Abstract
Dry eye is one of the most common chronic diseases in ophthalmology. It affects quality of life and has become a public health problem that cannot be ignored. The current treatment methods mainly include artificial tear replacement therapy, anti-inflammatory therapy, and local immunosuppressive therapy. These treatments are mainly limited to improvement of ocular surface discomfort and other symptoms. In recent years, regenerative medicine has developed rapidly, and ophthalmologists are working on new methods to treat dry eye. Mesenchymal stromal cells (MSCs) have anti-inflammatory, tissue repair, and immune regulatory effects, and have become a promising tool for the treatment of dry eye. These effects can also be produced by MSC-derived exosomes (MSC-Exos). As a cell-free therapy, MSC-Exos are hypoimmunogenic, serve more stable entities, and compared with MSCs, reduce the safety risks associated with the injection of live cells. This article reviews current knowledge about MSCs and MSC-Exos, and highlights the latest progress and future prospects of MSC-based therapy in dry eye treatment.
Introduction
Dry eye is a chronic and progressive disease, which can cause various forms of ocular discomfort and/or visual dysfunction. Globally, the incidence of dry eye ranges from 5.5% to 33.7% 1 . It is estimated that the total cost of dry eye patient management calls in the US health care system is US$54.4 billion per year 2 . Dry eye has become the most common chronic disease in ophthalmology, so it has become a public health problem that cannot be ignored.
Dry eye is characterized by an unstable tear film or unbalanced ocular surface microenvironment. The main functions of the tear film include lubrication, nutrition, cleansing, maintaining the optical properties of the cornea, and resisting bacterial infections of the cornea and conjunctiva. The human tear film contains secretions from specific tissues on the ocular surface (Fig. 1). Meibomian gland secretions are composed of tear film lipids, the aqueous layer is mainly secreted by the lacrimal gland, and mucin by the conjunctival goblet cells 3 . Dysfunction of these tissues may cause abnormal secretions, resulting in tear film instability 4 . At present, it is believed that local inflammation and immune response are the important mechanisms leading to pathological damage in dry eye 5 . Therefore, the medical treatment of dry eye usually focuses on topical drugs such as cyclosporine and glucocorticoids to reduce inflammation and immune response 6 . However, dry eye is a chronic progressive disease. These treatments can temporarily relieve the ocular surface symptoms, but they cannot completely resolve them. In recent years, regenerative medicine has developed rapidly, and scientists have explored other dry eye treatment methods such as stem cell–based therapy.
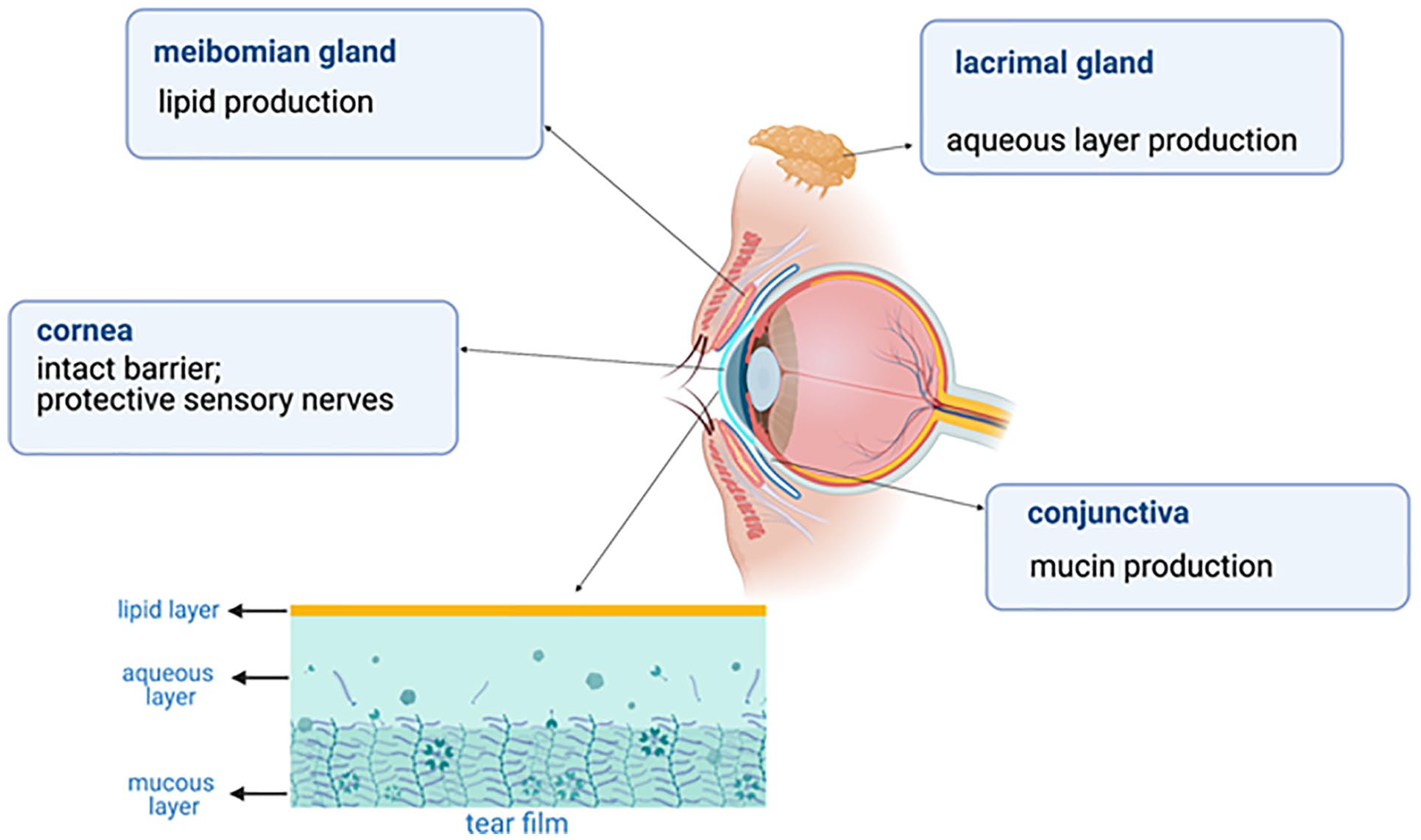
The composition of the tear secretion unit, the function of each tissue, and the structure of the tear film.
Mesenchymal stromal cells (MSCs) isolated from different tissues have been shown to show distinct differentiation potentials. MSCs can be isolated from bone marrow, placenta, fat, umbilical cord, and other tissues7,8. The anti-inflammatory, immune regulatory, and tissue regeneration potential of MSCs has been widely studied. MSCs can interact directly with target tissues through intercellular contact9,10. In addition to direct effects, they can also produce indirect effects through paracrine functions to modify the microenvironment.
Despite the clear advantages of MSC transplantation, the risks of allograft and cell rejection and small vessel lodging and obstruction remain 11 . However, exosomes can avoid these potential risks. Exosomes are small membrane vesicles (30~150 nm) formed by intracellular endocytic vesicle membrane compression and are released into the extracellular environment by fusion with the membrane 12 . MSC-derived exosomes (MSC-Exos) have functions similar to MSCs and are more secure, stable, and easy to store than cells. Therefore, MSC-Exos therapy as a cell-free therapy has been developed as a safer and more advantageous alternative to MSC therapy, and some research results have demonstrated these benefits.
Recently, MSCs and MSC-Exos have been widely used in the treatment of clinical diseases, and stem cell therapies have attracted increasing attention from ophthalmologists. The purpose of this article is to explore the latest research findings, potential applications, and challenges related to MSC-based treatment of dry eye. In view of the evidence that MSCs have roles in inflammation, immune regulation, and tissue regeneration, the treatment of dry eye based on MSCs shows promise, as described in this article.
Dry Eye
Pathology of Dry Eye
The pathogenesis of dry eye is complex and remains unclear. In 2017, the Tear Film and Ocular Surface Dry Eye Workshop (TFOS DEWS) II redefined dry eye as the following: a multifactorial disease of the ocular surface characterized by a loss of homeostasis of the tear film, and accompanied by ocular symptoms, in which tear film instability and hyperosmolarity, ocular surface inflammation and damage, and neurosensory abnormalities play etiological roles
3
.
The current research on the mechanism of dry eye is mainly focused on the following aspects.
Inflammation in Dry Eye
Inflammation has long been considered one of the main pathogenic factors in dry eye 13 . Measurement of the levels of tear cytokines in patients with dry eye provides evidence of ocular surface inflammation. Previous studies showed that the levels of interleukin (IL)-1, IL-4, IL-6, matrix metalloproteinase-3 (MMP-3), MMP-9, IL-8, IL-10, IL-17A, tumor necrosis factor (TNF)-β, and TNF-α in patients with dry eye were higher than those in controls, and were correlated with the severity of dry eye14–18. Among them, the expression of IL-6 was particularly high in dry eye. After IL-6 binds to the receptor, it can activate phosphorylation signal transduction, induce the transcription of activator-3, make Th17 cells secrete IL-17 and other inflammatory factors, and aggravate the inflammatory response and cell apoptosis 19 . In addition, IL-17 promotes the release of MMP-3 and MMP-920,21. MMPs involved in wound healing and inflammation. During the development of dry eye, MMPs break down the corneal barrier by disrupting the tight junctions between corneal epithelial cells.
In 2017, Rhee and Mah 22 proposed a vicious circle of dry eye: multiple factors in the ocular and external environment cause tear film instability, resulting in tear hyperosmolarity, leading to corneal and conjunctival apoptosis, triggering neurogenic inflammation, and then stimulating the lacrimal glands and meibomian gland, further aggravating tear film instability. Therefore, anti-inflammatory therapy has always been an essential part of dry eye treatment.
Immune Response of Dry Eye
T cells and related factors play a vital role in pathogenesis of dry eye. The ocular surface tissues in dry eye express high levels of C-C motif chemokine ligand (CCL) 3, CCL5, CCL20, and other Th cell chemokines, which can induce the migration of Th cells to the ocular surface. This may be one of the pathways for pathological CD4+T cells to reach and infiltrate the ocular surface. A study by De Paiva et al. 23 showed that both Th1 and Th17 cells exist in the goblet cell–rich area of the conjunctiva, accompanied by high expression of interferon-γ (IFN-γ) and IL-17. Strategies that inhibit Th17 cell function or target IL-17 can significantly reduce the severity and progression of dry eye 24 . In dry eye, Th17 cell homing is mediated by CCL20. IFN-γ can significantly reduce mucin secretion of goblet cells, which is important for maintaining immune tolerance on the ocular surface24,25. Regulatory T cells (Tregs) suppress the immune response by suppressing autoreactive T cells and dry eye.
Other Pathologies of Dry Eye
Dysfunction of any link in the nerve conduction pathway may trigger dry eye disease. The cornea is densely populated with sensory nerves. Changes in the ocular surface microenvironment (such as increased tear osmotic pressure and release of tear inflammatory factors) reach the nerve center through afferent signals, and efferent innervation stimulates ocular surface gland secretion and blink activity. Normal blinking allows the tear film to spread over the entire ocular surface 26 . However, abnormal corneal nerve function will further aggravate ocular surface injury, leading to the persistence of dry eye–related inflammation 27 . In addition, studies have demonstrated that conjunctival goblet cell secretion granules are regulated by peripheral sympathetic or parasympathetic nerves. Normal conjunctival nerve function is an important factor in regulating goblet cell secretion and maintaining ocular surface function 27 . In addition, previous studies have shown that oxidative stress can induce damage to ocular surface tissue in dry eye. Tear film components also reportedly change seasonally with the increase of 8-hydroxy-2′-deoxyguanosine (8-OHdG) and malondialdehyde (MDD) 28 .
MSCs and MSC-Exos
MSCs have been widely studied due to their easy access and wide range of sources. The physiological functions of MSCs have made MSC therapy a potential treatment for many diseases. The Mesenchymal and Tissue Stem Cell Committee of the International Society for Cellular Therapy has proposed minimum standards for defining human MSCs for laboratory scientific investigation and preclinical research. First, MSCs must be plastic-adherent under standard culture conditions. Second, MSCs must express CD105, CD73, and CD90, lacking expression of CD45, CD34, CD14, CD11b, CD79alpha, or CD19, and human leukocyte antigen (HLA)-DR surface molecules. Third, MSCs must differentiate in vitro into osteoblasts, adipocytes, and chondrocytes 29 .
Sources of MSCs
MSCs are pluripotent stromal cells that exist in various tissues throughout the development process. To date, MSCs have been isolated from adipose tissue, umbilical cord, dental tissue, bone marrow, cornea, skeletal muscle, synovium, and periosteum 8 . Among them, bone marrow (BM)-MSCs, adipose-derived (AD)-MSCs, umbilical cord (UC)-MSCs, and cornea-derived MSCs are widely used in laboratory and clinical research (Fig. 2).

The derivation of the four types of MSCs and their therapeutic application in dry eye. MSC: mesenchymal stromal cell.
The Biological Characteristics of MSCs and MSC-Exos
Immunogenicity of MSCs
MSCs have low immunogenicity; they are able to escape the immune system recognition mechanisms and evade the host’s immune defense 30 . According to the results of previous studies, MSCs have low expression of major histocompatibility complex (MHC) I and MHC II, and do not express CD40, CD40L, B71, B72, or other costimulators. These characteristics enable MSCs to escape the surveillance of the immune system and enter the body without being rejected by the immune system, an advantage known as immune privilege. Therefore, the infusion of MSCs can reduce the autoimmune side effects caused by engraftment. However, recent studies have demonstrated generation of antibodies against and immune rejection of allogenic MSCs, suggesting that MSCs do not have absolute immune privilege 30 .
Immunoregulatory and anti-inflammatory effects of MSCs
In the resting state, MSCs tend to be immunosuppressed and display characteristics of immune homeostasis. When MSCs are exposed to various pro-inflammatory factors, such as TNF-α and IL-1α, their immunosuppressive properties are significantly enhanced, leading to their differentiation into an immunosuppressive phenotype. MSCs can regulate a variety of immune cells and exert immune regulatory and anti-inflammatory effects.
MSCs can inhibit T cell proliferation, activation and differentiation, and induce T cell apoptosis and Treg recruitment31,32. Previous studies have shown that MSCs can increase the proportion of CD8(+)CD28(−) T cells and enhance their ability to inhibit the proliferation and activation of primary CD4(+) T cells. MSCs reduce the production of IFN-γ by activating CD4(+) T cells and induce apoptosis of activated CD4(+) T cells 33 . Present studies have shown that the effect of MSCs on immune balance is related to IFN-γ-induced enzyme indoleamine 2,3-dioxygenase (IDO) and other molecules34–36. Many studies have found abnormal regulation of IDO in patients with autoimmune diseases such systemic lupus erythematosus and Sjögren’s syndrome (SS)35,37. IDO represses effector T cells by finely modulating innate and adaptive immune responses by degrading the tryptophan to kynurenine and other downstream metabolites 38 . The kynurenine metabolic pathway can be stimulated by IFN-γ and other cytokines to activate IDO in SS, thereby interfering with serotonergic and glutamatergic neurotransmission, thus affecting the ocular surface34,39.
MSCs can also induce cell cycle arrest by downregulating CuclinD2 and upregulating p27Kip1 in T cells, resulting in the inability of activated T cells to divide. In addition, MSCs inhibit the proliferation and cytotoxicity of natural killer (NK) cells, suppress differentiation and maturation of dendritic cells (DCs), and the infiltration and apoptosis of neutrophils (Fig. 3). This is mainly achieved through the secretion of cytokines, including IL-10, human leukocyte antigen-G5 (HLAG), transforming growth factor-β (TGF-β), prostaglandin E2, IDO, and inducible nitric oxide synthase (iNOS)30,40. Therefore, MSCs have been usedto treat several autoimmune diseases based on theirimmunoregulatory and anti-inflammatory properties41,42.
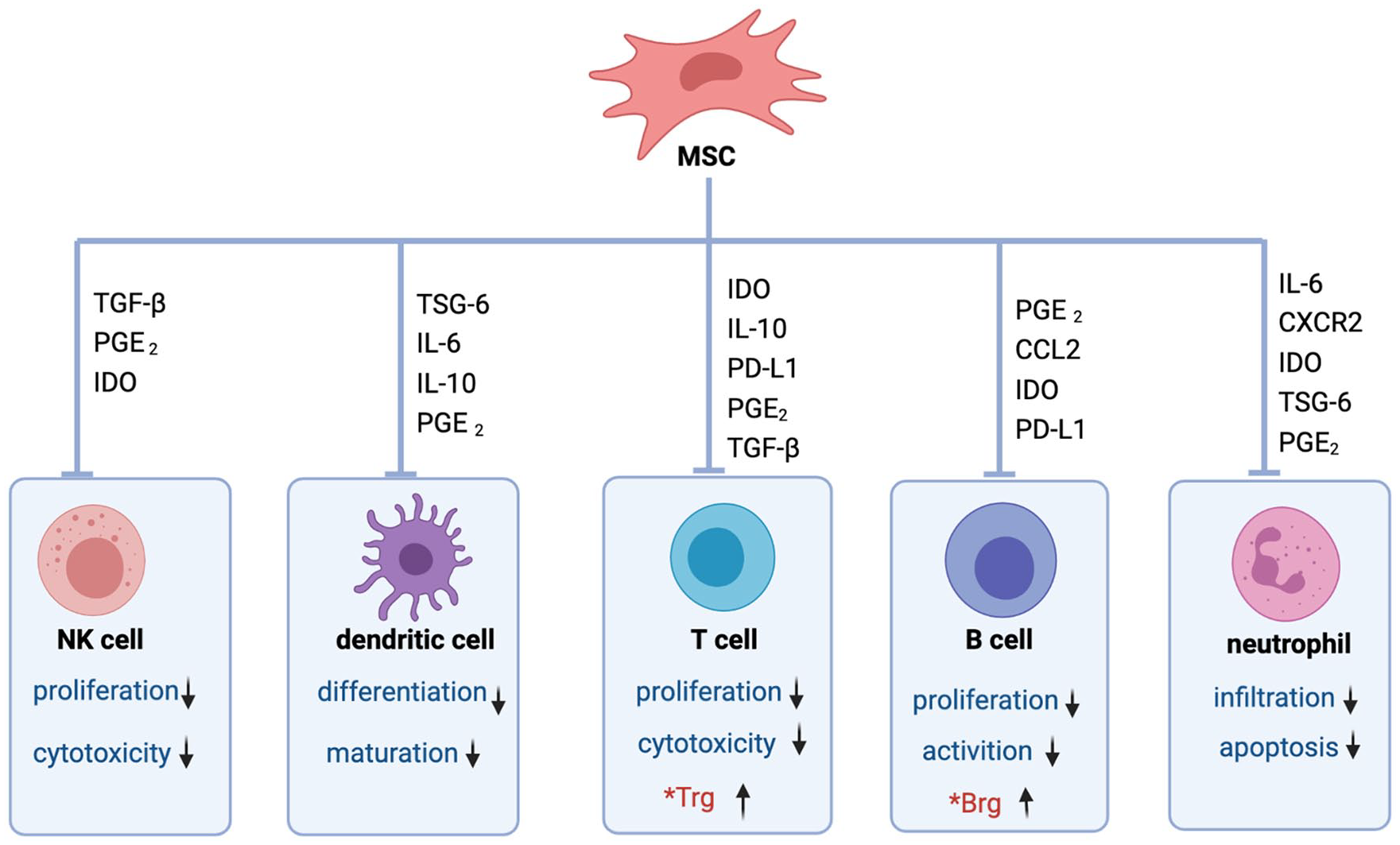
MSCs regulate the immune response through soluble factors and direct cell-to-cell contact. MSCs inhibit the proliferation and cytotoxicity of NK cells, suppress dendritic cells’ differentiation and maturation, inhibit the proliferation and cytotoxicity of T cells, inhibit the proliferation and activation of B cells, inhibit the infiltration and apoptosis of neutrophil, and also induce differentiation of Tregs and Bregs. Bregs: regulatory B cells; CCL: C-C motif chemokine ligand; IDO: indoleamine 2,3-dioxygenase; PD-L1: programmed cell death ligand; IL: interleukin; MSCs: mesenchymal stromal cells; NK: natural killer; PGE2: prostaglandin E2; TGF-β: transforming growth factor-β; Tregs: regulatory T cells; TSG-6: tumor necrosis factor-α stimulated gene/protein 6.
Tissue regeneration by MSCs
MSCs also have potential for tissue repair, with important possibilities in the treatment of several diseases. To date, regenerative medicine has been widely used to treat many organs in clinical practice, including transplantation of tissue-derived MSCs and treatment by stem cell–derived cytokines, using the potent tissue repair potential of MSCs. When tissues are damaged, stressed or necrotic cells release pro-inflammatory cytokines and chemokines. MSCs can secrete numerous chemokine receptors, and after binding to chemokines, MSCs migrate to damaged tissues. This characteristic is known as homing ability43,44, but the specific mechanism of this ability in MSCs has not been fully described. When MSCs migrate to injured tissue, they differentiate into osteoblasts, fibroblasts, or other cells needed for tissue repair under the regulation of the local environment 45 . Concurrently, MSCs secrete many cytokines related to tissue repair, including vascular endothelial growth factor (VEGF), fibroblast growth factor 2 (FGF-2), insulin-like growth factor 1 (IGF-1), and hepatocyte growth factor (HGF)9,46. These characteristics of MSCs make collaborative contributions to tissue repair.
MSC-Exos
The exosomes are composed of nucleic acids, proteins, and lipids. Fundamentally, an exosome is an antigen-presented vesicle with a long circulating half-life. Studies have shown that MSC-Exos themselves can act as therapeutic entities in tissue repair, immunomodulation, and in anti-inflammatory function 47 . MSC-Exo carries anti-inflammatory cytokines and miRNAs and exerts anti-inflammatory effects. Previous studies have shown that exosomes can suppress the TLR4-MyD88-NF-κB pathway, reduce the levels of pro-inflammatory factors (IL-1β, IL-6, TNF-α, and IL-12), and increase the levels of anti-inflammatory factors (IL-10 and TGF-β)48,49. MSC-Exos can also regulate T cells, B cells, NK cells, DCs, and other immune cells50–52.
Furthermore, exosomes have the advantages of high permeability, stability, and low tumorigenicity, which compensate for the limitations of MSC treatment 53 . Exosomes can freely pass through various biological barriers without blocking microvascular circulation, and they have stability, making them ideal drug carriers 54 . In addition, MSC-Exo is considered nonimmunogenic so that it can be used in clinical applications on a large scale. However, there are few toxicological studies on exosomes, and their application in vivo remains to be fully evaluated (Fig. 4).

The functions of MSC-derived exosomes, their advantages, and challenges in clinical application. MHC: major histocompatibility complex; MSCs: mesenchymal stem cells.
Application of MSCs in Dry Eye Treatment
According to the dry eye consensus of the Asia Dry Eye Society, tear film instability is the core mechanism of dry eye. Tear film stability is maintained by various ocular tissues such as those of the lacrimal gland, conjunctiva, cornea, and eyelid 55 , dysfunction of which will affect tear film homeostasis and may lead to dry eye syndrome. As explained earlier, this may in turn damage the abovementioned tissues and exacerbate the syndrome in a vicious circle.
Many studies have been conducted on MSCs in the dry eye treatment. The goal of MSCs in dry eye treatment is to regenerate new tissue to repair the corneal, conjunctival, and other damage caused by inflammation, and to restore tear film stability. Recently, several preclinical in vivo studies have shown that MSCs have significant potential in the treatment of dry eye (Table 1).
A Summary of Methods Used in In Vivo Studies on MSCs in the Treatment of Dry Eye Syndrome.

AD-MSCs: adipose-derived mesenchymal stem cells; BM-MSCs: bone marrow–derived mesenchymal stem cells; MSCs: mesenchymal stem cells; N/A: not applicable; UC-MSCs: umbilical cord–derived mesenchymal stem cells.
Some studies have also applied MSCs in clinical practice to explore their therapeutic effect on human dry eye61,62. Michael et al. injected allogeneic AD-MSCs into the lacrimal glands of five patients with aqueous-deficient dry eye. They found that the tear secretion increased and the tear film osmolarity decreased after treatment 63 . Weng et al. 64 used intravenous MSCs to treat dry eye and found that MSCs were a safe and potentially effective method for the treatment of cGVHD53 secondary dry eye. As of February 2022, there were three clinical trials on the ClinicalTrials.gov website (Table 2). In previous studies, MSC treatment of ocular surface diseases mainly included tissue transplantation 65 , periorbital injection57,63, topical drops 66 , and intravenous injection 67 (Fig. 2).
Ongoing Clinical Trials Using Mesenchymal Stem Cells in Dry Eye Treatment, Sourced From the Clinical Trials Database ClinicalTrials.gov (February 2022).
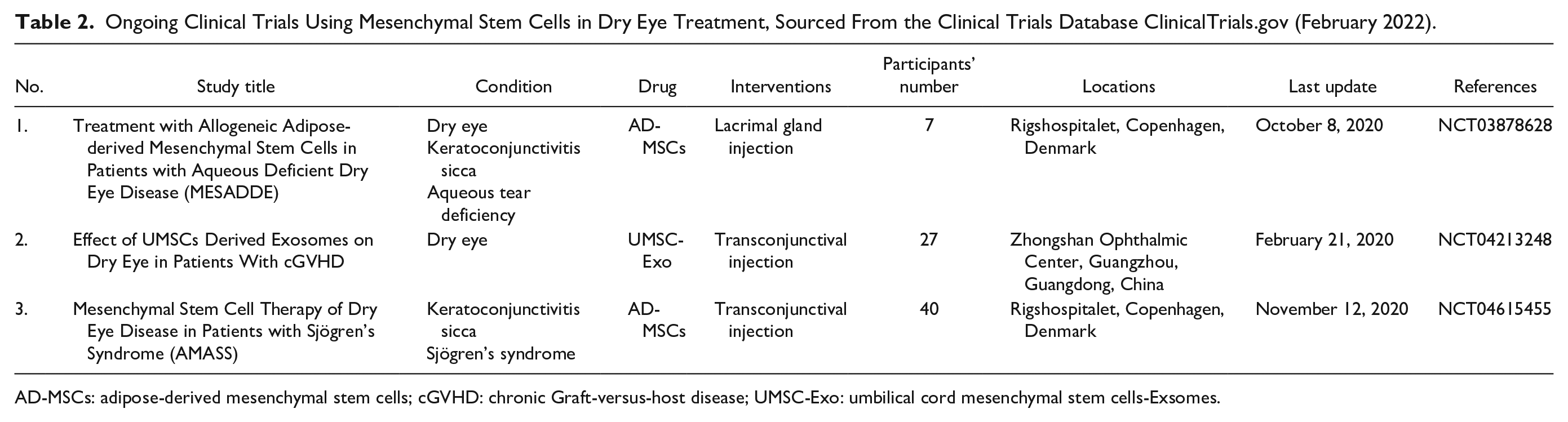
AD-MSCs: adipose-derived mesenchymal stem cells; cGVHD: chronic Graft-versus-host disease; UMSC-Exo: umbilical cord mesenchymal stem cells-Exsomes.
The Role of MSCs in Corneal Repair
Corneal inflammation, nerve damage, and limbal stem cell deficiency (LSCD) occur with dry eye, seriously affecting patients’ vision and quality of life. Since MSCs have anti-inflammatory, regenerative, and neuroprotective properties, scientists have explored their applications in corneal repair63,68–71. In addition, MSCs have immunoregulatory capacity and they can induce immunosuppression in vivo and in vitro, making them a promising option for ocular surface reconstruction and dry eye72–74.
Application of AD-MSCs in corneal tissue
Adipose tissue has been the main source of MSCs for research. Previous studies have shown that AD-MSCs have a significant impact on wound healing and regenerative medicine. Galindo et al. 75 used amniotic membrane containing human AD-MSCs for corneal transplantation to treat rabbit LSCD, and found that the ocular surface has good tolerance to human AD-MSCs and that human AD-MSCs have anti-inflammatory and anti-angiogenic effects. Other studies have used AD-MSCs or the acellular products of AD-MSCs after in vitro culture for corneal transplantation, including allograft, xenograft, and autologous AD-MSC corneal transplantation, demonstrating their potential for this purpose76–78. Conversely, a previous in vivo study found that local or intravenous administration of AD-MSCs during corneal transplantation did not improve graft-versus-host response and instead increased inflammation and neovascularization 79 . The discrepancy may be due to the use of different injection sites and different amounts of MSCs.
AD-MSCs may also act indirectly through paracrine signaling. It is known that AD-MSCs can produce paracrine factors, such as VEGF, IGF, and TGF-β, with the potential to improve tissue wound healing80,81. In the treatment of corneal injury, paracrine factors of AD-MSCs can promote corneal repair by reducing immune infiltration 82 . A study which cocultured AD-MSCs with human corneal epithelial cells found that AD-MSC secretions inhibited TGF-β-induced corneal epithelial–mesenchymal transition 83 .
Application of BM-MSCs in corneal lesions
MSCs were originally isolated from bone marrow, and current research focuses on BM-MSCs. Many studies to date have focused on corneal treatment with BM-MSCs, including subconjunctival injection, intravenous injection, and corneal transplantation. Among them, a clinical trial of corneal transplantation using allogenic MSCs conducted by Calonge et al. 68 in 2018 showed that BM-MSCs could promote the proliferation of corneal epithelium, which verified the effectiveness of BM-MSCs in the treatment of LSCD. A number of studies have shown that in the repair of corneal injury, BM-MSCs exert anti-inflammatory, anti-fibrosis, and proregeneration effects through the secretion of TSG-6 and HGF67,70,84,85. Sharad K. M et al. treated corneal injury in mice with intravenous BM-MSCs and found that MSCs have the capacity to inhibit the generation of myofibroblasts, and the corneal transparency is also restored due to high levels of HGF secretion. HGF was significantly upregulated on stimulation with recombinant human IL-1β 86 . In addition, BM-MSCs can reduce corneal oxidative stress 87 .
Application of UC-MSCs in corneal lesions
UC-MSCs are derived from umbilical cord blood, which is easier than other MSC sources to collect and more abundant in supply, alleviating ethical disputes.
Azmi et al. 88 cocultured human UC-MSCs with corneal epithelial cells and found that the MSCs promoted epithelial cell growth and function by downregulating the expression of HLA class I and diphenylamine IFN-γ stimulated human telomerase perpetuators. Park et al. 71 found less ocular damage and decreased cell apoptosis in mice intravenously injected with UC-MSCs. UC-MSCs tend to aggregate and attach to the damaged tissue area, and differentiate into cell types according to their specific microenvironment. Compared with BM-MSCs, the expression profile of UC-MSCs is closer to that of embryonic stem cells, which can differentiate into corneal epithelial, stromal, and endothelial cells. UC-MSCs also have low immunogenicity and low incidence of rejection and graft-versus-host disease. Therefore, many studies have used UC-MSCs for corneal transplantation 89 .
However, the current methods for isolating and culturing stem cells from the umbilical cord are cumbersome, resulting in low yields of primary cells, and limiting the application of UC-MSCs in corneal therapy 90 .
Application of cornea-derived MSCs in treatment of corneal lesions
Cornea-derived MSCs are mainly distributed in the anterior part of the corneal stroma near the limbal stem cells. They are MSCs with multidirectional differentiation potential. Research has shown that they can induce differentiation into corneal cells in vitro 91 , but there is some controversy about this. In 2005, a team from the University of Pittsburgh isolated and identified corneal stromal mesenchymal stem cells and found them to have a greater potential to differentiate into corneal cells in vitro than BM-MSCs, AD-MSCs, or UC-MSCs 92 . Multiple studies have also shown that coculture of cornea-derived MSCs with limbal epithelial cells is superior to culture with limbal epithelial cells alone 93 .
The therapeutic effect of cornea-derived MSCs is not yet clear. Chronic corneal inflammation in patients with dry eye often leads to corneal interstitial damage and fibrosis, scarring, and reduced transparency. Stem cells derived from the limbal stroma may avoid the formation of corneal scars 94 , but the mechanism by which MSCs act against fibrosis has not yet been elucidated. With the progression of dry eye, further damage to the cornea may lead to the generation of corneal neovascularization. Eslani et al. 95 used human corneal stem cells to treat mouse cornea and found that cornea-derived MSCs can induce macrophage apoptosis by secreting factors such as pigment epithelial–derived factor, thereby inhibiting corneal neovascularization.
The Role of MSCs in Conjunctival Repair
Goblet cells in the conjunctival epithelium secrete mucosal protein, which is the main component of the tear film mucoprotein layer. The loss of goblet cells will destroy the stability of the tear film, while the activation of inflammatory factors and the reduction of tear nutrient factors will change the differentiation of the conjunctiva and reduce goblet cells23,96. The loss of goblet cells in the conjunctival epithelium is a widely recognized feature of dry eye97–99. Severe dry eye, such as in Stevens–Johnson syndrome, may feature a complete loss of conjunctival goblet cells. Previous studies have shown that the number of conjunctival goblet cells increases after treatment with MSCs in animal dry eye models57,66. Lee et al. 100 used periorbital injections of BM-MSCs to treat a mouse model of dry eye induced by an intraorbital injection of concanavalin A, and found increased tear secretion and goblet cell numbers and decreased levels of inflammatory factors on the ocular surface after treatment.
The Role of MSCs in Lacrimal Gland Repair
Lacrimal glands play an important role in the physiological and pathological processes of the ocular surface. Abnormalities such as lacrimal gland inflammation and apoptosis can lead to decreased tear secretion. Some studies have shown that MSCs can promote lacrimal gland regeneration and increase tear secretion 101 . Villatoro et al. 102 injected AD-MSCs into the lacrimal glands of dogs with dry eye and found improvement in dry eye signs and good tolerance. Michael et al. conducted a clinical trial in which allograft AD-MSCs were injected into the lacrimal glands of patients with severe lacrimal defects. The results similarly showed an improvement in signs and symptoms with good tolerance 63 .
The Role of MSCs in Meibomian Gland Repair
The meibomian gland secretes meibum, the main component of the tear lipid layer which acts as a barrier to the evaporation of tear fluid. In meibomian gland dysfunction (MGD), the quality and quantity of the meibum will change, thereby negatively affecting the tear film stability, leading to reduced tears and dry eyes. Beyazyıldız et al. used MSCs to treat a benzalkonium chloride–induced rat model of dry eye and found that the ocular surface condition of the rats was improved after treatment. MSC infiltration into cells could be seen in the eyelid gland, but their therapeutic effect on the gland has not yet been demonstrated 66 .
Application of MSC-Exos in Dry Eye Treatment
Over the last decade, research on MSC-Exos has made great progress in the field of regenerative medicine. Various regulatory functions of MSC-Exos have been reported, including differentiation, inflammation, and immunosuppression73,103. In addition, exosomes secreted from stromal stem cells have been shown to have anti-fibrosis, anti-inflammatory, and regenerative effects on injured corneas104,105. Shen et al. 106 found greater proliferation and less apoptosis in rabbit corneal stromal cells cultured with rabbit adipose MSC-Exos than in those without MSC-Exos. Wang et al. 58 treated dry eye in mice using mouse AD-MSC-derived exosomes (mADSC-Exos) and found that mADSC-Exos could decrease the cytokines, promote corneal epithelial repair, and increase tear secretion by inhibiting the NLRP3-IL-1β signaling pathway.
Previous laboratory studies have shown that cornea-derived MSCs can prevent scar formation after corneal injury in mice, stimulate the regeneration of hyaline interstitial tissue, and this function depends on exosomes to deliver miRNAs 105 . In addition, the human cornea-derived MSCs can be absorbed by corneal epithelial cells and accelerate the healing of corneal injury 104 . Zhou et al. used MSC-Exos to treat chronic Graft versus host disease-associated dry eye in mice and refractory dry eye in patients.The study demonstrated that MSC-Exos could relieve dry eye symptoms and found that Mir-204 contained in MSC-Exos played a role in the treatment 107 . Previous studies have shown that microRNAs including mioRNA-466, miRNA-205, miRNA-122, and others can promote the healing of injured corneas108–110. However, no further clinical or laboratory studies have been conducted using these miRNAs to treat dry eye.
At present, exosome therapy has many limitations, which hinder its application in dry eye treatment. Specific problems are as follows: (1) Due to technical limitations, it is difficult to obtain MSC-Exos with high purity and high yield. Therefore, technology needs to be further developed to improve separation efficiency and productivity. (2) The direct relationship between the specific content of MSC-Exos (lipid, protein, and nucleic acid) and their positive therapeutic effects remains unclear. The therapeutic effect, safety, and effective dose of various loaded substances need to be further studied. (3) MSC-Exos as a drug delivery carrier is a current focus of research, and drug delivery efficiency is another challenge that needs to be overcome.
Conclusion
In recent decades, MSCs have demonstrated great potential and availability in human and animal studies on the treatment of ocular surface diseases, and some laboratory studies have shown that MSCs can effectively treat dry eye. However, there is a lack of clinical studies such as randomized controlled trials to confirm their efficacy in the treatment of dry eye and to clarify their mechanisms which may include tissue regeneration, anti-inflammatory, and immunomodulatory synergistic effects. Although MSC therapy is promising in the treatment of dry eye, limited survival time and the implantation of bioactive agents are bottlenecks in disease treatment. Sustained maintenance and improvement of the survival and secretion of MSCs remain challenging in this field and require further study.
Since their biological characteristics are very similar to those of their source cells, MSC-Exos are considered an effective cell-free treatment strategy. The exosomes derived from MSCs are easy to store and can be directly transported to the diseased site through targeted modification or local injection, circumventing many limitations of cell therapy. However, due to technical limitations, clinical transformation associated with MSC-Exo therapy remains a major challenge. Future research will require the development of efficient technical methods that can deliver sufficient doses of drugs into MSC-Exos while maintaining its physical integrity and biological activity. Although further research is needed, MSC-based therapy of dry eye is feasible and its scope for treatment of damaged tissue is exciting.
Footnotes
Acknowledgements
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Bureau of Quanzhou (2020CT003), and the Medical Innovation project of Fujian Province (2021CXA027).
