Abstract
Donor-derived recipient-specific anti-HLA antibodies (RSAs) are rarely investigated in allogeneic hematopoietic stem cell transplantation (allo-HSCT). In this retrospective study, 185 haploidentical donor-recipient pairs were consecutively analyzed. Anti-HLA antibodies were detected in eight donors (4.3%) and 31 recipients (16.8%), with only one donor (0.5%) harboring RSAs. Donors were predominantly young, male, and transfusion-naïve, which likely contributed to the low antibody prevalence. No association was observed between donor antibody positivity and graft rejection or severe acute graft-versus-host disease (GVHD). However, recipients of antibody-positive grafts showed higher rates of Epstein–Barr virus (EBV) reactivation and transfusion refractoriness. Our findings suggest that donor-derived HLA antibodies, particularly RSAs, are infrequent and clinically limited under current haploidentical donor selection and immunosuppressive strategies, but may subtly influence post-transplant immune recovery.
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) remains a potentially curative therapy for hematologic malignancies. In the setting of haploidentical-related donor transplantation, high titers of donor-specific antibodies (DSAs) have been recognized as a major risk factor for graft rejection and poor survival 1 . In contrast, donor-derived recipient-specific anti-HLA antibodies (RSAs), generated by the donor and transferred through graft infusion, remain largely unexplored. Reports suggest that donor-derived HLA antibodies may persist post-transplant, influence transfusion outcomes, or even contribute to graft-versus-host disease (GVHD) or transplant-associated thrombotic microangiopathy (TA-TMA)2,3. However, systematic evaluation in haploidentical HSCT (haplo-HSCT) cohorts is currently lacking, particularly regarding the presence of HLA antibodies in donors (especially RSAs targeting recipient HLA loci) and their potential impact on post-transplant outcomes.
This retrospective study consecutively enrolled 185 haploidentical donor–recipient pairs who underwent transplantation at our center between November 2023 and January 2025. Prior to transplantation, high-resolution HLA typing was performed for both donors and recipients, covering class I (A, B, C) and class II (DRB1, DQB1, DPB1) loci. In parallel, the immunization status was assessed using a solid-phase assay. Initial screening for anti-HLA class I and/or II antibodies was conducted with a Luminex flow fluorometer (LS, Luminex Screen, One Lambda, USA), and samples testing positive underwent further characterization with single antigen beads (LSA; Immucor, USA). Antibody reactivity was reported as mean fluorescence intensity (MFI). All recipients had hematologic malignancies and received either myeloablative conditioning (MAC) or reduced-intensity/toxicity conditioning (RIC/RTC), followed by a standard GVHD prophylaxis regimen including anti-thymocyte globulin and/or post-transplant cyclophosphamide, cyclosporine, mycophenolate mofetil, and methotrexate. Patients were monitored closely during follow-up for transplant-related complications.
Among the 185 patients included, the majority were diagnosed with leukemia or myelodysplastic syndromes (MDS), including acute myeloid leukemia (AML) in 36.2%, acute lymphoblastic leukemia (ALL) in 30.8%, and MDS in 23.2%. Donors were predominantly offspring (69.2%) or parents (18.9%), and ABO blood group incompatibility was observed in 42.7% of transplants. A total of 76.2% of patients received MAC, and peripheral blood stem cells (PBSCs) were used as the sole stem cell source. The median CD34⁺ cell dose infused was 6.6 × 10⁶/kg (range: 1.3–35.5 × 10⁶/kg). The data cutoff date for follow-up was May 31, 2025. With a median follow-up of 10.8 months (range: 0.3–28.8 months), the incidence of aGVHD was 20.6%, including 9.2% of grades III and IV. The incidence of platelet transfusion refractoriness (PTR) after transplantation was 23.9% (46 of 185 patients). Cytomegalovirus (CMV) viremia and Epstein–Barr virus (EBV) viremia were observed in 33.5% and 62.2% of patients, respectively. The overall survival rate was 87.6%, with a relapse rate of 10.8% and a non-relapse mortality of 7.6%. Baseline characteristics and transplantation outcomes are detailed in Supplementary Table S1.
Among the 185 donor–recipient pairs, anti-HLA antibodies were detected in 8 donors (4.3%) and 31 recipients (16.8%). Donors and recipients differed significantly in age (median 32 (10–59) vs 47 (13–71) years), sex, transfusion history, and pregnancy history (all P < 0.05). Donors were generally younger, predominantly male, and none had a history of blood transfusion. The prevalence of anti-HLA antibodies was significantly higher in recipients than in donors. Among the eight donors with detectable HLA antibodies, three exhibited antibodies against HLA class I, whereas six had antibodies directed against HLA class II antigens. One donor had antibodies to both HLA class I and class II. The maximal MFI of class I antibodies was 5391 (median 2600), while that of class II antibodies was 2982 (median 1838). Among antibody-positive individuals, the detection rate of RSAs was significantly lower than that of DSAs: only 1 of 8 antibody-positive donors (12.5%) had RSAs, a much lower frequency than previously reported in European cohorts2,4, whereas 13 of 31 antibody-positive recipients (41.9%) had DSAs. The incidence and subtypes of HLA antibodies in donors and recipients are detailed in Table 1.
Incidence and subtypes of HLA antibodies in donors and recipients.
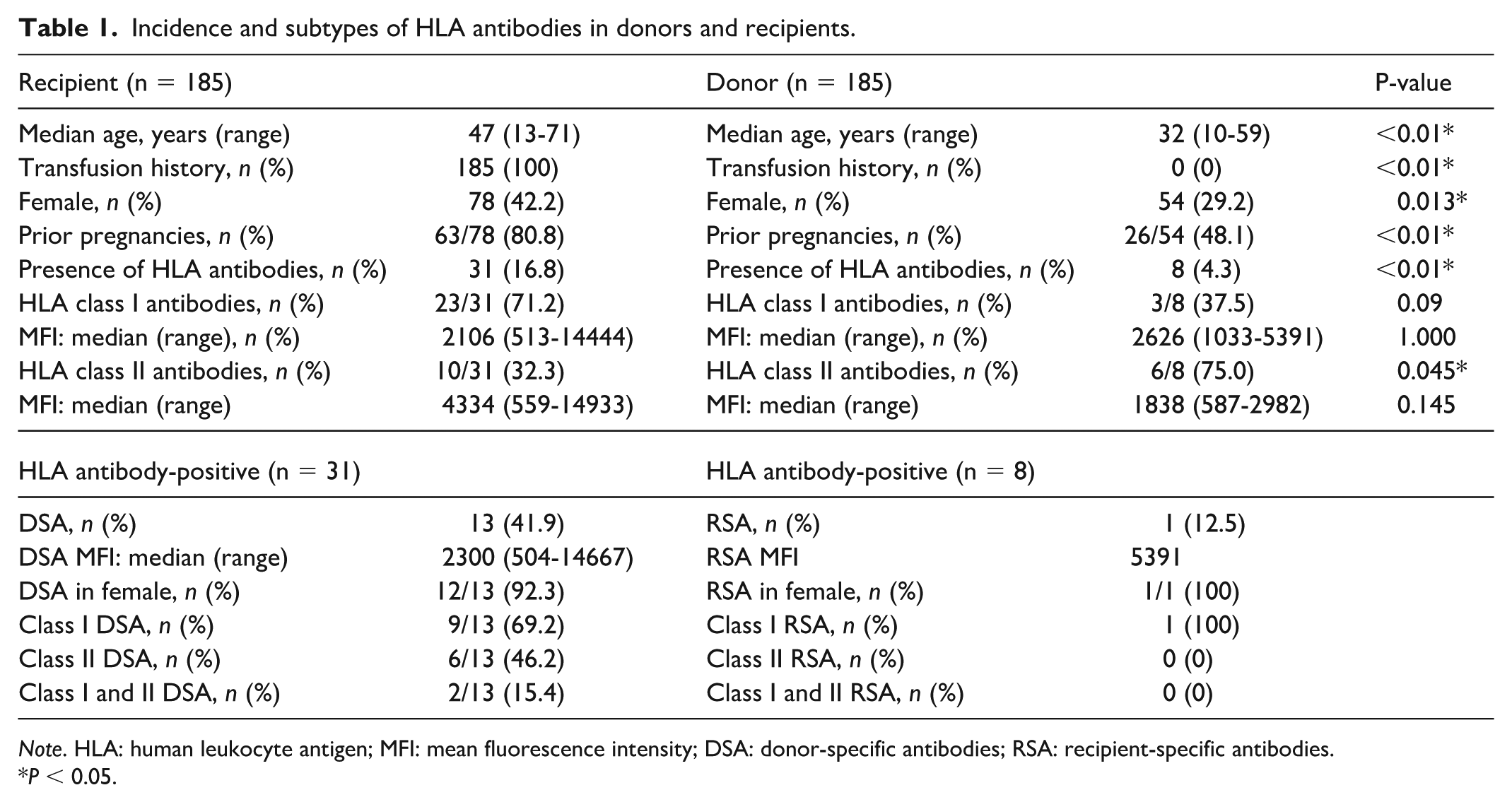
Note. HLA: human leukocyte antigen; MFI: mean fluorescence intensity; DSA: donor-specific antibodies; RSA: recipient-specific antibodies.
P < 0.05.
Further analysis of the eight patients whose donors were HLA antibody-positive revealed that none of the corresponding recipients had anti-HLA antibodies. Among these eight recipients, three (37.5%) developed aGVHD, with two (25%) experiencing grade I–II and one (12.5%) grade III disease. No patient developed chronic GVHD. Three patients (37.5%) exhibited PTR. One AML recipient with an RSA-positive donor developed only grade I cutaneous aGVHD without other notable clinical events. Regarding viral reactivation, seven cases (87.5%) had EBV reactivation and two cases (12.5%) had CMV reactivation. Seven patients survived, while one AML patient died on day +42 post-transplant due to severe infection, septic shock, and multiorgan failure. Clinical characteristics and outcomes of the eight patients with donor HLA antibody positivity are summarized in Supplementary Table S2.
Our study represents one of the first systematic evaluations of donor-derived anti-HLA antibodies in a haploidentical transplantation cohort from China. We observed a very low prevalence of donor HLA antibody positivity (4.3%) and a single case (0.5%) of RSA, remarkably lower than that reported in unrelated or mismatched European cohorts, where donor anti-HLA antibodies were detected in up to 30%–40% of donors and RSAs in 50%–75% of antibody-positive cases2,4. Several factors likely contribute to this discrepancy. Most donors in our cohort were young males without transfusion or pregnancy history, whereas previous studies identified female sex and prior pregnancy or transfusion exposure as the dominant drivers of HLA sensitization3,5. Ethnic or genetic factors may also underlie the lower prevalence observed in our cohort, given population-level differences in HLA allele frequencies and immune responsiveness; this hypothesis warrants validation in larger multiethnic studies. These findings reinforce the notion that the prevailing donor selection strategy in haploidentical transplantation (favoring young, related, male donors) may inherently minimize the risk of donor-derived HLA immunization and antibody transmission.
Importantly, no association was observed between donor HLA antibody positivity and graft rejection or severe acute GVHD in our cohort. Given that only eight donors were HLA antibody-positive, these results should be interpreted descriptively rather than inferentially. Delbos et al. 3 reported that donor class II anti-HLA immunization, particularly against HLA-DPB1, independently increased the risk of acute and chronic GVHD (hazard ratios 1.7 and 2.5, respectively) in unrelated donor transplantation. In contrast, all 185 patients in our haploidentical cohort received GVHD prophylaxis based on anti-thymocyte globulin and/or post-transplant cyclophosphamide, regimens known to mitigate both cellular and humoral alloreactivity6,7. The overall incidence of aGVHD was 20.6%, including 9.2% of grades III and IV, representing an acceptable outcome for haploidentical transplantation. Moreover, the single RSA-positive donor in our study was associated with only mild cutaneous grade I aGVHD, supporting the hypothesis that donor-derived antibodies exert limited clinical effects under robust immunosuppression.
Nevertheless, we found that recipients of grafts from HLA antibody-positive donors showed a numerically higher incidence of EBV reactivation (87.5% vs 61.0%) and PTR (37.5% vs 24.3%), whereas the incidence of CMV reactivation appeared slightly lower (25% vs 33.9%). However, none of these differences reached statistical significance (all P > 0.05). Although causality cannot be established, this observation suggests that donor-derived HLA antibodies may transiently interfere with post-transplant immune recovery. Regarding the relationship between donor HLA antibodies and PTR, Fasano et al. 8 previously demonstrated that donor-derived HLA antibodies can persist for more than 100 days after HSCT and contribute to transfusion refractoriness. However, the maximal MFI of donor-derived HLA class I antibodies in our cohort was 5391, which is below the limited published thresholds (≥5000–10,000) reported to be associated with clinically significant transfusion refractoriness or antibody-mediated graft effects8,9. Although the incidence of PTR was numerically higher in the donor HLA antibody-positive group than in the antibody-negative group, the difference was not statistically significant, likely due to the small number of positive donors. In addition, because thrombocytes express HLA class I but not class II antigens, among the eight donors with HLA antibodies, only three had antibodies restricted to HLA class I, and one of these developed PTR 33.3%. Therefore, we cautiously speculate that donor-derived HLA antibodies were unlikely to be directly responsible for PTR in this study. A limitation of our study is that HPA (human platelet antigen) antibody testing was not performed in donor samples. As immunization may generate both anti-HLA and anti-HPA antibodies, undetected donor-derived HPA antibodies could theoretically influence post-transplant platelet refractoriness 10 . Future studies incorporating combined HLA and HPA antibody screening are warranted to better delineate their respective roles in transfusion outcomes. Previous studies have also proposed a mechanistic link between donor- or recipient-derived HLA antibodies and TA-TMA, likely mediated through classical complement activation and subsequent endothelial injury, which has also been implicated in viral reactivation2,11. In our cohort, no TA-TMA events were observed among the eight recipients whose donors were HLA antibody-positive, possibly reflecting the very low prevalence of donor-derived HLA antibodies and RSAs (only one case, 0.5%). Nevertheless, the potential interplay between RSAs, complement activation, and endothelial dysfunction warrants further investigation in larger, prospective studies. Taken together, these findings suggest that the clinical effects of RSAs may primarily manifest through subclinical immune modulation rather than overt graft failure or GVHD.
Our results also highlight the relevance of donor characteristics and prior sensitization in predicting immunologic risk. It has been noted that multiparous women could produce RSAs targeting offspring HLA antigens, potentially leading to immune-mediated tissue injury if transferred with the graft3,11. Although the clinical consequences of passive antibody transfer remain uncertain, this theoretical risk justifies consideration of donor antibody screening, particularly in multiparous female donors or those with extensive transfusion histories. Future studies incorporating complement-binding assays, antibody isotyping, and longitudinal antibody monitoring could further clarify whether donor-derived antibodies have transient or persistent biological effects.
In conclusion, our study investigated the incidence and clinical characteristics of donor-derived HLA antibodies, including RSAs, in haploidentical transplant recipients with hematologic malignancies. Donor HLA antibodies were infrequent, and RSAs were even rarer, likely reflecting the younger and predominantly male donor population. Under the current donor selection strategy, donor HLA antibodies do not appear to be associated with severe post-transplant complications. Based on these findings, routine donor HLA antibody screening is not warranted for all haploidentical donors. However, targeted testing may be reasonable in selected situations, such as in female donors with prior pregnancies or donors with transfusion histories, where the likelihood of sensitization is higher. However, given the limited number of antibody-positive cases, their potential roles in endothelial activation, infection susceptibility, and immune reconstitution warrant further investigation in larger multicenter prospective studies integrating serologic and mechanistic analyses.
Supplemental Material
sj-docx-1-cll-10.1177_09636897261417187 – Supplemental material for Incidence and clinical relevance of donor-derived recipient-specific anti-HLA antibodies in haploidentical hematopoietic stem cell transplantation: a retrospective study
Supplemental material, sj-docx-1-cll-10.1177_09636897261417187 for Incidence and clinical relevance of donor-derived recipient-specific anti-HLA antibodies in haploidentical hematopoietic stem cell transplantation: a retrospective study by Xianbo Huang, Yanling Ren, Shasha Wang, Chen Mei, Yu Xu, Jie Jin, Hongyan Tong and Jiejing Qian in Cell Transplantation
Footnotes
Acknowledgements
We extend our gratitude to the patients for their consent to share their medical records for publication. We also acknowledge the dedication and expertise of the medical staff and physicians involved in patient care.
Ethical Considerations
This retrospective study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Review Committee of the First Affiliated Hospital, College of Medicine, Zhejiang University (Approval No. IIT20250786A, approved on June 25, 2025).
Author Contributions
J.Q., H.T., and J.J. conceived and designed the study. X.H. drafted the manuscript. X.H., J.Q., Y.R., S.W., Y.X., and C.M. collected and managed clinical data. X.H. and J.Q. verified the data and performed statistical analyses. All authors reviewed and approved the final version of the manuscript.
Data Availability Statement
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Consent Statement
Written informed consent was obtained from all patients and donors.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
