Abstract
Administration of mesenchymal stromal cells (MSCs) is a promising strategy to treat cardiovascular disease (CVD). As progenitor cells may be negatively affected by both age and comorbidity, characterization of MSC function is important to guide decisions regarding use of allogeneic or autologous cells. Definitive answers on which factors affect MSC function can also aid in selecting which MSC donors would yield the most therapeutically efficacious MSCs. Here we provide a narrative review of MSC function in CVD based on a systematic search. A total of 41 studies examining CVD-related MSC (dys)function were identified. These data show that MSC characteristics and regenerative potential are often affected by CVD. However, studies presented conflicting results, and directed assessment of MSC parameters relevant to regenerative medicine applications was lacking in many studies. The predictive ability of in vitro assays for in vivo efficacy was rarely assessed. There was no correlation between quality of study reporting and study findings. Age mismatch was also not associated with study findings or effect size. Future research should focus on assays that assess regenerative potential in MSCs and parameters that relate to clinical success.
Keywords
Introduction
Cardiovascular disease (CVD) is one of the leading causes of mortality in the Western world 1 . Genetic makeup, exposure to risk factors, and lifestyle factors all play a role in the pathogenesis of CVD. Aging is a main driver of CVD progression 2 , inducing nonreversible changes in the vasculature and the heart. These changes range from remodeling of the vessel wall to tissue damage at places of turbulent blood flow and predispose previously healthy individuals to the development of CVD 3,4 . Aging also affects the regenerative potential of the cardiovascular system. Aged individuals have fewer circulating endothelial progenitor cells (EPCs), which has been implicated in the development and accumulation of vascular damage 5,6 . Direct effects of aging on tissue repair capabilities of resident tissue cells have also been shown; for instance, aged endothelial cells are less effective at migration, more prone to become senescent, and they have an altered secretion profile that contributes to the development of hypertension and atherosclerosis 7 –9 . In turn, CVD also affects the progenitor cell compartment and regenerative potential of tissue cells. In many CVDs, an age-independent decrease in EPCs has been reported 10 –12 . Decreased migratory potential and increased senescence are also present in endothelial cells of CVD patients.
Eventually, end organ damage develops through a final common pathway of chronic tissue hypoxia, impaired or disturbed angiogenesis, inflammation, and eventually fibrosis.
Though progress has been made, therapies for CVD with the potential to reverse disease-induced damage are still lacking. Cellular therapy approaches that aim to promote angiogenesis and enhance organ function in CVD are currently being investigated. Preclinical and clinical research, while promising at first, demonstrated no clear benefits of autologous bone marrow mononuclear cells (BM-MNCs) in various CVDs 13,14 . Interestingly, advanced age is one of the strongest predictors of a lack of clinical response to autologous BM-MNC therapy 15 . EPCs are still being evaluated clinically, but technical difficulties in acquiring and expanding cells to the numbers required for cellular therapy hamper efforts 16 . Mesenchymal stromal cells (MSCs), first discovered as nonhematopoietic BM precursor cells, gained interest given their regenerative properties in a wide variety of disease contexts and the relative ease of procurement and expansion. In preclinical and clinical studies, MSCs were shown to promote angiogenesis, reduce fibrosis, have immunomodulatory properties, and ultimately restore tissue function. MSCs exert their regenerative effects through close cross talk with local tissue cells and immune effector cells by both direct cell–cell contact and release of paracrine factors 17 . Transdifferentiation remains controversial and is unlikely to account for the observed regenerative effects 18 .
During the past decades, MSCs gradually transitioned from the bench to the bedside in CVD; phase III clinical trials examining potential application of MSCs as a therapeutic agent in CVD are ongoing. For instance, MSCs have been shown to be effective at improving left ventricle ejection fraction, reduction in scar size, and neoangiogenesis when administered to patients with ischemic cardiomyopathy 19 . In patients with critical limb ischemia (CLI), MSCs were shown to improve perfusion and wound healing 20,21 . As systemic anti-inflammatory effects are thought to contribute to clinical effects of MSCs in CVD 22 , novel applications also include diseases that have both ischemic and autoinflammatory features such as systemic sclerosis (SSc) 23 .
Generally, MSCs for clinical applications are isolated from BM, adipose tissue (AT), or the umbilical cord, but MSC-like cells that fulfill the International Society for Cellular Therapy’s (ISCT) minimal criteria for MSCs 24 have also been identified in other tissues including organ tissue.
Variability in therapeutic efficacy between donors prompts careful consideration of the cell source to maximize clinical benefits. Currently, in MSC-based therapy, both autologous and allogeneic cells are clinically used. Investigating regenerative potential of MSCs prior to clinical administration is vital to aid identifying donors whose MSCs will yield the largest clinical effect. Donor characteristics such as gender and age have been reported to affect MSC efficacy. MSCs derived from older donors proliferate slower, have reduced differentiation capacity, and display more features of cellular senescence when compared with cells derived from younger individuals 25,26 .
Several studies suggest that autologous MSCs may also be functionally affected by disease. In unselected BM-derived cells, CVD dysfunction has been shown 12,27 , and dysfunction of MSCs has also been reported in CVD. If MSC dysfunction occurs in certain disease states, then this could reduce clinical efficacy of autologous MSC treatment and render administration of allogeneic cells more favorable. However, where MSCs were once considered completely immunoprivileged, studies have shown that immune responses directed against MSC may develop under certain circumstances 28,29 . For instance, senescence induced by prolonged culturing leads to loss of immunosuppressive ability. Additionally, donor characteristics such as gender and age might affect MSC efficacy, further complicating the search for a suitable cell source. Accordingly, in assessing CVD-related MSC dysfunction, age and features of cellular senescence are important factors to consider.
Here we review the currently available literature on CVD-mediated dysfunction in MSCs to assess the presence and extent of CVD-mediated dysfunction in MSCs harvested from clinically relevant sources. Additionally, the role of aging in CVD-mediated dysfunction will be explored, using the available evidence. We will discuss the implications of our findings for clinical MSC-based cell therapy.
Materials and Methods
Search Strategy
To identify studies that examined the effect of CVD on MSC (dys)function, a search was conducted in Medline/PubMed and Embase. The search strategy contained the following components: (1) MSCs and (2) disease and/or dysfunction. The search was performed on December 18, 2016. Studies were selected by 2 reviewers. The references of included studies and the studies citing them were hand searched.
We included primary studies that examined cells obtained from individuals with CVD that (partially) fulfill the minimal criteria for multipotent MSCs set by the International Society for Cell and Gene Therapy (ISCT) 30 . The MSCs had to be obtained from clinically relevant sites, that is, BM, AT, and umbilical cord tissue or cord blood (UCB).
We excluded studies that examined MSCs derived from animal sources. We also excluded nonprimary studies (reviews were used to snowball and enhance the keyword search).
A total of 45 studies were identified. Four studies were excluded. Two studies were excluded because they examined the effect of patient serum on healthy MSCs and the other 2 studies were excluded because the full text was not available.
Study Characteristics and Data Extraction
The following study characteristics were recorded: subject characteristics such as age and gender, MSC source, type of disease, N, passage number, growth medium, protein source (fetal bovine serum [FBS] vs. platelet lysate or alternatives), whether the cells had been freshly used or postthaw, details of the various assessments conducted, and their results. Data from reported assays were extracted to enable quantitative summaries. Raw data were gathered from the paper text or derived from digitally traced figures, using Plot Digitizer Version 2.6.8.
For analyses, “MSC dysfunction” was defined as “a difference between healthy and CVD MSCs” in one of the prespecified categories that focused on general MSC characteristics and/or regeneration: flowcytometric (FACS) marker expression of the ISCT minimal set, trilineage differentiation, proliferation, cytokine secretion, immunomodulation, angiogenesis/tissue repair, and senescence. In studies that did not report dysfunction in one of these categories but did find alterations or impairment in another area, an additional category (“any other dysfunction”) was created and scored.
Assessment of Risk of Bias
No validated “risk of bias” instrument exists for in vitro studies that utilize cells. For this purpose, findings were coded as 0 (no dysfunction) or 1 (dysfunction present). Reporting was similarly coded as 0 (not reported) and 1 (reported).
We assessed the correlation between the reporting of the study characteristics listed above and positive study results with Fisher’s exact test. The correlation between continuous variables (age of CVD or control participants, delta age) and reporting was assessed with logistical regression.
Studies containing in vivo animal experiments were assessed using the Hooijmans risk of bias tool 31 . Funnel plots were constructed using ratio of means (meanCVD/meancontrol). The inverse of the square root N was used to estimate the standard error 32 .
Other Statistical Analyses
Quantitative analyses are reported as ratio of means (meanCVD/meancontrol). As reliable estimates of variance were not available for a large portion of the included studies, no meta-analysis was performed. Distribution of study results was, however, reported by weighted mean on the square root of the sample size (N). To assess the correlation between delta age or passage number and effect ratio, data were analyzed by using mixed effects models using restricted maximum likelihood estimation.
All statistical analyses were conducted using IBM SPSS Statistics Version 24.0 or in “R” software (Version 2.15.3). A P value of <0.05 was considered statistically significant.
Results
The scope and quality of the studies widely differed. Some studies only examined the ISCT MSC minimal criteria—the presence of extracellular MSC markers and the absence of hematopoietic markers, and sometimes fewer than all 3 lineages of differentiation. Others conducted a thorough assessment of multiple MSC parameters, including senescence, immunomodulation, and tissue repair capabilities, sometimes including animal models as well. Table 1 lists the characteristics of the studies. A summary of the effect of disease on MSC function is provided in Table 2 for in vitro studies, and Table 3 details studies with an in vivo component. Below, we will discuss the studies conducted per CVD entity.
Study Characteristics.

Abbreviations: CVD, cardiovascular disease; AID, autoimmune disease; CAD, coronary artery disease; HF, heart failure; CLI, critical limb ischemia; PAD, peripheral artery disease; DM, diabetes mellitus; DMG, gestational diabetes mellitus; CKD, chronic kidney failure; SSc, systemic sclerosis; lcSSc, limited cutaneous SSc; dcSSC, diffuse cutaneous SSc; BM, bone marrow; AT, adipose tissue; sc, subcutaneous; UCB: umbilical cord blood; CABG: coronary artery bypass graft; HSCT, hematopoietic stem cell transplant.
In Vitro Assessment of MSCs.
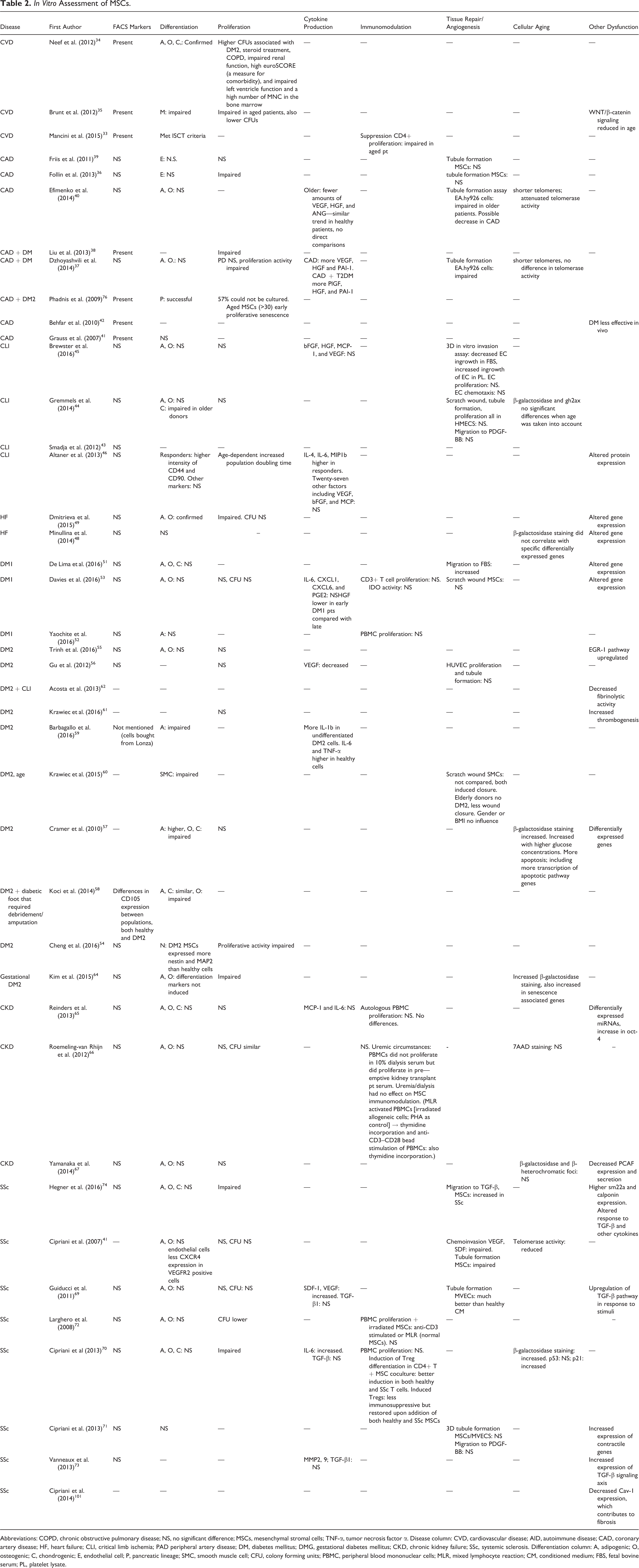
Abbreviations: COPD, chronic obstructive pulmonary disease; NS, no significant difference; MSCs, mesenchymal stromal cells; TNF-α, tumor necrosis factor α. Disease column: CVD, cardiovascular disease; AID, autoimmune disease; CAD, coronary artery disease; HF, heart failure; CLI, critical limb ischemia; PAD peripheral artery disease; DM, diabetes mellitus; DMG, gestational diabetes mellitus; CKD, chronic kidney failure; SSc, systemic sclerosis. Differentiation column: A, adipogenic; O, osteogenic; C, chondrogenic; E, endothelial cell; P, pancreatic lineage; SMC, smooth muscle cell; CFU, colony forming units; PBMC, peripheral blood mononuclear cells; MLR, mixed lymphocyte reaction; CM, conditioned medium; FBS, fetal bovine serum; PL, platelet lysate.
In Vivo Assessment of Mesenchymal Stromal Cells (MSCs).

Note. Table only contains preclinical studies.
aNumber of patients is not mentioned in 2 studies; CAD, coronary artery disease; DM, diabetes mellitus; CLI, critical limb ischemia; CKD, chronic kidney disease. LAD, left anterior descending artery; im, intramuscularly; iv, intravenously; ip, intraperitoneally.
Effect of Disease on MSC Characteristics and Regenerative Potential
General CVD
Three studies examined MSCs procured from patients undergoing elective cardiovascular surgery; no control group was present. Since the findings are presented in light of a correlation with specific patient factors, and not as a difference between individuals with cardiac disease and healthy controls, we discuss these studies separately.
Mancini et al. determined CVD risk/atherosclerosis status based on the planned procedure; 41 patients who underwent coronary bypass grafting (CABG) were considered to have atherosclerosis, whereas the 9 patients scheduled for a valve replacement were “nonatherosclerotic.” Mancini et al. subsequently related CVD risk to the ability of AT-MSCs to suppress anti-CD3/CD28-bead activated CD4+ T cell proliferation. Diabetes mellitus type 2 (DM2) and atherosclerosis were independently associated with a reduction in the immune suppressive capacity of AT-MSCs. ISCT minimal criteria were also determined but not compared between groups 33 .
Neef et al. found in their study examining BM-MSCs derived from 51 patients that colony forming unit (CFU) numbers were increased in DM2, steroid treatment, chronic obstructive pulmonary disease (COPD), impaired renal function, high euroSCORE (a measure for comorbidity), impaired left ventricle function, and a high number of MNCs in the BM. However, in multivariate analysis, only a high MNC number in the BM and steroid treatment were predictive of a high CFU. There was no correlation between MNC numbers and age, myocardial infarction (MI) or cardiovascular risk status, so confounding by MNC number was not present. ISCT minimal criteria were not found to be altered 34 .
Brunt et al. performed gene expression analysis in BM-MSCS from 22 patients who underwent undefined cardiovascular surgery. The array contained 84 genes that are linked to the WNT/β-catenin signaling pathway that is involved in embryonic development and tissue repair. They found that 34 differentially expressed genes were associated with cardiovascular risk factors 35 .
Coronary artery disease
Coronary artery disease (CAD) MSCs did not differ from healthy MSCs with regard to ISCT minimal criteria. The majority of the studies did not include healthy controls (5/8).
Three studies showed a detrimental effect of CAD on proliferation; however, one study did not find any differences 36 –39 .
Two studies did not find differences between diseased and healthy MSCs of BM or AT origin in a tubule forming assay 36,39 , whereas 2 others did find impairments. For instance, Efimenko et al. found impairment of tubulogenesis in AT-MSC from CAD patients, possibly due to decreased secretion of proangiogenic factors and increased production of antiangiogenic factors. Efimenko et al. also found a shorter telomere length and less telomerase activity in MSCs from CAD patients in comparison with healthy controls 40 .
Dzhoyashvili et al. also found impaired tubulogenesis in CAD AT-MSCs; both conditioned medium from MSCs derived from patients with CAD only or CAD + DM2 performed worse than healthy MSCs. There was no difference between CAD only and CAD + DM2 MSCs. Vascular endothelial growth factor (VEGF) expression secretion was increased in CAD and CAD + DM2 MSCs as compared with healthy MSCs 37 . Dzhoyashvili et al. then show that impairment of tubulogenesis is caused by an increase in anti-angiogenic factors rather than a decrease in pro-angiogenic factors—expression of the anti-angiogenic cytokine thrombospondin-1 was higher in CAD patients as well. Although they were not able to confirm higher thrombospondin-1 secretion in the culture medium, thrombospondin-1 mRNA levels negatively correlated with the results of the tubulogenesis assay. Both expression and secretion of PAI-1 were elevated in CAD and CAD + DM2 MSCs. Inhibition of PAI-1 partially restored angiogenesis 37 .
Three studies evaluated the efficacy of MSC administration in an animal model of myocardial ischemia, but these did not include healthy controls. The studies demonstrate that CAD MSCs do display regenerative features in vivo; no conclusions regarding dysfunction can be drawn 41,42 . Liu et al. found that DM2 in addition to CAD confers less increase in ejection fraction, myocardial contractile ability, and more myocardial apoptosis than CAD only or vehicle control. The authors speculate that this is due to a decrease in VEGF and B-cell lymphoma 2 (Bcl-2) secretion, present in only the CAD and DM2 group, as determined by significantly lower Bcl-2 and VEGF levels in the myocardium posttransplantation 38 .
Critical limb ischemia
CLI does not appear to affect the angiogenic potential of MSCs, not even MSCs harvested from the BM of an amputated leg.
Smadja et al. first examined the angiogenic effect of CLI BM-MSCs versus control MSCs in a hind limb ischemia model. There was no difference between CLI and control with regard to limb perfusion 43 .
Gremmels et al. extensively examined MSCs derived from iliac crest BM. They report no differences between CLI MSCs and healthy MSCs with regard to gene expression, in vitro tubulogenesis, scratch wound closure, and migration toward platelet-derived growth factor (PDGF)-BB. ISCT minimal criteria were mostly similar, though chondrogenic differentiation was impaired in CLI donors. Senescence, measured as β-galactosidase, was increased in CLI patients. However, these differences were age dependent; after correction for age, no significant difference was found between healthy controls and CLI patients. Senescence was not correlated with angiogenic potential. Most notably, the in vivo angiogenic potential of MSCs-CLI MSCs was similar to healthy MSCs 44 . Brewster et al. examined MSCs derived from 8 amputated legs versus iliac crest BM from healthy volunteers. Interestingly, even though the legs had not been perfused for some time, the resulting MSCs functioned just as well as healthy MSCs with regard to proliferation, cytokine production and endothelial cell proliferation, migration, and invasion. The only parameter that differed was MSC invasion, which was lower in the CLI group that had been cultured in FBS—there were no differences between MSCs cultured in platelet lysate. There were no differences between MSCs from CLI patients with DM2 and MSCs from CLI patients without DM2 45 .
Altaner et al. assayed MSCs acquired from patients who participated in a clinical trial that examined the effect of autologous BM-MNC administration in CLI. While no controls were included, Altaner et al. created groups based on clinical response to BM-MNC administration, enabling correlations between the clinical efficacy of BM-MNCs and the in vitro assays of BM-MSCs. Various in vitro differences were identified; responders had increased expression of the cell surface markers CD44 and CD90. Responders also secreted more interleukin (IL)-4, IL-6, and macrophage inflammatory protein-1b (CCL4). Protein profiling of MSC lysates showed that responders expressed more Zinc finger protein (SNAI1), and nonresponders expressed more E-cadherin and pancreatic and duodenal homeobox-1 (PDX-1) 46 . The influence of MSC frequency in the administered BM-MNC product on therapeutic efficacy was not assessed. Given the low frequency of MSCs in unfractionated BM 47 , the consequences of these findings for MSC-based therapy are uncertain.
Heart failure
Two studies examined BM-MSC function in patients with ischemic heart failure as compared with healthy controls. In both studies, gene expression patterns, assayed with real-time polymerase chain reaction arrays containing either 85 or 84 prespecified transcripts, were found to reflect adaptation of cardiac tissue to ischemia. Minullina et al. used the heart failure samples to assess the relation of differentially expressed genes and patient-specific factors due to significant differences between the healthy and disease group (gender balance, age). Body mass index was the only factor that could be linked to gene expression; nonobese individuals had an increase in gene expression associated with extracellular matrix (ECM) homeostasis such as Collagen Type I Alpha 2 Chain (COL1A2) and connective tissue growth factor (CTGF) and several matrix metalloproteinases (MMPs) in nonobese individuals. Transcript levels in obese individuals were similar to healthy controls. Interestingly, β-galactosidase activity, a measure for cellular senescence, did not correlate with differential gene expression in heart failure, although the authors do not report whether healthy controls were also examined 48 .
In addition to gene expression analyses, Dmitreva and colleagues conducted functional assays. Differentiation was unaffected. They found a lower population doubling time at p3 in the HF group, but no differences in CFU. This difference might be caused by a higher number of a-SMA+ CD105+ cells in the heart failure samples—possibly reflecting fibroblast contamination 49 .
Two clinical trials comparing autologous versus allogeneic MSCs as treatment in heart failure have been conducted. While these were not geared toward assessing regenerative potential, some conclusions can be drawn. The POSEIDON study does not clearly show benefits of one over the other; both groups display favorable structural cardiac changes. However, left ventricular function was only significantly improved in the allogeneic group, whereas functional outcome measures only significantly improved in the autologous group as compared with the allogeneic group 28 .
The POSEIDON-DCM study in nonischemic dilated cardiomyopathy demonstrated that patients treated with allogeneic MSCs had a greater improvement in clinical parameters. Differences in circulating immune cells were also found; in the allogeneic MSC group, patients had less terminally differentiated effector memory T cells and more memory B cells with a suppressed phenotype. T cell activation (measured as CD3+ CD25+ T cells) did not differ. Importantly, patients who had received allogeneic MSCs had an increase in EPC colony forming units, whereas patients who had received autologous MSCs had not 50 .
Diabetes mellitus type 1
Three studies evaluated diabetes mellitus type 1 (DM1) BM-MSCs versus healthy controls. No difference between DM1 cells and healthy controls was found with regard to differentiation and proliferation 51 –53 . Immunomodulation as measured with peripheral MNC or selected T cell proliferation was similar in 2 studies 52,53 . In a mouse model for diabetes, Yaochite et al. found that intrasplenically injected DM1 MSCs resulted in similar glucose control as treatment with healthy MSCs, showing that DM1 MSCs and healthy MSCs have similar therapeutic efficacy in DM1 52 .
Gene expression profiling was conducted in 2 studies (microarrays), and differentially expressed genes were found in DM1 MSCs 51,53 . De Lima et al. most notably found differences in pathways involved in migration, so migratory capacity was assessed—DM1 MSCs performed better than healthy MSCs. Functional pathway analysis revealed upregulation of the sympathetic nervous system. Davies et al. found increased transcription of pathways involving growth, development and response to stress and wounding in late DM1. There were no differences in the scratch wound assay (MSCs only) 51 . Davies et al. also found that baseline secretion of cytokines (IL-6, CXCL1, CXCL6, and PGE2) was similar to healthy control levels. Hemocompatibility was also similar, even though late DM1 cells express more CD55 (complement receptor) on the surface of the cells. Late DM1 even had less clot formation, possibly correlating with the lower expression of CD59 as compared with healthy cells 53 .
Diabetes mellitus type 2
Nine studies examined the (dys)function of AT-derived MSCs (AT-MSCs) in patients with diabetes mellitus type 2 compared with nondiabetic controls. Results are mixed—some studies point toward disease-related dysfunction, whereas others find no differences at all. Proliferation of AT-MSCs was reported in several studies to be similar or even better than healthy controls, but another study found slower proliferation 54 –57 . Similar mixed results were found in differentiation assays: osteogenic and chondrogenic differentiation was unaffected in one study 55 but impaired in 2 other studies 57,58 , and adipogenic differentiation was reported to be increased in one study 58 but completely absent in another 59 . The 2 studies that looked into gene expression found that DM2 AT-MSCs express genes implicated in the pathogenesis of DM2, demonstrating that AT-MSCs may be affected 55,57 . Tubulogenesis was unaffected 56 . Scratch wound healing (smooth muscle cells) was more efficient with DM2 CM 60 .
In vivo angiogenesis was evaluated by 2 studies, using the murine ischemic skin flap model. Trinh et al. observed a bigger necrotic surface area, less neoangiogenesis, and more CD45+ cell infiltrate in animals treated with DM2 AT-MSCs, but DM2 cells still performed better than vehicle control. Trinh et al. found EGF-1 to be involved in DM2-mediated MSC dysfunction, a pathway also affected in the DM2 disease phenotype. Inhibiting EGR-1 expression increased skin survival in the ischemic skin flap model 55 . Gu et al., however, did not see differences in the ischemic skin flap model with regard to necrosis 56 . This could be due to the heterogeneity of the protocols—different mouse species were used as well as different cell doses and injection protocols. Expression of cytokine mRNAs and secretion of cytokine proteins were evaluated in 4 studies. VEGF mRNA expression was higher in DM2 AT-MSCs in one study but lower in another study 58 . Gu et al. who determined elevated VEGF mRNA in DM2 AT-MSC subsequently showed that VEGF protein levels were lower in CM, but still performance in the ischemic skin flap model was similar 56 . Reports are similarly mixed about IL-6; some studies find higher baseline IL-6 levels (both protein and mRNA), others find lower IL-6 levels as compared with controls. It is also unclear whether transforming growth factor beta-β and bFGF secretion are affected.
Two studies point toward a possible thrombogenic effect of diabetic AT-MSCs. Krawiec et al. evaluated healthy, DM2 and elderly AT-MSCs in a murine aorta interposition model. Seeding of the constructs was successful and no differences were seen between groups. However, DM2-MSC-seeded grafts were thrombogenic—almost all grafts had to be explanted within 1-wk postimplantation—28% patency at 8 wk. Subsequent assays showed that DM2 AT-MSCs produced less fibrinolytic factors as compared with healthy MSCs 61 . Acosta et al. studied the secretion of fibrinolytic mediators by MSCs from DM2 patients after observing 2 instances of distal microvascular thrombosis in a clinical trial that examined intra-arterial administration of AT-MSCs for CLI. DM2 MSCs indeed displayed impaired fibrinolytic properties and secreted more plasminogen activator inhibitor type 1 and less tissue plasminogen activator than healthy MSCs, regardless of type blood serum 62 .
In DM2, one study reported higher senescence in DM2 AT-MSCs as compared with healthy controls. Senescence even increased with higher glucose content of the medium and could be rescued with insulin supplementation to the culture medium 57 .
The above studies in DM2 all examined AT-MSCs. In a review of 14 clinical studies in which autologous BM-MSCs were administered to 132 DM2 patients, van de Vyver describes that the results of MSC therapy are variable and that some patients do not respond at all 63 . This underscores the need for comparative studies in DM2 and healthy BM-MSCs.
Gestational diabetes mellitus
Kim et al. found that UCB-MSCs from mothers with diabetes mellitus gravidarum (DMG) proliferate slower than UCB-MSCs derived from healthy mothers. Osteogenic and adipose differentiation were negatively affected. Kim et al. also observed signs of mitochondrial dysfunction at both the gene expression and protein level in DMG UCB-MSCs. Increased β-galactosidase activity was seen in DMG MSCs 64 .
Chronic kidney disease
BM-MSCs from chronic kidney failure (CKD) patients were assayed extensively by Reinders et al. from FACS markers and differentiation to miRNA profiles and PBMC proliferation, but no differences were found between healthy MSCs and CKD MSCs 65 . Similarly, for AT-MSC, no differences in these parameters found between healthy controls and CKD MSCs. Roemeling-van Rhijn et al. assayed healthy and CKD AT-MSCs under both normal and uremic conditions with regard to suitability for cell therapy: aside from general MSC parameters, long-term genetic stability and apoptosis were taken into account. They also determined MSC-mediated suppression of PBMC proliferation. No differences between healthy and CKD MSCs were found 66 . Similarly, Yamanaka et al. did not find any differences between CKD and controls in ISCT minimal criteria, proliferation, and senescence. However, they do report a defect in the in vivo angiogenic potential of AT-MSCs. They also found a reduced gene and protein expression of PCAF, a regulator of HIF-α, which might underlie this angiogenesis defect 67 .
Systemic sclerosis
SSc is not traditionally considered a CVD, however, given the disease’s profound vascular manifestations as well as the presence of fibrosis, which is ubiquitous in CVD, research conducted in SSc MSCs might provide valuable insights—SSc can be considered a model disease in this sense 68 . Even though in SSc loss of angiogenic potential is a key feature, SSc MSCs retain their angiogenic and immunomodulatory properties, as shown in 2 studies, though 1 study reports impaired tubulogenesis 69,70 . In a coculture assay, Cipriani et al. furthermore showed that healthy endothelium downregulates SSc MSC transcription of contractile genes that are associated with a profibrotic phenotype 71 . Some studies report disease-specific profibrotic features in SSc MSCs, for example, higher expression of contractile genes upon TGF-β signaling, and general increased response to TGF-β in terms of receptor upregulation and increased downstream signaling 72 –74 . Whether a priori TGF-β receptor upregulation is present is contested by contrasting findings. Interestingly, even healthy MSCs display some response to profibrotic stimuli, though they also downregulate their TGF receptors, whereas SSc MSCs upregulate these. SSc MSCs generally display a more senescent phenotype than healthy controls, as demonstrated by 2 studies with age-matched controls 70,75 .
Aging and MSC Dysfunction
Aging in MSC dysfunction can be considered as an additive effect “on top of” potential CVD-mediated dysfunction or as an independent factor influencing regenerative capacity. The latter can only be detected in studies that included healthy MSCs.
In studies lacking healthy controls, generally a degree of age-dependent dysfunction was observed in post hoc analyses. Proliferation was impaired in aged patients in 2 studies, but one study found no such impairment 34,35,76 . Extracellular markers were not affected. With regard to regenerative capacity, Mancini et al. found that age impaired immunomodulation in AT-MSCs from CVD patients 33 .
Brunt et al. furthermore reported impairment of myogenic differentiation in older donors. Increasing age was also associated with less activity of the WNT/β-catenin signaling pathway in MSCs. However, though the authors assert that the WNT/β-catenin alterations are mainly age dependent, the differentially expressed genes in aged patients are all but one similar to differentially expressed genes in individuals with high cardiovascular risk score as compared with people with low cardiovascular risk score 35 .
In studies with controls, conflicting results were seen in the small number of studies that separately analyzed the contribution of biological or cellular aging to possible dysfunction. MSCs derived from elderly CAD patients had less angiogenic potential than MSCs derived from elderly healthy patients. This was shown to be due to an age-dependent decrease in VEGF production in combination with a CAD-mediated increase in anti-angiogenic factors 40 . On the other hand, in a study in CLI, all differences between healthy and diseased individuals were found to be due to advanced age in the CLI group 44 . Additionally, aged MCs from DM2 patients had a strong prothrombogenic effect whereas age-matched healthy MSCs did not 61 .
The relationship between MSC donor age and treatment efficacy in heart failure was assessed by Golpanian et al. in a retrospective analysis of pooled data from 2 clinical trials. Donor age did not affect the outcomes in MSC recipients. However, in pooling, the data autologous/allogeneic recipients were mixed, and no separate analysis comparing donor age and allogeneic/autologous cells was conducted 77 .
Not all studies were fully age-matched, possibly introducing confounders. Therefore, we collated all the study results and tested for an association between age-mismatched groups (age matched: average ages within 10 yr) and MSC dysfunction using the χ2 test. We found that there was no association between age matching and proliferation, cytokine secretion, FACS marker expression, immunomodulation, altered differentiation, angiogenesis, senescence, or any in vitro dysfunction in general.
Linear regression analysis did not reveal a significant association between delta age and tissue repair/angiogenesis ratio of means (Fig. 1C). Logistic regression did not reveal significant associations between MSC dysfunction and the difference in average ages (delta age; CVD control) between groups (P = 0.583). There was also no association between MSC dysfunction and average age of CVD patients (P = 0.232), nor age of healthy controls (P = 0.597).

Risk of bias analyses. (A) Funnel plot depicting the distribution of senescence ratio of means. (B) Funnel plot depicting the distribution of angiogenesis ratio of means. (C) Association of delta age versus angiogenesis ratio of means (circle: in vitro; triangle: in vivo) P = 0.65. (D) Analysis of passage number versus angiogenesis ratio of means (circle: in vitro; triangle: in vivo) P = 0.10.
Risk of Bias
As no standardized criteria exist for assessing risk of bias in in vitro studies, we assessed whether the reporting or lack thereof of certain parameters is associated with positive results. These parameters encompass reporting of the study population—such as gender, age, and reporting of culture and experimental procedures such as the media used, seeding density, and the passage number used.
Various types of controls were utilized (see Table 1), from screened organ or BM transplant donors to individuals undergoing surgery not related to the CVD in question. The N of each control type was too low to allow for direct correlation with study outcomes; however, the use of screened transplant donors trended toward an association with fewer findings of dysfunction (P = 0.096).
There was no clear association between reporting of study characteristics and in vitro findings, mainly because most features were generally well reported (Table 4). Nonreporting of the seeding density was significantly associated with differentiation alterations (P = 0.018). With regard to senescence, nonreporting of the seeding density trended toward a significant association (P = 0.061), but the total N of studies that examined senescence was low.
Reporting of Study Characteristics.

Analysis of a compound measure for reporting (“all parameters reported”) showed that findings of dysfunction in proliferation and angiogenesis are possibly correlated with not reporting at least one of the parameters. However, only 5 studies reported all parameters. Similarly, not all studies reported on all MSC functional characteristics. The passage number did not affect ratio of means with regard to tissue repair/angiogenesis (Fig. 1D).
The Hooijmans tool was used to assess risk of bias in animal studies. Some of the studies included blinded outcome assessment, but many of the details specified in the Hooijmans tool were lacking (data not shown). Four of the 6 studies that included healthy controls found no difference between healthy and CVD MSCs.
To assess publication bias, we created funnel plots using ratio of means data from studies assessing angiogenesis and senescence (Fig. 1A and B). No clear trend was observed that indicates publication bias with regard to either favorable or unfavorable effects of CVD on MSC function. It must be noted however that there was high heterogeneity in study results and trend toward overrepresentation of extreme findings in either direction.
Discussion
Assessment of MSC (Dys)function
We provide the first comprehensive literature review on MSC dysfunction in CVD (See figure 2 for a summary of our findings). Reliable assessment of disease-related (dys)function is essential to gain more insight into the potential clinical efficacy of MSCs in the various CVDs.

Summary of findings.
Our data show that in most studies, MSCs from CVD patients perform less well than healthy control MSCs in in vitro assays, but there were also studies in which multiple tests were used and no differences were found between healthy and CVD MSCs. In vitro assays may not fully capture the complex interactions between host and donor cells. The emphasis on the ISCT minimal criteria in many of the studies thus far is remarkable, as the ISCT minimal criteria do not bear any relationship with clinical efficacy. After all, the primary modalities of action of MSCs are through influencing the local environment by secretion of paracrine factors, and direct cell-to-cell contact, not through differentiation. No differences between diseased MSCs and control MSCs were found with regard to the ISCT extracellular marker set. In CLI, DM2 and DMG, there are indications that trilineage differentiation abnormalities are present.
Assays that do capture important aspects of MSC regenerative properties, such as immunomodulation, wound repair, and angiogenesis assays, were not often performed, and which aspect was examined varied widely. In some disease states such as DM1, MSCs were minimally affected, whereas in others profound MSC dysfunction was present in multiple aspects of regeneration, for example, in DM2 and SSc. Specific studies into disease-related dysfunction less focused on regenerative potential were also conducted, such as identification of disease-related alteration of cellular pathways or differentiation into cell types implicated in disease. For instance, in SSc and CVD, an impairment in differentiation to, respectively, endothelial and myogenic differentiation was found. As these lineages were not consistently assessed across all conditions, it is unknown whether this is unique for these disease entities. Given the profound role of immunomodulation in MSC-mediated tissue repair, it is surprising that such a small number of studies (7/41) conducted assays to assess this aspect of MSC function. Moreover, the assays used do not resemble in vivo immune function, nor do they recapitulate MSC-mediated immunomodulation, so the question remains whether the absence of dysfunction in these assays represents in vivo immunomodulatory capacity 78 .
Only few of the studies performed included in vivo testing of MSCs. Additionally, these studies often showed conflicting results, for instance, in DM2, where one study found DM2-associated impairment of angiogenic potential in a skin flap model and another found no impairment. Interestingly, some studies that found in vitro alterations, including reduced cytokine secretion or increased senescence in diseased MSCs, did not find a difference in in vivo efficacy 44,56 . However, generally, in vitro and in vivo assays were rarely combined. In vivo testing would be most useful to assess disease-related MSC (dys)function, as this allows simultaneous testing of multiple aspects of MSC function, such as tissue repair, angiogenesis, and immunomodulation. While, indeed, in vitro findings do not necessarily predict in vivo efficacy, cross-referencing may shed light on the relevance of detected in vitro abnormalities. Gremmels et al. conducted a thorough analysis of the predictive value of in vitro assays in predicting in vivo angiogenesis. The extent in which MSC CM stimulated endothelial proliferation was most predictive for in vivo results 44 . Such analyses are essential to gain more insight into how to gauge MSC potency. This does not only guide the decision to use autologous or allogeneic cells but can also aid in individual donor selection.
The discrepancy between in vitro and in vivo assessments of MSC function may also reflect a difference between animal models and the human body—for example, in a murine study, inappropriate differentiation of cells has been shown in cardiac tissue 79 , but, to date, this has not been observed in humans yet 28,80 . The diseased environment may also drive MSCs toward a regenerative phenotype, as has been shown previously 81 . On the other hand, it has also been shown that implantation in a diseased tissue can derail MSCs 82 . Additional indications for a negative influence of disturbed homeostasis in CVD were found in studies, in which diseased conditions such as hyperglycemia and uremia were modeled in vitro or in animal models—progenitor cell dysfunction was found 83 –85 . Adding patient-derived serum to healthy control MSCs also induces dysfunction in several diseases 86,87 . In line with these findings, Cramer et al. report glucose dose-dependent dysfunction in both healthy and diseased MSCs, but interestingly Roemeling-van Rhijn et al. did not find a deleterious effect of uremia in AT-MSCs 57,66 .
Aging-related dysfunction in regenerative potential, such as impaired proliferation, differentiation, immunomodulation, and angiogenesis, was reported in CVD MSCs in a small number of studies, corresponding with findings in healthy, aged MSCs 33,35,40,61 .
Features of premature cellular aging and increased senescence were assessed in some, but not all, CVD. Cellular aging was reported in CVD MSCs in several conditions, which could reflect a true premature aging phenotype in the progenitor cell compartment. For instance, individuals with CVD have shorter telomeres than their matched controls, and telomere length has been shown to inversely correlate with cardiovascular events 88 . Features of increased cellular senescence, another consequence of aging, can also be detected in CVD patients 89 . Unfortunately, few studies assessed cellular aging parameters, and of these, even fewer correlated the results with patient age, precluding firm conclusions regarding the relationship between premature aging, patient age, and MSC dysfunction. In our analysis of ratio of means, we did not find an association between delta age and senescent phenotype, neither was delta age associated with tissue repair/angiogenesis and immunomodulation ratio of means.
We assessed correlations between CVD patient age, healthy control age, and the age difference between patient and controls and study results. None of these parameters were associated with reported MSC dysfunction. Therefore, there is no evidence that disease-related dysfunction in CVD is due to biological age rather than the disease itself.
Limitations
Our review evaluated cultured MSCs—bearing relevance for possible clinical applications—and does not allow conclusions on the native MSC population—if such a circumscript population exists at all. Since it has been established that cells can lose distinctive phenotypes while in culture 90 , possible differences might have been masked by the prolonged culturing required to generate a sufficient number of MSCs. However, in most studies reviewed here, cells are not cultured much beyond passages used for therapeutic applications, enabling conclusions about the final cellular therapy product.
Not all MSC harvesting locations were equally explored in each CVD; thus, it is not always clear whether reported MSC dysfunction reflects local dysfunction or a global disease phenomenon. It is not known to what extent harvesting location influences cellular characteristics, some studies found differences whereas others, mostly in oncologic populations, found no differences 91 –93 . It has been shown that not all HSC compartments are equally perfused and that MSCs are predominantly arranged around blood vessels 94,95 . Differences in perfusion might thus account for differences in exposure to noxious stimuli and thereby influence cell characteristics in varying degrees. In CKD, no gene expression differences were found in BM-MSCs, but distinct alterations were present in AT-MSCs 65,67 .
It remains unknown why many conflicting findings have been reported. Reporting of study population characteristics is essential to identify possible confounders. For instance, healthy controls are not always completely healthy individuals but are sometimes recruited from patients who undergo orthopedic surgery. The target population for coronary bypass grafts and joint replacement overlaps, possibly creating a confounding effect. Furthermore, donors of control samples are not always assessed medically, and especially in the case of highly prevalent conditions such as the metabolic syndrome diabetes mellitus 2 or other insidious disease, this could also create confounding in otherwise matched samples. While it was not possible to analyze the relationship between each type of donor and study findings, there were no differences between studies that included screened donors versus studies that did not include screened donors.
General cell culture parameters have been shown in other studies to influence cell characteristics—for instance, culturing conditions influence paracrine secretion and gene expression in MSCs 96,97 . MSC passage number is inversely correlated with differentiation protocols 98 , and the initial seeding density influences proliferation 99 . Furthermore, immunomodulatory capacity has even been shown to be affected by confluence rather than initially seeded cell density 100 , though analysis of this variable was not possible as “confluence at the start of assays” is not a regularly reported parameter. However, reporting of specific study characteristics was not associated with MSC dysfunction. Bias may also have been introduced through mismatched control groups, as there were a few instances in which healthy gender- and age-mismatched controls were used in comparison with diseased, aged individuals; and in general, individuals in the healthy control populations were younger. However, we did not find a correlation between mismatching and findings of dysfunction, nor was there a correlation between study participant delta age and effect size or dysfunction.
Implications for Cellular Therapy
Disease-related MSC dysfunction in CVD may have important consequences for cellular therapy. One solution could be to use allogeneic donors; another solution could be to pretreat diseased cells to abrogate disease-specific dysfunction.
When opting for allogeneic donors, selection of these donors for age and comorbidity is essential. A general health screening, similar to the screening that BM transplant donors receive, should be implemented and has already been implemented in many centers. However, it should also be kept in mind that, as discussed above, diseased MSCs may function better in vivo than was expected based on in vitro findings.
The effect of preconditioning, or culturing under specific circumstances to achieve a certain effect in the cell or its efficacy, was explored in several studies, and profound effects were found. For instance, proliferation could be enhanced by using different concentrations of serum or even alternatives to “serum” (e.g., umbilical cord blood–derived serum or platelet lysate) 34,45 . Platelet lysate did not affect angiogenic potential of MSCs 45 . Culturing of CLI BM-MSCs under hypoxic circumstances stimulates cells to proliferate and secrete beneficial paracrine factors, also leading to better wound repair as assessed with a scratch wound healing assay 44 . Administration of insulin to DM2 AT-MSCs could rescue many disease features 57 . In contrast, hypoxic culturing of AT-MSCs derived from CKD patients did not result in abrogation of the disease phenotype, suggesting that depending on the specific CVD, different strategies should be applied 67 . Other approaches focused on repairing disease-induced dysfunction of cellular pathways. Brunt et al. were able to increase nuclear translocation of β-catenin in aged MSCs by treating them with lithium. This also enhanced myogenic differentiation, showing that influencing the WNT/β-catenin pathway can rescue disease phenotypes in CAD, such as the approach of Trinh et al. to downregulate the EGR-1 pathway in DM2 AT-MSCs and the approach of Behfar et al. to use a cocktail of small molecules that enhance “cardiopoiesis.” Both approaches improved in vivo efficacy of MSCs in an animal disease model 42,55 . Administration of specific growth factors may also be an option—in SSc, Cipriani et al. showed that administration of VEGF to SSc BM-MSCs enhances tubule formation (but still does not reach normal levels) and administration of PDGF-BB decreases the expression of contractile proteins 70,75 . Given the variable results of precondition strategies across the spectrum of CVD, any such measure taken should be carefully evaluated.
Conclusion
MSC characteristics are affected by CVD in various degrees, but studies are conflicting as to in what extent this affects regenerative potential, if at all. Linear regression analysis of pooled data did not show an association between age and in vitro dysfunction, suggesting that age is not a confounder of importance in these studies. Future studies should focus on identifying which in vitro assay best predicts in vivo or preferably clinical efficacy, and these assays should become a vital part of any study aiming to assess (dys)function of MSCs.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Support by the ZonMw Translational Adult Stem Cell Research Programme (Grants Nos. 116003005 and 116001026) is gratefully acknowledged.
