Abstract
Systemic lupus erythematosus (SLE) is a chronic systemic autoimmune disease associated with impaired organ functions that can seriously affect the daily life of patients. Recent SLE therapies frequently elicit adverse reactions and side effects in patients, and clinical heterogeneity is considerable. Mesenchymal stromal cells (MSCs) have anti-inflammatory, tissue repair, and immunomodulatory properties. Their ability to treat autoimmune diseases largely depends on secreted extracellular vesicles, especially exosomes. The effects of exosomes and microRNAs (miRNAs) on SLE have recently attracted interest. This review summarizes the applications of MSCs derived from bone marrow, adipocyte tissue, umbilical cord, synovial membrane, and gingival tissue, as well as exosomes to treating SLE and the key roles of miRNAs. The efficacy of MSCs infusion in SLE patients with impaired autologous MSCs are reviewed, and the potential of exosomes and their contents as drug delivery vectors for treating SLE and other autoimmune diseases in the future are briefly described.
Introduction
Systemic lupus erythematosus (SLE) is a chronic, systemic inflammatory disease that affects the urinary, motor, circulatory, respiratory, digestive, and nervous systems (Fig. 1). It develops in various populations, especially in women of childbearing age and is particularly prevalent among Africans 1 . It is generally characterized by alternating periods of remission and relapse, with little progressive deterioration 2 . The cellular and molecular mechanisms involved in the pathogenesis of SLE are highly complex and are probably affected by intricate interactions among genetic, environmental, and hormonal factors 3 .

Main clinical manifestations of systemic lupus erythematosus. Systemic lupus erythematosus is a chronic, systemic inflammatory disease that affects the urinary system, motor system, circulatory system, respiratory system, digestive system, and nervous system. Different patients are often affected by different systems and, so the clinical manifestations are also different. Clinically, skin and mucosal injuries (e.g., lupus malar rash, oral ulcer, photosensitivity, alopecia, maculopapular rash, etc.) and lupus nephritis are most common, but arthritis and serositis may also occur, and the condition is critical when neuropsychiatric manifestation is present. The clinical heterogeneity of the disease highlights the important influence of genetic factors, autoimmunity, and external environment on the disease development model, and brings challenges to the diagnosis and treatment.
A complete cure and universally effective treatment for SLE are not available. Most therapeutic schedules are designed to control and reduce disease activity. Therapeutic pharmaceuticals used to relieve lupus symptoms include non-steroidal anti-inflammatory, antimalarial, immunosuppressant drugs, and glucocorticoids 4 . However, these drugs carry a high risk of serious adverse reactions. In particular, long-term medication with glucocorticoids is often associated with serious adverse events involving the endocrine, musculoskeletal, hematopoietic, and cardiovascular systems. Osteonecrosis of the femoral head is a common and serious complication of glucocorticoids 5 .
Considering the severity of the disease, other feasible treatments for SLE have been investigated. Extracellular vesicles (EVs) derived from mesenchymal stromal cells (MSCs) 6 , exosomes and their micro (mi) RNAs might serve as cell-free approaches to treating SLE. Here, we review the application of potential of exosomes derived from MSCs and miRNAs to treating SLE.
Mesenchymal Stromal Cells
Mesenchymal stromal cells are a subset of non-hematopoietic adult stromal cells 7 with remarkable proliferative ability and potential for multiple differentiation, as well as repair, anti-inflammatory, and immunomodulatory properties (Fig. 2). Techniques for isolating and culturing MSCs from tissues, including bone marrow, adipose tissue, synovium, umbilical cord (UC), and gingiva, are gradually being refined.

General characteristics of mesenchymal stromal cells. Mesenchymal stromal cells can be isolated from a variety of organs and tissues such as bone marrow, adipose tissue, umbilical cord, synovial membrane and gingiva. In addition to their good proliferative capacity and typical tri-lineage differentiation (osteogenic, chondrogenic, and lipogenic differentiation) potential, the potential for differentiation to other cells such as neurocyte, endothelial, cardiocyte, and hepatocyte, which are essential for their tissue repair function, is gradually being explored. In addition, MSCs have received extensive attention for their anti-inflammatory and immunomodulatory roles (both acquired and innate immunity) through secretion of cytokines and extracellular vesicles.
Anti-Inflammatory and Restorative Functions of MSCs
Mesenchymal stromal cells exert anti-inflammatory effects by alleviating inflammation and reducing cytokine production 8 . Exosomes from bone marrow–derived MSCs (BMMSCs) express miR-146a-5p, which inhibits the expression of interleukin-1 receptor-associated kinase 1 (IRAK1) 9 , regulates the Th17/Treg cell imbalance, and reduces pro-inflammatory cytokine production. Furthermore, MSCs eliminate the inflammatory effects of neutrophils, inhibit the formation of neutrophil extracellular traps (NETs), and reduce thrombosis, inflammation, and fibrosis induced by NETs 10 . Mesenchymal stromal cells can migrate and aggregate to sites of inflammation and promote injury repair. For example, allogeneic MSCs can migrate to an injured kidney after infusion. In addition to their immunomodulatory role and suppressing the autoimmune response, MSCs can be differentiated into mesangial cells to treat lupus nephritis (LN) 11 .
Immunoregulatory Properties of MSCs
Mesenchymal stromal cells exert unique immunosuppressive effects in innate and adaptive immunity by inhibiting the proliferation and activities of immune cells such as macrophages, dendritic cells (DCs), natural killer (NK) cells, B lymphocytes, and T lymphocytes 11 .
Mesenchymal stromal cells derived from human gingiva (GMSCs) reduce the infiltration of CD8+ T, Th1, Th17, and other pro-inflammatory cells, and increase the proportion of immunosuppressive cells such as regulatory T (Treg) cells 12 . Mesenchymal stromal cells interfere with B cell proliferation and migration, as well as antibody and cytokine production 1 . They can also indirectly inhibit B cell function by inhibiting T cells 13 . Macrophages can take on two main phenotypes: inflammatory M1 and anti-inflammatory M2 macrophages. Most MSCs injected intravenously into mouse models of asthma travel through the bloodstream and eventually reach the lungs, where they die and are phagocytosed by macrophages. Such macrophages can differentiate into the immunosuppressive M2 phenotype and decrease the M1/M2 ratio in vivo, which is one mechanism through which MSCs play a regulatory role 14 .
Extracellular Vesicles Released From MSCs
In addition to classical soluble factors such as cytokines that play a role in paracrine signaling, the role of EVs released from MSCs (MSC-EVs) in intercellular communication should not be ignored 15 . Extracellular vesicles are a class of cell-derived heterogeneous membrane structures that can be apoptotic bodies, microvesicles, microparticles, or exosomes 16 . The immunosuppressive effects of MSCs are mainly manifested through their EVs 17 . Exosomes are rich in proteins, transcription factors, lipids, DNA, miRNAs, mRNAs, and cytokines. They act on target cells by binding to receptors in target tissues or by fusion with plasma membranes 18 .
Diagnostic Value of Exosomes and Their miRNAs
Exosomes are involved in cell migration, the immune response, cell differentiation, antigen presentation, and tumor invasion, among which miRNAs play important roles19,20. Non-coding miRNAs can regulate pathways associated with gene expression by interacting with specific targets after their extracellular transport by EVs 21 . Micro RNA entry into exosomes is not random and is mediated by unique miRNA sorting mechanisms 22 , such as the Endosomal Sorting Complex Required for Transport (ESCRT) 23 . Exosomes and their miRNAs have potential as diagnostic and therapeutic agents in autoimmune diseases 24 . For example, primary fibrosis in LN is closely associated with exosomal miR-129 isolated from urine, suggesting a predictive role of miR-129 in disease progression 24 . The expression of miR-21 and let-7A in urinary exosomes is obviously downregulated in patients with active LN compared with those who have inactive LN, but this rebounds during remission, suggesting that they could serve as biomarkers of active LN activity 25 . Using MSC-EVs to deliver miRNAs to specific cells is a potential mechanism of alleviating tissue damage 26 . Elevating the expression of programmed cell death ligand 1 via gene transfection in EVs of MSCs enhances their immunosuppressive ability 27 . This indicated that enhancing EV function has potential clinical applications.
Advances in exosomes and their miRNAs as biomarkers have suggested their potential therapeutic value. Exosome-based therapies avoid the need to administer live cells 6 . Therefore, exosome-based therapies might be suitable alternatives to conventional cell therapy for SLE. Exosomes derived from antigen-presenting cells such as DCs and MSCs are currently regarded as potential cell-free approaches to treating autoimmune diseases 28 .
MSCs in SLE Treatment
Allogenic MSC Transplantation in SLE
Bone marrow, adipose tissues, gingival tissues, UCs, and the synovium are sources of allogenic MSCs that can be used to treat SLE.
Bone marrow–derived MSCs and exosomes
For 40 years since their initial description, BMMSCs have become the most widely studied MSC population and are considered the gold standard for clinical MSC application 29 . The production of autoantibodies in patients with SLE depends on the activation of T-cell-assisted B cells. Therefore, regulating the activity of upstream T cells might restrict excessive B-cell activity in SLE 13 . Clinically, the immunological regulation of MSCs is applicable not only as therapy for SLE but also for connective tissue diseases (CTDs), graft-versus-host disease (GVHD), and other immune disorders. For example, a clinical trial found that BMMSCs relieved symptoms in 25 patients with GVHD without significant adverse events 30 .
Not only do BMMSCs inhibit the differentiation of T cells into T follicular helper (Tfh) cells, they also inhibit differentiated Tfh cell proliferation and interleukin (IL)-21 production, thus alleviating LN in mice 31 . Human BMMSCs inhibit T-cell-mediated B-cell proliferation, plasma cell differentiation, and antibody production in vitro 13 . In addition, MSCs secrete exosomes containing miRNAs that inhibit cytokine secretion in macrophages by regulating Toll-like receptor (TLR)-related signaling pathways 32 . Transfer RNA–derived small RNA (tsRNA)-21109 in BMMSC exosomes inhibits macrophage polarization toward the M1 phenotype in vitro, thus contributing to the development of a new specific therapeutic target for SLE 33 . Allogeneic BMMSC transplantation also enhances the function of autologous BMMSCs and improves osteopenia in a mouse model of lupus 34 .
However, bone marrow might not be the optimal source of MSCs. In addition to an invasive and painful isolation procedure, MSCs are scant in bone marrow aspirates, and their qualitative and quantitative properties are affected by donor age 35 .
Adipose tissue–derived MSCs and exosomes
Differentiation potential, proteomic characteristics, gene expression, and immunological characteristics differ between adipose tissue–derived MSCs (ADSCs) and BMMSCs. Adipose tissue–derived MSCs are easier to isolate and clinically safer to collect than BMMSCs 36 . Furthermore, ADSCs more effectively inhibit peripheral blood T cells, are more regenerative, and have more adaptable electrokinetic properties than BMMSCs 37 . Combining ADSCs with exosomes is more effective than injecting them alone and could be considered as a future stromal cell treatment option38,39. The results of treating a patient with SLE in Canada using autologous ADSCs were significant and stable 40 . A clinical trial in Iran found that ADSC transplantation is effective against LN, whereas a single dose might be insufficient to maintain remission 41 .
Adipose tissue–derived MSCs inhibit the initial differentiation of T cells into Th17 cells and reduce IL-17 secretion by regulating the protein kinase B/mammalian target of rapamycin complex 1/Ribosomal protein S6 kinase beta-1/Hypoxia-inducible factor 1-alpha/Th17 (AKT/mTORC1/p70S6K/HIF-1A)/Th17 signaling pathway in MLR/lpr mice 42 . Adipose tissue–derived MSCs also induce the differentiation of macrophages into the M2 phenotype via the exosomal-mediated transfection of active signal transducer and activator of transcription 3 (STAT3) 43 . Controlled clinical trials of autologous ADSCs for osteoarthritis found that intra-articular injections of ADSCs improved joint function and pain relief in patients 44 . Exosomes of ADSCs in patients with osteoarthritis are rich in miR-145 and miR-221, which can downregulate pro-inflammatory factor expression in periosteum cells in vitro 45 .
The properties of ADSCs can be enhanced in several ways. For example, incubating them with glial cell-derived neurotrophic factor promotes their migration and differentiation 46 . Conditioning ADSCs with interferon-gamma (IFN-γ) improves their immunosuppressive potential in conditioned media and EVs 47 .
Umbilical cord–derived MSCs and exosomes
Umbilical cords might be an ideal substitute for ADSCs and BMMSCs, as they are discarded after birth and thus can be easily collected. The amplification rate is higher in vitro for umbilical cord–derived mesenchymal stromal cells (UC-MSCs) than BMMSCs 48 . More importantly, UC-MSCs have similar or superior immunomodulatory characteristics to MSCs derived from other sources48,49.
Clinical trials have revealed that UC-MSCs are significantly effective for treating SLE (Table 1). In fact, MSC therapy can reduce abnormal apoptotic cell accumulation in SLE through phagocytosis, resulting in the promotion of prostaglandin E2 (PGE2)-mediated immunosuppression 50 . Moreover, MSCs promote CD1c+ DC upregulation in peripheral blood and FLT3L in serum, inhibiting the inflammatory response to lupus 51 . Furthermore, UC-MSCs can prevent SLE by synthesizing transforming growth factor (TGF)-β1 that promotes the production of IL-10+ B regulatory cells (Bregs) and correct the Treg/Th17/Th1 imbalance in mice 52 .
Clinical Trial and Application of MSCs in Systemic Lupus Erythematosus.

ADSCs: adipose tissue–derived stem cells; BMMSCs: bone marrow–derived mesenchymal stromal cells; IL: interleukin; MSC: mesenchymal stromal cell; SLE: systemic lupus erythematosus; UC-MSC: umbilical cord–derived mesenchymal stromal cell.
Overactivated complement C5a and C5B-9 might be involved in the progression of LN, and UC-MSCs can improve LN in mice by secreting factor H to inhibit C5 activation 58 . Current investigations into human UC-MSCs (hUC-MSCs) mainly focus on the application of exosomes. For example, hUC-MSCs induce T-cell senescence and ameliorate lupus by regulating sirtuin 1 (SIRT1)/tumor protein P53 (p53) signaling in CD4+ T cells via miR-199a-5p transfer 59 . Human UC-MSCs can also release exosomes to induce the anti-inflammatory polarization of macrophages and improve SLE-associated diffuse alveolar hemorrhage in mice 60 . Exosomes released by UC-MSCs significantly inhibit the production of pro-inflammatory cytokines such as IFN-γ, IL-2, and TNF-α and increase the production of anti-inflammatory cytokines such as IL-10 61 .
Several clinical trials have confirmed the safety and effectiveness of UC-MSCs in patients with SLE. However, repeated injections of MSCs are necessary to avoid disease recurrence 62 .
Gingival MSCs and exosomes
A population of human gingival stromal cells (GMSCs) has self-renewal and multipotent differentiation capabilities 63 . Furthermore, GMSCs have better stem cell properties and immune characteristics than BMMSCs 64 , are not dependent on growth factor and serum supplements, are non-tumorigenic, and have phenotypic stability and high telomerase activity in long-term culture in vitro 65 . The phenotype and immunoregulatory functions of GMSCs isolated from patients with active SLE and healthy controls might be similar 65 . However, clinical trials of GMSCs for treating SLE have not substantially progressed.
Gingival MSCs exert preventive and therapeutic effects on lupus, and their exosomes promote M2 polarization of macrophages66,67. Gingival MSCs convert ADP or ATP to adenosine via elevated CD39 and CD73 68 expression. They also promote transformation from an ATP-driven pro-inflammatory to an adenosine-driven anti-inflammatory environment, thus regulating naive CD4+ T-cell differentiation 68 . Furthermore, GMSCs limit the development of proteinuria as well as autoantibodies, and decrease the frequency of plasma cells and severity of LN by directly inhibiting B-cell activation, proliferation, and differentiation 69 . Abnormal activation of the mTOR signaling pathway plays a central role in cell aging and GMSC-EVs can significantly inhibit mTOR/PS6 signaling; thus, GMSC-EVs might help to alleviate abnormal cell senescence in lupus tissues 70 .
Synovium-derived MSCs (SMSCs) and exosomes
Arthritis is a generally non-invasive, common clinical manifestation of SLE. Advances in imaging technology have clarified that chronic synovitis is more prevalent than was previously estimated 71 . Immune cells, especially T cells, play a critical role in the pathogenesis of lupus arthritis 72 . Synovium-derived MSCs have powerful immunomodulatory activities 73 , such as the inhibition of T-cell proliferation. Human synovium–derived mesenchymal stromal cell (hSMSCs) co-cultured with T cells inhibit their proliferation while having high proliferative capacity and limited senescence 74 . Intra-articular hSMSC injections restore the Th17/Treg and Th1/Th2 cell balance in animal models of rheumatoid arthritis 75 . Therefore, SMSCs might have similar therapeutic potential against SLE arthritis. However, the effects of SMSCs on NK, antigen-presenting, and B cells await exploration.
Exosomes of hSMSCs have abundant miR-129-5p, which significantly reduces cartilage cell inflammation and apoptosis. Human SMSCs injected into patients with osteoarthritis results in miR-129-5p in exosomes specifically targeting high mobility group box 1 (HMGB1) and attenuating the HMGB1-mediated inflammatory response 76 .
The pathogenesis of lupus also involves upregulated HMGB1 77 ; therefore, HMGB1 might be a potential target for treating SLE. Lupus is often treated using glucocorticoids, but prolonged exposure can lead to necrosis of the femoral head. Infused SMSCs prevent glucocorticoid-induced femoral head necrosis in rats by promoting bone tissue maintenance and regeneration via exosome secretion 78 . In addition, the extracellular matrix associated with SMSCs enhances anti-inflammatory properties and the proliferative potential of articular cartilage via the SIRT1 pathway 79 . Therefore, SMSCs are more suitable for treating joint lesions than other sources of MSCs.
Currently, MSCs derived from various sources have shown immunomodulatory effects in animal models, and allogeneic MSCs have exerted substantial clinical effects in SLE treatment. However, the mechanism through which MSCs alleviate SLE remains unclear. Mesenchymal stem cells have immune-regulatory, anti-inflammatory, and tissue repair functions. Specific MSCs might exert superior effects on individual systemic symptoms of SLE, like those of SMSCs in arthritis. Therefore, the feasibility and safety of sampling MSCs must be determined to obtain ethical approval before their clinical application can be promoted. Moreover, the effects and potential side effects of MSCs obtained from various sources should be compared. Like MSCs, exosomes derived from them might be a potential therapeutic approach.
Autologous MSC Transplantation in SLE
Application of autologous MSCs
Therapy is more suitable with autologous cells to prevent immune reactions to allogeneic cells in humans 80 . Although autologous MSCs to treat SLE does not elicit side effects and increased CD4+CD25+Foxp3+ Treg cells, disease activity is not affected, and later stages of the disease were not prevented 53 . Lupus MSCs are ineffective against SLE mice, and autologous MSCs are ineffective in treating human SLE 80 . Thus, autologous MSCs might not produce desirable effects in patients with SLA 80 .
MSC dysfunction in patients with SLE
Mesenchymal stem cells from patients with SLE have abnormal proliferation, immune regulation, and phenotypes 81 , as well as an altered intracellular constitution, especially pertaining to miRNA expression.
MicroRNA signaling in BMMSCs from patients with SLE is unique; abnormal miR-663 elevation might be associated with disease activity. Specifically, miR-663 targets TGF-β1 and inhibits BMMSC proliferation and migration, thus inhibiting any decrease in the proportion of Tfh cells or increase in the proportion of Treg cells normally mediated by BMMSCs 82 . Intracellular miR-153-3p overexpression reduces UC-MSC proliferation and migration, inhibits the UC-MSC-mediated decrease in the population of Tfh cells, and increases that of Tregs by inhibiting PELI1 83 . The onset of lupus is associated with a triggered Treg/Th17 imbalance in BMMSCs due to decreased Let-7f expression 84 . Furthermore, MSCs in patients with SLE have abnormal differentiation and senescence phenotypes, a senescent morphology, and increased proportions (%) of apoptotic cells. The proliferation potential of MSCs is limited in vitro, and gene expression associated with the senescence secretion phenotype is significantly increased 85 .
Gingival MSCs isolated from patients with active SLE have phenotypes and persistent immunomodulatory functions like those of healthy controls 65 . The application of autologous MSCs to SLE requires further investigation.
Conditioned MSCs
Because autologous cell therapy can prevent allogeneic reactions in humans 80 , effort has been directed to repair autologous MSCs using manipulations in vivo and ex vivo (Table 2).
Effects of Pre-Treated MSCs Combined With Other Cell Types and Drugs.
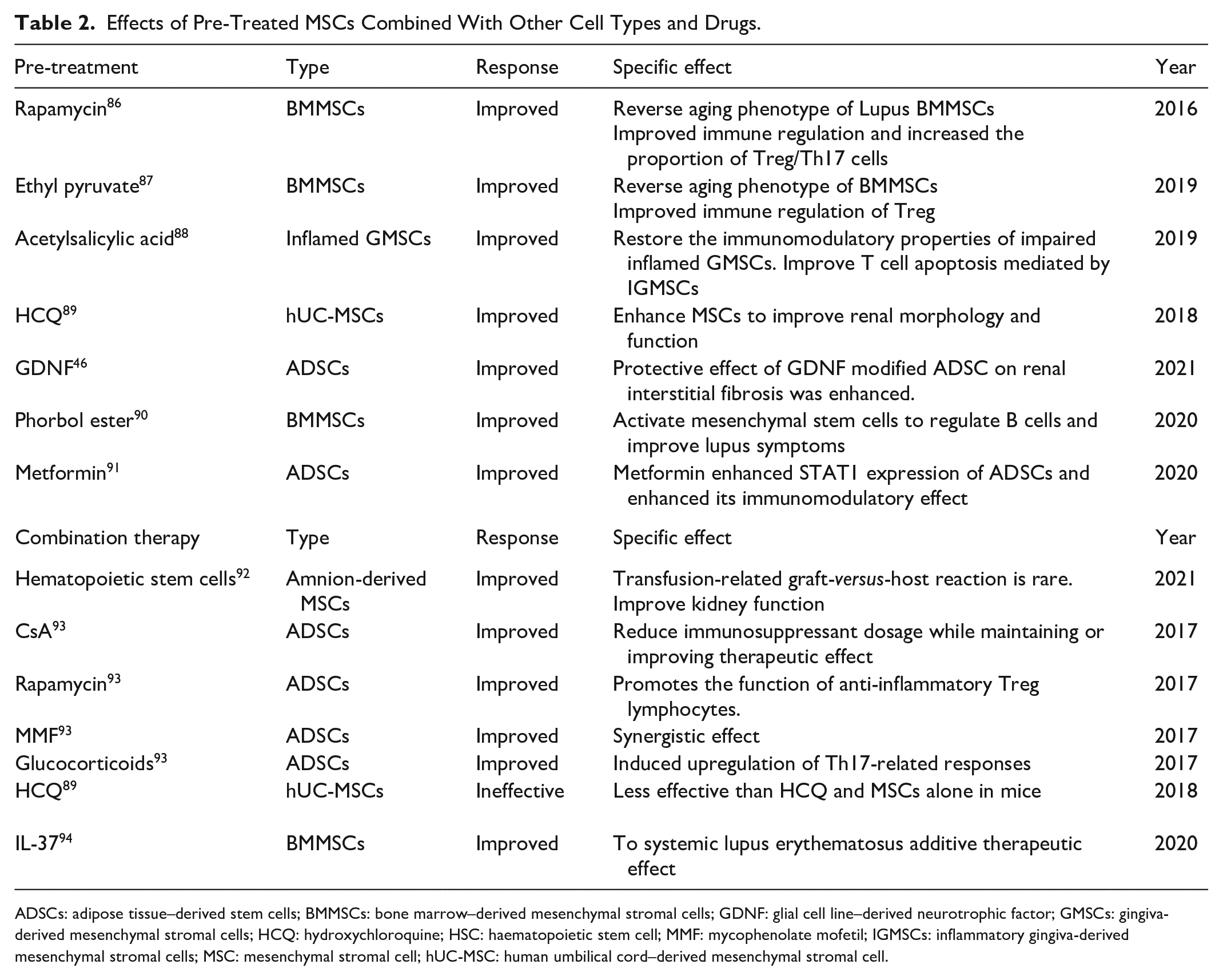
ADSCs: adipose tissue–derived stem cells; BMMSCs: bone marrow–derived mesenchymal stromal cells; GDNF: glial cell line–derived neurotrophic factor; GMSCs: gingiva-derived mesenchymal stromal cells; HCQ: hydroxychloroquine; HSC: haematopoietic stem cell; MMF: mycophenolate mofetil; IGMSCs: inflammatory gingiva-derived mesenchymal stromal cells; MSC: mesenchymal stromal cell; hUC-MSC: human umbilical cord–derived mesenchymal stromal cell.
Altering aging-related genes or signaling pathways can partially or completely reverse the aging phenotype and the defective immunoregulatory features of MSCs in patients with SLE 95 . Therefore, reversing MSC senescence might improve its therapeutic effects on SLE 81 .
The activated mTOR pathway is involved in MSC senescence in patients with SLE. Treating autologous MSCs derived from patients with SLE with the mTOR signaling inhibitor rapamycin (RAPA) ex vivo then infusing them might improve the immune-regulation ability of BMMSCs. Notably, RAPA increases the proportion of Treg/Th17 cells and regulates the secretion of related cytokines 86 . In addition, treating BMMSCs with ethyl pyruvate reverses BMMSC aging by blocking the HMGB1/TLR4/NF-κB signaling pathway, thus improving Treg-cell-related immune regulation 87 . Let-7f-5p can somewhat reduce the inflammatory response of SLE-BMMSCs by targeting NOD-like receptor protein 3 (NLRP3) 96 . Other MSC pre-conditioning methods might be applied to restore the functions of autoimmune MSCs. Such modulators might increase the possibility of applying autoimmune MSCs to autoimmune diseases. For example, incubating inflammatory GMSCs (IGMSCs) that have impaired immune-regulation ability with acetylsalicylic acid upregulates Fas ligand expression, thus enhancing the apoptosis of T cells mediated by IGMSC 88 .
Development of Combination Therapy
Among the drugs that are currently prescribed to treat SLE, immunosuppressants are often limited due to the risk of side effects. A combination of MSCs and immunosuppressants has shown promise for patients with refractory SLE. Combination therapies decrease the amount of immunosuppressants required to reduce immune activity in SLE 93 .
Mesenchymal stromal cells exert synergistic effects on immunosuppressants such as the calcineurin inhibitor CsA, the mTOR inhibitor RAPA, mycophenolate mofetil (MMF), dexamethasone (Dex), and prednisone. The combination of MSCs and immunosuppressants significantly alters the activation and balance of T-lymphocyte subsets and reverses the adverse effects of immunosuppressants such as the prednisone-, Dex-, or MMF-induced increase in T-cell differentiation into Th17 cells. However, MSC activity and function have not been significantly inhibited 93 . The results of combinations of immunosuppressants on MSCs differ based on the included drugs. For example, a combination of tacrolimus and RAPA antagonizes, whereas Dex does not alter the immunosuppressive effects of transplanted MSCs 93 .
In conclusion, although combining drugs with MSC transplantation is a promising new therapeutic strategy, further studies are warranted to ensure their effectiveness and safety (Table 2).
Prospects and Challenges of Cell-Free Therapy
Although live MSCs have gradually been applied in clinical practice as therapy, dangers include degrees of immune rejection, low cell survival rates, senescence, and carcinogenic potential 97 . Adequate funding and technology are also required to prepare and preserve live cells, which limits the widespread application of MSCs as therapy in hospital environments. However, the therapeutic effects of MSC-derived EVs, especially exosomes, are similar to those of MSCs. Being cell-free, exosomes have more advantages than MSCs, are easier to preserve and manage, have no immunogenicity, and can easily cross the blood–brain barrier 98 , which is considered a promising new treatment option. However, many aspects still require optimization before large quantities of such exosomes can serve as human therapies 17 .
The first issue is the high cost and low efficiency of current methods of extracting exosomes from cells, and related processes have not been standardized 99 . Differential ultracentrifugation can generate the purest exosomes, but it is time-consuming 100 . Artificial exosomes might guarantee production and facilitate widespread commercialization. Attempts are ongoing to increase EV production by modifying culture conditions and conducting specific interventions for MSCs 101 . Another issue is the efficiency of exosome homing to specific lesion sites. Extracellular vesicles such as exosomes can be homed to a portion of injured tissue dependent on the expression of CD44, CD29, or CD73 102 . However, most intravenously injected exosomes accumulate at sites where the mononuclear phagocyte system (MPS) is active 103 . Artificial modification of exosome membranes to increase the proportions of them that reach target cells is essential to improve bioavailability 17 . Because the pathogenesis of lupus is different caused by different factors, conventional drugs are only effective for a certain type of patients. Most current studies of potential therapeutic mechanisms of miRNAs in MSC exosomes target a specific miRNA, which provides great support for the development of targeted therapy. However, whether transfecting several miRNAs together into exosomes or inhibiting them will increase their effectiveness or cause side effects is unknown. A single miRNA can target multiple mRNAs, so exosomes carrying specific miRNAs delivered intravenously might elicit side effects 104 . Further studies are needed to decode the complexity of exosome components to better understand the safety of their combined effects on target tissues. Bioengineering efforts are also needed to achieve the amplification and safe production of compositionally homogeneous exosomes containing specific miRNAs or inhibitors 105 of miRNAs 106 . The optimal dose and frequency of exosome administration should also be determined to maximize efficiency 107 .
Conclusions
The anti-inflammatory repair potential, immunomodulatory, and specific immunological properties of MSCs hold promise for using allogeneic MSCs to treat SLE with abnormal autologous MSC function. Investigations into the effects of chemical and other manipulations to improve MSCs before delivery and the application of exosomes as drug delivery vectors in treating SLE are currently underway. Roles for exosomes and their miRNAs in the diagnosis and treatment of SLE have been demonstrated. Tissue engineering, biomaterials, and stem cell biology offer a brighter future for patients with SLE through the large-scale preparation of specific exosomes/miRNAs.
Footnotes
Acknowledgements
We are grateful to the Second Affiliated Hospital of Fujian Medical University for providing infrastructure facilities.
Author Contributions
YJL, ZC, and SL: concept and design of manuscript. YJL drafted and finalized the manuscript. ZC and SL drafted the manuscript. ZC, HBM, and SL revised the manuscript and provided critical advice about the content. All authors contributed to the article and approved the submitted version.
Availability of Data and Materials
Not applicable
Ethical Approval
Not applicable.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Bureau of Quanzhou (grant 2020CT003), the Natural Science Foundation of Fujian Province (grant 2020J01219), and the Science and Technology Bureau of Quanzhou (grant 2017Z009).
