Abstract
Major skin burns are difficult to treat. Patients often require special care and long-term hospitalization. Besides specific complications associated with the wounds themselves, there may be impairment of the immune system and of other organs. Mesenchymal stromal cells (MSCs) are a recent therapeutic alternative to treat burns, mainly aiming to accelerate the healing process. Several MSC properties favor their use as therapeutic approach, as they promote angiogenesis, stimulate regeneration, and enhance the immunoregulatory function. Moreover, since patients with extensive burns require urgent treatment and because the expansion of autologous MSCs is a time-consuming process, in this present study we chose to evaluate the therapeutic potential of xenogeneic MSCs in the treatment of severe burns in rats. MSCs were isolated from mouse bone marrow, expanded in vitro, and intradermally injected in the periphery of burn wounds. MSC-treated rats presented higher survival rates (76.19%) than control animals treated with PBS (60.86%, p < 0.05). In addition, 60 days after the thermal injury, the MSC-treated group showed larger proportion of healed areas within the burn wounds (90.81 ± 5.05%) than the PBS-treated group (76.11 ± 3.46%, p = 0.03). We also observed that CD4+ and CD8+ T cells in spleens and in damaged skin, as well as the percentage of neutrophils in the burned area, were modulated by MSC treatment. Plasma cytokine (TGF-β, IL-10, IL-6, and CINC-1) levels were also altered in the MSC-treated rats, when compared to controls. Number of injected GFP+ MSCs progressively decreased over time, and 60 days after injection, few MSCs were still detected in the skin of treated animals. This study demonstrates the therapeutic effectiveness of intradermal application of MSCs in a rat model of deep burns, providing basis for future regenerative therapies in patients suffering from deep burn injuries.
Introduction
Extensive skin burns are difficult to treat. Nowadays, the treatment of extensive skin burns remains a major challenge for health professionals. In addition to the disrupted skin barrier, there may also be impairment of the immune system and development of pathophysiological alterations in several internal organs, which contribute to the poor outcome of these patients. Severe and extensive burns require specialized health care and long hospital stays, resulting in high social costs (1). Assessment of patient severity is based on localization, extent, depth, and type of the burns (flame, chemical, scald), prior condition of patient, added to injuries caused by smoke inhalation (25). Indeed, numerous studies have shown a close relationship between mortality rates and extension of burned areas (8,14). In severe cases, therapeutic alternatives are limited, and mortality rates may reach 100%.
Currently, there is still no definition of the most efficient and cost-effective treatment for burned patients. In severe cases, therapeutic alternatives are limited and may not be available (8,27). Despite prompt restoration of fluid and electrolyte balance, respiratory support, and early debridement, adequate handling of the wound itself is fundamental to improve patient outcomes. Proper covering of the wound not only reduces fluid loss but also decreases the risk of subsequent infections (13,46). A skin autograft is considered the standard of care treatment for these wounds, because it provides fast coverage and improves wound healing. Patients with extensive burns, however, lack enough healthy skin for grafting (28). For those, therapeutic alternatives are warranted. Skin substitutes with dermal matrix are currently available as therapeutic options for extensive burns, when autografting is not possible. These skin substitutes may be acellular or contain differentiated cells, often autologous keratinocytes and fibroblasts. Although success has been reported with this approach, cultivation of autologous cells is time consuming, and allogeneic fibroblasts or keratinocytes may be rejected shortly after placed (21).
Mesenchymal stromal cells (MSCs) have been investigated as a therapeutic alternative for burn wounds. In the last years, MSCs have been used efficiently to treat several conditions, such as hematological and immunomediated diseases (7,30), heart dysfunctions (51), bone injury (61), and skin ulcers (5,17), in both experimental models and humans (7,17,30,61). Mesenchymal cells are considered suitable for the treatment of skin ulcers and burn wounds not only for their capacity to originate different cell types but also for their paracrine potential, secreting cytokines (TGF-β, IL-10, IL-6), chemokines (CCL2, CCL5, CXCL12), and growth factors (VEGF, IGF, bFGF, SDF, HGF), which may contribute to the healing process (36,54). Another interesting property of MSCs is immunomodulation, which may directly or indirectly affect different components of the immune system, leading to control of inflammation. Moreover, MSCs have little immunogenicity, enabling the use of off-the-shelf allogeneic cells (4,36,52).
Wu and collaborators demonstrated that injections of green fluorescent protein-labeled MSCs (GFP+ MSCs) along the periphery of ulcers were able to speed reepithelialization, promote angiogenesis, and increase the expression of vascular and endothelial growing factors. In addition, when application of neonatal allogeneic dermal fibroblasts was compared with vehicle control medium, expression of angiopoietin-1 increased, in both normal and diabetic mice (59). Mesenchymal cells have successfully healed acute and chronic wounds in experimental models and in patients (17). Nevertheless, only a few studies have reported long-term engraftment of injected cells in the injured tissue or their possible differentiation into skin cells, corroborating the paracrine and angiogenic potential of these cells as the main operating mechanisms in the wound-healing process (12).
Recent investigations suggest that MSC therapy may also enhance healing of burns in rats (31,43,48) and pigs (32). In these experimental models, MSCs were able to improve healing and also reduce the number of inflammatory cells in the injured site. Moreover, MSCs promoted angiogenesis and accelerated the development of granulation tissue (31,32,43,48). Mesenchymal cells were also demonstrated effective in burned patients refractory to conventional therapies. In 2005, Rasulov and collaborators described topical application of allogeneic MSCs in a woman who had accidently burned 40% of the body surface. The procedure enhanced self skin engraftment, with significant impact upon clinical outcome (42). In a different case report, repeated topical applications of autologous MSCs were able to increase wound healing and normalize C-reactive protein (CRP) plasma levels in a patient with a deep burn wound on one of the upper limbs, caused by unintentional exposition to γ rays (6).
Clinical and experimental evidence supports the use of both autologous and allogeneic MSCs in the treatment of burn wounds. However, MSC expansion demands time, and autologous cells may not be readily available for immediate treatment. Therefore, allogeneic MSCs, derived from healthy donors, previously expanded in culture and cryopreserved, may be a more suitable alternative for the initial and acute care of extensively burned patients.
Mesenchymal cells present low expression of MHC molecules (52), enabling allogeneic and xenogeneic applications. In the present study, we investigated safety and therapeutic efficacy of intradermal application of mouse MSCs in a rat model of extensive burn wounds. By such xenogeneic approach, we intended to impose an extra level of mismatched histocompatibility to the study. Therefore, if therapeutic outcomes were satisfactorily achieved, we would be able to confirm MSC hypoimmunogenicity and possibly aim clinical translation. Indeed, the results from this study may enable the development of future strategies to improve regenerative therapies using allogeneic MSCs for severely burned patients.
Materials and Methods
All animal procedures followed the guidelines of the Brazilian College of Animal Experimentation (COBEA) and were reviewed and approved by the Ethics Committee on Animal Experimentation (CETEA) of the Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), where the experiments were registered as No. 115/2007.
The Wistar rats used in this work were provided by the main animal facility at the University of São Paulo, Campus of Ribeirão Preto. The experimental animals were maintained in a conventional animal facility at the School of Pharmaceutical Sciences of Ribeirão Preto.
A total of 134 male rats were used in the experiments. Fifty-six were followed until the end of the study or natural death. Fourteen different animals were used for the wound-healing evaluations, 18 for the detection of GFP+ cells, six for the microbiological analyses and 40 for other analyses: myeloperoxidase assay, isolation of skin and spleen cells, histopathology, and cytokine measurements.
In order to perform the survival analysis and body weight measurements, 30 rats were used for the phosphate-buffered saline (PBS) and 26 for the MSC treatments. Seven rats from each group were used in the wound-healing analysis with ImageJ software, while for the microbiological evaluations, three additional rats from each group were used. Both groups of animals were kept alive and analyzed for a minimum of 0, 7, 15, 30, 45, and 60 days postburning.
Five additional rats from each group were sacrificed at 7, 15, 30, and 45 days postburning for the myeloperoxidase assay, isolation of skin and spleen cells, histopathological analyses, and cytokine measurements. For the detection of GFP+ cells by flow cytometry, three additional rats from each group were sacrificed at 0, 15, and 60 days postburning.
Harvesting and Isolation of Mesenchymal Stromal Cells
Femurs and tibias of at least three 5- to 7-week-old male FVB GFP+ mice (The Jackson Laboratory, Sacramento, CA, USA) were flushed for bone marrow harvesting. The obtained mixed cell suspension was then centrifuged for 10 min at 314 × g, and the pellet was resuspended in α-MEM medium (Gibco, Grand Island, NY, USA) supplemented with 0.58 g/L L-glutamine (Gibco), 3.70 g/L NaHCO3, 10.000 UI/L penicillin (Gibco), 0.05 g/L and 5.96 g/L streptomycin (Gibco), HEPES (Gibco), and 15% (v/v) fetal bovine serum (FBS; Thermo Scientific, Rockford, IL, USA). Cultures were maintained in 75-cm2 culture flasks (Greiner Bio One, Austria), incubated at 37°C and 5% CO2 for isolation of MSCs.
MSCs spontaneously adhere to polystyrene. Therefore, after 72 h in culture, nonadherent cells were removed by exchanging the whole medium volume, while attached cells were maintained in culture. Upon reaching approximately 80% confluence, cultures underwent trypsinization, with bovine trypsin/EDTA at 0.25% m/v (Gibco) to detach the adherent cells and allow subculture. For all procedures, MSCs in third–fourth passage were used.
Mesenchymal Stromal Cell Immunophenotyping
Cell suspension aliquots (200 μl) containing 1 × 106 MSCs/ml from passages 3-4 were resuspended in PBS and then incubated for 20 min, at room temperature, in the dark, with 5 μl of phycoerythrin-conjugated monoclonal antibodies 0.2 mg/ml, either control isotypes or specific for CD45, CD11b, CD31, CD105, CD29 (eBioscience, San Diego, CA, USA). After incubation, cells were centrifuged for 5 min at 706 × g, washed twice with PBS/EDTA, resuspended in 200 μl PBS/EDTA, and immediately analyzed by FACSCalibur flow cytometer (Becton Dickson BD, San Jose, CA, USA). Immunophenotypic characteristics of MSCs were analyzed at the Flow Cytometry Laboratory of the Ribeirão Preto Regional Blood Center (Ribeirão Preto/FMRP-USP, São Paulo, Brazil). Data of 20,000 events/sample were acquired based on size parameters (FSC) for granularity (SSC). Analyses were performed using the Cell Quest Pro software (BD, San Jose, CA, USA). The MSCs used in this study were characterized, in accordance with the literature, by high expression of surface molecules such as CD105 and CD29, and absent or low expression of CD45, CD11b, and CD31 (15) (data not shown).
Characterization of the Extensive Burn Rat Model
In order to create an extensive burn experimental model, the Precision Workshop from the University of São Paulo developed a device with a digital screen to make standardized burn wounds (Fig. 1A). This device is able to keep constant temperature on a metal plate during the contact with the trichotomized skin of Wistar rats. Prior to applying the burning plate, the dorsal hair of the rats was shaved, followed by application of depilatory cream (Depilart, L. Esper & Cia, São Paulo, Brazil). The extensive burn wounds were then created with light pressure of the metal plate (4.5 × 2.5 cm) heated to 200°C against the animal's dorsal area for 25 s (Fig. 1B). Deep burns affecting the three skin layers were later histologically confirmed. Each animal received a sequence of four contiguous burns, adding up to a total burned area of approximately 45.0 cm2. The burned rats were maintained on individual cages without any dressings on the skin wounds.
Experimental burn design. (A) Burn device with metal plate used to make the experimental wounds. (B) Initial burn aspect on dorsal surface of the rat. (C) Body weight curves in animals with experimental burns treated with intradermal application of 5 × 106 MSCs (n = 7) or PBS (n = 7). (D) Survival curves in animals with experimental burns treated with intradermal application of 5 × 106 MSCs (n = 26) or PBS (n = 30). *p < 0.05.
In Vivo Burn Wound-Healing Assay and Mesenchymal Cell Transplantation
Wistar rats were anesthetized with 1 μg/g solution at a concentration of 1:1 (v/v) ketamine at 10% (Agener Union, São Paulo, SP, Brazil) and 20 mg/ml xylasin (Dopaser-Hertape Calier, Minas Gerais, Brazil) intraperitoneally. Then burn lesions were created as detailed. Twenty minutes later, the still anesthetized animals were divided into two groups of seven rats. Groups were respectively treated with 5 × 106 MSCs in 2,000 μl of PBS or only 2,000 μl of PBS, divided in 10 intradermal injections distributed along the periphery of the burns. Animals were housed individually, with food and water ad libitum.
Burn Wound-Healing Analysis
Digital photographs of the wounds were taken on days 0, 7, 15, 30, 45, and 60 after creation of burns. For all pictures, the camera was placed at a specific distance from the animals (30 cm) and under consistent settings. Using a ruler as a measuring reference, wound margins were traced on images, and areas were determined by ImageJ (National Institutes of Health, Bethesda, MD, USA) image analysis software. Percentage (P) of wound healing (area of reepithelialization) was calculated through the equation: P = 100 – (Af /Ai · 100), where Af and Ai correspond to the final and initial areas of the wounds, respectively. Such approach has been proposed by Masson-Meyers et al. (35).
Histopathological Analyses
Animals were sacrificed by decapitation on days 7, 15, 30, 45, and 60 after cell or sham treatment, and skins were removed, fixated in 10% neutral-buffered formalin, and embedded in paraffin. Sections (5 μm) were stained with hematoxylin and eosin (H&E; Merck KGaA, Darmstadt, Germany), mounted, and evaluated under light microscope. All histological analyses were performed by a pathology specialist, from the Pathology Laboratory of the Ribeirão Preto Medical School, University of São Paulo, who was blinded to animal treatment and to sacrifice periods. Histopathological evaluations included monitoring of vascularization, granulation tissue, total polymorphonuclear inflammatory cells, and collagen fibers. Vascularization was determined by presence of blood vessels that were identified when they presented the typical pattern containing three layers from the luminal side: the tunica intima, the tunica media, and the tunica adventitia. Granulation tissue was quantified by thickness of granulation layer. Inflammatory cells were estimated by quantification of polymorphonuclear inflammatory cells, and collagen fibers were measured by quantification of filamentary and acellular structures stained by eosin. Score parameters were established, ranging from 0 to 3, where 0 indicates absence of histological findings or thin granulation layer (<1%); 1 indicates minor histological findings or moderate granulation layer (<25%); 2 indicates moderate histological findings or thick granulation layer (<50%), and 3 indicates considerable histological findings or very thick granulation layer (>50%) (39,59). Results from control and MSC-treated animal groups were compared.
Myeloperoxidase Assay
Skin samples were collected, weighed, and homogenized with a Polytron® PT 3100 (568 × g) homogenizer (Kinematica AG, Littau-Luzern, Switzerland). After centrifugation of the samples, accumulation of neutrophils was measured by myeloperoxidase (MPO) assay. The pellet was lysed with 15 ml 0.2% NaCl (Merck KGaA Darmstadt, Germany) solution for 30 s, followed by addition of 1.6% NaCl containing 5% glucose. The suspension was centrifuged, the pellet was resuspended in 0.05 M NaPO4 buffer (Sigma-Aldrich, St. Louis, MO, USA), pH 5.4, containing 0.5% hexadecyltrimethylammonium bromide (Sigma-Aldrich), and rehomogenized. Aliquots of 1.0 ml of the suspension were transferred to tubes, followed by three cycles of freezing–thawing with liquid nitrogen. The aliquots were centrifuged for 15 min at 1,100 × g and 5.0 μl of the supernatant, diluted in 45.0 μl 0.08 M NaPO4, was used for the assay in 96-well plates. Myeloperoxidase activity in the supernatant was detected with 25 μl 1.6 mM tetramethylbenzidine (Sigma-Aldrich) followed by 100 μl 0.5 mM H2O2 (Merck KGaA). Absorbance was measured with a 96-well in an EL800 plate reader (Bio-Tek, Winooski, VT, USA) at 450 nm, and results were compared with a standard curve of mouse peritoneal neutrophils. Results were reported as mean ± SEM of the total number of neutrophils per gram of skin.
Isolation of Skin Cells
For isolation of cells from the wounded skin, excised wounds were cut in small pieces and incubated with 10 mg/ml Liberase blendzyme 1 (Roche Applied Science, Indianapolis, IN, USA) in a 37°C shaking water bath for 1 h. Then RPMI-1640 medium (Gibco), supplemented with 10% FBS, 2 mmol/L L-glutamine, and 100 U/ml penicillin and streptomycin (Gibco) was added to the tube containing the cells, and the suspension was filtered through a 70-μm nylon cell strainer (BD). Cells were pelleted and resuspended in RPMI-1640 medium.
Isolation of Cells From Spleen
Each spleen was placed into a Petri dish and mashed with the plunger end of a syringe. Cell suspension was collected, and erythrocytes were lysed with Tris (Promega, Madison, WI, USA) 0.17 M and NH4Cl (Merck KGaA) 0.16 M buffer. Next, RPMI-1640 medium (Gibco), supplemented with 10% FBS, 2 mmol/L L-glutamine, 100 U/ml penicillin and streptomycin (Gibco) was added to the tube containing splenocytes and centrifuged at 300 × g for 10 min at 4°C. The supernatant was discarded and the pellet resuspended in RPMI-1640 medium.
Flow Cytometry of CD3/CD4 or CD3/CD8 Cells
Spleen or skin cell aliquots containing 1 × 106 cells in 200 μl PBS were incubated for 20 min at room temperature in the dark with 5 μl of fluorescein-isothiocyanate (FITC) or phycoerythrin-conjugated monoclonal antibodies 0.2 mg/ml, either control isotypes or specific for CD3 and CD4, or CD3 and CD8 (eBioscience, San Diego, CA, USA).
After incubation, cells were centrifuged for 5 min at 706 × g, washed twice with PBS/EDTA, resuspended in 200 μl PBS/EDTA, and immediately analyzed by FACSCalibur flow cytometer (BD). Single cell suspensions were analyzed at the Flow Cytometry Laboratory of the Regional Blood Center (Ribeirão Preto/FMRP-USP, São Paulo, Brazil). For skin cells, data of 1,000,000 events/sample were acquired based on size parameters (FSC) for granularity (SSC) and for spleen cells, data of 100,000 events/sample. Data analyses were performed using the Cell Quest Pro software (BD).
Cytokine Measurements
Experimental animals were periodically sacrificed by decapitation without anesthesia. Immediately after the procedure, whole blood was collected in the presence of sodium citrate anticoagulant USP, formula A (JP Pharmaceutical Industry, Ribeirão Preto, SP, Brazil) and centrifuged at 491 × g for 10 min at 4°C. The cytokines interleukin-10 (IL-10), IL-6, transforming growth factor-β (TGF-β) (eBioscience), and cytokine-induced neutrophil chemoattractant 1 (CINC-1) (R&D Systems, Minneapolis, MN, USA) were quantified by ELISA in the plasma supernatants, according to the manufacturer recommendations.
Microbiological Analyses of Wounds
Bacterial contamination of the burned skin was evaluated at days 0, 3, 7, 15, 30, 45, and 60 after cell or sham treatment. A sterile swab was used to collect microbiological material in a wound area of about 1 cm2. Subsequently, swabs were inserted into sterile tubes containing 1 ml of saline solution, and the material was spread on Cystine Lactose Electrolyte-Deficient agar (CLED). Microbiological growth was quantified after 48 h of incubation. Analyses were performed at the Clinical Analysis Laboratory from the School of Pharmaceutical Sciences of Ribeirão Preto, University of São Paulo.
Detection of GFP+ Cells by Flow Cytometry
Groups of three animals were sacrificed on days 0, 15, and 60 after MSC treatment to evaluate MSC homing efficiency. All dorsal burned skin was removed with a margin of 0.5 cm of normal skin around the experimental burn area. The tissue was digested with 10% Liberase blendzyme 2 (Roche) in RPMI unsupplemented medium (Gibco) and analyzed by flow cytometry, aiming to detect the presence of GFP+ cells. Cells from the PBS-treated group were used as negative controls. Approximately 1 × 106 events were analyzed by flow cytometry FACS Calibur (BD) (item 2.8).
Statistical Analyses
Statistical analyses were based on comparison of the variable P among the experimental groups. Groups were compared (PBS-treated vs. MSC-treated groups) by Mann–Whitney test. Differences were considered statistically significant when p < 0.05. Analyses were performed with GraphPad PRISM version 5.0 (GraphPad Inc., La Jolla, CA, USA) data analysis software.
Results
Characterization of the Experimental Model of Extensive Burn in Rats
Extensive tissue damage was observed after experimental burns. Visually, the burns extended through almost the entire dorsal surface of the animals (Figs. 1B and 2A). Histological sections showed damage affecting all skin layers (data not shown).
Treatment with Mesenchymal Cells Increases Survival Rates After Burn Exposure
In order to observe variations in body mass during the experimental period, both control (n = 7) and MSC-treated animals (n = 7) were weighed before (T0) and also at 7, 15, 30, 45, and 60 days after treatment. No differences were found between mean body weight of the animals in both control and MSC-treated groups, as shown in Figure 1C.
We also compared survival rates between MSC-treated (n = 26) and control (n = 30) groups after the burning process. Highest mortality rates were observed on day 4 after burn injury in both animal groups. However, after day 7, survival rates were higher in the in MSC-treated (76.19%) than in the PBS-treated (60.86%) groups (p < 0.05) (Fig. 1D). No additional deaths were observed, in both groups, in further time points (Fig. 1D).
Treatment with Mesenchymal Cells Decreases Wound-Healing Time
Photographic images of the wounds were used to analyze dynamics of the healing process. In both animal groups, total wound areas were increased at 7 days after the burn injury (Fig. 2B). This expected enlargement is due to progression of the necrotic tissue in the wound margins and has been previously reported (50). In the subsequent evaluation time points, beyond the seventh day, progressive reduction of the burn wound areas was observed in both groups. Beyond 45 days after injury, healed areas of the wounds were significantly larger in the MSC-treated (72.95 ± 5.17%, n = 7) than in the PBS-treated animals (57.74 ± 2.02%, n = 7) (p < 0.05) (Fig. 2B), indicating a beneficial effect of the cell therapy. Sixty days after the burning procedure, MSC-treated animals showed 90% (90.81 ± 5.05%, n = 7) of epithelialization of the wound area, against approximately 80% (76.11 ± 3.46%, n = 7) in the controls (p < 0.05) (Fig. 2B).
Kinetics of wound healing of MSC- or PBS-treated animals. (A) Representative burning aspect in animals from both PBS-and MSC-treated groups along time points (day 0), 7, 15, 30, 45, and 60 after initial burn injury. (B) Percentage of wound healing in both PBS- (n = 7) and MSC-treated (n = 7) groups at 0, 7, 15, 30, 45, and 60 days postburning. Area measurements provided by ImageJ software. Bars represent means ± SEM and *p < 0.05.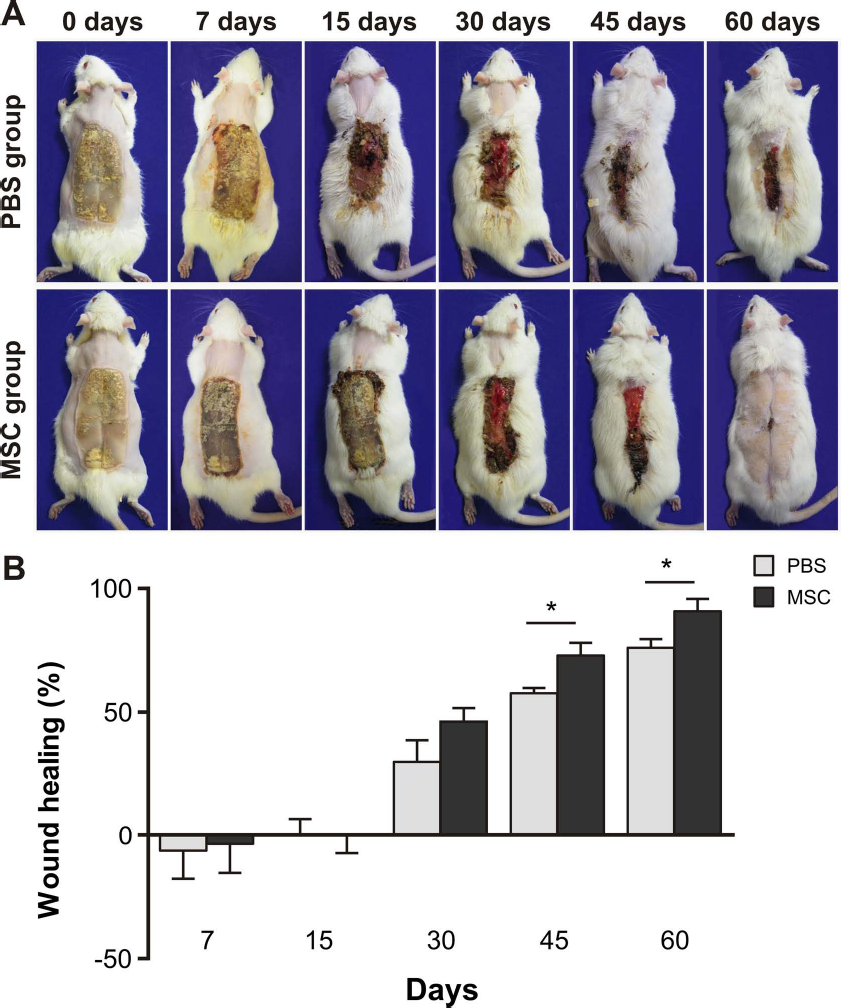
Treatment with Mesenchymal Cells Induces Formation of Blood Vessels and Granulation Tissue, and Decreases Inflammatory Cells in the Wound Areas
In order to obtain a general idea of the wound-healing process microscopically, three histological samples from the skins of both PBS-treated (control) and MSC-treated animals were analyzed under light microscope at 7, 15, 30, 45, and 60 days after treatment. To this end, a histological scoring system was used, aiming to evaluate the number of blood vessels, granulation tissue, and inflammatory cells in skin/wound sections from control and MSC-treated animals. The injured areas of the MSC-treated animals exhibited larger number of blood vessels (on day 15) compared with those from control animals (Fig. 3A). Thirty days after the burning procedure, the MSCs-treated group also presented higher amount of granulation tissue (Fig. 3B), indicating a more advanced wound-healing process when compared with the control group (Fig. 3B). On day 45, the MSC-treated animals presented larger number of inflammatory cells in the injured areas when compared with controls (Fig. 3C). When collagen deposition was assessed, no differences between experimental groups were observed (Fig. 3D).
Histological scores of burn wounds after intradermal injections of PBS or MSCs, over time. (A) Blood vessel scores, (B) granulation tissue scores, (C) inflammatory cell scores, (D) collagen fiber scores. Histological analyses were performed from three skin specimens from each experimental group at each evaluation period, stained with hematoxylin and eosin (H&E) evaluated under a light microscope, and score parameters were established, ranging from 0 to 3, where 0 indicates absence of histological alteration; 1 indicates minor histological alterations; 2 indicates moderate histological alterations, and 3 indicates considerable histological alterations. Results shown as means ± SEM, *p < 0.05.
Treatment with MSCs Modulates Systemic and Local T-Lymphocyte Populations
Aiming to identify other specific inflammatory cells, T-lymphocyte frequencies were quantified in the spleens and burned skin from five rats in each experimental group. MSC-treated animals presented higher frequencies of splenic CD4+ T lymphocytes at 7 days (Fig. 4A), and decreased splenic CD8+ T-cell counts at 15 days after MSC or sham injections (Fig. 4B). Furthermore, differences in CD4+/CD8+ lymphocyte ratios in the spleens were also detected between MSC-treated and control animals at 7 and 15 days after burning (Fig. 4C).
Kinetics of T-cell subsets in spleen and wound tissue skin at different postburn time points in MSC- and PBS-treated animals. (A) Percentages of CD4+ T cells in the spleen, (B) percentages of CD8+ T cells in the spleen, (C) CD4/CD8 lymphocyte ratios in the spleen, (D) CD4+ T-cell percentages in burned wound tissue, (E) CD8+ T-lymphocyte percentages in burned wound tissue. Cell counts by flow cytometry from cells CD3 and CD4 or CD3 and CD8 doubly stained. Results displayed as means ± SEM, from n = 5 samples per experimental group. *p < 0.05.
We also evaluated T-lymphocyte subpopulations in the injured tissue. Seven days after wound creation, the MSC-treated group presented less CD4+ (Fig. 4D) and CD8+ (Fig. 4E) infiltrated T cells than the control group. Decreased CD4+ T-cell counts were also observed in the wound tissue on the 15-day and 45-day time points (Fig. 4D). These observations suggest that MSCs may have modulated CD4+ and CD8+ T cells on the injury site.
Changes in Cytokine Profile After Treatment with Mesenchymal Cells
Cytokine levels were analyzed in plasma from five rats in each experimental group. The MSC-treated group showed higher levels of TGF-β 15 days after the experimental procedure (Fig. 5A) and higher levels of IL-10 (Fig. 5B) at the 30-day time point, indicating increased levels of anti-inflammatory cytokines. However, proinflammatory cytokine concentrations were also higher in the MSC-treated group, as noticed in the IL-6 and CINC-1 plasma levels 30 days after treatment (Fig. 5C, D).
Kinetics of cytokine levels in plasma of burned rats treated with PBS or MSCs. (A) TGF-β levels, (B) IL-10 levels, (C) IL-6 levels, (D) CINC-1 levels. Measurements by ELISA in plasma from burned rats. Results shown as means ± SEM, from n = 5 samples per experimental group; *p < 0.05 and ***p < 0.02.
Treatment with Mesenchymal Cells Induces Late Influx of Neutrophils to the Site of Injury
Skin specimens from five animals belonging to each experimental group were analyzed under myeloperoxidase (MPO) assay, as a method to quantify the neutrophil influx into the burned areas. MPO levels remained low and stable in both animal groups at all time points, up to 30 days after burn creation. At the 45-day time point, however, MPO concentrations significantly increased in the MSCs-treated group (Fig. 6), indicating late neutrophil influx.
Quantification of tissue neutrophils. Neutrophils were quantified in wounded skin fragments from PBS- and MSC-treated animals through myeloperoxidase (MPO) assay, at different time points after burning injury. Results displayed as means ± SEM, from n = 5 samples per experimental group. *p < 0.05.
Microbiological Analyses of Burn Wounds
Microbiological Content of Burn Wounds

Engraftment of Mesenchymal Cells in the Burn Wounds
The presence of intradermally injected GFP+ MSCs in the wound tissue was investigated at different time points after treatment. For this purpose, the dorsal burned skins of three animals from both MSC-treated and control groups were analyzed for presence of GFP+ cells. Immediately after MSC applications (0 day), approximately 1.5% (1.58 ± 0.09, n = 3) of the skin cells were GFP+. The percentage of GFP+ MSCs recovered from the skins of MSC-treated rats progressively decreased along time, and 15 days after MSC treatment only 0.37% of the skin cells were GFP+ (0.37 ± 0.08, n = 3). Nevertheless, 60 days after injections, approximately 0.30% (0.31 ± 0.06, n = 3) of GFP+ cells were still detected in the dorsal skin of animals. Figure 7 shows the skin analysis from a representative animal.
MSC detection in skin. Percentage of flow cytometry-detected MSC GFP+ cells in burn wound tissue at 0, 15, and 60 days after MSC treatment. Figure from representative animal.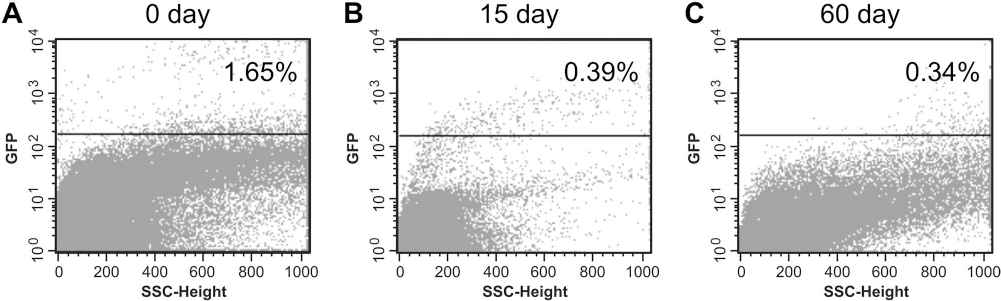
Discussion
In the current literature, only a few experimental burn models precisely reflect the clinical setting. In this study, we developed an experimental model in rats, which is comparable to human third-degree burns. In this model, a large body surface is affected, involving the three layers of skin, therefore classifying injuries as both extensive and severe. Differently than small-sized burns, with mainly local effects, extensive burns result in systemic alterations that surpass the injuries themselves, leading to hypermetabolism, systemic inflammatory response syndrome (SIRS), among other pathophysiological alterations (1,9,24).
The wound-healing process is considerably impaired in patients with severe burns. The wound itself acts as a mechanical barrier, preventing migration of cytokines and growth factors, essential for successful healing of the area (16). In addition, the compensatory anti-inflammatory response developed after the injury promotes systemic immunosuppression, which affects the inflammatory phase, essential for the development of an adequate wound-healing process (56). Since mesenchymal cells have the potential to modulate inflammation, stimulate tissue repair, and possibly differentiate into and replace local cell types, they have been investigated as potential tools for regenerative medicine (10,29,36,53).
In our study, we observed that 45 days after the burn injury, wounds treated with MSCs presented better healing than those from the control group. Histological parameters such as density of vessel formation, amount of granulation tissue, and severity of inflammation indicated a beneficial effect of MSCs upon the healing process. These results corroborate previous reports from the literature showing that MSCs are able to enhance healing after burns (6,31,32,42,43,48).
At the 15-day postinjury time point, we observed that MSC-treated animals presented higher vessel density than controls. We believe that this result may be due to the angiogenic potential of MSCs, enhancing local blood supply, and thus contributing to tissue regeneration (26,59). We also observed that 30 days after the injury, MSC-treated animals presented more granulation tissue, which also indicates progression of the healing process. Granulation tissue is temporarily found on the surface of the wounds as part of recovery process (58). Our results agree with numerous reports from the literature showing that MSC therapy is able to accelerate the formation of granulation tissue in skin ulcers (3,26,43,48,55).
Inflammation was also investigated as a mechanistic target for MSCs. In the initial phases of wound healing, local inflammation stimulates development of scar tissue, which is important to limit the extent of injury and circumscribe necrotic and undesirable material (45,58). It has already been shown that the increased local immune response that follows burn injuries is strongly associated with scar formation (11,19). On the other hand, scars may physically impair later beneficial events of the healing process. Mesenchymal cells are able to modulate the activity of immune cell types such as NK cells, dendritic cells, neutrophils, lymphocytes, among others, and possibly interfere with wound outcomes, including scar formation (34). In fact, less inflammation and decreased skin fibrosis has been described in a mouse model of bleomycin-induced skin injury treated with bone marrow-derived MSCs (60).
We observed a neutrophil inflammatory infiltrate in the skins of MSC-treated animals. T-lymphocyte counts, on the other hand, were decreased in several time points, which we believe may be related to the improved healing of MSC-treated animal wounds. Furthermore, at some time points, MSC-treated animals presented decreased CD4+ and CD8+ T-lymphocyte frequencies in the wound tissue, suggesting that MSCs contributed to locally modulate these two lymphocyte subtypes. In our study, we were not able to determine the consequences of this specific effect, as the exact role of these cells in the wounded area is still not established.
Studies using MSCs as an alternative therapeutic approach to different immunomediated pathologies, such as graft-versus-host disease, have already demonstrated the in vivo immunomodulatory properties of these cells (7). Nevertheless, this is the first study to show that local treatment with MSCs decreases the amount of infiltrating lymphocytes in severely burned skin. We believe that this local immunomodulatory effect may be beneficial to the cicatrization process following deep burns.
In patients with severe thermal burns, immunological abnormalities are characterized by early exacerbation of the inflammatory response, altered CD4/CD8 ratios, and secretion of large quantities of proinflammatory cytokines, occasionally leading to development of SIRS (18,22,40). These abnormalities contribute to the poor outcome of patients and high mortality rates (51).
Mesenchymal cells are known to secrete large quantities of growth factors and anti-inflammatory cytokines such as TGF-β and IL-10, which play important role in systemic and local immunomodulation (36). Accordingly, in our study, TGF-β and IL-10 may have contributed not only to the reduction of mortality rates but also to improve the wound-healing process after the extensive burn injuries. We also observed beneficial effects of MSCs upon the postinjury inflammatory response. At some time points in the MSC-treated group, CD4 and CD8 lymphocyte counts, besides CD4/CD8 ratios, remained closer to normal rates (37), characterizing a more adequate immune response.
Burned patients commonly develop the compensatory anti-inflammatory response syndrome in later periods, characterized by systemic immunosuppression that predisposes individuals to the development of subsequent infections and sepsis (49,56,63). In our study, although we detected bacterial colonization in wounds from both animal groups, late mortality was not observed. Unexpectedly, late after burns, MSC-treated rats presented increased levels of proinflammatory IL-6 and CINC-1 in the plasma, which may be related to peak neutrophil infiltration in the wound tissue at the 45-day time point. This may indicate that, at late stages, MSCs are able to reverse the systemic immunosuppression secondarily installed after severe burns, allowing the immune system to respond to challenges such as microbial infections. This is a very speculative supposition, and further studies are needed to specifically investigate such aspects.
The late detection of neutrophils in the injured skin may be also related to the remodeling process and mostly due to collagenase secretion than to control of bacterial colonization. The MMPs secreted by neutrophils are important for the degradation of extracellular matrix components, thereby decreasing local fibrosis during the final stages of cicatrization (20). This is not a universally accepted concept, since high production of collagenase by neutrophils is associated with poor healing of chronic ulcers in patients with diabetes (33). In our study, however, presence and activity of MMPs in the wounds were not investigated. This is another question that remains for future studies.
In the experimental model used in this study, systemic and local alterations induced by MSCs were observed since the initial periods after burn creation and up to later time points. Few studies have determined the exact duration of MSC-mediated effects, most of them reporting progressive reduction in number or even complete disappearance of implanted cells, across time and resolution of injuries (17,31,59,60). Accordingly, we observed decreasing numbers of GFP+ MSCs in the skin of the wounded animals over time, and at the 60 days after injections, few MSCs were detected in the wound tissue. Nevertheless, although short-lived, MSCs were able to improve wound healing and to modulate the immune system.
The mechanisms involved with low survival of MSCs in the injury site are still not established. MSCs have been considered hypoimmunogenic or “immune privileged” enabling transplantation across MHC barriers. However, despite satisfactory results obtained in patients following allogeneic MSC infusions (7,23,41,42), several studies have reported evidence of immune-mediated rejection, such as generation of antibodies against donor MSCs (2,38,62). Additionally, proinflammatory factors released in the site of tissue damage may polarize MSCs to either a proinflammatory or immunosuppressive phenotype and determine the outcome of the MSC-mediated process of tissue repair (44,47,57). Moreover, MSCs with more inflammatory profile may act as antigen-presenting cells, and thereby stimulate immunogenicity and subsequent cell rejection (2). In our study, only few MSCs were still detectable at 60 days after intradermal application; thus, because of the xenogeneic barrier, we believe they were likely rejected. In fact, although at first seeming discouraging, MSC rejection may be important in clinical practice, since it indicates that the immune system is able to control the presence of allogeneic or xenogeneic cells and therefore avoid undesirable proliferation or malignancy. Mechanisms underlying such supposition may be affected by the species of MSC origin, immunological status of the host, and presence or absence of inflammation (2), but these are issues still to be explored.
In summary, our results show that therapy with xenogeneic MSCs was able to decrease mortality, modulate the immune response, and enhance wound healing/epithelialization of severe burns. These results thus corroborate the regenerative and immunomodulatory potential of this cell population and support the use of unrelated, possibly off-the-shelf, MSCs for the treatment of extensively burned patients.
Conclusions
This study demonstrates the therapeutic efficacy of intradermal administrations of xenogeneic MSCs in an experimental deep burn model in rats. Treatment with MSCs was able to both improve wound healing and modulate the immune response in rats, despite low cell engraftment rates. This is an important step toward future applications of MSCs as a regenerative therapy for patients affected by deep burns.
Footnotes
Acknowledgments
We would like to thank and honor the memory of Professor Júlio C. Voltarelli, mastermind and pioneer of stem cell therapy for autoimmune and inflammatory disorders in Brazil. We are grateful to the staff of the Central Animal Facility and School of Pharmaceutical Sciences Animal Facility, both from the University of São Paulo campus Ribeirão Preto, for animal supply and care, especially to Mr. Reinaldo Fernando Batista and Precision Workshop from the University of São Paulo. We also thank Sandra Navarro Besciane for the graphic design and Camila C. B. O. Menezes for the flow cytometry analysis, Marcelo Dias Baruffi and Willian Abraham da Silveira for the microbiological tests, Mariana R. Davanso for the technical support, and Ricardo Caliari Oliveira for the English review. This study received financial support from the Brazilian Research Foundations CNPq (No. 573754-2008-0) and FAPESP (No. 2007/57482-6 and No. 2008/578773). The authors declare no conflicts of interest.
