mainly to the differences in methodology, such as the transplantation approach and cell type. The present investigation was the first to evaluate the combined intrathecal stem cell therapy with SCs and MSCs in both subacute and chronic SCI patients with neuropathic pain. Saberi et al 29 , in a study on 33 patients with complete SCI, found that intramedullary injection of SCs was safe with slight improvements in sensory and motor function after 2 years of follow-up. Preclinical findings have supported the combinatorial therapy with activated SCs and MSCs30,31. Yang et al 30 grafted multichannel scaffolds seeded with activated SCs, MSCs, or both activated SCs and MSCs into the transection gap of adult rats with complete transection of the thoracic spinal cord. They found significant functional and electrophysiological recovery in the combination group compared with the others 4 weeks after transplantation. They also observed significant neuronal differentiation of MSCs in the combination group compared with the group that received MSC alone.
The results of the present study showed a significant improvement in the pain items among SCI patients with neuropathic pain who underwent cell therapy. This finding might be attributable to distinct pathophysiologic mechanisms underlying the neuropathic and nociceptive pain categories. As the most common category among SCI patients, nociceptive pain results from the activation of peripheral nociceptors by noxious stimuli. The initial trauma could lead to many conditions, such as muscle spasm, contracture, and weakness, as well as joint and muscle overuse that can cause nociceptive pain. Neuropathic pain, however, is associated with a more complex mechanism that involves central, peripheral, and supraspinal mechanisms 32 . Reflective of this is the challenging treatment with current agents and monotherapy, which has demonstrated a limited therapeutic yield7–9.
Preclinical evidence suggests that MSCs alleviate neuropathic pain after SCI through multiple interconnected mechanisms, primarily involving immunomodulation and neuroprotection. MSCs possess potent anti-inflammatory properties by modulating glial cell activity, which is critical in central sensitization associated with neuropathic pain. They achieve this by inhibiting the activation of microglia and astrocytes and downregulating the expression of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6, which are implicated in pain sensitization and neuroinflammation. MSCs secrete anti-inflammatory proteins, such as TSG-6, which further suppress inflammatory signaling pathways like TLR2/MyD88/NF-κB in spinal microglia, thereby reducing neuroinflammation and alleviating neuropathic pain33,34. In addition, MSCs secrete neurotrophic factors like GDNF, which support neuronal survival, facilitate the repair of damaged neurons, and modulate pain signaling pathways, thereby reducing pain 35 . They also alleviate pain by downregulating purinergic P2X4 and P2X7 receptors, which decreases spinal microglia activation and subsequently mitigates chronic neuroinflammation 35 . Furthermore, MSCs exert direct neuroprotective effects by reducing oxidative stress and apoptosis in the spinal cord, modulating apoptotic markers such as the Bax/Bcl-2 ratio, and lowering the expression of inflammatory mediators such as cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS). These effects help prevent cellular damage and preserve neuronal integrity. Finally, MSCs promote the remyelination of injured nerves by differentiating into oligodendrocytes, which restores normal nerve function and reduces pain transmission. Taken together, these multifaceted actions of MSCs—modulating immune responses, reducing neuroinflammation, protecting neurons, and promoting tissue repair—represent a promising therapeutic strategy for managing neuropathic pain following SCI 36 .
Schwann cell transplantation alleviates neuropathic pain in SCI primarily by promoting the remyelination of damaged nerve fibers. In SCI, demyelination impairs nerve signal transmission and contributes to neuropathic pain by altering the molecular and structural integrity of the nerve fibers. For instance, the breakdown of myelin basic protein (MBP) fragments from the myelin sheath, facilitated by matrix metalloproteinase-1 (MT1-MMP), has been demonstrated to induce mechanical allodynia37,38. In addition, disruption of ErbB signaling in Schwann cells, which is vital for proper myelination, can lead to demyelination and subsequent mechanical hypersensitivity 39 . Schwann cells promote remyelination by supporting axonal regrowth, reducing inflammation, and restoring normal nerve conduction. A key player in this process is neuregulin-1 (Nrg1), a growth factor isoform expressed on the axonal surface, which binds to ErbB receptor tyrosine kinases on Schwann cells and serves as a crucial signaling molecule for myelination. The binding of NRG1 to ErbB2/3 receptors activates downstream signaling pathways, such as ERK1/2, PI3K/Akt, and Rac1/Cdc42, which facilitate cytoskeletal reorganization, cell survival, and myelination. The strength of the NRG1/ErbB signaling pathway is critical in determining the extent and efficiency of myelin production by Schwann cells39,40.
Co-transplantation of Schwann cells with MSC enhances remyelination and pain relief through synergistic mechanisms. MSCs release neurotrophic factors, such as Nrg1, which further activate the ErbB2/3 signaling pathways in Schwann cells, thereby increasing their capacity for myelination 41 . Moreover, MSCs contribute to creating a less inflammatory environment by secreting anti-inflammatory cytokines, including transforming growth factor-beta (TGF-β), which reduce the activation of glial cells and the production of pro-inflammatory cytokines like TNF-α and IL-1β34,41. In addition, MSCs upregulate neuroprotective factors such as BDNF and GDNF that support neuronal survival and facilitate tissue repair, thus fostering a regenerative environment 35 . The combination of Schwann cells with MSCs offers structural support for regenerating axons and promotes an optimal setting for remyelination and nerve repair. This combined therapeutic strategy is thought to accelerate the repair processes significantly, leading to more effective alleviation of neuropathic pain associated with SCI.
Furthermore, in line with preclinical studies, both between-group and within-group analyses indicated a significantly better response to cell therapy in patients with subacute injury compared with the chronic subgroup 6 months after the injection. Prior studies on rodent models of SCI have shown better cell survival when cells are administered in the subacute phase of SCI. This might be due to the reduction in the acute inflammatory response in addition to the absence of glial scar tissue, which forms in the chronic phase and impedes axonal regeneration and growth42–44. Some clinical investigations have reported a higher rate of neurological improvement among patients with subacute SCI45,46. A meta-analysis, however, demonstrated that no significant association exists between the timing of transplantation and improvement in some outcome measures 23 . Nevertheless, currently, there is a lack of evidence in this respect, and future high-quality clinical trials are highly warranted to evaluate the differences in response to cell therapy between subacute and chronic SCI patients. Moreover, according to our findings, both cervical and thoracolumbar levels of injury had improvements in various pain items, yet this was more significant among the patients with cervical SCI. Similarly, Muthu et al 23 , in their meta-analysis, found significant improvements in AIS grade for both cervical (P = 0.002) and thoracolumbar (P = 0.041) subgroups with a greater effect in the cervical level of injury. However, further studies are highly required to validate these findings.
Another remarkable finding of the present study was the significant improvement in QoL, which was observed among patients who underwent cell therapy at the 6-month follow-up. Interestingly, patients in the treatment group also showed substantially higher scores in QoL items compared with the individuals in the control group 6 months after the injection. Many prior studies have shown a significant association between chronic pain conditions, in particular, neuropathic pain, and poorer QoL among patients5,6,39,40 Attal et al, in a nationwide survey of 3816 subjects, found significantly lower QoL and sleep with higher anxiety and depression scores among individuals with neuropathic pain than those with non-neuropathic pain or without pain. 41 Burke et al 6 , in another nationwide survey including 643 subjects with SCI-related chronic pain, found a significantly poorer QoL among individuals with neuropathic pain compared with respondents who reported nociceptive or no pain. They also found a significant association between pain interference items and WHOQOL-BREF domains. Accordingly, the cell therapy’s impact on pain reduction has likely contributed to the improvement in QoL of patients with neuropathic pain in the present study. Moreover, patients showed significant reductions in all QoL domains. The psychological domain changed most in comparison with other domains. In this regard, previous investigations have shown strong associations between the QoL of patients with chronic SCI-related pain and various psychologic aspects such as pain catastrophizing, SCI helplessness, and depression41–43.
The present study has some limitations, some of which are common among clinical trials evaluating stem cell therapy in SCI. First, this study was not placebo-controlled or blinded due to its methodological nature. We extracted cells from an autologous source, with patients undergoing sampling from bone marrow and the sural nerve. Consequently, patients were aware of the sampling process. Performing bone marrow aspiration and sural nerve harvesting, which are invasive procedures, without a medical purpose would have been ethically inappropriate. Second, it is uncertain whether the clinical improvements in subacute patients are due to treatment itself or the spontaneous recovery that follows the acute injury, which could be a potential confounding factor. In respect of this, a multiple regression was performed, and changes in outcome measures were controlled for most potential confounders, such as chronicity, duration of the injury, and baseline value of the outcome measure. Third, the follow-up period was 6 months. Therefore, future studies are needed to evaluate the safety and efficacy of this intervention in long-term follow-up. Fourth, this study was performed in one center, which mandates future multicenter studies to assess the efficacy of this intervention in different clinical care settings. Fifth, the present study did not include single-cell arms to compare the effects of individual administration of MSCs or SCs with their combination or placebo. Hence, future clinical trials with multiple arms could provide evidence on the additional clinical benefit achieved through the combination of these cell types compared with their isolated use in patients with SCI. Despite the above limitations, this study has strengths. First, the sample size was larger than prior studies assessing the efficacy of cell therapy in SCI and resultant neuropathic pain, specifically. Second, the present study was the first to assess the efficacy of intrathecal combined cell therapy with SCs and MSCs in SCI-related neuropathic pain.
Conclusions
Single intrathecal combined cell therapy with SCs and BMSCs safely led to notable improvements in various aspects of pain, in addition to QoL of patients with SCI-related neuropathic pain. Further investigations with long-term follow-up period are highly warranted. Moreover, future investigations should consider evaluating combinatorial approaches by using this therapeutic strategy along with other existing treatments for SCI-induced neuropathic pain.
Footnotes
Author Contributions
MA: Conceptualization, Methodology, Investigation; RT: Conceptualization, Methodology, Writing-review and editing; MH: Writing original draft, Investigation; RH: Investigation; IM: Investigation; MC: Data curation; MH: Formal analysis; AZ: Investigation; RM: Supervision; SO-Y: Supervision, Project administration.
Ethics Approval
The present study was a randomized, active-controlled, parallel-group phase II clinical trial designed in accordance with the Declaration of Helsinki and was approved by the ethics committee of the Shahid Beheshti University of Medical Sciences (IR.SBMU.MSP.REC.1401.053) and Aja University of Medical Sciences (IR.AJAUMS.REC.1402.215). This clinical trial was also registered at the Iranian Registry of Clinical Trials (IRCT20200502047277N8).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
All procedures in this study, including study intervention, cell culture, and biopsy specimen analysis, were performed after obtaining written informed consent. Patients were completely informed of the study intervention and its experimental nature, as well as potential adverse events, such as infection, autonomic dysreflexia, motor or sensory deficit, and spasticity.
Consent for Publication
Not applicable.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by Shahid Beheshti University of Medical Sciences. The funding source had no role in writing of the manuscript or decision to submit it for publication and any other part of the study.
