Abstract
The survival rates of children with high-risk acute myeloid leukemia (AML) treated with hematopoietic stem cell transplant (HSCT) range from 60% to 70% in high-income countries. The corresponding rate for Brazilian children with AML who undergo HSCT is unknown. We conducted a retrospective analysis of 114 children with AML who underwent HSCT between 2008 and 2012 at institutions participating in the Brazilian Pediatric Bone Marrow Transplant Working Group. At transplant, 38% of the children were in first complete remission (CR1), 37% were in CR2, and 25% were in CR3+ or had persistent disease. The donors included 49 matched-related, 59 matched-unrelated, and six haploidentical donors. The most frequent source of cells was bone marrow (69%), followed by the umbilical cord (19%) and peripheral blood (12%). The 4-year overall survival was 47% (95% confidence interval [CI] 30%–57%), and the 4-year progression-free survival was 40% (95% CI 30%–49%). Relapse occurred in 49 patients, at a median of 122 days after HSCT. There were 65 deaths: 40 related to AML, 19 to infection, and six to graft versus host disease. In conclusion, our study suggests that HSCT outcomes for children with AML in CR1 or CR2 are acceptable and that this should be considered in the overall treatment planning for children with AML in Brazil. Therapeutic standardization through the adoption of multicentric protocols and appropriate supportive care treatment will have a significant impact on the results of HSCT for AML in Brazil and possibly in other countries with limited resources.
Keywords
Introduction
Acute myeloid leukemia (AML) represents 15%–20% of acute leukemias in children, and the risk of treatment failure is based on genetic risk and response to therapy 1 –4 . Although the initial remission rate exceeds 90%, more than 40% of children with AML die of refractory/relapsed disease or treatment-related toxicity 5 . Recently, event-free survival (EFS) and overall survival (OS) rates greater than 60% and 70% in 5 years, respectively, have been reported in high-income countries (including transplant patients and those who did not need a transplant). The best therapeutic results are achieved by integrating intensive chemotherapy, optimal supportive care, and hematopoietic stem cell transplant (HSCT) adapted to each individual patient’s risk of relapse 6 –9 .
Patients with low-risk or intermediate-risk AML are typically not candidates for HSCT in first remission (CR1), whereas patients with high-risk disease or poor response to induction therapy are referred for HSCT in CR1. Allogeneic HSCT offers the best chance of long-term cure for children with relapsed AML 4,10 –12 .
In countries classified by the World Health Organization as middle-income countries, such as Brazil, HSCT has been implemented at several centers, particularly in wealthier regions. Sociocultural and economical limitations pose great challenges to diagnosing AML in these patients and treating them with chemotherapy and HSCT. Many patients do not undergo a proper cytogenetic and molecular evaluation and classification of their disease at diagnosis, and they may not be treated using standardized treatment protocols. In these countries, there is a limited number of hospital beds available for chemotherapy or transplantation, and optimal supportive care and management of comorbidities may not be available, compromising cure rates and increasing the risks associated with these complex and expensive procedures 13 –18 .
Pediatric AML has an estimated incidence of 400 cases per year in Brazil 19 . In a recent Brazilian cohort, 5-year OS was reported to be less than 40% 18 . Results for HSCT have been reported for adult patients in Brazil, but the survival rates for children, adolescents, and young adults are unknown 20 . The objective of this study was to describe the outcomes of children with AML treated with chemotherapy followed by HSCT.
Materials and Methods
Patient Data
This was a multicenter retrospective study using datasets (demographic, disease, and transplant-related characteristics from coded patients) provided by the Brazilian Pediatric HSCT Working Group (BPHWG). The participating institutions reported to the BPHWG all consecutive allogeneic HSCTs performed between 2008 and 2012 to treat AML in patients younger than 21 years at the time of transplant (except those with Down syndrome or promyelocytic leukemia).
Ethics Statement
The protocol was approved by the Ethics Committee of the coordinating institution (Hospital Pequeno Príncipe, Curitiba) (code CAAE: 53705016.7.1001.0097). Informed consent was waived because we used coded datasets provided by the BPHWG.
Endpoints and Definitions
High-risk cytogenetics at this study include t(6;11)(q27;q23)/MLLT4-KMT2A, t(10;11)(p12;q23)/MLLT10-KMT2A, t(10;11)(p11.2;q23)/ABI1-KMT2A, t(6;9)(p23; q34)/DEK-NUP214, t(8;16)(p11;p13)/KAT6A-CREBBP, t(16;21) (q24;q22)/RUNX1-CBFA2T3, t(5;11)(q35;p15.5)/NUP98-NSD1, inv(16)(p13.3q24.3)/CBFA2T3-GLIS2, t(11;15)(p15;q35)/NUP98-KDM5A, t(3;5)(q25;q34)/NPM1-MLF1, FLT3-ITD, and monosomy 7 6 . The primary endpoint was OS. Secondary endpoints were hematopoietic recovery, relapse, progression-free survival (PFS), non-relapse mortality (NRM) rates, and acute Graft versus host disease (aGVHD) and chronic GVHD (cGVHD). OS was defined as the time from HSCT to death from any cause. Surviving patients had their survival times censored at the last follow-up. Relapse was defined as the presence of morphologic or cytogenetic evidence of disease in the peripheral blood, marrow, or extramedullary sites. PFS was defined as the time from HSCT until relapse, progression, or death without evidence of disease. Treatment-related mortality was defined as mortality unrelated to disease progression or relapse. Neutrophil engraftment time was defined as the interval between the transplant and the first of three consecutive days with an absolute neutrophil count of ≥0.5 × 109/L. Platelet engraftment time was defined as the interval between transplant and the last of seven consecutive days with a platelet count of ≥20 × 109/L without platelet transfusion support. Primary graft failure was defined as the failure to achieve an absolute neutrophil count of ≥0.5 × 109/L for three consecutive days or donor chimerism of <5%. Secondary graft failure was defined as initial donor engraftment followed by graft loss, as evidenced by a persistent decline in the absolute neutrophil count to ≥0.5 × 109/L and/or donor chimerism of <5%. aGVHD and cGVHD were classified according to the criteria of Glucksberg et al. 21 and the NIH 2014 criteria 22 . Donor human leukocyte antigen (HLA) matching was defined as the use of HLA-matched sibling or alternative donors, including HLA-matched or mismatched unrelated donors and mismatched family members (haploidentical donors). Matched sibling donors were HLA-A, HLA-B, or HLA-DR/DRB1 typed. Unrelated bone marrow matching was evaluated at a minimum of eight HLA alleles and unrelated umbilical cord blood matching at a minimum of six HLA alleles. Patients were labeled as myeloablative conditioning (MAC) recipients if they received a total body irradiation (TBI) of ≥8 Gy fractionated or a busulfan dose of >8 mg/kg. Reduced-intensity conditioning (RIC) regimens differ from MAC conditioning because the dose of alkylating agents or TBI is reduced by at least 30%. Most often, these RIC regimens combine fludarabine with an alkylating agent, melphalan, busulfan, thiotepa in reduced doses, or fludarabine with reduced dose TBI 23 .
Statistical Analysis
Statistical analyses were performed using the R statistical software, version 3.6.0 (www.r-project.org). A two-sided P-value of <0.05 was considered to indicate statistical significance. Descriptive analyses were reported as absolute and relative values. Continuous data were expressed as the median and standard deviation. Comparisons were performed using the chi-square test or Fisher’s exact test, as appropriate, for categorical variables and the Wilcoxon or Kruskal–Wallis test for continuous variables.
The analysis of the incidence of transplant-related mortality considered relapse as a competitive event. The analyses of the incidence of graft failure, neutrophil recovery, and platelet recovery considered death as a competitive event. For PFS analyses, relapse or death were considered events. The incidences of aGVHD and cGVHD were estimated with death and primary graft failure considered as competitive events. Incidence curves were compared using Gray’s test.
The Cox proportional hazards regression model (random effects) was used to assess the impact of potential risk factors on mortality in multivariable analyses, providing hazard ratios (HRs) with their 95% confidence intervals (CIs). Variables were added sequentially to the model and kept if P was less than 0.05. We then included potential confounders in the model and reassessed the results. The proportional hazard assumption was checked by analyzing the Schoenfeld residuals.
OS and disease-free survival were estimated using the Kaplan–Meier method.
Results
Patients
A total of 114 patients younger than 21 years with AML underwent allogeneic HSCT at 10 Brazilian institutions between January 2008 and January 2012. The median age at the time of transplant was 10 years (range 0.8–20 years), 60% of the patients were male, and 80% were white.
Clinical Characteristics
Cytogenetic data were available for 81 patients, and 31 presented alterations that classified them as high risk 7 (Table 1). Positive cerebrospinal fluid cytology at diagnosis was reported for 13 of 105 patients (12%); data were unavailable for the remaining nine patients. The French–American–British (FAB) classification subtype was reported for 100 patients, and the most frequent FAB subtype was M5 (reported for 23% of cases).
Patients and Disease Characteristics.
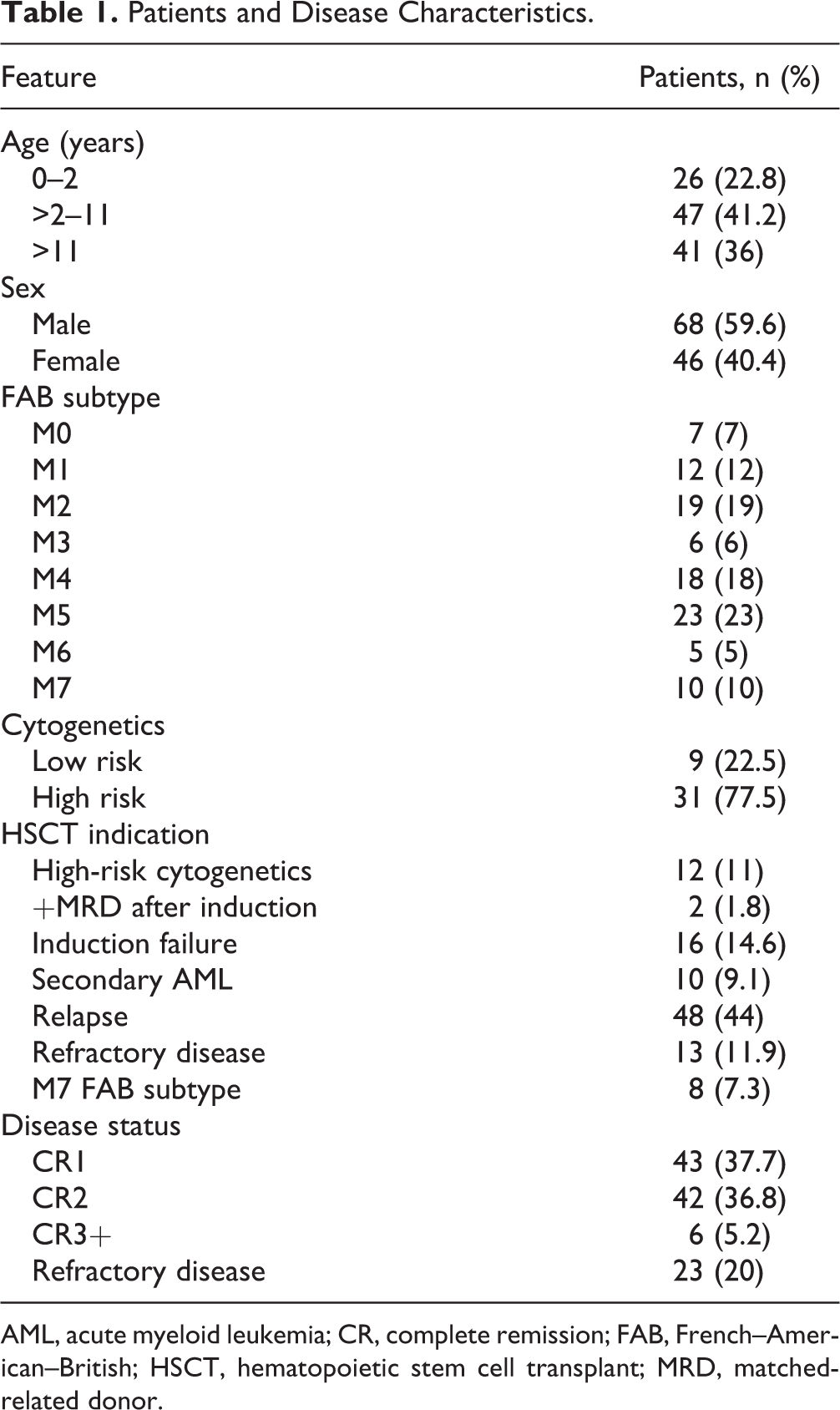
AML, acute myeloid leukemia; CR, complete remission; FAB, French–American–British; HSCT, hematopoietic stem cell transplant; MRD, matched-related donor.
HSCT Indications
The main indications for HSCT were relapse in 48 patients (42%), persistent disease after induction in 16 (14%), and refractory disease in 13 (11.9%). For those patients who experienced remission, the indications for HSCT included cytogenetics in 12 (11%), secondary AML in 10 (9%), M7 FAB subtype in eight (7.3%), and positive matched-related donors (MRDs) after induction in two (1.8%). Data on the indication for HSCT were unavailable for five patients.
Remission Status Before Transplant
Twenty-three patients (20%) had not reached morphologic remission before undergoing HSCT (i.e., they had active disease). At the time of HSCT, 43 patients (37.7%) were in CR1, 42 (36.8%) were in CR2, and 29 (25.4%) had advanced disease or relapse (23 had active disease and six were in CR3+).
Conditioning Regimen
Myeloablative conditioning was used in 104 patients (91%) and a reduced-intensity regimen in 10 (8.7%). Sixty patients (53%) received busulfan in combination with cyclophosphamide, five (4.4%) received busulfan in combination with fludarabine, and seven (6%) received busulfan in combination with melphalan. TBI combined with cyclophosphamide was used in 11 patients (10%) and TBI combined with fludarabine in 17 (15%). Fourteen patients (10%) received other conditioning regimens.
Supportive Care and Infection Prophylaxis
Antibiotic prophylaxis was used in 92% of the patients, with levofloxacin or ciprofloxacin being used for most of them (66.6% of 114 patients). Antifungal prophylaxis, mostly with fluconazole (in 86.8% of cases), was administrated to 111 patients. The main transplant characteristics are shown in Table 2.
Transplant Characteristics.

GVHD, Graft versus host disease; TBI, total-body irradiation; ATG, anti-thymocyte globulin; BU, busulfan; CY, cyclophosphamide; FLU, fludarabine; MEL, melphalan; CSA, cyclosporine; MTX, methotrexate; PTCY, post-transplant cyclophosphamide.
Donors and Stem Cell Source
There were 49 MRDs, six haploidentical donors, and 59 matched-unrelated donors (MUDs), 22 of whom had 6/6 or 8/8 compatibility. The most frequent source of cells was bone marrow (69%), followed by the umbilical cord (19%) and peripheral blood (12%).
Engraftment
After undergoing HSCT, six patients (5.3%) were not evaluable for engraftment. One hundred and one patients (88.6%) showed engraftment at a median of 19 days for neutrophil recovery (12–162 days) and at a median of 22 days (9–156 days) for platelet recovery. Primary rejection was observed in eight patients (7%), four of whom (50%) received a second HSCT.
GVHD
GVHD prophylaxis included a calcineurin inhibitor (cyclosporine or tacrolimus) in 90% of the patients; this was combined with methotrexate in 50% of these cases.
Of 101 evaluable patients, 49 had aGVHD. The median time to occurrence of aGVHD was 27 days (range 10–99 days), and the main site was skin (in 29 patients [59.2%]). The incidence of grade I or II aGVHD was 43% (95% CI 33%–52%), being significantly higher (P = 0.038) in patients who received transplants from unrelated donors (52%; 95% CI 38%–63%) than in patients who received transplants from related donors (31%; 95% CI 17%–43%). For grade III or IV aGVHD, the incidence was 18% (95% CI 10%–24%), and there was no significant difference (P = 0.083) between recipients of transplants from unrelated donors (23%; 95% CI 12%–33%) and recipients of transplants from related donors (10%; 95% CI 13%–18%).
The incidence of cGVHD was 23% (95% CI 15%–30%), with severe disease being observed in six patients. The median time to onset of cGVHD was 195 days (range 52–406 days). There was no significant difference between recipients of transplants from related donors (31%; 95% CI 17%–43%) and recipients of transplants from unrelated donors (17%; 95% CI 7.2%–26%; P = 0.076).
Fungal and Viral Infection
Fungal infection was observed in 19 patients (17%), with 11 of these cases (58%) being caused by Candida spp. Viral infections occurred in 67 of the patients (59%), with 36 of these infections (54%) being caused by cytomegalovirus (CMV). In addition to CMV, the other viral infections reported were Epstein–Barr virus, adenovirus, herpes simplex virus 1 and 2, herpes virus type 6, varicella-zoster virus, polyomavirus, parainfluenza, influenza A, H1N1 influenza, metapneumovirus, and respiratory syncytial virus (Table 3).
Transplant Outcomes.
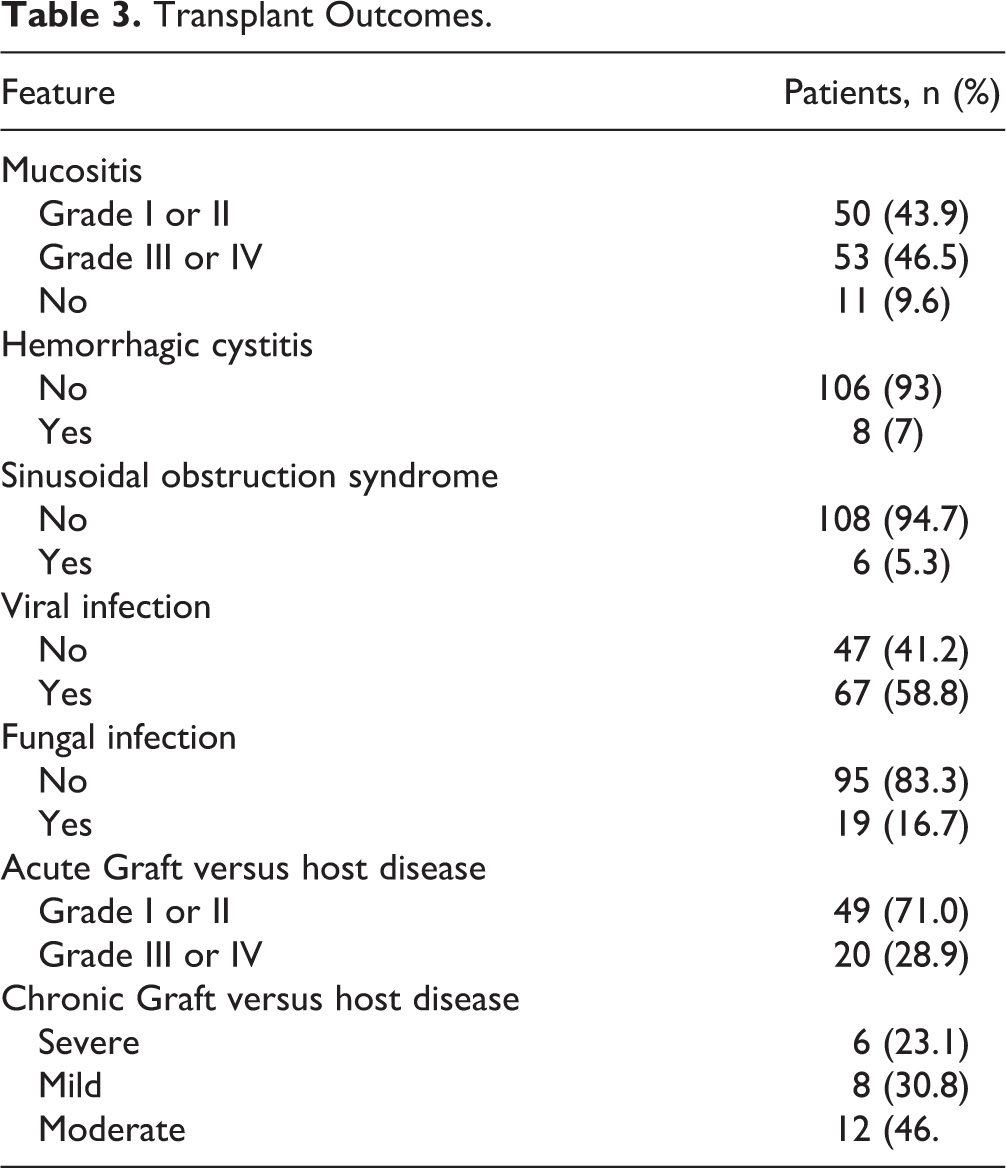
Relapse
Forty-nine patients (48.5%) experienced a relapse. The median time from HSCT to relapse was 122 days (range 17–2424 days). After relapse, 12 patients received a second HSCT, and four received donor lymphocyte infusions (DLIs). Of the patients who experienced relapse, only six (12.2%) survived (at a median follow-up of 6 years).
Survival
The median follow-up time for surviving patients was 4.4 years. Sixty-five patients died, at a median of 159 days after HSCT (range 10–2660 days). The major causes of death were related to relapse (n = 40), infection (n = 19), and GVHD (n = 6).
The 1-year NRM was 19% overall, 10% for recipients of transplants from related donors, and 25% for recipients of transplants from unrelated donors (P = 0.097).
The 4-year OS was 47% (95% CI 30%–57%), and the 4-year PFS was 40% (95% CI 30%–49%) (Fig. 1). There was no significant difference in survival between recipients of transplants from related donors and recipients of transplants from unrelated donors.

Overall survival (A) and progression-free survival (B).
In univariable analysis, 4-year OS was significantly associated with the disease status before transplant (P = 0.001), fungal infection (P = 0.012), and FAB subtype (P = 0.017) of which M7 AML had the lowest survival. The following variables were assessed in the model: age, disease status before transplant, stem cell source, donor type (matched related or alternative), and conditioning regimen (myeloablative or reduced intensity). In the multivariable analysis, only disease status before the transplant was independently associated with 4-year OS and PFS (Tables 4 and 5). There was a significant association (P < 0.0001) between 4-year OS and pretransplant disease status of CR3+ (HR 5.63; 95% CI 2.02–15.67) and between 4-year OS and active disease (HR 3.69; 95% CI 1.83–7.43). A similarly significant association (P < 0.0001) was observed for 4-year PFS, with worse outcomes being recorded for patients in CR3+ (HR 6.71; 95% CI 2.21–20.38) and those with active disease (HR 3.08; 95% CI 1.58–5.99) (Figs. 2–3).
Cox's Proportional Hazards Model for Time (OS) to Event (Death) (N = 114).
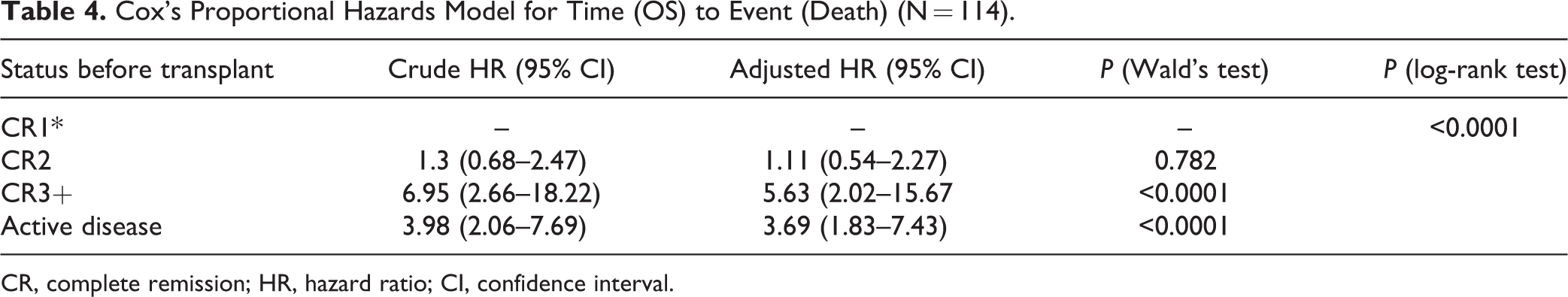
CR, complete remission; HR, hazard ratio; CI, confidence interval.
Cox's Proportional Hazards Model for Time (OS) to Event (disease progression) (N = 114).
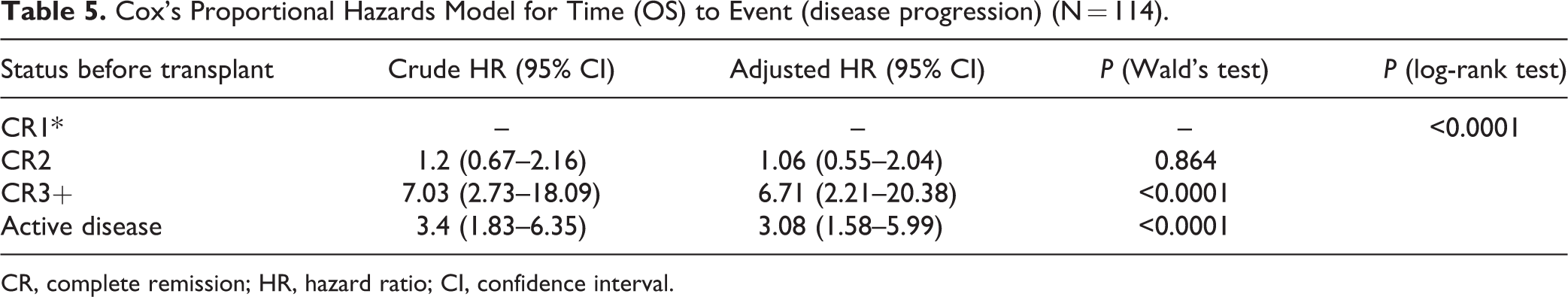
CR, complete remission; HR, hazard ratio; CI, confidence interval.

Overall survival (A) and progression-free survival (B) according to disease status.

Overall survival (A) and progression-free survival (B) according to donor type.
Discussion
In this study, we sought to evaluate the impact of HSCT on the outcomes of children with AML in Brazil. We analyzed data on 114 children with AML who underwent HSCT consecutively at 10 Brazilian institutions. Primary rejection (failure to achieve an absolute neutrophil count of ≥0.5 × 109/L for three consecutive days or donor chimerism of <5%) was observed in eight patients (7%). Acknowledging that some patient also received RIC in this study, 7% is still quite high compared with the current literature (retrospective studies have estimated the overall incidence of graft failure around 5.5%) 24 –26 . The 4-year OS of children with AML who received transplants between 2008 and 2012 was slightly more than 40%. Patients in CR1 or CR2 had significantly higher survival than those in CR3+ or those with persistent disease at the time of HSCT.
In our study, the outcomes of HSCT in children with AML appeared to be inferior to those reported in the United States and Europe. A report by Bitan et al. from the Center for International Blood and Marrow Transplant Research (CIBMTR) on 141 pediatric patients with AML who underwent the transplant in CR1 showed a 5-year PFS of 54% after myeloablative conditioning 27 . Data from the British MRC10 and MRC12 trials showed a 5-year OS of 68% in children who received transplants of marrow from matched sibling donors 28 . The Nordic Society of Paediatric Haematology and Oncology (NOPHO) reported a 3-year EFS of 61% in children who underwent the transplant in CR1 8 . Locatelli et al. analyzed the outcome of 243 children with high-risk AML in CR1 who were enrolled in the AIEOP-2002/01 protocol and underwent either allogeneic (n = 141) or autologous (n = 102) HSCT. The 5-year probability of disease-free survival was 73% 29 . Finally, an AML SCT-BFM study aimed at standardizing pediatric HSCT for AML across centers in Germany and Austria reported 4-year EFS and OS of 61% and 70%, respectively 30 .
Improving the outcomes of HSCT for children with AML in Brazil and other countries with limited resources requires overcoming several barriers. The first of these is the lack of HSCT units and dedicated beds to meet the increasing demand for cellular therapies, including HSCT. The CIBMTR has shown that the number of transplants performed worldwide has increased substantially during the past two decades. However, most of these transplants have been performed in North America, Western Europe, Japan, or Australia. Far fewer transplants have been performed in lower-income countries in South America, Africa, the Middle East, and East Asia. The ratio of inhabitants to transplants performed in Brazil is unbalanced, and many patients do not have access to HSCT even when donors are available 31 . Data from the Brazilian Transplant Registry indicate that uninsured patients may have to wait for an organ transplant for more than a year after donor identification 32 . Because most of the Brazilian HSCT centers are located in the Southern or Southeast regions, expenses associated with travel and housing can be prohibitive for many families. The ability of families to care for children after transplant might also be undermined by sociocultural factors such as poor living conditions and a lack of understanding of the complexity of the medical care required 33 .
NRM due to infectious complications and GVHD were higher in our study than in most recently published international series. In our study, the rate of death not associated with AML was 19%. Zaucha-Prazmo et al. reported an NRM rate of 11% in 82 children with AML in Poland 34 . In a study by Svenberg et al. of 201 children who underwent HSCT consecutively at the Karolinska University Hospital, Sweden, between January 2003 and December 2013, the NRM rates were 12% and 14% at 1 and 5 years, respectively 35 . In a study by Nemecek et al. of 58 children with AML who underwent HSCT between January 1990 and December 1999, children in CR2 (n = 12), those in untreated first relapse (n = 11), and those with refractory disease (n = 35) had estimated NRMs of 0%, 27% (range 0%–54%), and 17% (range 5%–30%), respectively 36 .
Factors related to the evaluation and treatment of patients with AML before referral to transplant centers might also influence the outcomes after transplant 37 . Centers that manage pediatric AML usually lack access to comprehensive relapse-risk assessment before or after induction remission, and not all cases of AML undergo genetic characterization before treatment is initiated. This precludes accurate stratification of children who might be considered for HSCT soon after remission is attained. Post-remission chemotherapy for such patients would increase the morbidity because of the infectious, cardiac, and renal complications associated with the treatment, in addition to delaying HSCT. A lack of effective methods to measure residual disease after remission induction chemotherapy at many institutions also contributes to inadequate patient selection for HSCT and referral. Even though multiparametric flow cytometry laboratories are available at most Brazilian leukemia treatment centers, multiparametric flow cytometry processes must be standardized for MRD investigations in order to provide reliable and reproducible results that ensure quality in the clinical application of the method 38 .
Finally, since the 1980s, several interventions have been implemented to reduce NRM and improve outcomes after HSCT. More effective approaches to preventing GVHD, fungal infection, and CMV disease and to HLA typing and matching have been introduced. Pharmacokinetics-based targeting of busulfan dosing has been adopted. For patients receiving transplants, enhancements have been made in the last decades. At the same time, relevant advances have occurred in related fields, including critical care medicine, nephrology, and transfusion medicine 39 . Prophylaxis for bacterial, fungal, and viral infections was not uniformly performed for the patients in our study. It is possible that the mortality associated with infection would be decreased by applying uniform guidelines for infection prophylaxis.
A critical point to be evaluated in the Brazilian healthcare context, in which beds for HSCT are still insufficient, is whether HSCT should be offered to children with AML who are expected to have a dismal prognosis after HSCT. In our study, patients with persistent disease after relapse had extremely poor outcomes. Similarly, only one of 10 patients with megakaryoblastic leukemia survived after undergoing the transplant. Finally, patients with secondary AML also had very poor prognoses.
The outcomes of HSCT can be improved by establishing communication between transplant centers and pediatric oncology units that are the point of the first contact with children newly diagnosed with AML, identifying the children with AML who would require HSCT early in the treatment course, and implementing uniform guidelines for infection prophylaxis before and during HSCT. It is also critical to involve community support groups to facilitate transport and to provide food, housing, and psychosocial and educational support for patients and families.
Our study has several limitations because of the differences in practices among transplantation centers, including differences in patient selection for HSCT, the choice of stem cells, conditioning regimens, prophylaxis for GVHD and infection, and general supportive care measures. Once resources in this setting are a significant variable, time from eligibility to transplant could be of particular importance. Unfortunately, as this study is a retrospective work from a database, this information was not available. Also, during the study period, according to the Brazilian National Transplant System, a public organization, 312 individuals younger than 21 years with AML underwent HSCT, but 114 (36.5%) were included in this study. Because, pediatric transplant services have different eligibility criteria for age, many patients with AML were transplanted in adult HSCT units during the study period, and we did not have access to data on patients treated on adult HSCT services.
Conclusion
In conclusion, the results of our study, with patients transplanted from 2008 to 2012, suggest that the outcomes of HSCT for children with AML in first and second complete remission are acceptable, and this should be considered in the overall treatment planning for children with AML in Brazil.
Footnotes
Acknowledgments
We are grateful to all of the families and patients involved in the study, to the Brazilian Pediatric HSCT Working Group, to the Brazilian Society of Bone Marrow Transplant (SBTMO), and to the Brazilian Society of Pediatric Oncology (SOBOPE) who supported this work. The authors thank Keith A. Laycock, PhD, ELS, for scientific editing of the manuscript.
Author Contributions
Conceptualization: ALMR, CB, AS, RCR, BCF; formal analysis: ALMR, CB, AS, LA, GLP, RCR, BCF; funding acquisition: BCF; investigation: ALMR, CB, AS, VARC, VGZ, SN, LED, JFF, AKV, LGDJ, AAG, LA, RCR, BCF; methodology: ALMR, CB, AS, LA, RCR, BCF; resources: BCF; supervision: CB, AS, RCR, BCF; writing—original draft: ALMR, CB, AS, LL, RCR, BCF; writing—review and editing: ALMR, CB, AS, LL, RCR, BCF.
Ethical Approval
This study was approved by the Ethics Committee of the coordinating institution of Hospital Pequeno Príncipe, Curitiba (code CAAE: 53705016.7.1001.0097). Informed consent was waived because we used coded datasets provided by the BPHWG.
Statement of Human and animal Rights
This study was approved by the Ethics Committee of the coordinating institution of Hospital Pequeno Príncipe, Curitiba (code CAAE: 53705016.7.1001.0097). Informed consent was waived because we used coded datasets provided by the BPHWG.
Statement of Informed Consent
Informed consent was waived because we used coded datasets provided by the BPHWG.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Raul C. Ribeiro, MD, was partially funded by NCI grant CA21765, ALSAC, and the St. Jude Department of Oncology. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
