Abstract
The global population of individuals afflicted with diabetes mellitus has been increasing year by year, and this disease poses a serious threat to human health as well as the economies worldwide. Pancreatic or islet transplantations provide one of the most effective and long-term therapies available to treat diabetes, but the scarcity and quality of pancreatic islets limit their use in treatments. Here, we report the development of a one-step, monolayer culture, and chemical-based protocol that efficiently mediates the differentiation of human adipose-derived stem cells (hADSCs) into insulin-producing cells (IPCs). Our data indicate that hADSCs in monolayer culture that are allowed to differentiate into IPCs are superior to those in suspension cultures with respect to insulin secretion capacity (213-fold increase), cell viability (93.5 ± 3.27% vs. 41.67 ± 13.17%), and response to glucose stimulation. Moreover, the expression of genes associated with pancreatic lineage specification, such as PDX1, ISL1, and INS (encoding insulin), were expressed at significantly higher levels during our differentiation protocol (6-fold for PDX1 and ISL1, 11.5-fold for INS). Importantly, in vivo studies demonstrated that transplantation with IPCs significantly mitigated hyperglycemia in streptozotocin-induced diabetic rats. Our results indicate that this one-step, rapid protocol increases the efficiency of IPC generation and that the chemical-based approach for IPC induction may reduce safety concerns associated with the use of IPCs for clinical applications, thereby providing a safe and effective cell-based treatment for diabetes.
Keywords
Introduction
Diabetes mellitus (DM) is a disorder associated with metabolic syndrome that is characterized by hyperglycemia caused by insufficient insulin secretion or insulin resistance1,2. According to an International Diabetes Federation report published in 2021 3 , 536.6 million individuals worldwide are afflicted with diabetes. DM is also associated with secondary complications such as cardiovascular disease, stroke, retinopathy, nephropathy, and neuropathy4,5. Moreover, by the year 2030, DM is projected become the seventh leading cause of death worldwide 6 .
Type 1 DM is caused by autoimmune attack on β cells with a consequent reduction of insulin secretion 7 . In type 2 DM, insulin resistance results in increased insulin demand that ultimately leads to β-cell exhaustion and depletion 8 . All type 1 DM patients and many type 2 DM patients require exogenous insulin delivery to maintain blood glucose homeostasis and avoid DM-associated complications, but the challenges associated with managing insulin dosing may lead to poor overall control of hyperglycemia. Pancreatic or islet transplantation is one of the most effective and long-term therapies available to patients with insulin-dependent DM9,10, but the scarcity of pancreatic islets as well as their quality limit their use in treatments. Therefore, alternative strategies are needed to identify novel sources for expanding β cells.
Recent advances in regenerative medicine and cell-based therapy approaches have provided options for DM treatment. Human embryonic stem cells11,12, induced pluripotent stem cells13,14, and mesenchymal stem cells (MSCs)15,16 have been reported to have the potential to differentiate into β-like insulin-producing cells (IPCs) for treatment of diabetes in animal models. However, issues have arisen such as ethical aspects of the use of human embryonic stem cells and the risk of teratoma formation after transplantation of those cells or induced pluripotent stem cells17,18. Therefore, investigators have explored alternative strategies for producing IPCs via differentiation using other cell sources, such as MSCs. MSCs are widely used in regenerative medicine because of their capacity for multi-differentiation, immunomodulation, and tissue repair19–21. Among MSCs used in regenerative medicine, human adipose-derived stem cells (hADSCs) have become an important component of treatment owing to their accessibility, abundance, and minimally invasive collection procedure compared with other sources22,23. Moreover, many studies have suggested that ADSCs should be considered for replacement, repair, and regeneration of damaged cells for many clinical indications including DM for both autologous and allogeneic stem-cell therapy22–24.
Activin A is a member of the transforming growth factor β superfamily that can facilitate the differentiation of endocrine and exocrine cells in the pancreas and promote the formation of pancreatic islets 25 . Another potential inducer of pancreatic islet formation is exendin-4, a long-acting agonist of the GLP-1 (glucagon-like peptide 1) receptor, which is involved in both the replication and function of β-cell26,27. One study reported that use of a low-attachment suspension culture method could generate IPC spheroids in the presence of activin A and exendin-4 28 . Additionally, Wartchow et al. compared three different formulations of differentiation-related factors and found that IPCs generated by Timper’s protocol had the best insulin secretion profile and could ameliorate hyperglycemia in streptozotocin (STZ)-induced diabetic rats 29 , indicating that IPCs generated via the activin A/exendin-4 formulation have the potential to treat diabetes. However, spheroids that generate spontaneously during culture on low-attachment plates have some disadvantages such as variability in spheroid size and a lack of nutrients and oxygen in the spheroid core, resulting in a skewed distribution of proliferating, non-proliferating, and apoptotic cells that can lead to cell death during culture or after transplantation30,31.
To improve the efficiency of generating IPCs via differentiation from hADSCs, we investigated the efficacy of using an activin A/exendin-4 formulation to induce differentiation under different culture conditions. For this purpose, we used clear tissue culture–treated plates for monolayer culture and ultra-low attachment plates for suspension culture. Cell morphology was assessed via phase-contrast microscopy, and cell viability was assessed with a cell counter. Furthermore, we analyzed human insulin concentration in samples of culture medium that secreted from IPCs with a chemiluminescence immunoassay and performed a glucose challenge assay to test the sensitivity of IPCs to glucose. We also used quantitative reverse transcription-PCR (qRT-PCR) to determine the expression of genes associated with pancreatic lineage specification in IPCs. Finally, experiments were carried out to evaluate the efficacy of IPCs for the treatment of diabetes. Our results demonstrate that the use of a one-step, monolayer culture and chemical-based protocol can efficiently produce IPCs that may have applications in the treatment of DM.
Materials and Methods
Cell Preparation
The hADSCs were obtained from a healthy donor, and the study was approved by the Research Ethics Committee of Hualien Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation (IRB No: IRB106-88-A). hADSCs were isolated and cultured on plates according to our published methods32,33. In brief, adipose tissue (2–5 g) was harvested from the subcutaneous fat of the abdominal wall by lipoaspiration during abdominal surgery from healthy donors. The adipose tissue was placed in Ca2+/Mg2+-free phosphate-buffered saline (PBS) and immediately transferred to the laboratory on ice, removed from the transport medium, placed in a Petri dish, and cut into small pieces (1–2 mm3) in the presence of Ca2+/Mg2+-free PBS. The tissue pieces were dissociated with 0.1% collagenase I (Gibco, Carlsbad, CA, USA) and incubated for 60 min at 37°C. The resulting cells were cultured for 2 days in serum-free medium (K-SFM; Gibco, Grand Island, NY, USA) supplemented with 10% fetal bovine serum (HyClone, Logan, UT, USA), N-acetyl-
Flow Cytometry
hADSCs or IPCs were harvested and suspended in PBS. Then, cells were incubated with antibodies conjugated with phycoerythrin (PE) or fluorescein isothiocyanate (FITC) in the dark at 4°C for 30 min, washed with PBS to clear unbound antibody, and analyzed with a flow cytometer (FC500; Beckman Coulter, Brea, CA, USA). Antibodies against CD14-PE, CD19-PE, CD34-PE, CD73-PE, CD105-PE, HLA-DR-PE (human leukocyte antigen D receptor), CD90-FITC, and CD45-FITC were purchased from BD (Franklin Lakes, NJ, USA), and the isotype controls for IgG1-PE, IgG1-FITC, and IgG2a-PE were purchased from BD.
Multipotent Differentiation Assays for hADSCs
The differentiation of hADSCs into adipocytes, osteocytes, or chondrocytes was carried out according to our published methods
34
. For differentiation into adipocytes, hADSCs were seeded in 6-well clear tissue culture–treated plates (Corning, Corning, NY, USA) at 1 × 105 cells per well and grown to 70% confluency in culture medium overnight. Then, the medium was replaced with DMEM (Gibco) containing 0.5 mM 3-isobutyl-1-methylxanthine, 1 µM dexamethasone, 10 µg/ml insulin, and 100 µM indomethacin (all from Sigma-Aldrich). The medium was replaced every 3 days, and cells were maintained in culture for 14 days. Differentiation into adipocytes was detected by staining with Oil red O (Lifeline Cell Technology, Frederick, MD, USA). For differentiation into osteocytes, hADSCs were seeded, cultured, and induced as described above. The differentiation medium was DMEM containing 10 mM β-glycerophosphate (Sigma-Aldrich), 0.1 µM dexamethasone, 0.2 mM ascorbic acid (Sigma-Aldrich), and 2 mM
Differentiation of hADSCs Into IPCs in Monolayer and Suspension Cultures
hADSCs were seeded at a density of 2.4 × 105 cells per well in 6-well clear tissue culture–treated plates for monolayer culture and in 6-well ultra-low attachment plates (Corning) for suspension culture. hADSCs were cultured in medium overnight. The next day, the medium was removed and the cells were washed twice with serum-free DMEM/F12 medium (Gibco). Differentiation was then carried out for 7 days in fresh serum-free DMEM/F12 medium containing 17.5 mM glucose, 10 mM nicotinamide, 2 pM activin-A, 10 nM exendin-4, 100 fM hepatocyte growth factor, 10 nM pentagastrin (all from Sigma-Aldrich), 2% B-27 serum-free supplement (Gibco), and 1% N-2 supplement (Gibco). The medium was not replaced during the differentiation period.
Measurement of Insulin Concentration in Culture Medium of Human Cells
Culture medium for hADSCs or IPCs was collected, and human insulin concentration was measured with a chemiluminescence immunoassay using an ADVIA Centaur XPT system (Siemens Medical Solutions USA, Inc., Malvern, PA, USA).
Glucose Challenge Assay
The glucose challenge assay was performed as described previously 35 . Briefly, hADSCs were seeded at a density of 2.4 × 105 cells per well in 6-well clear tissue culture–treated plates or 6-well ultra-low attachment plates, and cells were divided into low-glucose and high-glucose groups. Nondifferentiated hADSCs were used as a control group. After 7 days of differentiation as described above, the differentiation medium was removed and cells were washed twice with PBS. Then, the low-glucose and high-glucose groups were established by adding DMEM containing 5.5 mM glucose (Gibco) or DMEM containing 25 mM DMEM (Gibco), with subsequent incubation for 24 h. Culture medium was collected, and human insulin concentration was measured with a chemiluminescence immunoassay.
qRT-PCR
To quantify gene expression, cells were harvested after 7 days of differentiation. Total RNA was isolated using the Quick-RNA™ MiniPrep kit (Zymo Research, Irvine, CA, USA), and cDNA was generated using the PrimeScript™ RT Reagent kit (Takara, Tokyo, Japan). qRT-PCR was carried out with the SYBR® Premix Ex Taq™ II kit (Takara) using a LightCycler® 480 Real-Time PCR System (Roche, Mannheim, Germany). Samples were normalized using the housekeeping gene HPRT 28 , and data were analyzed using the delta–delta Ct method. The sequences of PDX1 (pancreatic and duodenal homeobox 1) primers were as follows: sense, 5’-TGATACTGGATTGGCGTTGTTT-3’; antisense, 5’-TCCCAAGGTGGAGTGCTGTAG-3.’ The sequences of ISL1 (insulin gene enhancer protein 1) primers were as follows: sense, 5’-CAACTGGTCAATTTTTCAGAAGGA-3’; antisense, 5’-TTGAGAGGACATTGATGCTACTTCAC-3.’ The sequences of INS primers were as follows: sense, 5’-GCAGCCTTTGTGAACCAACA-3’; antisense, 5’-TTCCCCGCACACTAGGTAGAGA-3.’ The sequences of the CD47 primers were as follows: sense, 5’- GGCAATGACGAAGGAGGTTA-3’; antisense, 5’-ATCCGGTGGTATGGATGAGA-3’. The sequences of the HPRT (hypoxanthine phosphoribosyltransferase 1) primers were as follows: sense, 5’-TCAGGCAGTATAATCCAAAGATGGT-3’; antisense, 5’-AGTCTGGCTTATATCCAACACTTCG-3.’
Establishment of STZ-Induced Diabetes in Rats and Cell Transplantation
The animal study was approved by the National Taiwan University College of Medicine and College of Public Health Institutional Animal Care and Use Committee (No: 20200050). Male Wistar rats (4–5 weeks old) were purchased from BioLASCO (Taipei, Taiwan) and maintained under controlled light and environmental conditions (12 h light/12 h dark cycle at a temperature range of 22°C–24°C) with free access to commercial chow and water. Diabetes in rats (weight, 200–250 g) was induced experimentally by intraperitoneal injection of STZ (Sigma-Aldrich; 50 mg/kg in citrate buffer, pH 4.5) on three consecutive days. Blood glucose level in samples of tail vein blood was measured using an ACCU-CHEK glucose meter (Roche Diagnostics, Indianapolis, IN, USA). Rats with a blood glucose level greater than 200 mg/dl were considered to have diabetes. Two weeks after STZ injection, blood glucose level was stable and reflected hyperglycemia (~600 mg/dl), and rats were randomly divided into three groups: STZ + normal saline group: six diabetic rats received normal saline (0.9%) at 200 µl per rat by intravenous injection; STZ + hADSCs group: six diabetic rats received 3 × 106 hADSCs per 200 µl normal saline (0.9%) per rat by intravenous injection; STZ + IPCs group: six diabetic rats received 3 × 106 IPCs per 200 µl normal saline (0.9%) per rat by intravenous injection. The control group comprised six untreated STZ Wistar rats that also received 200 µl normal saline (0.9%) by intravenous injection. Blood glucose was measured on days 0, 1, 3, 5, 7, and 14 after cell transplantation.
Statistical Analysis
All data represent the mean ± standard deviation. For the insulin secretion, cell viability, and qRT-PCR experiments as well as animal studies, statistical significance among different experimental groups was determined by the Student’s unpaired t-test using Microsoft Office Excel 2007. The statistical significance of differences between values in various experiments was set at P < 0.05, P < 0.01, or P < 0.001.
Results
Morphological and Cytological Characterization of hADSCs
hADSCs were isolated and cultured according to our published methods32,33. hADSCs were spindle shaped and appeared to be a homogeneous population as assessed with phase-contrast microscopy (Figs. 1A and 2A). Immunophenotyping by flow cytometry revealed that the hADSCs were positive (≥95%) for the surface markers CD73, CD90, and CD105, but essentially negative (≤2%) for CD14, CD19, CD34, CD45, and HLA-DR (Fig. 1B). Moreover, hADSCs could differentiate into adipogenic, osteogenic, and chondrogenic lineages (Fig. 1C). These results indicated that the hADSCs complied with MSC criteria of the International Society for Cellular Therapy 36 .

Morphological and cytological characterization of hADSCs. (A) Morphology of hADSCs as observed with phase-contrast microscopy. (B) hADSCs were positive for CD73, CD90, and CD105 and negative for CD14, CD19, CD34, CD45 and HLA-DR. The black-line peak indicates the isotype control. (C) Multipotent differentiation assays indicate the ability of hADSCs to differentiate into three lineages, namely adipocytes (Oil red O staining), osteocytes (alkaline phosphatase staining), and chondrocytes (alcian blue staining). Magnification: 100×. Scale bar: 100 µm. FITC: fluorescein isothiocyanate; hADSCs: human adipose-derived stem cells; HLA-DR: human leukocyte antigen D receptor; PE: phycoerythrin.

Morphological changes of hADSCs after differentiation into IPCs during culture in a monolayer or in suspension. (A) The morphology of hADSCs was observed and photographed with a phase-contrast microscope. (B, C) IPCs in monolayer culture had a spindle shape (B) whereas those in suspension culture formed spheroids (C). Magnification: 40×. Scale bar: 200 µm. hADSCs: human adipose-derived stem cells; IPCs: insulin-producing cells.
hADSCs Undergo a Change in Morphology After 7 Days of Differentiation in Monolayer or Suspension Culture
To assess the ability of hADSCs to undergo differentiation in culture over a period of 7 days, the cells were subjected to monolayer culture on clear tissue culture–treated plates or suspension culture using ultra-low attachment plates. After 7 days, the hADSCs in monolayer culture had undergone differentiation to yield a homogeneous population of spindle-shaped cells (Fig. 2B), whereas hADSCs in suspension culture were heterogeneous and formed spheroids of varying size (Fig. 2C). These data demonstrated that the morphology of differentiated cells differed depending on whether hADSCs underwent differentiation in monolayer or suspension culture.
hADSCs Differentiate into IPCs in Monolayer Culture Have Superior Insulin Secretion Capacity and High Cell Viability
An insulin chemiluminescence immunoassay revealed that the IPCs derived from suspension culture of hADSCs secreted only 239.6 ± 26.85 mIU/l insulin, whereas IPCs derived from monolayer culture secreted a 213-fold greater amount, that is, 51,223.47 ± 2808.03 mIU/l (Fig. 3A). In addition, the viability of the differentiated IPCs was much greater in the monolayer culture (93.5 ± 3.27%) than in the suspension culture (41.67 ± 13.17%). Taken together, these data showed that hADSCs that underwent differentiation into IPCs in monolayer culture secreted much more insulin and had a greater survival rate than those in suspension culture.

Insulin secretion and viability of IPCs in monolayer and suspension cultures. (A) After 7 days of culture in differentiation medium, human insulin concentration in the medium was measured with a chemiluminescence immunoassay (P < 0.001, Student’s unpaired t-test, n = 6). Differentiation medium alone served as the negative control. (B) Cell viability was measured with an ADAM-MC automatic cell counter (P < 0.01, Student’s unpaired t-test, n = 6). Data represent the mean ± SD. IPCs: insulin-producing cells; SD: standard deviation.
Secretion of Insulin by IPCs in Response to Glucose Stimulation
To further evaluate the insulin secretion in response to glucose stimulation of IPCs in monolayer and suspension culture, a glucose challenge assay was performed. The monolayer-derived IPCs secreted significantly more insulin than the nondifferentiated hADSCs or suspension-derived IPCs in both low-glucose medium (3.79 ± 1.14 µIU per 105 cells vs. 0.32 ± 0.02 µIU and 0.90 ± 0.04 µIU, respectively) and high-glucose medium (8.20 ± 1.44 µIU per 105 cells vs. 0.49 ± 0.03 µIU and 0.83 ± 0.04 µIU) (Fig. 4). Notably, culture in high-glucose medium caused the monolayer-derived IPCs to secrete a significantly greater amount of insulin relative to the low-glucose medium, that is, by 2.2-fold, but this was not the case for the suspension-derived IPCs. Taken together, these results indicated that IPCs differentiated in monolayer culture had a significantly greater functional response to glucose stimulation compared with IPCs differentiated in suspension culture. Therefore, subsequent experiments were carried out exclusively with monolayer-derived IPCs.

Response of IPCs to low- and high-glucose challenges. Insulin was secreted into medium in response to a low-glucose (5.5 mM) or high-glucose (25 mM) stimulation. Incubation was carried out for 24 h, with subsequent measurement of insulin secretion via a chemiluminescence immunoassay. IPCs differentiated in monolayer culture secreted a significantly greater amount of insulin than did the nondifferentiated hADSCs or hADSCs that were subjected to differentiation in suspension culture in both the low-glucose medium (P < 0.01, Student’s unpaired t-test, n = 3) and high-glucose medium (P < 0.001, Student’s unpaired t-test, n = 3). Data represent the mean ± SD. hADSCs: human adipose-derived stem cells; IPCs: insulin-producing cells; SD: standard deviation.
Expression of Pancreatic Lineage–Related Genes in IPCs
To unequivocally demonstrate that hADSCs cultured in a monolayer gave rise to differentiated IPCs, the expression of genes associated with pancreatic lineage specification was measured by qRT-PCR. The data revealed a significant upregulation of mRNAs transcribed from PDX1, ISL1 and INS (6-fold for PDX1 and ISL1, 11.5-fold for INS) on day 7 post-differentiation compared with nondifferentiated hADSCs (Fig. 5A–C), indicating that hADSCs in monolayer culture have the potential to differentiate into IPCs.
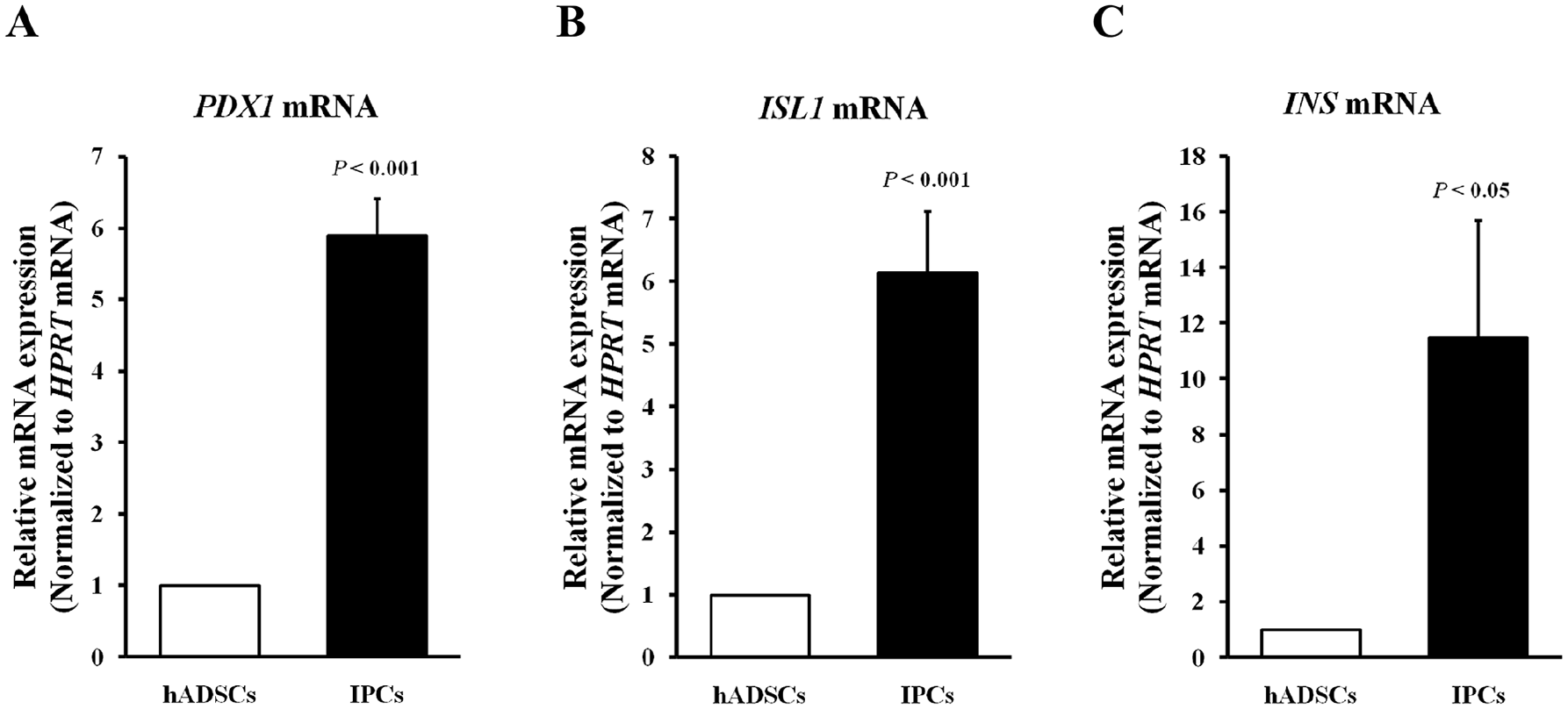
Quantitative reverse transcription-PCR analysis of pancreatic lineage-related genes in hADSCs and IPCs. Expression was measured for (A) PDX1 (P < 0.001, Student’s unpaired t-test, n = 3), (B) ISL1 (P < 0.001, Student’s unpaired t-test, n = 3), and (C) INS (P < 0.05, Student’s unpaired t-test, n = 3). All data were normalized to HPRT expression, and the fold change of each mRNA level was relative to the corresponding genes in hADSCs. Data represent the mean ± SD. hADSCs: human adipose-derived stem cells; IPCs: insulin-producing cells; mRNA: messenger ribonucleic acid; SD: standard deviation.
Transplantation of IPCs into Diabetic Rats Mitigates Their Hyperglycemia
To assess the ability of IPCs to mitigate hyperglycemia in vivo, IPCs were transplanted into STZ-induced diabetic rats (3 × 106 IPCs per rat) by intravenous injection, and blood glucose was measured on days 0, 1, 3, 5, 7, and 14 (Fig. 6A). Compared with the diabetic rats that received normal saline rather than IPCs, the diabetic rats transplanted with IPCs had significantly reduced hyperglycemia within 1 week, and the reduction was most notable (~17%) on day 3 after transplantation (589.33 ± 21.20 mg/dl vs. 486.83 ± 49.03 mg/dl; Fig. 6B). By day 14 post-transplantation, however, blood glucose returned to a level similar to that of the STZ + normal saline group. Moreover, for the diabetic rats that received the undifferentiated hADSCs (STZ + hADSCs group), hyperglycemia was only significantly reduced on day 1 post-transplantation (563.83 ± 35.05 mg/dl vs. 507.50 ± 34.03 mg/dl). Additionally, none of the diabetic rats died or experienced any serious adverse events as a result of transplantation with IPCs and hADSCs from cell transplantation to sacrifice, indicating that intravenous injection of 3 × 106cells per rat was safe. These results demonstrated that transplantation with IPCs via intravenous injection could lessen the severity of hyperglycemia by up to 17% and that this avenue of administration was safe.
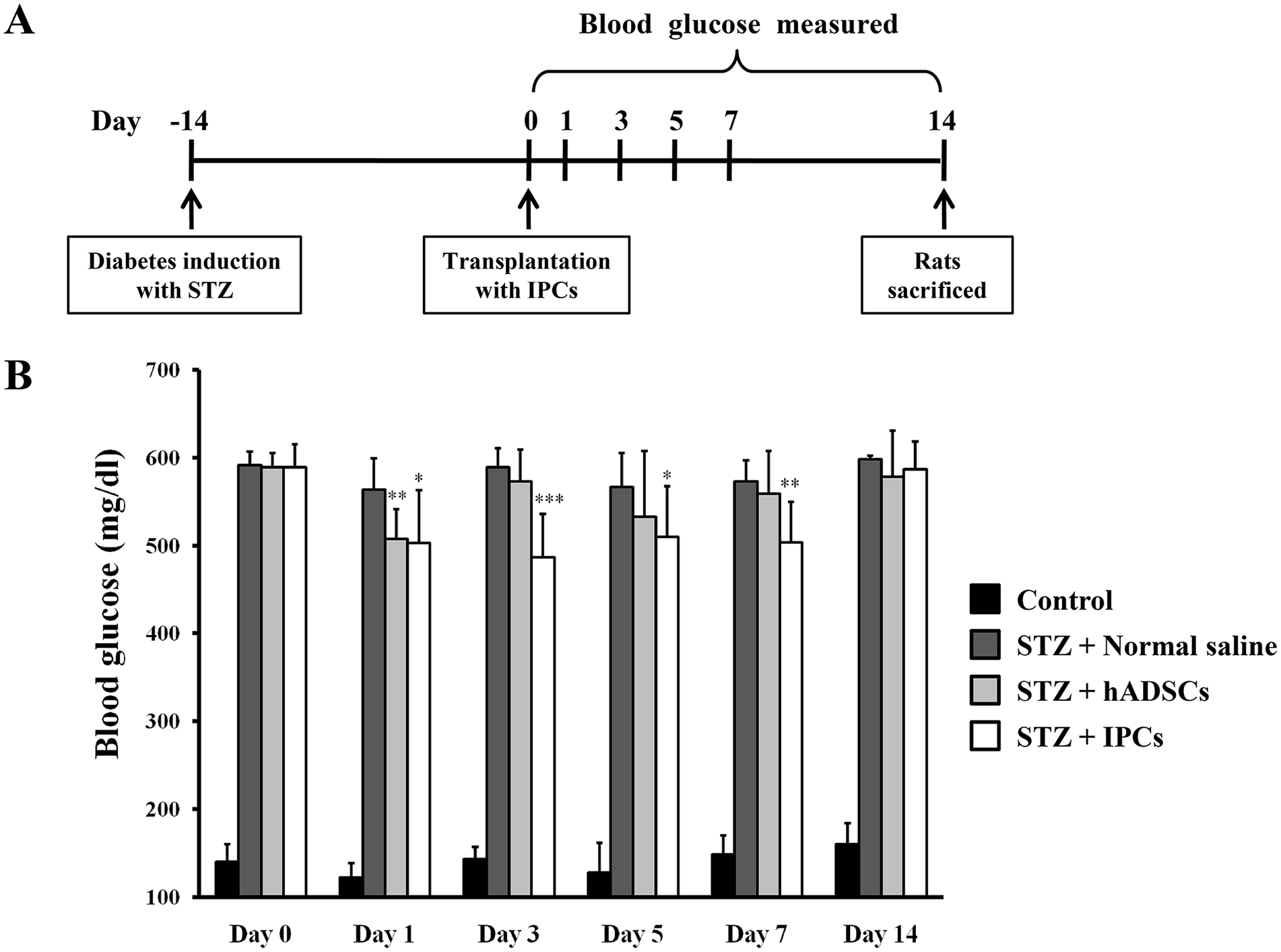
IPCs reduce the severity of hyperglycemia in STZ-induced diabetic rats. hADSCs underwent differentiation in monolayer culture to yield IPCs, which were transplanted into STZ-induced diabetic rats. (A) Timeline for transplantation of hADSCs or IPCs into the diabetic rats by intravenous injection. (B) Blood glucose was measured on days 0, 1, 3, 5, 7 and 14 (*P < 0.05, ** P < 0.01, *** P < 0.001, Student’s unpaired t-test, n = 6). Data represent the mean ± SD. hADSCs: human adipose-derived stem cells; IPCs: insulin-producing cells; SD: standard deviation; STZ: streptozotocin.
Discussion
It has been reported that three-dimensional suspension culture can more accurately mimic in vivo conditions than traditional two-dimensional monolayer culture37,38. However, our present showed that, compared with suspension culture, hADSCs in monolayer culture could differentiate into a homogenous population of IPCs (Fig. 2B) and had greater insulin secretion capacity (Fig. 3A), cell viability (Fig. 3B), and response to glucose stimulation (Fig. 4). Recently, Hogrebe et al. 11 developed a two-dimensional differentiation protocol for generating human pluripotent stem cell–derived β cells that could more effectively treat diabetes in mice compared to treatment with cells differentiated in suspension culture. In addition, Mandal et al. 39 used a monolayer culture system with a new chemical-based differentiation approach to generate IPCs and found that these differentiated cells from ADSCs exhibited higher PDX1 expression compared with cells differentiated in suspension culture. Although these studies did not discuss the advantages of monolayer culture over suspension culture, we speculate that cells in monolayer culture are relatively more equally distributed and thus have greater exposure to oxygen, nutrients, and differentiation factors in the medium, thus providing a better microenvironment for attached cells to differentiate and survive 31 .
The transcription factors PDX1 and ISL1 play essential roles in pancreatic organogenesis, development of the endocrine pancreas, and the survival, proliferation, and function of β cells40–42. Moreover, PDX1 and ISL1 coordinate with epigenetic modifications to regulate INS expression in response to changes in blood glucose concentration 43 . Genetics-based studies have shown that vector-encoded PDX1 can transduce stem cells to differentiate into IPCs44,45. However, genetics-based approaches require viral vectors for transduction and may raise safety concerns for clinical application 46 . Therefore, stepwise chemical-based methods have been proposed as a solution, and many studies have demonstrated that this approach can differentiate ADSCs into IPCs16,39,47. Our data indicate that using a one-step monolayer culture and chemical-based protocol can increase the expression of PDX1 and ISL1 by ~6-fold and INS by ~11.5-fold (Fig. 5) in only 7 days of induction, suggesting that our relatively rapid protocol could increase the efficiency of differentiation of hADSCs to yield IPCs with reduced cost and decreased probability of contamination within the IPC generation, thus providing more opportunities for cost-effective clinical use of IPCs.
Our animal study used non-immune-deficient rats and did not require the administration of immunosuppressive agents after cell transplantation. We hypothesized that this experimental design more closely mimics the in vivo condition of cell-based therapy and enables direct evaluation of the therapeutic effect of transplanted cells. As expected, the differentiated IPCs could modestly, yet significantly, reduce the severity of hyperglycemia in diabetic rats for 1 week, and the IPCs could reduce hyperglycemia better than hADSCs (Fig. 6B). Our supplementary data showed that the differentiated IPCs retained MSC surface markers (Fig. S1A) and had elevated expression of CD47 mRNA (Fig. S1B). MSCs have low immunogenicity because they do not express HLA-DR48,49; similarly, the IPCs produced in the present study also lacked HLA-DR (Fig. S1A), allowing speculation that IPCs had low immunogenicity. In addition, CD47 is an immunoglobulin-like protein that can interact with the receptor SIRPα on macrophages to negatively regulate phagocytosis50,51. Leung et al. 52 indicated that transgenic expression of murine CD47 on the cell surface of human embryonic stem cells could attenuate macrophage-mediated phagocytosis and improve cell survival after transplantation. Another study also showed that expression of mouse CD47 in rat insulinoma cells markedly reduced the susceptibility of these cells to phagocytosis and restored blood glucose in diabetic mice 53 . Thus, CD47 functions as a “do not eat me” signal to ensure that transplanted cells are not inappropriately phagocytosed by macrophages. Therefore, we speculate that IPCs are more effective regulators of blood glucose than are ADSCs. Moreover, in addition to their ability to secrete insulin, IPCs have low immunogenicity and thus may escape host immune-cell attack and thus prolong the therapeutic effect.
In addition to the potential for immune attack on therapeutic cells (thus reducing therapeutic efficacy), certain other factors such as mechanical stress, hypoxia, and nutrient deprivation during transplantation also need to be considered54,55. To test whether hADSCs and IPCs were subjected to mechanical stress during transplantation, we performed an experiment revealing that their viability was more than 95% (data not shown) after passage through 25G and 26G needles, indicated that the cells used in our experiments were essentially not affected by the route (i.e., needle) of transplantation. In addition, Zheng et al. 56 observed that hypoxia could induce the apoptosis of pancreatic β-cells via the unfolded protein response and CHOP/caspase-3 signal pathway. To avoid post-transplantation cell death due to hypoxia and/or nutritional deprivation, we selected the intravenous route of injection, which ensured maximal therapeutic efficacy. Taken together, our findings suggest that IPCs generated from our protocol may have applications in the treatment of patients with DM.
Long-term safety monitoring is one of the important issues of cell therapy. Although observation period after cell therapy is only 2 weeks, none of the diabetic rats died or experienced any serious adverse events as a result of transplantation with IPCs in our study. In addition, the results of a 2-year follow-up clinical trial indicated that using exendin-4/activin A formula differentiated IPCs combined with bone marrow-derived hematopoietic stem cells for the treatment of Type 1 DM had no untoward effect was observed, sustained improvement in HbA1c and serum C-peptide, decreased glutamic acid decarboxylase antibodies and reduced mean insulin requirement 57 , suggesting exendin-4/activin A formula differentiated IPCs may be a safe and viable treatment option for Type 1 DM. Nonetheless, further studies should be designed to assess the long-term safety of cell transplantation for human use.
Supplemental Material
sj-jpg-1-cll-10.1177_09636897221106995 – Supplemental material for A One-Step, Monolayer Culture and Chemical-Based Approach to Generate Insulin-Producing Cells From Human Adipose-Derived Stem Cells to Mitigate Hyperglycemia in STZ-Induced Diabetic Rats
Supplemental material, sj-jpg-1-cll-10.1177_09636897221106995 for A One-Step, Monolayer Culture and Chemical-Based Approach to Generate Insulin-Producing Cells From Human Adipose-Derived Stem Cells to Mitigate Hyperglycemia in STZ-Induced Diabetic Rats by Ruei-Yue Liang, Kai-Ling Zhang, Ming-Hsi Chuang, Feng-Huei Lin, Tzu-Chien Chen, Jhih-Ni Lin, Ya-Jyun Liang, Yi-An Li, Chun-Hung Chen, Peggy Leh Jiunn Wong, Shinn-Zong Lin and Po-Cheng Lin in Cell Transplantation
Footnotes
Ethical Approval
Ethical aspects of the study were approved by the Research Ethics Committee of Hualien Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation (IRB No: IRB106-88-A) and the National Taiwan University College of Medicine and College of Public Health Institutional Animal Care and Use Committee (No: 20200050).
Statement of Human and Animal Rights
All procedures involving human subjects were conducted in accordance with policies adopted by the Research Ethics Committee of Hualien Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation (IRB No: IRB106-88-A). All procedures involving animal subjects were conducted in accordance with policies adopted by the National Taiwan University College of Medicine and College of Public Health Institutional Animal Care and Use Committee (No: 20200050).
Statement of Informed Consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Gwo Xi Stem Cell Applied Technology Co., Ltd., Hsinchu, Taiwan (grant number: IP-RD-18-001, IP-RD-19-005 and IP-RD-20-003).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
