Abstract
Astragalus membranaceus (Ast) and ligustrazine (Lig) have a protective effect on lower hemorrhagic transformation induced by pharmaceutical thrombolysis. The cerebral ischemia rat model was induced with autologous blood clot injections. A combination of Ast and Lig, or a protein kinase C delta (PKCδ) inhibitor—rottlerin, or a combination of Ast, Lig, and rottlerin was administered immediately after recombinant tissue plasminogen activator injection. The cerebral infarct area, neurological deficits, cerebral hemorrhage status, neuronal damage and tight junctions’ changes in cerebral vessels, and the messenger RNA and protein levels of PKCδ, myristoylated alanine-rich C kinase substrate (Marcks), and matrix metallopeptidase 9 (MMP9) were determined after 3 h and 24 h of thrombolysis. The ultrastructure of the neuronal damage and tight junctions was examined under a transmission electron microscope. The expression levels of PKCδ, Marcks, and MMP9 were assessed by immunohistochemistry, western blot, and quantitative real-time polymerase chain reaction . Administration of Ast and Lig not only significantly decreased neurological deficit scores, infarct volumes, and cerebral hemorrhage but also inhibited the disruption due to neuronal dysfunction and the tight junction integrity in the cerebral vessel. Treatment with a combination of Ast and Lig effectively protected ischemia-induced microhemorrhage transformation through PKCδ/Marcks pathway suppression.
Introduction
Acute cerebral infarction is the primary type of ischemia; many treatments for this disease demonstrate limited efficacy for treating neurological function. Recombinant tissue plasminogen activator (rt-PA) is the only drug for ischemia approved by Food and Drug Administration, 1,2 but its efficacy is limited by a time-constrained treatment window, that is, approximately >4.5 h from ischemia onset. 3
Furthermore, hemorrhagic transformation (HT) after thrombolysis, which refers to the pathological process such as vascular reperfusion and coagulation disorder in the ischemic area, 4 has been documented as the most common complication of thrombolytic treatment in the clinical setting. 5 HT mechanism after drug thrombolysis is still unknown. In the early stage of ischemia, damage to the endothelial cell barrier leads to the exudation of water and other small molecules, 6 followed by the degradation of the basal layer barrier function, resulting in the occurrence of bleeding. HT involves the matrix metallopeptidases (MMPs), and MMP activation is partly responsible for the disruption of the blood–brain barrier (BBB). 7 In HT mechanism, MMP9 degraded the perivascular basement membrane and the extracellular matrix, thus, promoting the BBB breakdown mediated by neuronal apoptosis and brain injury. 8
In addition, previous studies have found that protein kinase C delta (PKCδ) plays a critical role in regulating cerebral ischemia-reperfusion injury 9 ; myristoylated alanine-rich C kinase substrate (Marcks) is a major substrate of PKC. 10 PKCδ-specific peptide inhibitors attenuate reperfusion injury and improve outcome following thrombolysis. 11,12
Astragalus membranaceus (Ast) and ligustrazine (Lig), Chinese herbal medicines, are widely used in the treatment of cerebral ischemia. Ast has been found to have antioxidant properties and protective effects on intestinal mucosal injury induced by ischemia-reperfusion in rats. 13 The underlying mechanism in neural protection of Lig relates to its antioxidant, antiapoptotic, and anti-inflammatory properties. 14,15 In our previous study, we found that treatment with a combination of Ast and Lig reduced ischemia-induced microhemorrhage transformation by maintaining the integrity of the BBB. However, the mechanism of the Ast and Lig combination to modulate PKCδ/Marcks in HT is unknown.
In the present study, a classical brain ischemia model was utilized to investigate the neuroprotective effects of Ast and Lig, to examine their role in the attenuation of ischemia-induced microhemorrhage, and in the preservation of neurological function by regulating PKCδ expression/Marcks and MMP9.
Methods
Animals
The animal procedures were performed according to the protocol approved by the Institutional Animal Care and Use Committee (IACUC) of Guangdong Provincial Hospital of Chinese Medicine, Guangzhou, China. This protocol was in accordance with the European Community guidelines (EEC Directive of 1986; 86/609/EEC). Adult male Sprague–Dawley (SD) rats (Medical Laboratory Animal Center of Guangdong, Guangzhou, China), weighing 270–330 g, were housed in the animal room at 22–24 °C on a 12 h light/dark circle with free access to food and water.
Induction of Focal Embolic Cerebral Ischemia
A single, intact, and autologous embolus was prepared according to the procedure that was modified from the previous study. 16 Femoral arterial blood (20 μl) was withdrawn and injected into a 20-cm PE-50 tube. Next, the PE-50 tube was connected to a syringe containing thrombin (Sigma Aldrich; St Louis, MO, USA). After 15 min, a 30- to 35-mm (1.0 μl), intact, and autologous clot was transferred into a 75 mm2 culture dish containing a small amount of saline. Subsequently, the clot was withdrawn into a PE-10 tube, which was connected to a PE-50 tube. After connecting the PE-50 tube to a Hamilton syringe, the embolus was ready for injection. The animals were anesthetized with 100 mg/kg ketamine (GuTian Pharmaceutical Co., Ltd., Fujian, China) and 10 mg/kg xylazine (Sigma Aldrich). A heating pad (RWD Life Science, Shenzhen, China) was used to maintain animals’ temperature at 37.5 °C. Following anesthesia, the rats were placed in the supine position, and a midline incision was made in the neck under an operating microscope (Leica; Wetzlar, Germany). The right common carotid artery, external carotid artery (ECA), and internal carotid artery (ICA) were isolated. The PE-10 tube containing the autologous embolus was then carefully introduced into the ECA lumen and advanced gently from the ECA into the ICA lumen as described previously. 17 To prevent the embolus from being injected into the posterior cerebral artery, the length of the advancing PE-10 tube was 15 mm (from the bifurcation of the ICA and ECA). The clot was gently injected into the ICA over 10 s. The sham surgery was identical to the procedure described for the induction of the focal embolic ischemia model with the exception that the autologous embolus was not injected.
Experimental Design
The animals were randomized into sham (n = 20), middle cerebral artery occlusion (MCAO, n = 30), thrombolysis (n = 30), thrombolysis + rottlerin (PKC-inhibitor, n = 30), Ast + Lig (n = 30), and thrombolysis + rottlerin + Ast + Lig groups (n = 30). The Ast for injection (dried A. membranaceus (Fisch) Bunge roots) was obtained from Chiatai Qingchunbao Pharmaceutical Company (Hangzhou, China; Batch: 1112043), and the Lig for injection was obtained from Zhengzhou Cheuk-Fung Pharmaceutical Company (Zhengzhou, China; Batch: 11101312).
In accordance with the previous studies, the Ast (the astragaloside content was 10 mg/ml) and Lig injection (the tetramethylpyrazine content was 10 mg/ml) dosages were 2 ml/kg and 1 ml/kg, respectively. 18 Saline was added to the compound to prepare 3 ml solutions for injection. Immediately after the surgeries and sham surgeries, saline (3 ml, sham group and MCAO group) and Ast (2 ml/kg) and Lig (1 ml/kg, 3 ml final volume, Ast + Lig group) combination were injected intraperitoneally. After 3 h of cerebral ischemia, rt-PA (5 mg/kg; Boehringer Ingelheim GmbH, Ingelheim am Rhein, Germany) was intravenously injected to induce pharmaceutical thrombolysis. After 2.5 h of cerebral ischemia, rottlerin (1 mg/kg, Sigma Aldrich, R5648) was administered intravenously, followed by rt-PA (5 mg/kg) injection using micropump. After 3 h and 24 h of cerebral ischemia induction, the rats from all groups (n = 6) were subjected to triphenyl tetrazolium chloride (TTC) staining to detect the focal cerebral infarcts. At 3 h and 24 h after the surgeries, rats from all of the groups (n = 6 at each) underwent neurological deficit assessments and were euthanized by decapitation, followed by cerebral hemorrhage measurements (n = 6 of sham, rt-PA, rt-PA + Ast + Lig group), transmission electron microscopic (TEM) examinations (n = 5 in each group), immunohistochemical assays (n = 3 in each group), quantitative real-time polymerase chain reactions (qRT-PCR) (n = 6 in each group) and Western blot analyses (n = 6 in each group). The experimental design is shown in Fig. 1.

Experimental design. The rats were intravenously injected with rt-PA for thrombolysis after 3 h of MCAO. Then the neurological deficits, TTC, infarct volumes, TEM assessments, cerebral hemorrhage, and the expressions of PKCδ, Marcks, and MMP-9 were conducted by IHC, WB, IF, or qRT-PCR at the indicated time points after thrombolysis. IF: immunofluorescence; IHC: immunohistochemistry; Marcks: myristoylated alanine-rich C-kinase substrate; MCAO: middle cerebral artery occlusion; MMP-9: matrix metalloproteinase 9; PKCδ: protein kinase C delta; qRT-PCR: quantitative real-time polymerase chain reaction; rt-PA, recombinant tissue plasminogen activator; TEM: transmission electronic microscope; TTC: 2,3,5-triphenyltetrazolium chloride; WB: Western blot.
Neurological Deficit Assessment
We employed a five-point system to assess the neurological deficits of the rats at 3 h and 24 h after the induction of cerebral ischemia as previously reported. 19
TTC Staining
The brains were removed and sliced into 2 mm cross-sections using a rat-brain matrix (RWD Life Science). Sample slices were incubated in 2% 2,3,5-TTC solution (Sigma-Aldrich) at 37 °C for 15 min.
Cerebral Hemorrhage Measurement
The animals were euthanized, perfused, and decapitated as described above. Cerebral hemorrhage was quantified according to the previously reported methods. 20 A standard curve was obtained using a “virtual” model of the hemorrhage. Incremental volumes of homologous blood (0, 2, 4, 8, 16, and 32 μl) were added to the perfused brain. The brain samples were homogenized and then centrifuged at 13,000 rpm for 30 min. Next, Drabkin reagent (1.6 ml; Sigma-Aldrich) was added to 0.4 ml supernatant aliquots, and the optical density was measured at 540 nm with a spectrophotometer. The amount of cerebral hemorrhage was quantified and compared with the standard curve to obtain hemorrhage volume (μl) data.
Immunohistochemistry
The rats were euthanized and transcardially perfused with saline and 4% paraformaldehyde as described above. The brains were removed and stored in 4% paraformaldehyde for 24 h. Tissues were then sliced with a Leica RM2235 microtome (Leica Microsystems) and mounted on slides for staining. The brain sections (3 μm) were used in Strept Avidin-Biotin Complex (SABC) Kit (BOSTER, SV0002) and incubated overnight at 4 °C with primary antibodies against PKCδ (1:250 dilution; ab133456; Abcam Technology, Cambridge, UK), Marcks (1:300 dilution; ab13811; Abcam Technology), and MMP9 (1:300 dilution; ab38898; Abcam Technology). Images were acquired from the MCA region of the cortex from a blinded observer. Imaging for fluorescence was performed using a Zeiss Axio Observer, and the Just Another Co-Localization Plugin (JACoP) for ImageJ was used to determine Pearson’s coefficient with background threshold adjusted to baseline.
Transmission Electronic Microscope (TEM) Examination
The animals were euthanized and transcardially perfused with 50 ml of saline followed by 100 ml of 4% paraformaldehyde and 2.5% glutaraldehyde in 0.1 mol/L cacodylic acid buffer (pH 7.3). The fixed brain was dehydrated through an ethanol series, embedded in epoxy resin, and then cut into ultrathin sections. The sections were mounted on copper grids, stained in uranyl acetate and citric acid lead, and then observed under a TEM (H-7650; Hitachi; Tokyo, Japan).
The rats were then transcardially perfused with precooled saline and glutaraldehyde (2.5% glutaraldehyde and 2% paraformaldehyde in 0.1 mol/L sodium dimethyl arsenate buffer). The parietal cortices of the right cerebral hemisphere were cut into blocks (1 mm × 1 mm × 1 mm). The blocks were placed in a solution containing lanthanum and glutaraldehyde and fixed for 2 h at 4°C. The blocks were then rinsed twice with 0.1 mol/L sodium cacodylate buffer and dehydrated with an acetone gradient, and then cut into ultrathin sections. The sections were observed under a TEM.
Western Blot Analysis
Western blot analysis was performed as previously described. 21 Protein concentration was measured by Enhanced Bicinchoninic Acid Assay Protein Assay Kit (Beyotime Institute of Biotechnology, Shanghai, China). Protein samples (20 μg/lane) were loaded on a 12% sodium dodecyl sulfate polyacrylamide gel electrophoresis to separate and electrophoretically transferred to a polyvinylidene difluoride membrane (Millipore Corporation, Billerica, MA, USA). The membrane was blocked with 5% bovine serum albumin (BSA; BIOSHARP, Hefei, AH, China) for 1 h at room temperature. The membrane was further incubated overnight at 4 °C with anti-Marcks antibody (ab217695; Abcam Technology), anti-PKC antibody (ab19031; Abcam Technology), anti-MMP-9 antibody (ab38898; Abcam Technology), and anti-β-actin antibody (ab8226; Abcam Technology). Then, the membrane was incubated with corresponding horseradish peroxidase-conjugated secondary antibodies (ZSGB Biotech Co., Ltd., Beijing, China) for 2 h at room temperature. The signal was developed using an enhanced chemiluminescence (ECL) kit (Beyotime Institute of Biotechnology, Shanghai, China) and then exposed to X-ray films. The films were scanned using an Epson Perfection 2480 scanner (Seiko Corp, Nagano, Japan). The relative quantity of protein was analyzed based on densitometry analysis by use of the Image J program and normalized to that of loading controls.
qRT-PCR
qRT-PCR was performed as previously described. 21 Total RNA was isolated from the brain samples with Trizol reagents (Invitrogen) according to the manufacturer’s instructions. cDNA for the templates was synthesized from the total RNA, and qRT-PCR was performed with ABI Prism TM 7500 machine by using a TransScript Green Two-Step qRT-PCR SuperMix Kit (TRANSGEN BIOTECH, AQ201-01, Beijing, China). The primers were synthesized by HYY Biotechnology (Guangzhou, China) and are shown in Table 1. The experiments were repeated twice. β-actin was used as internal control.
The Primers of PKCδ, MARCKS, MMP-9, and β-Actin Used in this Study.

Marcks: myristoylated alanine-rich C-kinase substrate; MMP-9: matrix metalloproteinase 9; PKCδ: protein kinase C delta.
Statistical Analysis
The data were statistically analyzed and graphed using GraphPad Prism 7 (GraphPad Software, San Diego, CA, USA). All data were presented as the means ± standard deviations. Statistical significances between groups were determined by two-tailed Student’s t-test and two-way analysis of variance test. *P < 0.05, **P < 0.01, and ***P < 0.001 were considered statistically significant.
Results
The Infarct Volumes of Each Group
The infarct volumes in the MCAO, thrombolysis, Ast + Lig-treated, rottlerin-treated, Ast + Lig + rottlerin-treated groups were 275 ± 20.3 mm3, 268.5 ± 16.0 mm3, 235.7 ± 22.0 mm3, 238.0 ± 26.0 mm3, 231.0 ± 19.0 mm3 at 3 h after thrombolysis and 278.4 ±19.9 mm3, 265.7 ±18.5 mm3, 230.8 ± 26.0 mm3, 239.0 ± 28.5 mm3, 229.8 ± 27.3 mm3 at 24 h after thrombolysis, respectively (Fig. 2A). No infarcted areas were detected in the sham group (Fig. 2B). At 3 h after thrombolysis, the infarct volume in Ast + Lig-treated group was decreased by 14.6% compared with the MCAO group and 13.2% compared with the thrombolytic group. At 24 h after thrombolysis, the infarct volume in Ast + Lig-treated group was decreased by 17.1% compared with the MCAO group and 14% compared with the thrombolytic group. The decline became more obvious at 3 h and 24 h when Ast + Lig was combined with PKCδ-specific inhibitor rottlerin. However, this difference was not significant (P > 0.05).
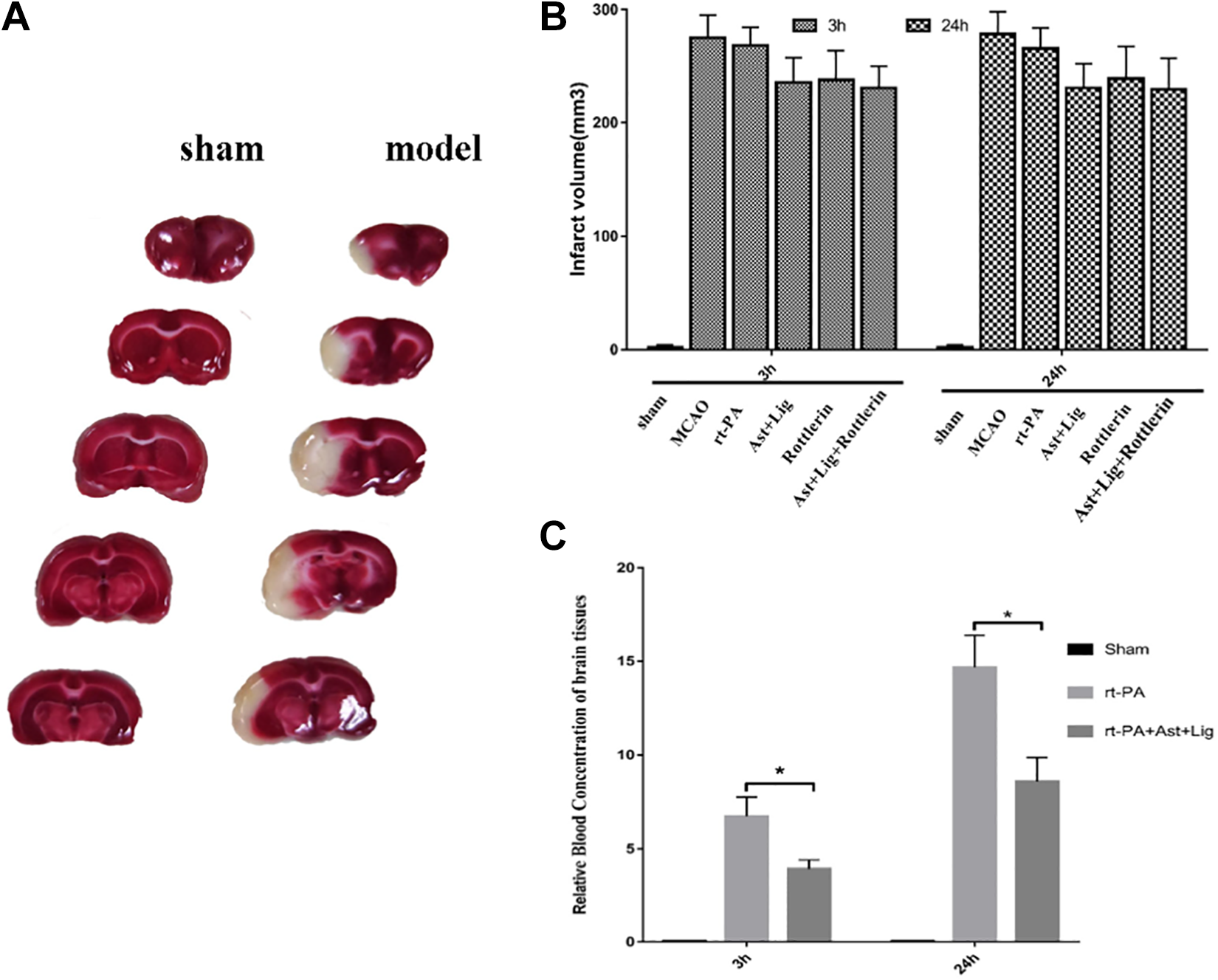
TTC staining. Infarct volumes and cerebral hemorrhage after thrombolysis for 3 h and 24 h. (A) The brain sections stained with TTC for 15 min showed a significant infarct area (white part) in the MCAO group. (B) According to the analysis and statistics of infarct volumes by Image J software, compared with the MCAO group, all groups showed a decrease without statistical significance (P > 0.05, n = 6 in each group). (C) Cerebral hemorrhage in the sham group, rt-PA group, and Ast + Lig group at 3 h and 24 h after thrombolysis. In the Ast + Lig group, the cerebral hemorrhage was significantly reduced in comparison with that in the rt-PA group (*P < 0.05, n = 6 in each group, mean ± standard deviation). Ast: Astragalus membranaceus; Lig: ligustrazine; MCAO: middle cerebral artery occlusion; rt-PA: recombinant tissue plasminogen activator; TTC: 2,3,5-triphenyltetrazolium chloride.
The Drug Combination Treatment Reduced rtPA-Mediated Brain Hemorrhage in Rats
The amount of cerebral hemorrhage in thrombolytic group (6.53 ± 1.03 and 14.561 µl after 3 h and 24 h of ischemia) continued to increase with time after adding Ast + Lig (3.81 ± 0.48 and 8.62 ± 1.26 µl after 3 h and 24 h of ischemia; Fig. 2C). Infusion of tPA (5 mg/kg) in rats subjected to ischemia increased brain hemorrhage, and the amount of cerebral hemorrhage will increase correspondingly with time when compared with rtPA + Ast + Lig-treated group. Moreover, the amount of microhemorrhage in the Ast + Lig-treated group was significantly less than that in the rtPA group at 3 h and 24 h (P < 0.05).
Drug Combination and PKCδ-Inhibitor Treatment Reduced the Neurobehavioural Deficits
The results showed that rats in cerebral ischemia model group suffered severe damage after 3 h and 24 h, with behavioral scores of 3.33 ± 0.33 and 3.33 ± 0.33, respectively. However, the neurological deficits were decreased significantly in the rt-PA, rt-PA + Ast + Lig, rt-PA + Ast + Lig + rottlerin, and rt-PA + rottlerin-treated groups (P < 0.05); Noticeably, co-Ast + Lig and co-Ast + Lig + rottlerin group exhibited lower neurological deficits compared with Ast + Lig group and Ast + Lig + rottlerin group (2.16 ± 0.60, 2.16 ± 0.48 vs 2.33 ± 0.49, 2.83 ± 0.40 at 3 h; 1.83 ± 0.31, 2.0 ± 0.37 vs 2.17 ± 0.40, 2.67 ± 0.33 at 24 h, Table 2), but these differences were not significant (P > 0.05).
Neurological Deficit Assessment.
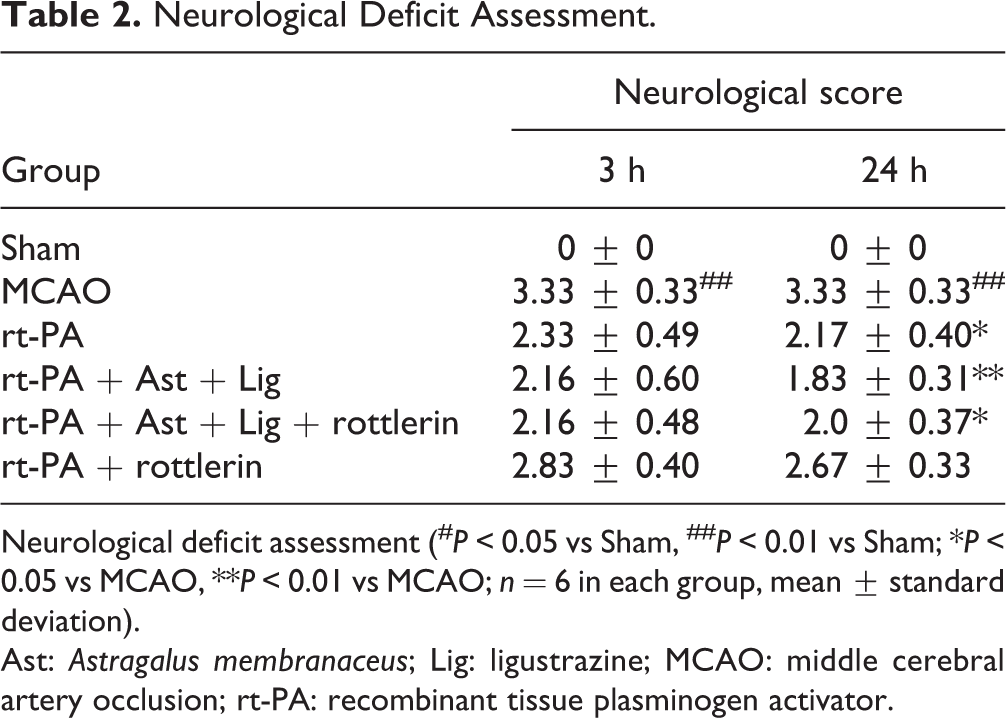
Neurological deficit assessment (# P < 0.05 vs Sham, ## P < 0.01 vs Sham; *P < 0.05 vs MCAO, **P < 0.01 vs MCAO; n = 6 in each group, mean ± standard deviation).
Ast: Astragalus membranaceus; Lig: ligustrazine; MCAO: middle cerebral artery occlusion; rt-PA: recombinant tissue plasminogen activator.
Treatment with the Drug Combination Combined with PKCδ Inhibitor Reduces Neuronal Damage and Maintains the Integrity of Tight Connections of Blood Vessels in the Brain
The neuron morphology of the brain tissue in the sham operation group was normal (Figs. 3A and 4A). In the MCAO group, there was a high degree of edema and degeneration around the neurons, increased electron density in the cytoplasm, mitochondrial vacuolization, increased nuclear heterochromatin, and a small number of autophagosomes appeared (Fig. 3B) at 3 h. Furthermore, there were obvious edema and neuronal necrosis, mitochondrial vacuolization, and nuclear fragmentation after 24 h (Fig. 4B). Neuronal necrosis was not reduced with thrombolytic drug (rtPA) treatment (Figs. 3C and 4C). However, with the addition of Ast + Lig and PKCδ inhibitor (rottlerin), the neuron damage was significantly reduced (Figs. 3D-F and 4D-F). Furthermore, in the inhibitor group, the damage was more severe than that in Ast + Lig addition-treated groups (Figs. 3E,F and 4E, F). The tight junctions of the cerebral vessels in the sham rats remained intact (Figs. 5A and 6A). However, the tight junctions were disrupted in the MCAO group (Fig. 5B). The gap between the tight junctions of the cerebral vessels was widened, and around the cerebral vessels, the cavity deepened with time (Fig. 6B). After the administration of Ast + Lig and PKCδ inhibitor (rottlerin), the tight junction damage in the cerebral vessels was reduced significantly (Figs. 5D-F and 6D-F). The tight junction damage was further reduced in the Ast + Lig-treated groups (Figs. 5E, F and 6E, F).
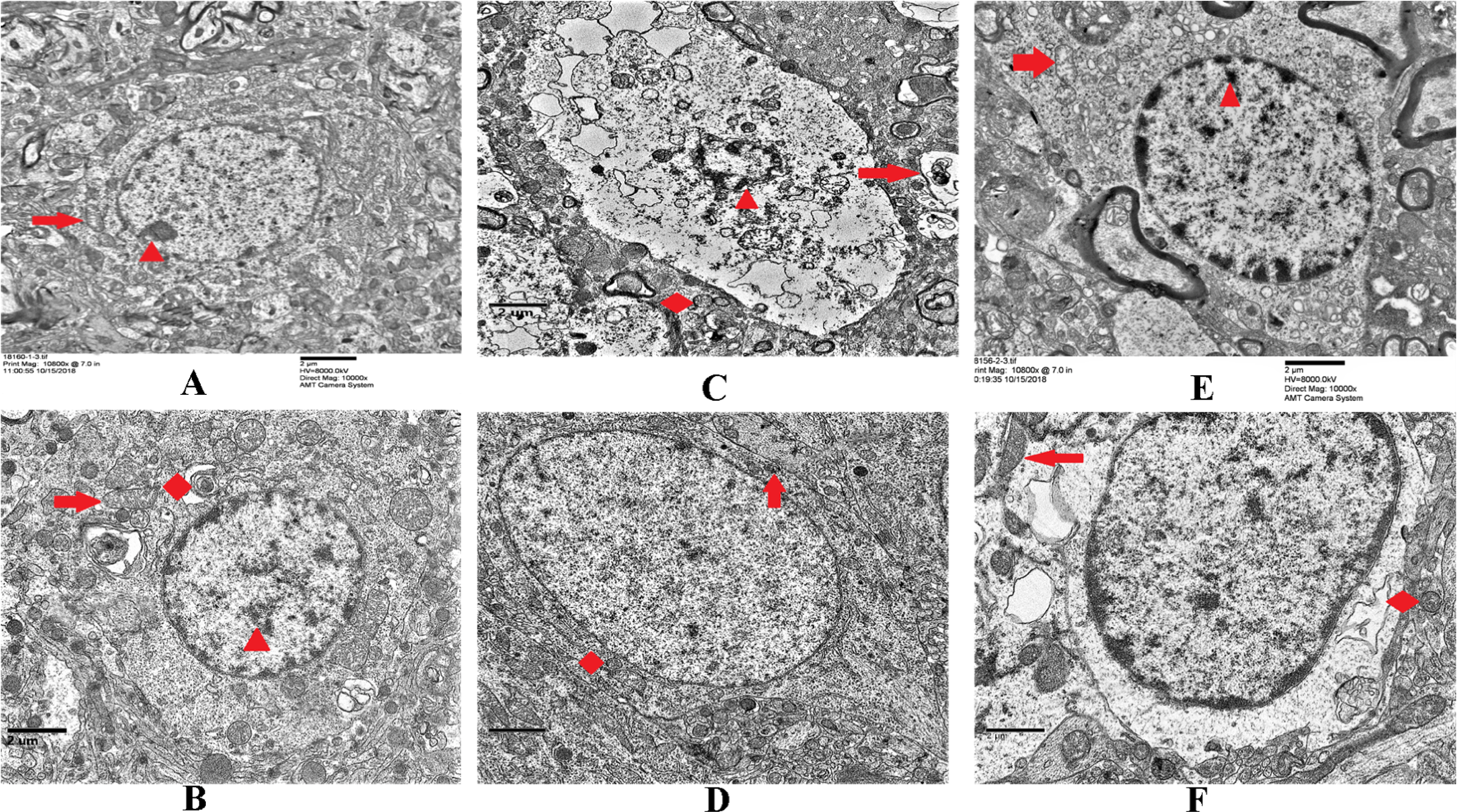
Neuron changes in each group at 3 h. (A and B) Sham group versus MCAO group. The arrow indicated that in the MCAO group, the neuron has high edema and degeneration, increased electron density in the cytoplasm, vacuolated mitochondria, increased nuclear heterochromatin, and a small number of autophagosomes in the cytoplasm. (C and D) rt-PA group versus Ast + Lig group. Compared with Ast + Lig group, there was more vacuolated cytoplasm, nuclear shrinkage, and necrosis in the rt-PA group. (E and F) Rottlerin group versus Ast + Lig + rottlerin group. Compared with the Ast + Lig + rottlerin group, the damage was significantly greater in the rottlerin group, including increased heterochromatin in the nucleus and mitochondrial swelling. Ast: Astragalus membranaceus; Lig: ligustrazine; MCAO: middle cerebral artery occlusion; rt-PA: recombinant tissue plasminogen activator.
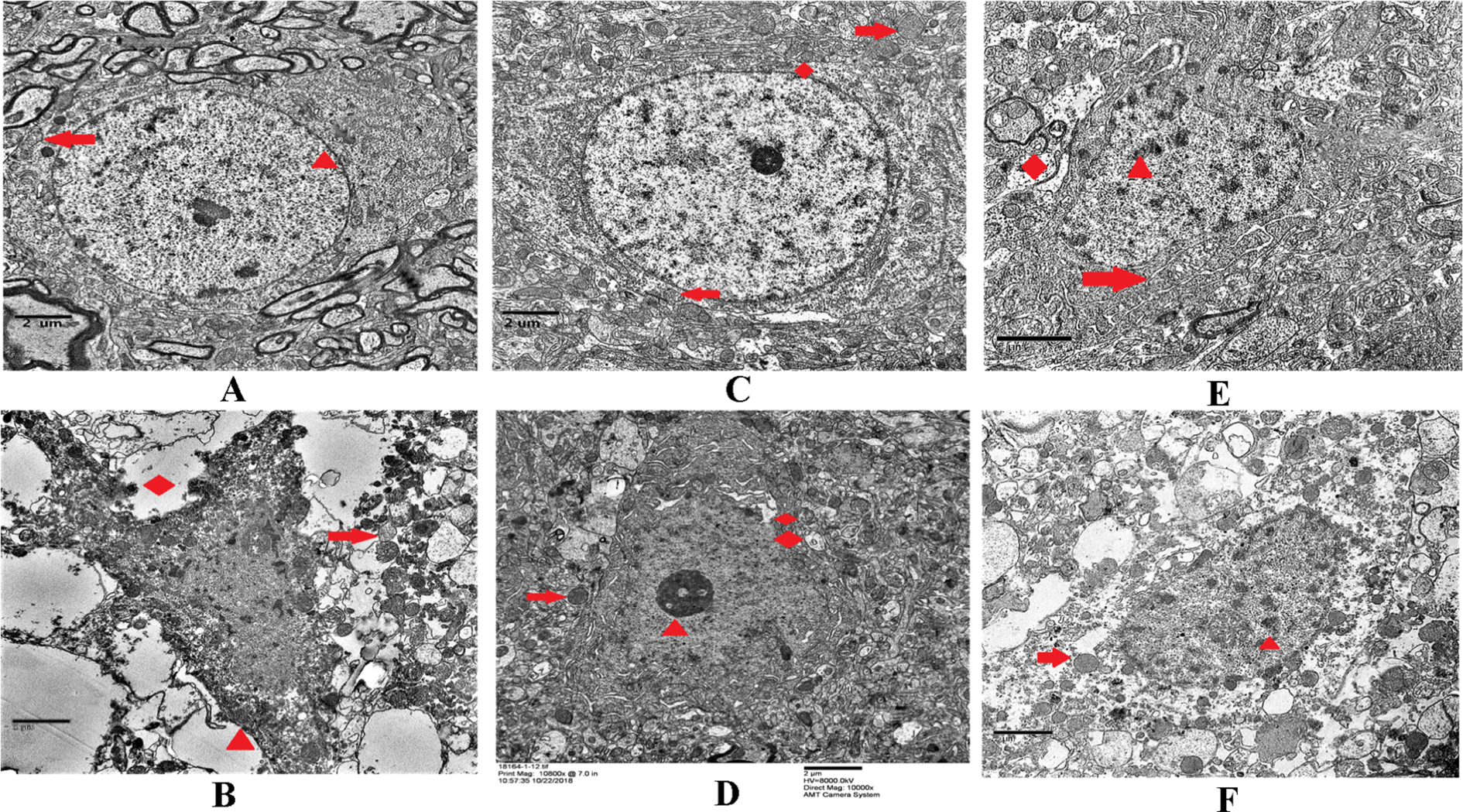
Neuron changes in each group at 24 h. (A and B) Sham group versus MCAO group. The arrow indicates that in the MCAO group, there was obvious edema, degeneration, and neuronal necrosis, as well as increased electron density in the cytoplasm, vacuolization of mitochondria, nuclear fragmentation, and a small number of autophagosomes in the cytoplasm. (C and D) rt-PA group versus Ast + Lig group. There are intact neuronal nuclei and partial neuronal necrosis and no obvious neuronal damage with the treatment of Ast + Lig. (E and F) Rottlerin group versus Ast + Lig + rottlerin group. Compared with Ast + Lig + rottlerin group, the rottlerin group showed significantly greater damage with increased heterochromatin in the nucleus, wider perinuclear space, nuclear deformation, mitochondrial swelling, and falling off into vacuoles. Scale bar = 500 nm. Ast: Astragalus membranaceus; Lig: ligustrazine; MCAO: middle cerebral artery occlusion; rt-PA: recombinant tissue plasminogen activator.

Tight junction changes of the cerebral vessel in each group at 3 h. (A and B) Sham group versus MCAO group. The arrows indicate that vascular endothelial cells are highly swollen and vacuolated, and the tight junction gap is widened in the MCAO group. (C and D) rt-PA group versus Ast + Lig group. It can be seen that the cerebral vessel wall is basically smooth and flat, and the junction was tight and dense in the Ast + Lig group. In the rt-PA group, there is vacuolization inside the vascular endothelial cells, and the junction is tight and loose. (E and F) Rottlerin group versus Ast + Lig + rottlerin group. Compared with the Ast + Lig + rottlerin group, the tight junction gap was widened, and the vascular lumen surface was irregular in the rottlerin group. Ast: Astragalus membranaceus; Lig: ligustrazine; MCAO: middle cerebral artery occlusion; rt-PA: recombinant tissue plasminogen activator.

Tight junction changes of the cerebral vessel in each group at 24 h. (A and B) Sham group versus MCAO group. The arrow indicates that the vascular endothelial cells are more swollen and vacuolated, and the tight junction gap is more widened in the MCAO group than the same group for 3 h. (C and D) rt-PA group versus Ast + Lig group. The cerebral vessel wall was basically smooth and flat, and the tight junction gap was dense in the Ast + Lig group. But in the rt-PA group, the vascular endothelial cells were swollen, and the tight junction gap was widened. (E and F) Rottlerin group versus Ast + Lig + rottlerin group. Compared with the Ast + Lig + rottlerin group, the tight junction gap became wider, and the vascular lumen surface was irregular in the rottlerin group. Scale bar = 500 nm. Ast: Astragalus membranaceus; Lig: ligustrazine; MCAO: middle cerebral artery occlusion; rt-PA: recombinant tissue plasminogen activator.
Treatment with the Drug Combination Combined with PKCδ-Inhibitor Prevents the Activation of PKC, Marcks, and MMP9 Induced by Cerebral Ischemia
Immunohistochemical results revealed that PKCδ, Marcks, and MMP9 were expressed in the brain samples in the sham group. However, these protein expressions were dramatically increased in the sections in the thrombolytic group. The protein expressions were mildly reduced in the Ast + Lig groups. The protein expressions in the inhibitor group were distinctly downregulated compared with the thrombolytic group (Figs. 7 and 8). We use Image-Pro @ Plus software to analyze the immunohistochemical staining imaging. The results of integrated option density/area were used to analyze the positive protein expression. We found that the expression levels of PKCδ, Marcks, and MMP 9 in the cerebral ischemia group were significantly higher compared with the other groups at 3 h and 24 h after thrombolysis. At 3 h after thrombolysis, MMP9 was notably decreased with Ast + Lig and rottlerin intervention (P < 0.05), and the decrease was more significant in the Ast + Lig combination group and the rottlerin group (P < 0.01). PKCδ expression also decreased when treated with Ast + Lig combined with rottlerin (P < 0.05); there was no significant decrease in the Ast + Lig group and the thrombolytic group compared with the MCAO group (P > 0.05). Marcks relative expression in the model group was increased compared with the relative expression in the sham group (P < 0.05), and no significant decrease was observed after the intervention of thrombolysis, specific inhibitor, and Ast + Lig (P > 0.05) (Fig. 9). At 24 h after thrombolysis, MMP9 was notably decreased in the Ast + Lig combination group and rottlerin + Ast + Lig group (P < 0.05), and the decrease was more significant with the intervention of Ast + Lig group and rottlerin group compared with the thrombolytic group (P < 0.05). PKC expression is higher in the thrombolytic group compared with the MCAO group (P > 0.05). PKC decreased more significantly in the Ast + Lig + rottlerin group than the pure rottlerin group (P < 0.01). Marcks relative expression in the model group was increased compared with the relative expression in the sham group (P < 0.01), and no significant decrease was observed after the intervention of thrombolysis, specific inhibitor, and in Ast + Lig groups alone (P > 0.05). Nonetheless, when combined with Ast + Lig and rottlerin, the decrease was significant (P < 0.05) (Fig. 9).

Immunohistochemical images of intervention with Ast + Lig and PKCδ. Ast: Astragalus membranaceus; Lig: ligustrazine; PKCδ: protein kinase C delta.

The positive protein expression of PKCδ, Marcks, and MMP9 around the cerebral vessels in each group. Marcks: myristoylated alanine-rich C-kinase substrate; MMP9: matrix metalloproteinase 9; PKCδ: protein kinase C delta.
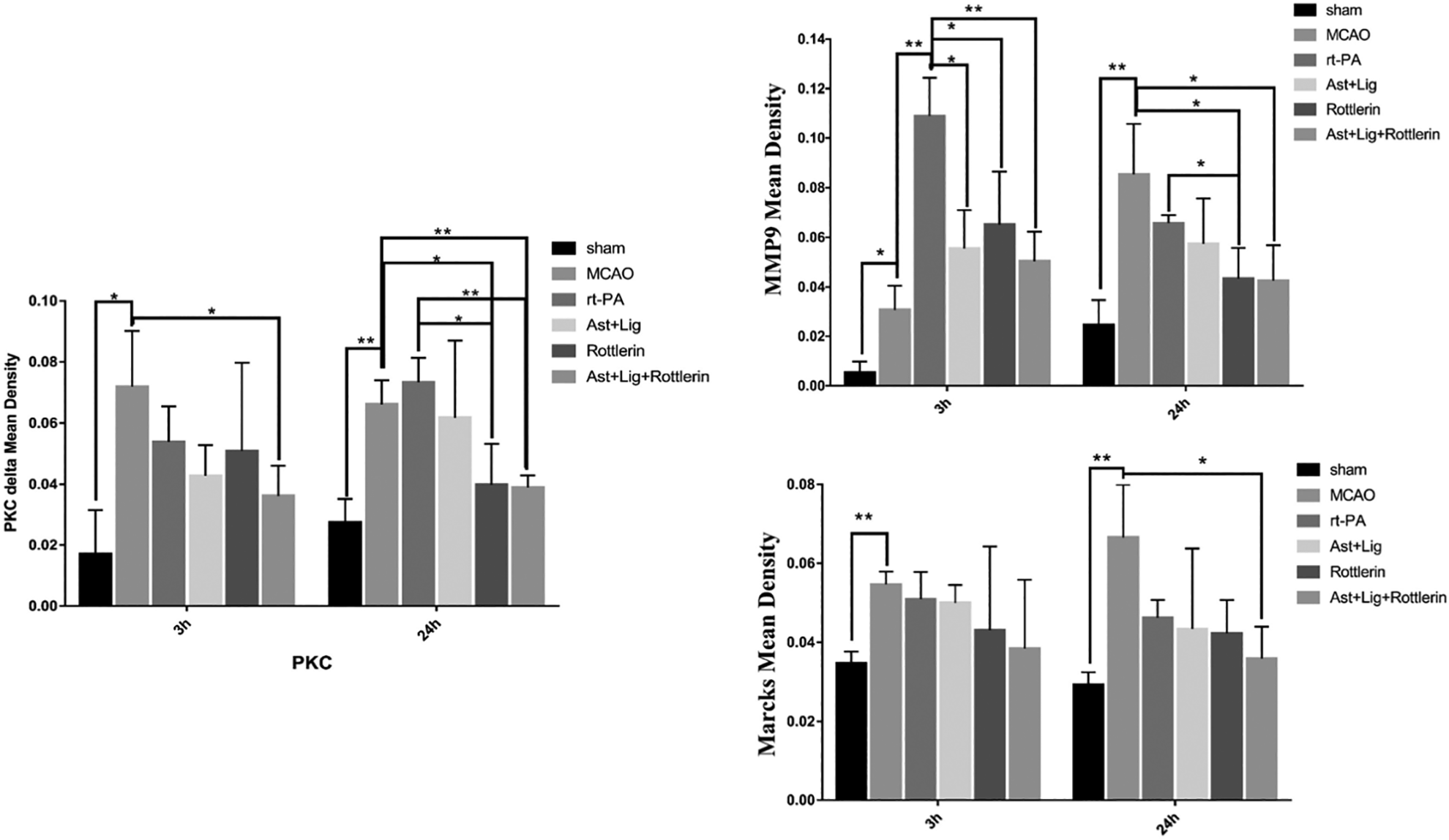
Results of each group after each statistical analysis. The treatments with Ast + Lig, inhibitor rottlerin and Ast + Lig + rottlerin all reduced expression levels of PKCδ, Marcks, and MMP9. And the decrease in Ast + Lig + rottlerin group was the most significant (**P < 0.01, *P < 0.05, n = 6 in each group). Scale bar = 50 µm. Band intensities normalized to β-actin intensity. Ast: Astragalus membranaceus; Lig: ligustrazine; Marcks: myristoylated alanine-rich C-kinase substrate; MMP9: matrix metalloproteinase 9; PKCδ: protein kinase C delta.
Western blot analyses revealed that the levels of PKCδ, Marcks, and MMP9 in the model group were significantly higher compared with the other groups not only at 3 h but also at 24 h after thrombolysis (Fig. 10). After thrombolysis, the MMP9 was notably decreased with the intervention of Ast + Lig and rottlerin (P < 0.05), and the decrease was more significant in the group with the addition of Ast + Lig compared with the rottlerin alone (P < 0.05). PKC expression also significantly decreased after the addition of Ast+ Lig and inhibitor in the rottlerin group (P < 0.05), and there was no significant decrease in the Ast + Lig and thrombolytic group compared with the MCAO group at 24 h (P > 0.05). Interestingly, at 24 h, the decrease was significant with the addition of rottlerin inhibitor (P < 0.01), and the decrease was more significant in Ast + Lig + rottlerin group than rottlerin group alone (P < 0.01). Marcks relative expression in the model group was increased compared with the sham group at 3 h and 24 h (P < 0.05) and was dramatically decreased in the Ast + Lig and Ast + Lig + rottlerin groups after thrombolysis for 3 h (P < 0.05), and no significant decrease was observed at 24 h (P > 0.05) (Fig. 11). In addition, we examined the expression of MMP9 through immunofluorescence, and the results showed that in the thrombolytic group, it was markedly increased compared with the sham group, which could be seen at 3 h and 24 h. The level of MMP9 was decreased with the addition of the combination of Ast + Lig (Fig. 12).

Western blot images. The protein expression of PKCδ, Marcks, and MMP-9 in MCAO rats after thrombolysis for 3 h/24 h. Marcks: myristoylated alanine-rich C-kinase substrate; MCAO: middle cerebral artery occlusion; MMP9: matrix metalloproteinase 9; PKCδ: protein kinase C delta.

Statistical analysis of the results of each group after 3 h/24 h of thrombolysis. The relative protein expression levels of PKCδ/Marcks and MMP9 in the model group were increased in comparison with those in sham group after thrombolysis for 3 h/24 h (P < 0.05). And all were decreased with the intervention of Ast + Lig and inhibitors after thrombolysis (P < 0.05), and the decrease was more significant in the Ast + Lig group than in the rt-PA group after thrombolysis for 3 h/24 h (P < 0.05). Compared with the intervention with rottlerin, the relative protein expression level of PKCδ was significantly decreased after the intervention with Ast + Lig after thrombolysis for 3 h (P < 0.05). Compared with the model group, there was no significant decrease both in the Ast + Lig group and rt-PA group (P > 0.05); the decrease was significant after the addition of inhibitors (P < 0.01), and the decrease was more obvious in the Ast + Lig + rottlerin group after thrombolysis for 24 h (P < 0.05). Treatments with Ast + Lig and Ast + Lig + rottlerin significantly reduce the expression of Marcks after thrombolysis for 3 h (P < 0.05). Band intensities normalized to β-actin intensity. Ast: Astragalus membranaceus; Lig: ligustrazine; Marcks: myristoylated alanine-rich C-kinase substrate; MMP9: matrix metalloproteinase 9; PKCδ: protein kinase C delta; rt-PA: recombinant tissue plasminogen activator.
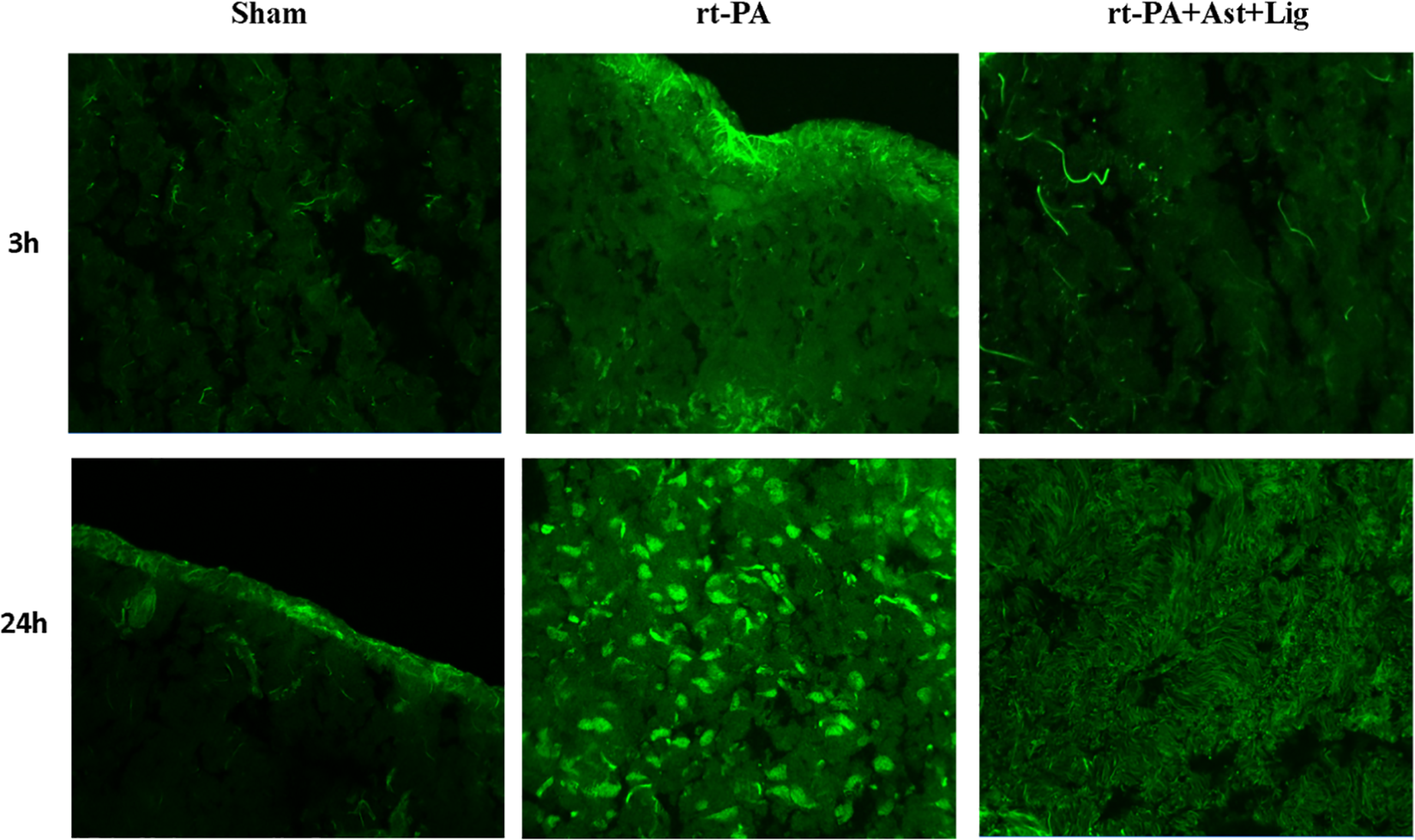
Immunofluorescence of brain tissue. Immunofluorescence diagram of brain tissue showed that the expression of MMP9 in rt-PA group was increased compared with that in the sham group, which could be seen within 3 h and was more significant at 24 h. The expression of MMP-9 in Ast + Lig group was decreased compared with that in rt-PA group (***P < 0.001, **P < 0.01, *P < 0.05, n = 6 in each group). Scale bar = 50 µm. As: Astragalus membranaceus; Lig: ligustrazine; MMP9: matrix metalloproteinase 9; rt-PA: recombinant tissue plasminogen activator.
In the model group, the mRNA expression levels of PKCδ, Marcks, and MMP9 were all increased at 3 h and 24 h after thrombolysis but were declined significantly after drug treatement (Fig. 13). At 3 h after thrombolysis, MMP9 level was significantly decreased with the intervention of rtPA, Ast + Lig and rottlerin (P < 0.01), and the decrease was more significant in the rottlerin group (P < 0.001). PKC expression also significantly decreased after the addition of Ast + Lig and inhibitor rottlerin group (P < 0.05); there was significant decrease in the Ast + Lig + rottlerin group compared with the rottlerin alone group (P < 0.001). The mRNA levels of Marcks were significantly lower in the drug-administered group compared with the MCAO group (P < 0.001), and there was lower in the group with Ast + Lig intervention (P > 0.05) (Fig. 13). At 24 h after thrombolysis, MMP9 was notably decreased in the Ast + Lig combination group and rottlerin + Ast + Lig group (P < 0.01). Importantly, the decrease was more significant with the intervention of Ast + Lig and rottlerin compared with the thrombolysis alone group (P < 0.01). PKC expression also decreased in the thrombolytic group; treatment with rottlerin significantly decreased when compared with the ischemia model group. However, no significant differences were found in the thrombolysis alone group (P > 0.05). The mRNA expression of Marcks in the rtPA group was decreased compared with the ischemia model group (P < 0.001), and significant decrease was observed after the intervention of Ast + Lig group compared with the rtPA alone group (P < 0.001). When combined with the rottlerin group solely, the decrease was significant in the combined rottlerin and Ast + Lig group (P < 0.001) (Fig. 13).

The mRNA expression levels of Marcks, PKCδ, and MMP9 in each group for 3 h/24 h. After thrombolysis for 3 h/24 h, the relative mRNA expression levels of PKCδ/Marcks and MMP9 were significantly increased in the model group and decreased in the rt-PA, Ast + Lig and inhibitor rottlerin groups (P < 0.01) not only at 3 h but also at 24 h. Compared with the rt-PA group, Ast + Lig and rottlerin showed more significant decrease of these 3 factors after thrombolysis for 24 h. After thrombolysis for 3 h, the relative mRNA expression of PKCδ/Marcks was significantly decreased after intervention with inhibitor rottlerin and Ast + Lig (P < 0.05); there was no significant decrease between Ast + Lig group and rt-PA group (P > 0.05), and the decrease occurred after the addition of inhibitors after thrombolysis for 24 h (P < 0.05). After thrombolysis for 3 h/24 h, the relative mRNA expression of MMP-9 decreased after intervention with Ast + Lig compared with the rt-PA group for 24 h (P < 0.01), and the relative mRNA expression of MMP9 significantly decreased after intervention with inhibitor rottlerin (****P < 0.0001, ***P < 0.001,**P < 0.01,*P < 0.05, n = 6 in each group). Ast: Astragalus membranaceus; Lig: ligustrazine; Marcks: myristoylated alanine-rich C-kinase substrate; MMP9: matrix metalloproteinase 9; PKCδ: protein kinase C delta; rt-PA: recombinant tissue plasminogen activator.
Discussion
Recently, the focus of drug discovery research has shifted from the originally well-accepted “one target, one drug” model to a “multitarget, multidrug” model that aims to systemically modulate multiple targets. Combination drugs that simultaneously impact multiple targets are more effective in controlling complex disease systems, such as ischemia, than drugs designed to act against individual molecular targets. 22 Both Ast and Lig are important constituents of Buyang Huanwu decoctions, which are widely used in Chinese medicinal decoctions for the treatment of cerebral ischemia. 23 –25
In our previous studies, we treated rats with cerebral ischemia with Ast with the addition of Lig and we have found that it has protective effects on the BBB integrity. The dose–effect relationships of Ast and Lig have previously been established. However, the protective mechanism of these compounds is still unclear. Therefore, we decided to assess the Ast and Lig effect in relieving ischemia-induced microhemorrhage and preserving neurological function by regulating PKCδ expression/Marcks pathway.
In the present study, we used focal embolic cerebral ischemia rat model to determine the effect of Ast and Lig combination following acute ischemic ischemia through the activation of the PKCδ/Marcks pathway. Our results showed that neurobehavioral deficits, infarct volumes, brain hemorrhage, tight junction damage, and neuronal damage were decreased in this model by rtPA, Ast + Lig, and rottlerin treatment. We detect the effect of Ast + Lig in the model after thrombolytic treatment to explore the mechanism underlying the protective effect. Further results showed that the treated rats exhibited decreased levels of PKCδ, Marcks, and MMP9. Furthermore, Ast + Lig combined with rottlerin protected the brain more effectively than the Ast + Lig or rottlerin used alone. Furthermore, Ast + Lig combined with rottlerin post-treatment remarkably reduced infarct volume and improved neurological function. These results indicate that Ast + Lig could be a potential therapy of HT after the thrombolytic treatment in a clinical setting.
Thrombolysis using rt-PA is presently a major component in therapy for acute ischemia, but restoration of the blood flow is often associated with exacerbation of tissue injury, especially in microcirculation, prone to concurrent HT. 26,27 However, traditional Chinese medicine has emerged as an interesting option for it.
Ast, the water-soluble component of A. membranaceus root (Huangqi in Chinese), has been widely used in China for the treatment of cerebrovascular conditions, such as ischemic ischemia. Lig, the monomer of ligustrazine (Rhizoma Chuanxiong in Chinese), can prevent cerebrovascular disease. 28 Many studies suggest that Ast and Lig have strong effects against neurological dysfunction, such as relieve the damage of the BBB in rats, 29 reduce cerebral edema post-ischemia/reperfusion, correlate with the suppression of MMP9, 30 anti-inflammatory and antiapoptotic in MCAO rats, 31 prevents neurotoxicity, 32 and so on. Although there are reports that Ast and Lig used alone have neurological protection effect in ischemia model rats, there is no report on the effect of Ast and Lig protecting against MMP9 activity in MCAO rats through the suppression of PKCδ/Marcks pathway. However, PKCδ/Marcks pathway has been found to play a crucial role in ischemic ischemia. 33 –35 PKCδ has been implicated in mediating cerebral ischemic and reperfusion damage. 36 Thrombolytics are widely used to intervene in acute ischemic ischemia, but re-establishment of circulation may paradoxically initiate a HT. There is study with mice lacking PKCδ showing that the absence of this enzyme markedly reduces HT following transient ischemia. 37 In addition, PKCδ is involved in cortical cerebral circulation by modulating postischemic cerebral blood flow by attenuating hyperemia and hypoperfusion. 38 Marcks, as a major PKC substrate, exists in high concentrations in neurons and has been implicated in cytoskeletal rearrangement, membrane trafficking, and neurotransmitter release. 39
The functioning of the brain depends on the integrity and normal functioning of the brain vessels. MMP9 is the most important transforming substance of hemorrhage, which is closely related to cerebrovascular diseases. 40,41 In humans, investigations with peripheral blood samples have shown high MMP9 levels in patients with ischemic ischemia and intracranial hemorrhage. More importantly, MMP9 levels are related to poor neurological outcomes, suggesting a deleterious role in human brain damage. 42 Activation of MMP9 can lead to neuronal damage such as BBB damage and vasogenic cerebral edema. 43 The level of MMP9 in vivo may determine whether thrombolytic treatment is effective and whether serious hemorrhagic complications occur.
Therefore, in the current study, expression levels of PKCδ, Marcks, and MMP9 in cerebral ischemia injuries were compared, and possible mechanisms of the brain-protective effect of Ast and Lig were suggested. Additionally, we wanted to investigate whether the combined therapy strategy can compensate for their own disadvantages for the treatment of ischemia or have more significant effects as compared with the drugs used alone and as combination in the Ast + Lig + rt-PA and Ast + Lig + rottlerin + rt-PA groups.
The protein and mRNA expression levels of PKCδ, Marcks, and MMP9 were activated in the middle cerebral artery embolization model of autologous thrombus in rats, and the expression was more obvious with time (the expression was higher in 24 h group). After thrombolytic intervention, the expression was decreased but not significantly. The treatment with Ast + Lig combination decreased significantly. On the basis of this, combination with the PKCδ inhibitor rottlerin showed obvious downregulation, which is more effective than the treatment with Ast + Lig or rottlerin alone group. Moreover, when thrombolytic treatment was used in rt-PA, the amount of cerebral hemorrhage was significantly increased, the destruction of cerebral tight connections was obvious, and the neuronal necrosis was also more serious. However, all these injuries were reduced in the Ast + Lig group, and with the combination of the PKCδ inhibitor rottlerin, the effect became better. Therefore, rt-PA thrombolytic treatment can increase the risk of HT and become more severe with time.
The Ast and Lig combination has an obvious effect in reducing the early cerebral hemorrhage after thrombolysis, and it is beneficial to the overall condition and neurological function recovery of rats after thrombolysis, and this effect becomes more prevalent in combination with PKCδ inhibitor rottlerin.
Although the detailed molecular mechanism underlying thrombolysis-induced HT interacting with PKCδ/Marcks and whether other molecules are also involved in PKCδ/Marcks pathway regulating MMP9 remains unknown and need to be explored in the future studies, our findings highlight that PKCδ/Marcks involved in HT by positively regulating MMP9 is highly important. In addition, we have not performed in vitro experiment.
In conclusion, the mechanism of Ast and Lig combination in preventing HT after thrombolysis in acute cerebral ischemia is potentially due to Ast and Lig can block the activation of PKCδ/Marcks pathway, inhibit the activity of MMP9, protect the integrity of tight connection of cerebral vessel, and neuronal dysfunction.
Footnotes
Acknowledgments
We thank Dr Dimas Primacahayadi for the language modification of our article.
Authors Contribution
RH-P, XL-T, KH, HJ-W, and YH performed the animal experiments and analyzed the data. RH-P, XL-T, KH, HJ-W, and YH discussed the results and contributed to the manuscript preparation. RH-P, XL-T, KH, and YH wrote the manuscript. RH-P, XL-T, and YH designed and supervised the project. All authors have read and approved the final manuscript.
Consent for Publication
All authors agree to publish.
Ethical Approval
Ethical approval to report this case series was obtained from Experimental Animal Ethics Committee of Guangdong Provincial Hospital of Chinese Medicine (Approval No. 2018033).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Experimental Animal Ethics Committee of Guangdong Provincial Hospital of Chinese Medicine (Approval No. 2018033) approved protocols.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study is supported by the National Natural Science Foundation of China (No. 81804038 to RP, No. 81873262 to YH), Guangdong Provincial Key Laboratory of Research on Emergency in Traditional Chinese Medicine (No.2017B030314176 to YH), the Project of Science and Technology Bureau of Guangdong Province (No. 2016A020226030 to RP), and the Special Research Foundation of Science and Technology for Traditional Chinese Medicine from Guangdong Hospital of Chinese Medicine (No. YN2016QJ17 to RP).
