Abstract
This study investigates retrospectively, in selected patients, the ischemic outcome (reversible ischemia, infarction, and hemorrhage) and neurologic outcome of acute stroke treated with intra-arterial thrombolysis and the predictive value of pretreatment single-photon emission-computed tomography (SPECT). Thirty patients with complete recanalization within 12 hours were analyzed. The extent of ischemia was outlined on SPECT, and two CBF parameters were calculated: the ratio of ischemic regional activity to CBF in the cerebellum and the asymmetry index. Reversible ischemia, infarction, and hemorrhage were identified by comparing SPECT and follow-up computed tomography. Nine patients (30%) had no or small infarction, 14 (47%) had medium or large infarction, and seven (23%) had hemorrhage. Forty-two lesions were identified (22 reversible ischemia, 13 infarction, and 7 hemorrhage). Duration of ischemia, urokinase dose, disease type, and occlusion site were nonsignificant factors, whereas neurologic outcome and CBF parameters were significant among the three patient groups and three types of ischemic lesions. Ischemic tissue with CBF greater than 55% of cerebellar flow still may be salvageable, even with treatment initiated 6 hours after onset of symptoms. Ischemic tissue with CBF greater than 35% of cerebellar flow still may be salvageable with early treatment (less than 5 hours). Ischemic tissue with with CBF less than 35% of cerebellar flow may be at risk for hemorrhage within the critical time window. Pretreatment SPECT can provide useful parameters to increase the efficacy of thrombolysis by reducing hemorrhagic complications and improving neurologic outcome.
Keywords
Intra-arterial thrombolytic therapy has been an effective alternative to restore CBF, reduce ischemic injury, and improve neurologic outcome in patients with acute ischemic stroke (del Zoppo et al., 1988; Mori et al., 1988; Hacke et al., 1988; Theron et al., 1989; Ezura and Kagawa, 1992; Zeumer et al., 1993; Ueda et al., 1994; Jansen et al., 1995; Sasaki et al., 1995). However, thrombolytic therapy of acute ischemic stroke is associated with significant morbidity and mortalities. The success and complication rate vary among different institutions. Many factors that may influence the ischemic outcome (reversible ischemia, infarction, and hemorrhage) and neurologic outcome (National Institutes of Health [NIH] stroke scores and clinical findings) have been proposed. These include patient characteristics, nature and duration of ischemia, technical variation, and clinical relevance. No firm data prove or disprove the existence of other factors or the degree of their influence on outcomes. There is no established protocol in dealing with intra-arterial thrombolytic therapy, particularly the guidelines for proper patient selection.
Single-photon emission-computed tomography (SPECT) as a noninvasive brain perfusion imaging modality is expected to have potential value in the assessment of tissue reversibility before treatment (Giubilei et al., 1990; Hanson et al., 1993; Baird et al., 1994). Although Alexandrov and colleagues (1997b) suggest the need for the clinical value of SPECT applicable to patient selection for thrombolysis, pretreatment assessment of parameters reflecting the perfusion status of the ischemic tissue has not been part of treatment protocol in the past. Our previous report indicates that the risk of hemorrhagic transformation in acute stroke patients treated by intra-arterial thrombolytic therapy depended on residual CBF of ischemic tissue judged by pretreatment SPECT (Ueda et al., 1994). Finally, complete recanalization was not achieved in all of the patients studied in prior reports, which suggests that technical factors may influence outcomes. This study investigates the ischemic and neurologic outcomes of acute stroke patients with early and complete recanalization using intra-arterial thrombolytic therapy and assesses the value of pretreatment SPECT in the prediction of outcomes. Specifically, we tested the hypothesis that the parameters provided by pretreatment SPECT can be helpful in patient selection and improving outcomes.
MATERIALS AND METHODS
Eighty-five selected patients treated in our institution between April 1989 and March 1996 using intra-arterial thrombolytic therapy for acute ischemic stroke were evaluated retrospectively. Of the 85 patients, 45 were studied with SPECT using technetium-99m-labeled hexamethylpropyleneamine oxime (99mTc-HMPAO) before the treatment. Thirty of these 45 patients who also had complete recanalization less than 12 hours after the onset of symptoms were included in the current study. These patients included 16 men and 14 women between 35 and 83 years of age (mean ± SD, 67 ± 10 years). The duration of ischemia is the time after onset rounded to the nearest hour.
The inclusion criteria for this study were as follows: (1) no apparent hypodense areas were observed on the admission computed tomographic (CT) scan; (2) it was preferred that the patient was treated within 6 hours (87%) but no later than 12 hours; (3) occluded arteries suggested by symptoms were demonstrated by cerebral angiography; and (4) informed consent was obtained from the patient or relatives. The exclusion criteria were as follows: (1) recent hemorrhagic stroke; (2) critical systemic condition; (3) serious disabling disease; (4) classic contraindications to thrombolytic therapy (recent surgical operation, gastrointestinal bleeding, or known bleeding tendency).
The CT was performed on admission, immediately after therapy, the following day, and 1 week, 2 weeks, and 1 month after therapy. When no clear hypodense areas were observed at the sites suggested by clinical symptoms, HMPAO-SPECT was performed using a rotational gamma camera (GCA6O2A, Toshiba, Tokyo, Japan) or a four-head gamma camera (SPECT2000H-40, Hitachi, Tokyo, Japan) with a low-energy high-resolution collimator. The patients were injected with 30 mCi of 99mTc-HMPAO. About 5 minutes later, emission projection data were acquired in 60 views over 360° for 12 seconds per view using a 64 × 64 matrix in the former camera and in 64 views over 360° for 20 seconds per view using a 64 × 64 matrix in the latter one. The pixel size was 4.0 × 4.0 mm. The slice thicknesses were 7 mm in the former camera and 5 mm in the latter one. The total time required to perform the SPECT (12 minutes) and measure the parameters (5 minutes) was less than 25 minutes. In patients with internal carotid artery territory occlusion, the residual CBF was evaluated. Of 16 axial sections, the one section most clearly demonstrating the ischemic region was selected. The regions of interest in the ischemic area in the cerebral hemisphere (a), the corresponding region on the contralateral side (b), and the whole cerebellar hemisphere on the ischemic side (c) were set, and the mean count was determined in each region of interest. Linearity adjustment was made by assuming the blood flow in the normal cerebellar hemisphere to be 55 mL/100 g/min (Shinohara et al., 1985) according to the method of Lassen and associates (1988). The CBF was assessed semiquantitatively by calculating two parameters: (1) the ratio of ischemic regional activity (R) to cerebellar activity (CE) (R/CE ratio = a/c), and (2) the asymmetry index (AI = 1 +[b − a]/[a + b]). The regions of interest were hand drawn in apparent whole ischemic areas based on qualitative inspection of the regional CBF maps. The contralateral, mirror image region of interest was drawn automatically. The regions of interest in cerebellum were hand drawn in whole hemisphere on the ischemic side to avoid influence by crossed cerebellar diaschisis. First, the residual CBF was evaluated in the whole ischemic region for each patient. Second, to evaluate injury thresholds, the regions of interest in patients who had apparent cortical infarction on the follow-up computed tomography (CT) scans were retrospectively determined separately in the infarcted region and noninfarcted region.
Arterial digital subtraction angiography was performed by the Seldinger method using a 5-French catheter using a 6-French femoral sheath. The tip of a Tracker-18 catheter (Target Therapeutics, Fremont, CA, U.S.A.) was advanced into the thrombus or upstream from the site of occlusion over a 0.356-cm (0.014-inch) Taper Dasher guide wire (Target Therapeutics). Urokinase (240,000 U) was dissolved in 20 mL of physiologic saline and was injected manually for about 10 minutes. The Tracker catheter was moved frequently through the occluded segment to disrupt thrombus mechanically. Angiography was performed by injection of contrast material through the Tracker catheter immediately after each infusion. This was repeated until recanalization of the occluded vessel was confirmed. The maximum dose of urokinase was 960,000 U. The patients were managed initially by intravenous injection of heparin (5000 U) and by administration of 10% glycerol (200 mL) and micromolecular dextran (500 mL) during the therapy. Cerebral angiography also was performed on the day after thrombolysis in most patients because the sheath catheter in the femoral artery usually was kept until the next day. No reocclusion were seen in second angiography. Ticlopidine (200 mg/d) administration was started the day after treatment when no intracranial hemorrhage or systemic bleeding tendency was observed.
The patient's neurologic status was evaluated on admission, the following day, and 1 week and 1 month after treatment according to the NIH Stroke Scale (Brott et al., 1989), which expresses the severity of neurologic status numerically from 0 (normal) to 42. An NIH score of 42 points was applied to patients who had died within 1 month of treatment. The outcome was evaluated 1 month after onset according to the following five-grade outcome scale:
Excellent: No neurologic deficits were observed, and the patient had returned fully to previous activities. Good: Mild neurologic deficits remained, but the patient had returned partly to previous activities. Fair: Rehabilitation was difficult, but no assistance was needed in activities of daily life. Poor: Assistance was needed in activities of daily life. Death.
Acute ischemic stroke was classified into cardioembolic infarction and atherothrombotic infarction according to the guidelines of the Cerebral Embolism Task Force (1986) based on the onset pattern, angiographic findings, and results of cardiovascular examinations such as EEG, echocardiography, and Holter electrocardiography.
For statistical analysis, the values were expressed as the mean ± SD, and differences among the three groups were examined by Scheffé's F test.
RESULTS
Patient classification and outcome
Based on the follow-up CT, the patients were classified into three groups: A, 9 patients with no evidence of infarction, small subcortical infarctions, or spotty basal ganglial infarctions (Fig. 1); B, 14 patients with medium-sized or large cortical infarctions (Fig. 2); and C, 7 patients with hemorrhagic transformation (Fig. 3).
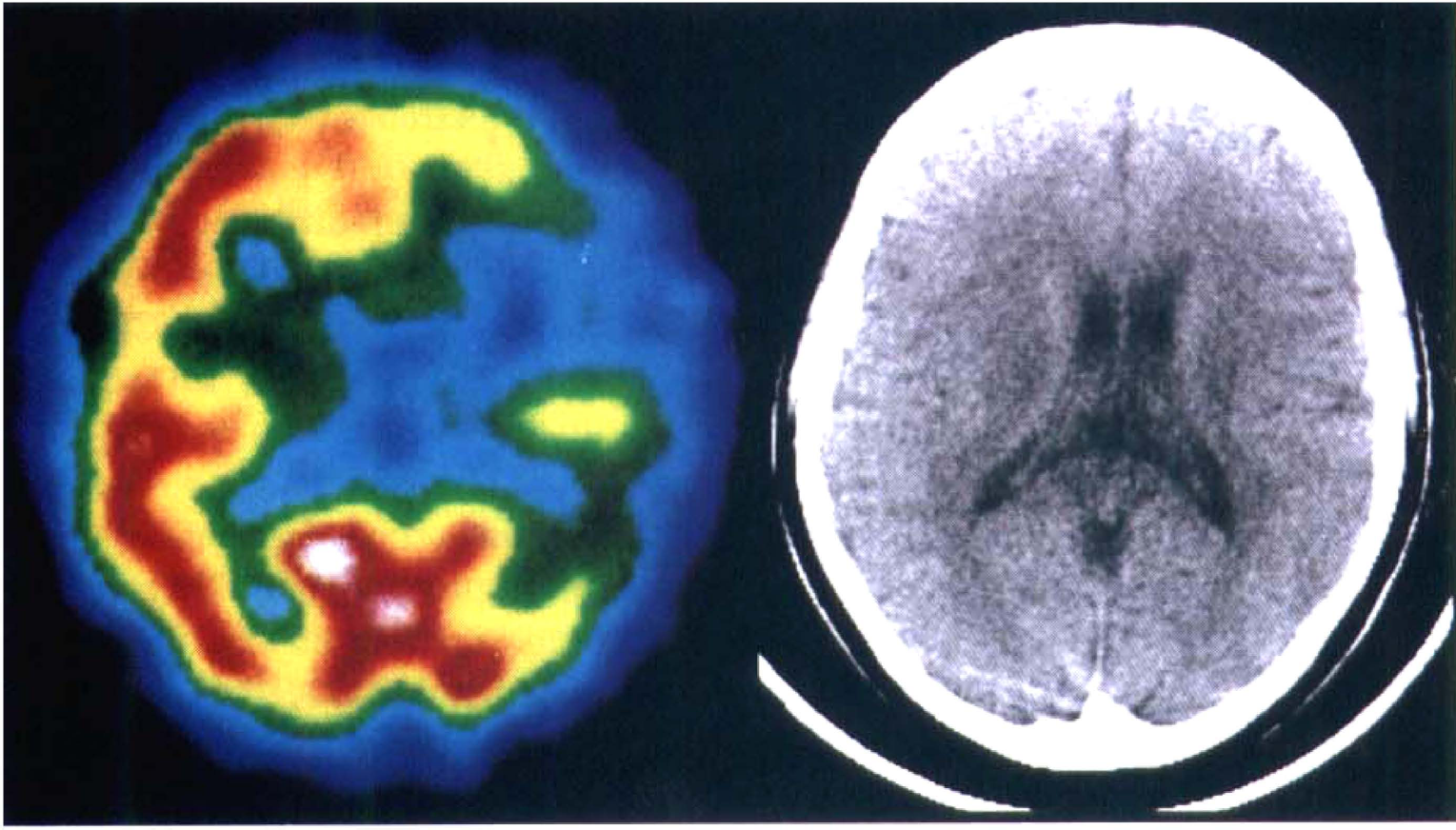
Pretreatment single-photon emission-computed tomography (SPECT) and posttreatment computed tomography (CT) scan in a group A patient who had complete recanalization of the left middle cerebral artery occlusion 4 hours after onset of symptoms. Left: SPECT shows left frontotemporoparietal perfusion deficit. The ratio of ischemic region to cerebellar flow was 0.51, and the asymmetry index was 1.35. Right: A CT scan 1 month later shows no apparent infarction.
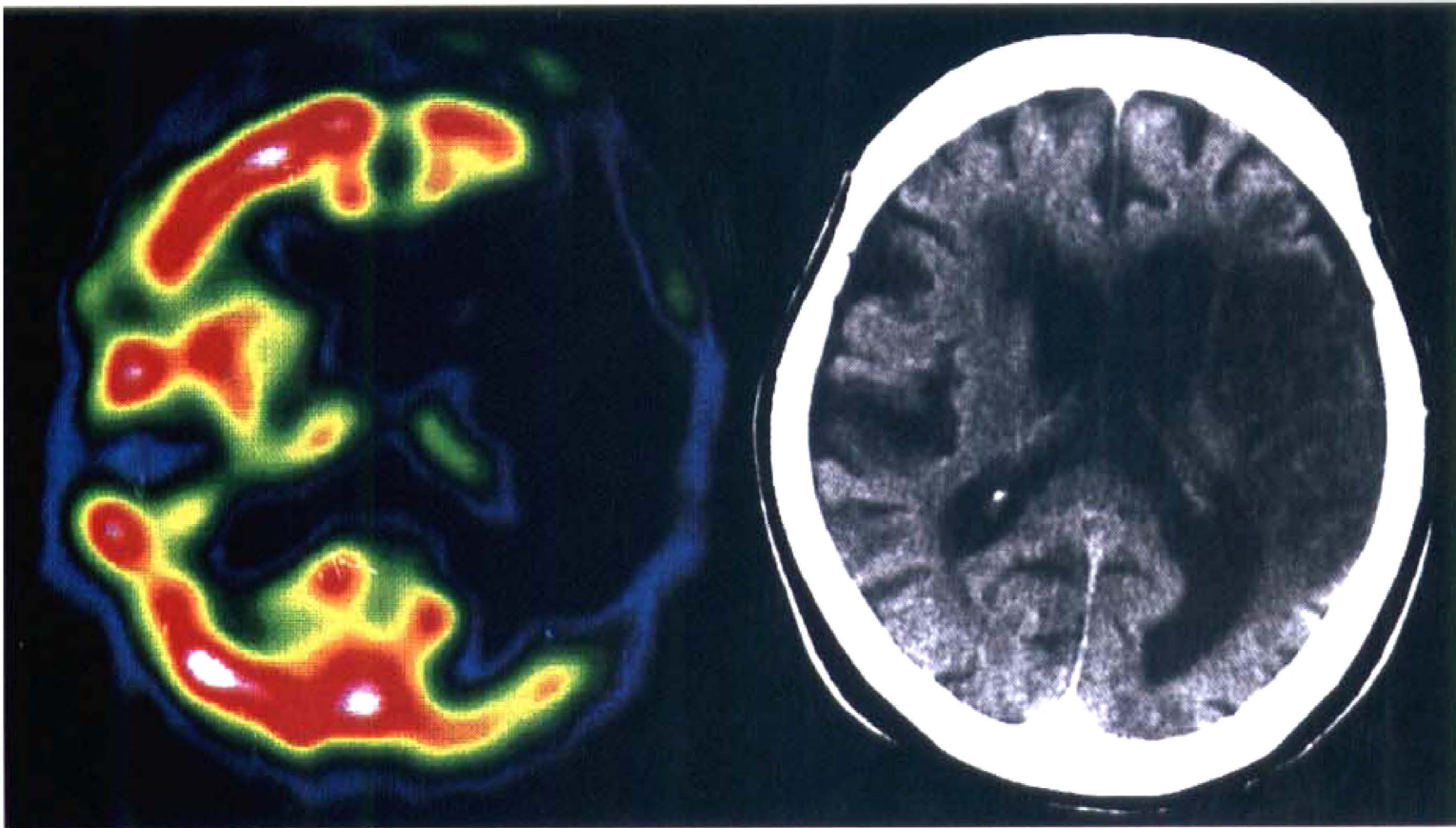
Pretreatment SPECT and posttreatment CT scan in a group B patient who had complete recanalization of the left middle cerebral artery occlusion 6 hours after onset of symptoms. Left: SPECT shows left frontotemporoparietal perfusion deficit. The ratio of ischemic region to cerebellar flow was 0.45, and the asymmetry index was 1.25. Right: A CT scan 1 month later shows a large infarction in the territory of the left middle cerebral artery.
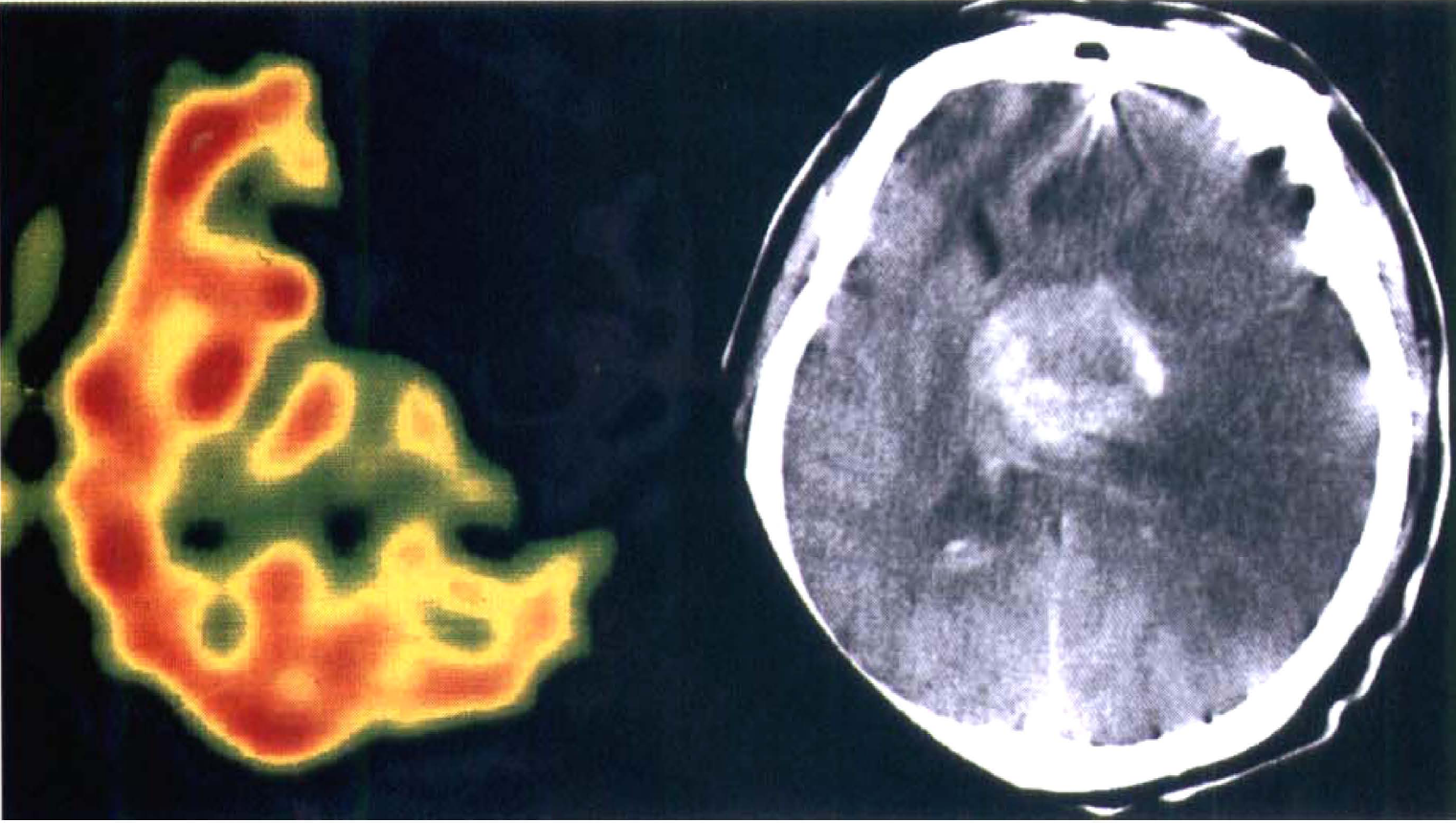
Pretreatment SPECT and posttreatment CT scan in a group C patient who had near complete recanalization of the left internal cerebral artery occlusion 3 hours after onset of symptoms. Left: SPECT shows left frontotemporoparietal perfusion deficit. The ratio of ischemic region to cerebellar flow was 0.12, and the asymmetry index was 1.71. Right: A CT scan obtained the day after treatment shows hemorrhagic transformation in the left basal ganglia with massive left hemispheric edema.
Clinical characteristics, treatment protocol, SPECT findings, and outcomes are summarized in Table 1. Neurologic outcome judged by the NIH score and clinical outcome judged by our five-grade outcome scale were significantly different among all three patients groups (P < 0.01). However, the duration of ischemia, disease type, sex, site of occlusion, and urokinase dose were not significantly different among these three patients groups. Duration of ischemia (less than 5 hours) became significant only in patients with severe ischemia (R/CE ratio < 0.55). SPECT findings can differentiate the hemorrhagic group from both the reversible ischemia and infarction groups, but the difference between the reversible ischemia and infarction groups for the ischemic duration between 2 and 12 hours was not statistically significant. When patients are separated into two subgroups using the 5-hour duration of ischemia as the cutoff, SPECT findings can readily differentiate all three groups. Hemorrhagic transformation detected on CT scan immediately after treatment or on the following day occurred in seven patients. Neurologic deterioration was observed in the following five patients: two with massive parenchymal hematomas, two with hematomas and extensive brain edema, and one with a medium hematoma in the basal ganglia. Hemorrhagic transformation significantly depended on residual CBF, age, and pretreatment NIH scores, even in patients with early and successful recanalization.
Patient data, treatment protocol, SPECT findings, and outcomes
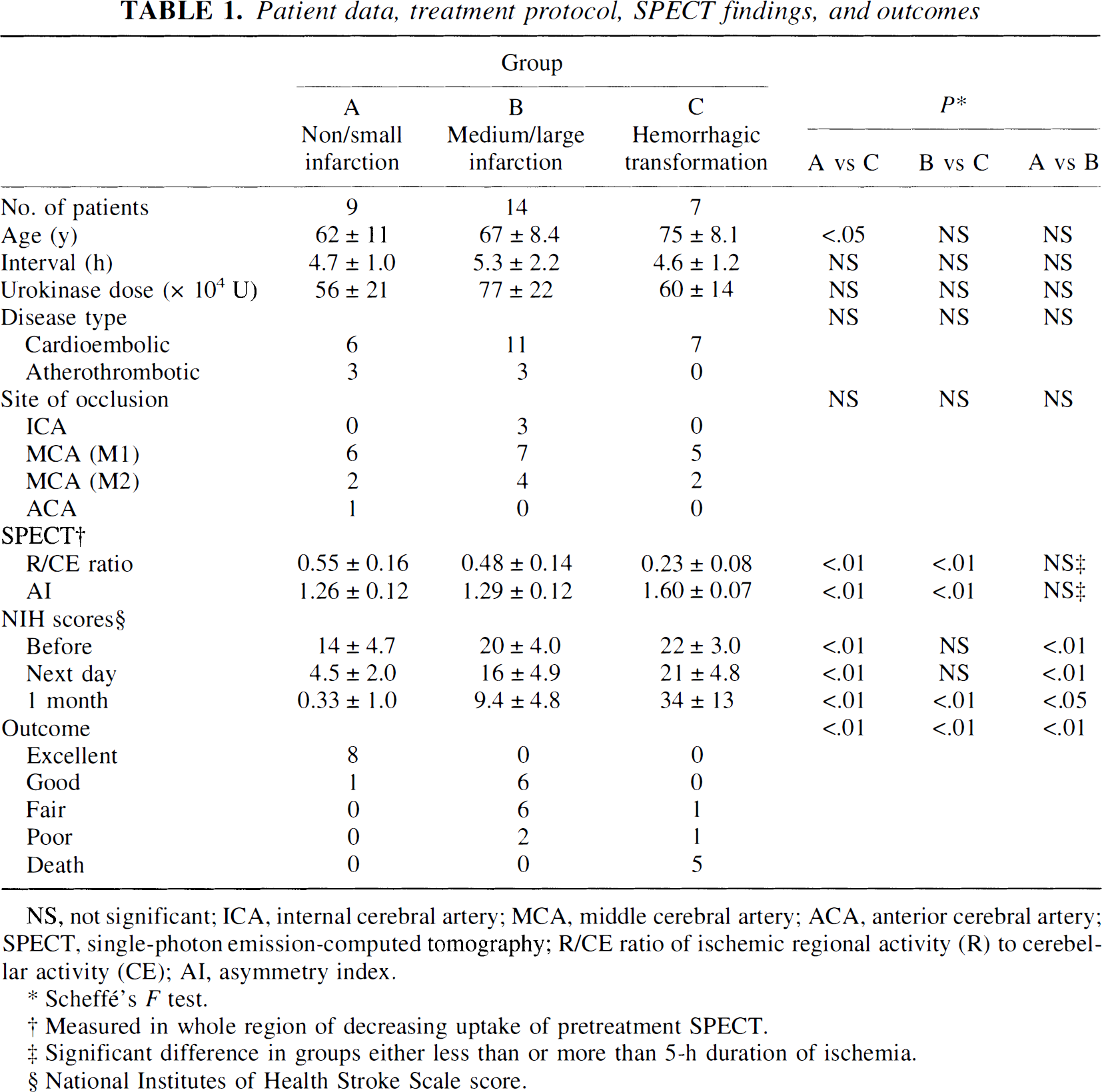
NS, not significant; ICA, internal cerebral artery; MCA, middle cerebral artery; ACA, anterior cerebral artery; SPECT, single-photon emission-computed tomography; R/CE ratio of ischemic regional activity (R) to cerebellar activity (CE); AI, asymmetry index.
Scheffé's F test.
Measured in whole region of decreasing uptake of pretreatment SPECT.
Significant difference in groups either less than or more than 5-h duration of ischemia.
National Institutes of Health Stroke Scale score.
Ischemic lesion classification and cerebral blood flow thresholds for ischemic injury
By comparing the ischemic lesions judged by pretreatment SPECT with noninfarcted, infarcted, and hemorrhagic regions judged by follow-up CT, 42 lesions (22 noninfarcted, 13 infarcted, and 7 hemorrhagic lesions) were identified in the 30 patients evaluated (Figs. 4 and 5).
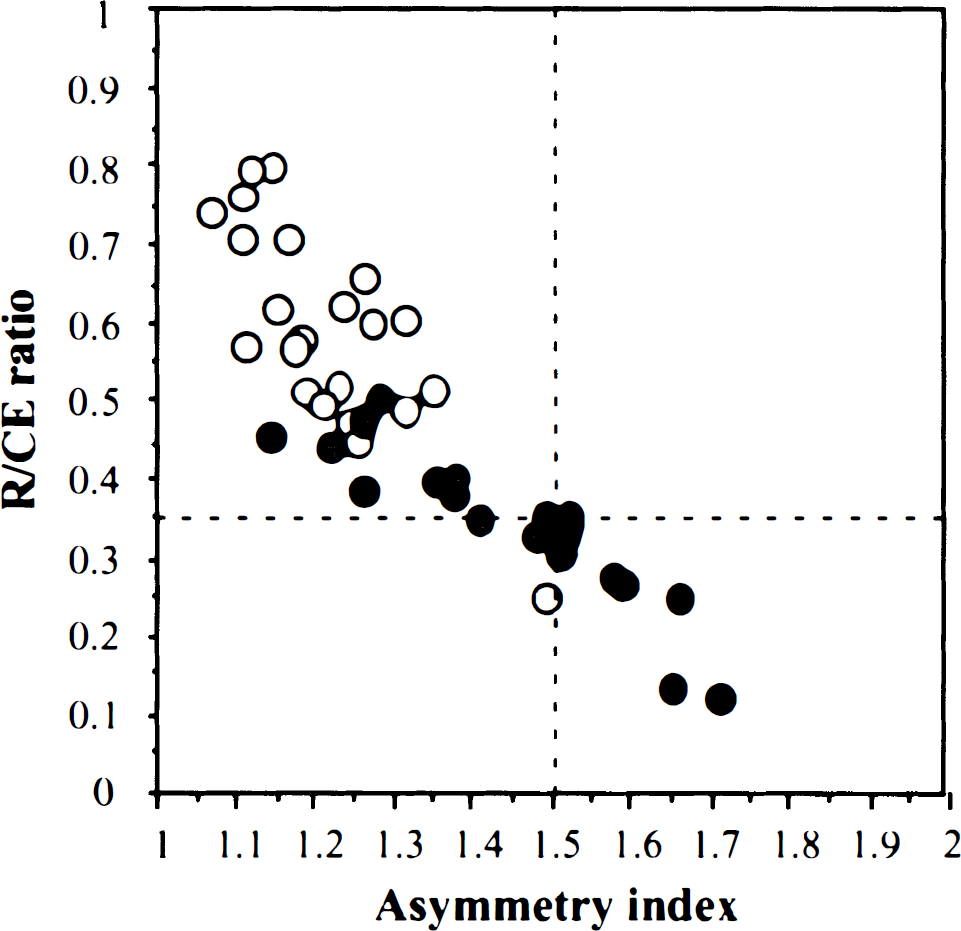
Outcome versus severity of ischema. Even with successful early recanalization using intra-arterial thrombolysis, outcome in acute ischemia differs among individuals and depends on primary pretreatment residual CBF. As judged by SPECT, there is significantly different pretreatment CBF between infarcted and hemorrhagic areas (P < 0.01) as well as noninfarcted and hemorrhagic areas (P < 0.01). ○ Non-infarction. ∘ Infarction. • Hemorrhage.
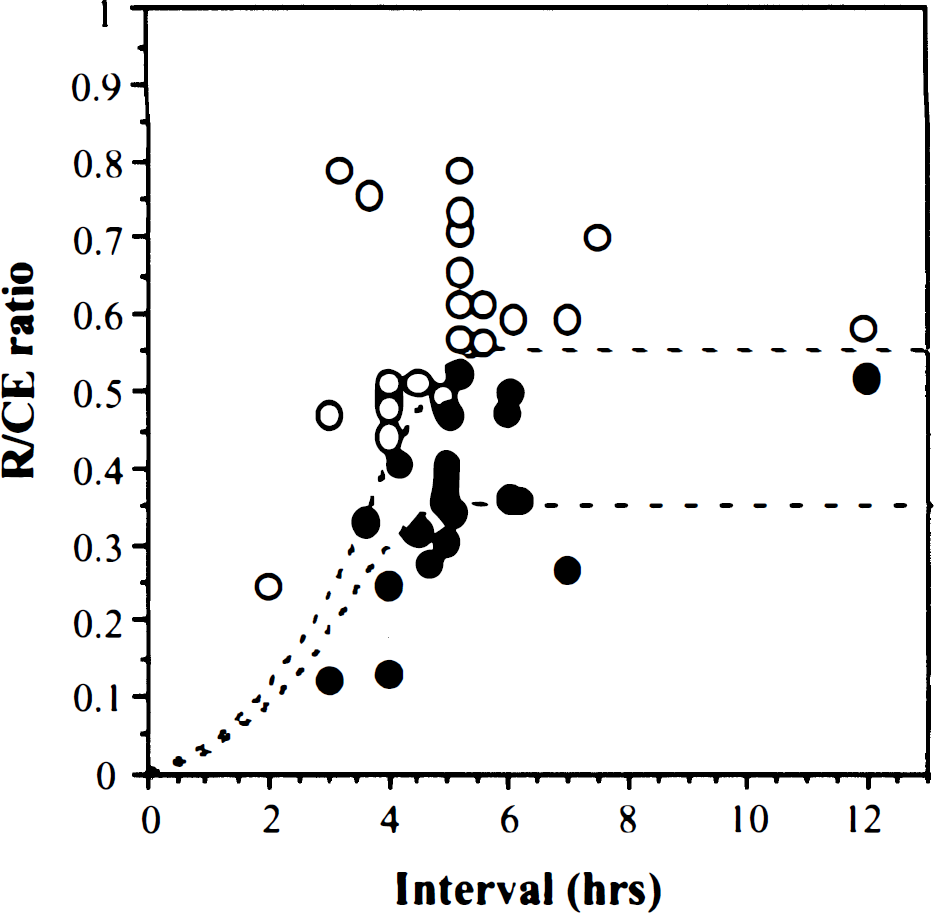
Outcome versus severity and duration of ischemia. A total of 42 lesions from 30 patients were selected. In patients with relatively high pretreatment CBF (ratio more than 0.55), the chance of recovery is excellent with a long therapeutic window (up to 12 hours) and rare incidence of hemorrhage. However, not all patients with low pretreatment CBF (CBF in the ischemic zone greater than 55% of cerebellar CBF) had a bad outcome if early recanalization could be achieved. ○ Non-infarction. ∘ Infarction. • Hemorrhage.
The average R/CE ratios in the noninfarcted, infarcted, and hemorrhagic lesions were 0.62 ± 0.15, 0.41 ± 0.06, and 0.23 ± 0.08, respectively (Fig. 4). The average AI values in the noninfarcted, infarcted, and hemorrhagic regions were 1.20 ± 0.12, 1.38 ± 0.13, and 1.60 ± 0.07, respectively. There was a significant difference in R/CE ratio and AI between each region (P < 0.01). In all seven patients with hemorrhagic transformation, the R/CE ratio and AI were less than 0.35 and more than 1.5, respectively.
Within 5 hours after onset of symptoms, ischemic outcome (reversible ischemia, infarction, and hemorrhage) appeared to depend on both pretreatment CBF and the duration of ischemia. After 5 hours from onset of symptoms, the ischemic outcome was influenced by only pretreatment CBF. Furthermore, the thresholds for the development of infarction or hemorrhage were likely to be about 0.55 and about 0.35 of R/CE ratio, respectively (Fig. 5).
These thresholds provide important information that can be potentially useful in the management of acute stroke patients with intra-arterial thrombolysis: (1) ischemic tissue with a flow index greater than 0.55 still may be salvageable, even when treatment is initiated 6 hours after onset of symptoms; (2) ischemic tissue with a flow index greater than 0.35 still may be salvageable with early treatment (less than 5 hours); and (3) ischemic tissue with a flow index below 0.35 may be at risk for hemorrhage, even when treatment is started within the critical time window.
DISCUSSION
Human and animal studies (Jones et al., 1981; Furlan, et al., 1996) indicate that each minute of ischemia is harmful, and the sooner the treatment of ischemic stroke is begun, the better are the chances to minimize the ischemic damage. Emergency treatment has been focused on the first 6 hours after the onset of ischemic stroke symptoms. It is generally believed that the 6-hour period provides a therapeutic window of opportunity during which medical intervention can be initiated to limit brain damage. Within this therapeutic window, it is presumed that most ischemic tissue remains viable and, theoretically, can be revived. The purpose in choosing a therapeutic window for early intervention has two distinctive goals: (1) to improve the ischemic outcome by rescuing the reversible ischemic tissue, and (2) to avoid hemorrhagic complications caused by reperfusing irreversibly damaged ischemic tissue. Improving the ischemic outcome by reducing the reperfusion injury can achieve a better neurologic outcome.
In recent studies using either intravenous or intra-arterial thrombolytic therapy, not all of the patients had excellent ischemic outcomes, with significant hemorrhagic complications occurring even in those treated within the therapeutic window. Analysis of the data from 261 patients in nine studies (del Zoppo et al., 1988; Mori et al., 1988; Hacke et al., 1988; Theron et al., 1989; Ezura and Kagawa, 1992; Zeumer et al., 1993; Ueda et al., 1994; Jansen et al., 1995; Sasaki et al., 1995) using intra-arterial thrombolytic therapy with 59% recanalization rate ranging from within 6 to within 24 hours after the onset of symptoms shows that neurologic improvement was achieved in only 42% of patients, but hemorrhage occurred in 21%. Interestingly, patients with earlier treatment (less than 6 hours) did not always have a better neurologic outcome and less hemorrhagic complications compared with those with late treatment (less than 24 hours). The inconsistent results among these studies were expected because many potential problems existed in their design and performance. Therefore, there is an urgent need to design a clinical trial with strict criteria and standardized protocols to compensate for variables that influence outcome (Ferguson and Ferguson, 1994; del Zoppo et al., 1994; Adams et al., 1996). Many issues were raised about the way previous studies were designed, including patient selection, classification of ischemic injury, vascular territories, drugs involved and their dosages, and duration and route of administration of thrombolytic agents.
Our preliminary data from a limited number of patients indicate that some of these issues may not always appear to have as much influence on neurologic outcome as we thought in patients with intra-arterial thrombolytic therapy (Table 1). These issues include the duration of ischemia, classification of ischemic injury, size of ischemia, urokinase dose, disease type, and site of occlusion. However, our data suggest that pretreatment CBF influences ischemic outcome, and pretreatment CBF, age, and pretreatment NIH score also affect hemorrhagic rate. Furthermore, duration of ischemia influences ischemic outcome only in those with low pretreatment CBF and does not influence the hemorrhagic rate.
Yuh and associates (1995) advocate pretreatment evaluation of tissue viability and reversibility judged by magnetic resonance (MR) perfusion imaging for better patient selection to improve the efficacy of thrombolytic therapy. Pretreatment evaluation of tissue perfusion has not been a part of the routine protocol for emergency treatment of acute stroke in the past. Our study design therefore provides an opportunity to evaluate the efficacy of intra-arterial thrombolytic therapy in a more ideal or perhaps less complicated situation in that all of our patients had early and complete recanalization (eliminate the technical variation) and pretreatment SPECT (add information reflecting tissue reversibility).
It is well known that the reversibility of ischemic tissue depends on the severity and duration of ischemia. Experimental studies show that brain injury can be limited by restoring the CBF within several hours (Jones et al., 1981). The ischemic tissue with a low CBF (<10 mL/100 g/min) as the ischemic core rapidly underwent irreversible damage. The concept of an ischemic penumbra surrounding the ischemic core was defined originally as a region of blood flow below that needed to sustain electric activity but above that required to maintain cellular ionic gradient, which may in time lead to irreversible cellular damage (Symon et al., 1977; Astrup et al., 1981; Hossmann et al., 1982 and 1994). The concept of the ischemic penumbra is indicative of potentially reversible ischemic tissues with early and appropriate therapeutic interventions (Fisher and Garcia, 1996). The classic CBF ranges for the penumbra is known to be around 10 to 20 mL/100 g/min in animal studies (Jones et al., 1981). Tamura and colleagues (1980) indicated that the flow threshold for middle cerebral artery occlusion in cats was 12 to 15 mL/100 g/min maintained for over 2 hours. Clinical positron emission tomography studies suggest that CBF in the penumbra ranges from 7 to 17 mL/100 g/min (Furlan et al., 1996). Heiss and coworkers (1997) report that flumazenil positron emission tomography can detect irreversibly damaged ischemic tissue in their experimental study and first clinical applications. Although CBF reflected by SPECT is not an absolute measurement, our data suggest that the pretreatment CBF of ischemic tissue plays the most important role in the influence of ischemic outcome including hemorrhagic complication, and the parameters provided by pretreatment SPECT have value in assessing the reversibility of ischemic tissue.
Although the therapeutic window in humans is generally believed to be the first 6 hours after the onset of symptoms, our data show a variation among different individuals. We believe that the therapeutic window for potentially salvageable ischemic tissue is likely dependent on the degree of collateral flow and metabolic status (Yuh et al., 1995). For patients with higher pretreatment CBF, the therapeutic window may be much longer (up to 12 hours in our series) than for those with low pretreatment CBF (less than 5 hours) (Figs. 4 and 5). Furlan and colleagues (1996) report a therapeutic window of up to 16 hours, which is much longer than the popularly proposed 3- to 6-hour window. Moreover, the therapeutic window in patients with occlusion of the posterior circulation may be relatively longer than that in patients with occlusion of the anterior circulation (Hacke et al., 1988). Zeumer and associates (1993) suggest that intra-arterial thrombolytic therapy for occlusion of the internal carotid artery or middle cerebral artery should be started within 5 hours. Our data are consistent with the data of Zeumer and others, suggesting that 5 hours rather than 6 hours is a preferable cutoff time for the therapeutic window. However, our data also indicate that the 5-hour therapeutic window applies only to those with low pretreatment CBF judged by SPECT.
Hemorrhagic transformation is believed to be caused by reperfusing irreversibly damaged tissue. Hemorrhagic transformation after thrombolytic therapy can be classified into hemorrhagic infarction and parenchymal hematoma. Hemorrhagic infarction and parenchymal hematoma with extensive brain edema often aggravate the neurologic symptoms and adversely influence neurologic outcome (Overgaard et al., 1990; Lyden and Zivin, 1993). Although the incidence of spontaneous hemorrhagic transformation including asymptomatic hemorrhage was reported to be 15% to 43% (Toni et al., 1996), there is evidence that intravenous thrombolytic therapy increases the hemorrhagic transformation rate (Multicentre Acute Stroke Trial-Italy (MAST-I) Group, 1995; The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group, 1995; The Multicenter Acute Stroke Trial-Europe Study Group, 1996). Delayed recanalization, particularly in intravenous recombinant tissue plasminogen activator (rt-PA) infusion, increases the chance of reperfusing ischemic tissue that has ischemic duration beyond that individual's therapeutic window. Although the difference in overall hemorrhagic events (rt-PA 42.8%; placebo 36.8%) in the European Cooperative Acute Stroke Study (Hacke et al., 1995) was not significant between the two groups, the occurrence of large parenchymal hemorrhages was significantly more frequent in the rt-PA treatment group (19.8% versus 6.5%).
On the other hand, early recanalization, especially in intra-arterial rt-PA or urokinase infusion, can cause a sudden rise in intravascular pressure, and parenchymal hematomas can develop if ischemic tissue has been irreversibly damaged, particularly in individuals who have short therapeutic windows. In intra-arterial infusion studies, hemorrhagic transformation was reported in a mean of 21% of patients within the reported range of spontaneous hemorrhage rate. Recently, in a randomized, controlled, double-blind trial of intra-arterial recombinant prourokinase, there was no difference between the frequency of brain hemorrhage with clinical deterioration when comparing the recombinant prourokinase (15%) and placebo (14%) groups (del Zoppo et al., 1998).
In the current study, hemorrhagic transformation occurred in 23%, and parenchymal hematoma was observed in 17%. Most hemorrhagic transformation occurred in patients within the 6-hour therapeutic window (71% between 3 and 5 hours), indicating that early intervention may not always be helpful and other factors also must be considered (Figs. 4 and 5). Moreover, the incidence of hemorrhagic transformation in patients with thrombotic middle cerebral artery occlusion was reported to be less than that in patients with embolic middle cerebral artery occlusion because the mean residual CBF was significantly higher in the former than in the latter (Ueda et al., 1997). We observed that patients with hemorrhagic transformation were significantly older, with a much lower residual CBF (R/CE ratio < 0.35) and higher pretreatment NIH score than those without hemorrhagic transformation.
Other factors also may influence hemorrhagic transformation. Levy and associates (1994) report that factors significantly related to the development of an intracerebral hematoma were rt-PA dose and diastolic hypertension. Ueda and others (1995) suggest that although intra-arterial thrombolysis should not theoretically exert a greater influence on systemic coagulation and fibrinolytic factors than intravenous thrombolysis, the administration of high-dose rt-PA induced greater changes in those factors. Alexandrov and associates (1997a) indicate that a high risk of cardioembolism, M1 occlusion, and absence of collateral flow helped to identify patients at risk for spontaneous hemorrhagic transformation.
Computed tomography has been a valuable pretreatment imaging tool in the evaluation of patients with acute stroke for possible thrombolytic therapy. The early signs of ischemia on CT scans are associated with severe ischemic stroke and a poor clinical outcome (von Kummer et al., 1994; Yokogami et al., 1996; Tarr et al., 1996). Based on our results, it may be reasonable to assume that early, abnormal CT findings are indicative of a markedly low CBF. However, normal CT findings do occur in patients with a poor outcome, suggesting that the severity of hypoperfusion may not always be appreciated by early CT scans and perfusion studies may be helpful in such situations.
The CBF study using HMPAO-SPECT has advantages in that it is readily available and can be performed quickly in emergency cases (Herderschee et al., 1991). In addition, the value of this technique in the management of stroke has been established in the past (Hanson et al., 1993; Baird et al., 1994). Shimosegawa and associates (1994) indicate that the lesion-to-contralateral radioactivity ratio for infarction threshold was 0.48 ± 0.14 in patients without thrombolytic therapy who underwent imaging within 6 hours after onset, findings which are similar to ours. Sasaki and colleagues (1996) demonstrated that reperfusion in only nine patients within 7 1/4 hours from onset significantly reduced the development of infarction in a R/CE ratio between 0.55 and 0.75. Ezura and coworkers (1996) indicate that moderate ischemia with residual relative regional CBF of between 0.35 and 0.70 might be suitable for intra-arterial thrombolysis by evaluation of only seven patients. Our data show that the pretreatment CBF judged by SPECT in 42 lesions of 30 patients with complete recanalization is significantly different among reversible ischemia, infarction, and hemorrhage (Figs. 4 and 5). The development of infarction and hemorrhagic transformation depend mainly on pretreatment CBF, and most patients with hemorrhagic transformation have had severe ischemic injury and irreversible damage within the so-called 6-hour therapeutic window. In this study, within 5 hours after onset of symptoms, the development of infarction or hemorrhage appeared to depend on both the residual CBF and on the duration of ischemia, whereas after 5 hours from onset, the thresholds for the development of infarction or hemorrhage were likely to be about 0.55 and about 0.35 of R/CE ratio, respectively.
Perhaps most importantly, our data show that the residual CBF by initial SPECT correlated with the neurologic outcome, supporting the notion that SPECT provides important information for determining whether thrombolytic therapy is worthwhile by predicting ischemic outcome, including associated hemorrhagic risk and neurologic outcome, at any given time point within or beyond the so-called 6-hour therapeutic window (Ueda et al., 1996). Patients having a high CBF (R/CE ratio > 0.55) should be treated, regardless of how long it has been beyond the 6-hour therapeutic window after onset (our data suggest that it can be up to 12 hours). On the other hand, the outcome of patients with low CBF could be improved only if they were treated within 5 hours.
With recent advances in fast imaging techniques, including echo-planar imaging, MR imaging may be another potentially useful means to assess tissue viability and reversibility before thrombolytic therapy. Diffusion MR imaging is reported to be useful clinically in the diagnosis of hyperacute ischemic stroke as early as 30 minutes after onset without treatment (Warach et al., 1995). Kohno and colleagues (1995) report that the CBF threshold in detecting abnormal signal intensity on diffusion MR imaging was 41 mL/100 g/min 2 hours after middle cerebral artery occlusion in rats, which is substantially higher than the infarction threshold. This suggests that diffusion imaging is a sensitive means of detecting of ischemia. However, whether the diffusion abnormality can be used to evaluate tissue reversibility at any given time point remains to be determined. Perfusion MR imaging is reported to provide information related to microcirculation of the brain parenchyma. Perfusion MR imaging is based mainly on hemodynamic phenomena (kinetic) rather than a biological process; as in spectroscopy and diffusion imaging, it can readily detect flow abnormalities and is ideal for evaluating hyperacute ischemic stroke (Maeda et al., 1997). Information related to tissue perfusion status can be available within 25 minutes in our institution using echo-planar imaging technique. More semiquantitative data similar to those acquired by SPECT techniques in our study are needed to judge the capability of perfusion MR imaging in assessing tissue reversibility before thrombolytic therapy (Yuh et al., 1995). Similarly, stable xenon-CT (Touho and Karasawa, 1996; Firlik et al., 1997) and dynamic CT are other methods that also may have the potential to provide information related to the reversibility of ischemic tissue for the treatment of acute stroke.
In conclusion, based on our limited data, outcomes of acute cerebral ischemia with early and complete recanalization using intra-arterial thrombolytic therapy are significantly different and are influenced markedly by pretreatment CBF assessed by SPECT. The CBF thresholds evaluated by SPECT provide important information that can be potentially useful in the management of acute stroke patients with intra-arterial thrombolysis: (1) ischemic tissue with a flow index greater than 0.55 still may be salvageable even with treatment initiated 6 hours after onset of symptoms; (2) ischemic tissue with a flow index greater than 0.35 still may be salvageable with early treatment (less than 5 hours); and (3) ischemic tissue with a flow index below 0.35 may be at risk for hemorrhage, even when treatment is started within the critical time window. Our study suggests that SPECT is a non-invasive, readily available, and convenient means that can provide useful parameters to predict patient outcome before treatment. When combined with clinical information and, in some patients, the duration of ischemia, it may be potentially useful to improve patient selection for thrombolytic therapy and therefore its efficacy.
Footnotes
Acknowledgements
The authors thank Dr. Yoshifumi Sugawara and the staff members of the Division of Radiology, Ehime University School of Medicine, Japan, who cooperated in this study. The authors also thank Ms. Phyllis Bergman for her expert assistance in the preparation of this manuscript.
