Abstract
Objective
Cerebral ischemia-reperfusion injury (CIRI) remains a major challenge in clinical practice, without effective and targeted approaches to address it. Curculigoside has been reported to exert beneficial effects in CIRI, yet the underlying novel mechanisms remain unclear. In this work, we aimed to explore whether Curculigoside exerted its protective role by regulating transient receptor potential melastatin 2 (TRPM2)-mediated microglial polarization.
Methods
A well-established middle cerebral artery occlusion (MCAO) model was constructed in Sprague-Dawley rats, with Curculigoside administrated via the caudal vein. In vitro, LPS/IFN-γ-stimulated BV2 microglial cells were used to investigate the effects of Curculigoside on polarization and neuroinflammation. High-throughput mRNA sequencing was used to predict key targets of Curculigoside. TRPM2 overexpression and autophagy inhibition assays were conducted to verify the underlying mechanism.
Results
Curculigoside treatment significantly alleviated neurological deficits, reduced cerebral infarction volume, and decreased brain edema, accompanied by inhibited M1 polarization of microglia and mitigated inflammatory injury during CIRI. Curculigoside reduced pro-inflammatory M1 polarization in LPS/IFN-γ–stimulated BV2 microglia, thereby reducing neuronal apoptosis. RNA-seq functional enrichment analysis identified TRPM2 as a key target, and Curculigoside was found to downregulate TRPM2 expression and regulate the mTOR-mediated autophagy pathway. Notably, the protective effects of Curculigoside on microglial polarization were reversed by TRPM2 overexpression or autophagy inhibition.
Conclusion
Curculigoside mitigates CIRI by promoting beneficial microglial polarization through the TRPM2/mTOR-autophagy axis, supporting its potential as a therapeutic agent for CIRI.
1. Introduction
Ischemic stroke, a frequently occurring cerebrovascular disease (CVD) among the elderly, is the result of the obstruction or stenosis of cerebral arteries. 1 At present, the only therapeutic method available for ischemia is reperfusion, and this reperfusion effect is accomplished through either mechanical thrombectomy or intravenous thrombolysis. 2 Paradoxically, it is important to note that a sudden reestablishment of blood circulation to the ischemic zone occasionally heightens the risk of suffering cerebral ischemia-reperfusion injury (CIRI).3,4 Besides, owing to the extremely short treatment time window and the potential hemorrhage risk, only a limited number of patients are capable of deriving advantages from the abovementioned strategies. 5 Thus, it is especially crucial to disclose the potential mechanism of CIRI, as it may offer novel clinical treatment modalities.
Multiple pathological cascades are triggered during CIRI, including oxidative stress, 6 glutamate excitotoxicity, 7 mitochondrial dysfunction, 6 apoptosis 8 and neuro-inflammation. 9 The neuro-inflammatory cascade is a complex and highly regulated biological process, it can be immediately activated following vessel occlusion via shearing stress from the endothelium. 10 Cumulative evidence suggests that resident microglia are of great significance in the pathogenesis of ischemic brain injury. 11 In the acute period during the first minutes to hours right after a stroke starts, the resident microglia primarily recruited to peri-infarct region and are involved in clearing the necrotized cells. While in the subacute phase, which ranges from hours to days after the insult, microglia become overactivated, which is accompanied by pro-inflammatory mediator release. 12 During the process of microglial polarization, classically-activated M1 microglial cells are capable of generating a vast amount of pro-inflammatory factors, which are considered detrimental to recovery. Modulating the polarization process of microglial cells is a significant strategy to repress neuroinflammation induced by cerebral ischemia.13,14 The recent studies demonstrated that autophagy is the critical factor balancing the beneficial and harmful effects of immunity and inflammation. 15 For instance, the activation of autophagy is capable of reducing neuroinflammation following CIRI and restraining the M1 polarization process in microglia. 16 Hence, gaining a better understanding of autophagy and the balance of microglial polarization in CIRI may provide valuable insights for the formulation of therapeutic strategies.
Transient receptor potential channel melastatin 2 (TRPM2), the second member of the TRPM family initially discovered in 1998, was formally named in 2002. 17 Many studies have reported the involvement of TRPM2 in a wide range of pathological processes, including vascular and neurodegenerative diseases.18,19 There is evidence that the activation of ion channel TRPM2 is specifically responsible for cerebral ischemic injury in males. 20 The other study has shown that TRPM2 downregulation protects the liver from ischemia reperfusion injury in vivo, which is accompanied by autophagy activation. 21 Besides, TRPM2 knockout in microglia has shown to play a beneficial role in pilocarpine-induced neuroinflammation via regaining autophagy by regulating classical AMPK/mTOR pathway. 22 These findings suggest the protective effect of TRPM2 in CIRI, while its role and relative mechanism need further exploration.
Curculigoside, a phenolic glycoside, is a key bioactive compound of Curculigo orchioides. Curculigoside possesses quite extensive pharmacological activities, such as anti-oxidant, 23 anti-inflammation 24 and neuroprotection. 25 Curculigoside alleviated CIRI and inhibited blood-brain barrier (BBB) breakdown in rats. 26 Meanwhile, Curculigoside has been reported to facilitate the maturation and stability of newly formed blood vessels in rats insulted with middle cerebral artery occlusion (MCAO). 27 Nevertheless, the accurate mechanism of Curculigoside on CIRI has not been thoroughly elucidated. The present work aimed to confirm the protective role of curculigoside in CIRI in vivo and in vitro, and to investigate whether it acts by regulating microglial polarization via TRPM2-mediated autophagy.
2. Materials and methods
2.1. Animals and treatment
Male Sprague-Dawley rats (weighing 250 ± 10 g) were supplied by Liaoning Changsheng Biotechnology Co., Ltd. (NO. SCXK (Liao) 2020-0001) and kept in standard environment (temperature: 21-25 °C, humidity: 45%-65%, 12 h light/dark cycle). After adaption, the rats were randomly assigned to four groups: Sham, middle cerebral artery occlusion (MCAO), MCAO + low-dose Curculigoside (L-Curculigoside, 20 mg/kg), and MCAO + high-dose Curculigoside (H-Curculigoside, 40 mg/kg). Randomization was performed using a computer-generated random number table by an independent researcher not involved in outcome assessment. The MCAO and Curculigoside-treated rats underwent left-side MCAO by inserting silicone-coated sutures into the internal carotid artery. The sutures were advanced until occluded the origin of the middle cerebral artery and maintained occlusion for 2 h followed by suture removal to initiate 24 h of reperfusion as previously described. 28 The rats in the sham group received identical surgical procedures without suturing. Curculigoside (purity ≥ 98%, CAS: 85643-19-2, Shanghai Aladdin Biochemical Technology Co., Ltd., Lot: C413287) was injected via the caudal vein at the doses of 20 and 40 mg/kg 30 min after reperfusion. Sham or MCAO rats received an equal volume of saline. For MCAO surgery, rats were anesthetized with 3% isoflurane for induction followed by 1.5–2% isoflurane maintenance via a nose cone. Anesthesia depth was monitored continuously by assessing respiratory rate, pedal reflex, and tail pinch response. Post-surgery, rats received analgesia with buprenorphine (0.05 mg/kg, subcutaneous injection) immediately after surgery and every 12 h to manage postoperative pain. The neurological deficits detection was performed 24 h after MCAO induction before detecting brain infarct volume. During this period, the mortality rate of the rat was monitored. To preserve tissue integrity, the brain tissue processing was initiated within 5 min on ice after euthanasia confirmation.
2.2. Neurological function evaluation
Neurological Severity Score (NSS) was used to evaluate neurological function of MCAO rats as previous report at 24 h after MCAO induction. 29 The scoring range: 0 points = no neurological deficit (normal movement and posture); 1 point = mild deficit (inability to fully extend the contralateral forelimb); 2 points = moderate deficit (turning to the contralateral side when crawling); 3 points = severe deficit (falling to the contralateral side when walking); 4 points = coma or death. The assessor performing the NSS evaluation was fully blinded to the rats’ treatment group assignment.
2.3. 2, 3, 5-triphenyl-2H-tetrazolium chloride (TTC) staining
The brains were harvested 24 h after MCAO induction and immediately frozen at -20 °C for 30 min to preserve integrity. Subsequently, the brain tissues were cut into 2 mm-thick coronal slices. These slices were then incubated with 1% TTC solution (Sigma, St. Louis, MO, USA) at 37 °C for 20 min in a dark environment and fixed with 4% formaldehyde solution (Beyotime Biotechnology, Nanjing, China) at room temperature for 24 h. The ischemic area remained unstained and Image-Pro Plus 6.0 was utilized for image analysis.
2.4. Histopathological analysis
2.4.1. Hematoxylin-eosin (H&E) staining
Freshly collected brains were fixed in 4% paraformaldehyde, dehydrated with graded ethanol, cleared with xylene, and embedded in paraffin. The paraffin-embedded brain tissues were sectioned into 5 μm-thick slices, which were dewaxed, rehydrated, stained with hematoxylin (Solarbio, Beijing, China), and counterstained with eosin (Solarbio, Beijing, China). Images were captured using a light microscopy (Olympus, Japan) at 200 X magnification.
2.4.2. Nissl staining
The brain sections previously prepared were utilized and subjected to Nissl dye according to the manufacturer’s instruction (Solarbio, Beijing, China). ImageJ was utilized to count the number of normal neurons with Nissl body to assess neuronal damage. The researcher performing the quantification was unaware of the sample grouping.
2.5. Cell culture and treatment
BV-2 cells were obtained from Wuhan Procell Biotechnology Co., Ltd. (Wuhan, China) and cultured in DMEM containing 10% FBS and 1% penicillin/streptomycin at 37°C with 5% CO2. The cells utilized in this study were verified to be mycoplasma-free through PCR detection before experimental initiation. The cells cryopreserved within passage 5 were used. For the induction of M1-like phenotype polarization, the BV-2 cells were co-cultured with 100 ng/mL lipopolysaccharide (LPS) and 20 ng/mL interferon-gamma (IFN-γ) for 24 h followed by 6 h treatment of 40/80 μM Curculigoside. The supernatant was collected and filtered through a 0.22 μM filter to obtain the conditioned medium.
HT22 cells were purchased from Wuhan Procell Biotechnology Co., Ltd. (Wuhan, China) and were verified to be mycoplasma-free through PCR detection before experimental initiation. The cells cryopreserved within passage 5 were used. HT22 cells were pretreated with 40/80 μM Curculigoside for 1 h and subjected to oxygen-glucose deprivation (OGD) for 3 hours followed by reoxygenation for 24 h. As a control, the OGD group was pretreated with DMSO before undergoing the same OGD and reoxygenation (OGD/R) procedure. The OGD procedure was performed using a glucose-free Dulbecco’s Modified Eagle Medium (DMEM, Gibco), and the cells were then placed in a hypoxic incubator maintained at 37 °C with a gas mixture of 94% N2, 5% CO2, and 1% O2 for 3 h to induce OGD.
The HT22 cells were exposed to the abovementioned conditioned medium for 24 h to evaluate the impact of the conditioned medium on cell viability.
BV2 microglia were polarized with 100 ng/mL LPS Combine with 20 ng/mL IFN-γ for 24 h, in sequence, the cells co-treated with 40/80 μM Curculigoside with/without 3-MA (autophagy inhibitor) for 6 h before collection.
The TRPM2 overexpression plasmid was constructed using the pcDNA3.1(+) vector (Hunan Fenghui Biotechnology Co., Ltd., China) and transfected BV2 microglia using Lipofectamine 3000 Reagent (Thermo Fisher Scientific, USA) before abovementioned induced polarization and Curculigoside treatment.
2.6. CCK-8
The abovementioned HT22 cells were used to detect cell viability. In brief, 10 μL of CCK-8 working solution (Beyotime Biotechnology, Nanjing, China) was added to each well and the cells were incubated for 2 h at 37 °C with 5% CO2. The sorbance at 450 nm was measured using a microplate reader (BioTek, USA).
2.7. TUNEL
The treated HT22 cells were fixed with 4% paraformaldehyde, permeabilized with 0.1% Triton X-100, and incubated with TUNEL reaction solution (One Step TUNEL Apoptosis Assay Kit, Beyotime Biotechnology, Nanjing, China) for 60 min at 37°C in dark. 4′,6-diamidino-2-phenylindole (DAPI, Aladdin, Shanghai, China) was used to counterstain nuclei, the slides were analyzed by fluorescence microscopy (Olympus, Japan).
2.8. RNA-sequencing
Brain tissue samples were procured from rats in MCAO group and MCAO+H-Curculigoside group, with 3 biological replicates per group (n=3). and total RNA extraction was executed using the RNAprep Pure Tissue Kit (Tiangen Biotech, Beijing, China). RNA quantity was gauged by a NanoDrop ND-1000 spectrophotometer. High-throughput mRNA sequencing was carried out on an Illumina NovaSeq 6000 platform (Novogene Bioinformatics Technology Co., Ltd., Beijing, China). Differential gene expression analysis was performed using DESeq2 software. The thresholds for defining differentially expressed genes (DEGs) were set as |log2 fold change (FC)| ≥ 1.5 and adjusted p < 0.05, with the Benjamini-Hochberg method.
2.9. Immunofluorescent staining
Brain tissue sections were permeabilized with 0.3% Triton X-100 (Solarbio, Beijing, China) and blocked with 5% bovine serum albumin (BSA, Solarbio, Beijing, China). Sections were incubated with primary antibodies: anti-NeuN (ab177487; 1:200, Abcam, USA), anti-Iba-1 (ab178846; 1:200, Abcam, USA), anti-GFAP (ab7260; 1:200, Abcam, USA), and anti-TRPM2 (A6137; ABclonal, Wuhan, China) overnight at 4 °C. Next, Alexa Fluor 488- and 594-conjugated goat anti-Rabbit secondary antibodies (1:500 dilution) for 1 hour at room temperature in the dark. Sections were then washed three times with PBS for 5 minutes each, and nuclei were counterstained with 4’,6-diamidino-2-phenylindole (DAPI, SENBEIJIA, Nanjing, China) for 5 min. Stained sections were imaged using an Olympus BX53 fluorescence microscope.
2.10. Flow cytometry
The polarized BV2 cells were harvested, washed with PBS, and stained with fluorescence-conjugated antibodies against CD86 (BioLegend, Cat. No. A17199A, dilution 1:150) and CD206 (BioLegend, Cat. No.C068C, dilution 1:150) for 30 min at 4°C in dark. After washing with PBS, the percentage of M1 (CD86+CD206-) and M2 (CD206+CD86-) microglia was analyzed using a flow cytometer (CytoFLEX, Beckman Coulter, China). FSC/SSC gates were used to exclude debris and doublets, defining the viable cell population (Figure S1). Subsequently, CD86/CD206 gates were applied to distinguish M1 (CD86+CD206-) and M2 (CD206+CD86-) microglia.
2.11. Reverse transcription-quantitative PCR (RT-qPCR)
Total RNA from cortex tissues on the ischemic side and BV2 cells were extracted with TRIzol® Reagent (Invitrogen, USA). Reverse transcription was performed with in ExicyclerTM96 real-time quantitative system with the SYBR Green PCR Kit (Solarbio, Beijing, China), which procedures were conducted the manufacturer’s instruction.
The primers for rats: TRPM2: forward 5’-GAAGGAAAGAGGGGGTGTG-3’; reverse 5’-CATTGGTGATGGCGTTGTAG-3’; IL-1β: forward 5’-AGCTGGAGAGTGTGGATCCC-3’; reverse 5’-CCTGTCTTGGCCGAGGACTA-3’; TNF-α: forward 5’- CTCAAGCCCTGGTATGAGCC-3’; reverse 5’- GGCTGGGTAGAGAACGGATG-3’; IL-10: forward 5’-AGGGTTACTTGGGTTGCC -3’; reverse 5’-GGGTCTTCAGCTTCTCTCC-3’; GAPDH: forward 5’-AAGGTCGGTGTGAACGGATT-3’; reverse 5’-TGAACTTGCCGTGGGTAGAG-3’.
The primers for mice: IL-1β: forward 5’-AAAATGCCTCGTGCTGTCTG-3’; reverse 5’-CAGAATGTGCCACGGTTTTC-3’; TNF-α: forward 5’-TGTGCTCAGAGCTTTCAACAA-3’; reverse 5’-CTTGATGGTGGTGCATGAGA-3’; IL-10: forward 5’-AGGCGCTGTCATCGATTTCT-3’; reverse 5’-ATGGCCTTGTAGACACCTTGG-3’; GAPDH: forward 5’-CAGCAACTCCCACTCTTCCAC-3’; reverse 5’-TGGTCCAGGGTTTCTTACTC-3’.
2.13. Western blot analysis
The cortex tissues on the ischemic side and abovementioned cells were harvested to extract proteins using cell RIPA lysis buffer (Solarbio, Beijing, China) with protease inhibitor PMSF (Solarbio, Beijing, China). Proteins (10 μg) were resolved by 10% or 12% SDS-PAGE, transferred to PVDF membranes and blocked with 5% non-fat milk in TBST at room temperature for 1.5 h. In sequence, the membranes were incubated with the corresponding primary antibodies (anti-iNOS (ab178945; dilution: 1:1000, Abcam, USA), anti-TRPM2 (A6137; dilution: 1:1000, ABclonal, Wuhan, China), anti-p-mTOR (AP0115; dilution: 1:1500, ABclonal, Wuhan, China), anti-mTOR (A2445; dilution: 1:1500, ABclonal, Wuhan, China), anti-LC3II/I (A27200PM; dilution: 1:1500, ABclonal, Wuhan, China), anti-p62 (A19700; dilution: 1:1000, ABclonal, Wuhan, China) overnight at 4 °C, HRP-labelled goat anti-mouse/rabbit secondary antibody at room temperature for 2 h. After visualization using enhanced chemiluminescence system (ECL, Liankebio Biotechnology, Hangzhou, China) for 10-60 s, the protein bands were quantified by using Gel-Pro-Analyzer.
2.14. Statistical analysis
Software GraphPad Prism 8.0 was used to conduct data analysis. All datasets were analyzed by one-way or two-way ANOVA as appropriate, followed by Tukey’s Honestly Significant Difference (HSD) test for multiple comparisons. The data was represented as mean ± SD and p < 0.05 was considered to be significant statistically.
3. Results
3.1. Curculigoside alleviates CIRI
Firstly, we confirmed the protective effects of Curculigoside on CIRI in vivo. As shown in Figure 1(a), Curculigoside treatment showed a trend toward reducing mortality compared to MCAO. Curculigoside administration notably reduced cerebral infarction volume measured by TTC staining (Figure 1(b), p<0.0001, p<0.0001), decreased brain water content (Figure 1(c), p<0.0001, p<0.0001) and improved neurological function (Figure 1(d), p=0.0092, p=0.0006). Besides, HE staining revealed that the MCAO group exhibited severe pathological alterations in the ischemic penumbra, including disordered tissue architecture and inflammatory cell infiltration, while Curculigoside treatment mitigated these changes (Figure 1(e)). Consistent with the pathological alterations, as shown in the images of Nissl staining, Curculigoside significantly attenuated neuronal injury as quantitatively analyzed by Nissl body counts and morphological integrity compared with the MCAO group (Figure 1(f), p=0.0023, p=0.0003). These results suggest that the protection of curculigoside against CIRI in vivo. Curculigoside alleviates CIRI.
3.2. Curculigoside modulated microglial activation and M1/M2 polarization after CIRI
In sequence, we conducted immunofluorescence analysis targeting NeuN (a neuron-specific nuclear protein), Iba-1 (a microglial cell marker), and GFAP (an astrocytic marker) to explore the underlying mechanisms of Curculigoside’s protective effects against CIRI. As shown in Figure 2(a), Curculigoside treatment exhibited a significant upregulation of NeuN Compared to the MCAO group. Its impact on GFAP expression was relatively mild (Figure 2(b)). Interestingly, the impact of Curculigoside on Iba-1 expression was notably more substantial than on the other markers (Figure 2(c)), suggesting that modulation of microglial activity might be a pivotal mechanism underlying the protective effects of Curculigoside. As indicated in Figure 2(d), inflammatory cytokine IL-1β (p<0.0001) and TNF-α (p<0.0001) were markedly elevated in the penumbra tissues of MCAO rats, whereas the anti-inflammatory cytokine IL-10 (p<0.0001) was downregulated, which could be reverse by Curculigoside administration (p=0.0002, p<0.0001/p=0.0029, p<0.0001/p=0.0316, p<0.0001). These findings indicate that Curculigoside exerts its neuroprotective effect against CIRI predominantly by modulating microglial activation and polarization. Curculigoside modulated microglial activation and M1/M2 polarization after CIRI.
3.3. Curculigoside inhibits microglia M1 polarization in vitro
LPS-and IFN-γ-treated BV-2 cell model was established to investigate the role of Curculigoside in modulating M1-like paradigm. As indicated in Figure 3(a), LPS and IFN-γ stimulation induced a marked shift towards the pro-inflammatory M1 microglial phenotype (CD86+CD206−), while Curculigoside treatment effectively counteracted this polarization (p=0.0003, p<0.0001). Meanwhile, Curculigoside significantly suppressed the expression of iNOS, a M1-like associated gene, in LPS-and IFN-γ-mediated BV-2 cells (Figure 3(b), p=0.0002, p<0.0001). Furthermore, as shown in Figure 3(c), we detected the mRNA levels of inflammatory factors (IL-1β, TNF-α and IL-10) in LPS-and IFN-γ-treated BV-2 cells and discovered that Curculigoside significantly inhibited the mRNA expression of pro-inflammatory factor IL-1β (p<0.0001/p<0.0001) and TNF-α (p<0.0001/p<0.0001), while accelerating the production of anti-inflammatory factor IL-10 (p<0.0001/p<0.0001) mRNA. These results indicate that Curculigoside possesses the ability to modulate BV2 microglial polarization, which might reveal its protective effects against CIRI. Curculigoside inhibits microglia M1 polarization in vitro.
3.4. Curculigoside inhibits microglial activation for neuroprotection
In sequence, to elucidate whether Curculigoside exerts a direct neuroprotective effect, we investigated its impact on HT22 cells challenged with OGD/R injury. As shown in Figure 4(a) and (b), compared to the cells in blank group, the OGD/R group showed reduced cell viability and increased apoptosis. Curculigoside treatment mitigated these effects, though the protection was moderate (p=0.0202, p=0.0302/p=0.0342, p=0.0092). In subsequent, HT22 cells were co-cultured with the supernatant of LPS + IFN-γ treated microglial cells to explore the role of microglial mediators. Interestingly, we found the supernatant of LPS + IFN-γ treated microglial cells decreased viability and elevated apoptosis of HT22 cells, which could be reversed by Curculigoside administration, suggesting that Curculigoside might protect neurons via inhibiting microglial activation. Curculigoside inhibits microglial activation for neuroprotection.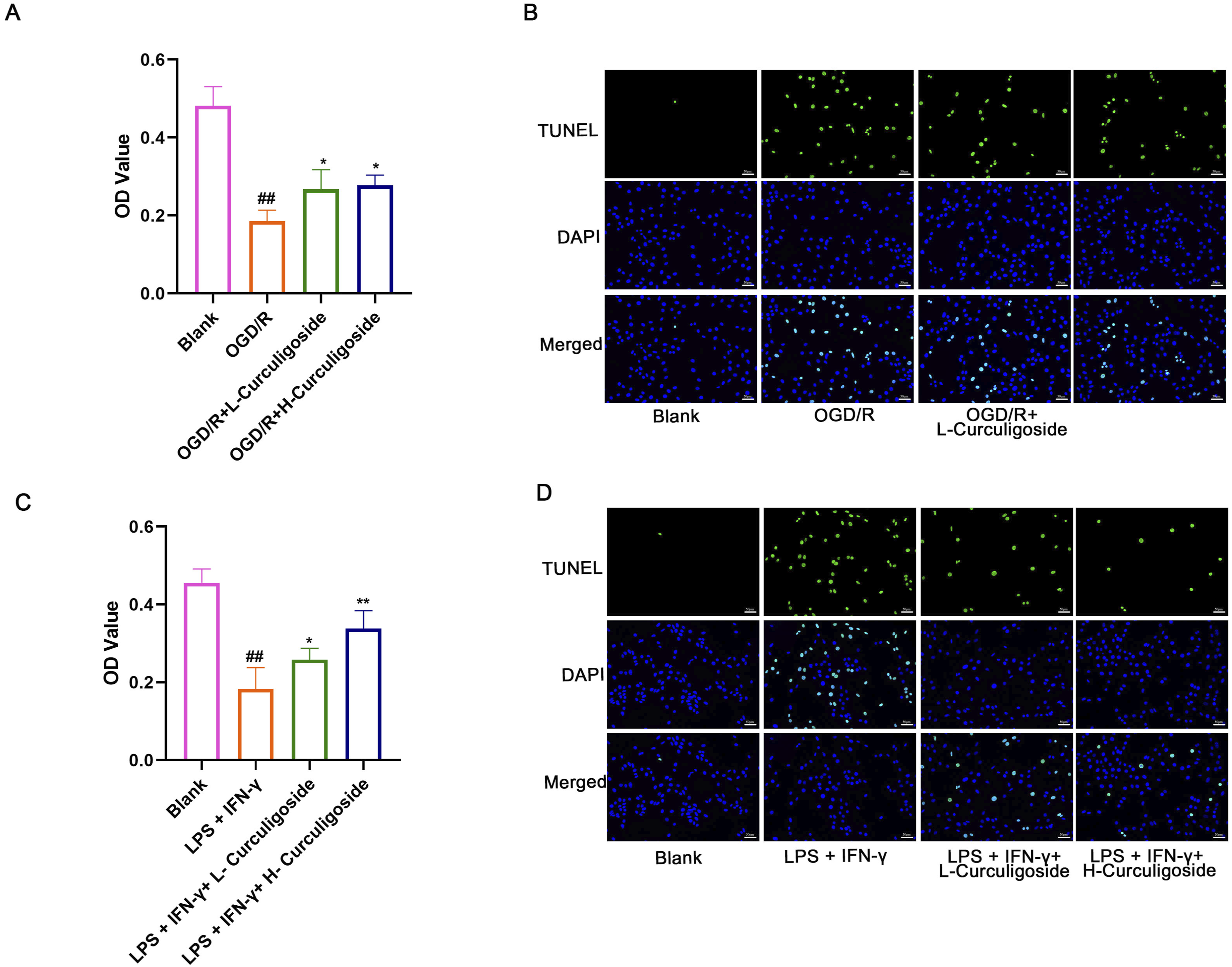
3.5. Curculigoside alters TRPM2 expression and regulates microglial activation associated pathway in CIRI
We next explored Curculigoside’s potential targets in CIRI by performing high-throughput mRNA sequencing on MCAO and MCAO+H-curculigoside brain tissues. The differentially expressed genes identified were further analyzed using Gene Ontology (GO) functional analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) to enrich pathways related to microglia/macrophage polarization (Figure 5(a)–(c)). Notably, TRPM2 was a key differentially expressed gene after Curculigoside treatment. RT-qPCR, western blot and immunofluorescence confirmed that curculigoside attenuated the upregulation of TRPM2 in brain tissues subject to CIRI (Figure 5(d)–(e), p<0.0001, p<0.0001/p=0.0454, p<0.0001), accompanied by reduced p-mTOR (p=0.0054, p<0.0001) and normalized autophagic markers, including LC3B-II/I (p=0.0011, p<0.0001) and p62 (p=0.0003, p<0.0001) (Figure 5(f)). These findings indicated that Curculigoside may exert neuroprotection by modulating TRPM2-mediated autophagy in microglial cells. Curculigoside alters TRPM2 expression and regulates microglial activation associated pathway in CIRI.
3.6. Curculigoside regulates microglia polarization via TRPM2-mediated autophagy
To explore whether Curculigoside participates in LPS + IFN-γ-induced microglia polarization via TRPM2-mediated autophagy, autophagy inhibitor 3-MA was used. As shown in Figure 6(a), Curculigoside treatment led to a reduction in TRPM2 (p=0.0009) and p-mTOR (p<0.0001), accompanied by normalization of LC3B-II/I ratio (p=0.0089) and p62 (p<0.0001) expression. Conversely, co-administration of 3-MA abrogated these regulatory effects (p=0.0010/p<0.0001/p=0.0510/p=0.0010), indicating the critical role of Curculigoside on autophagy regulation. In TRPM2-overexpressing cells, Curculigoside’s protective effects on inflammatory factor production were abolished (Figure 6(b) and (c)), accompanied by increased M1 proportion and decreased M2 markers proportion (Figure 6(d)). Collectively, these findings demonstrate that Curculigoside regulates microglial polarization by restoring TRPM2-mediated autophagic flux, identifying TRPM2 as a pivotal mediator of its neuroprotective mechanism. Curculigoside regulates microglia polarization via TRPM2-mediated autophagy.
4. Discussion
Timely and successful reperfusion is the most efficacious therapeutic strategy for ischemic stroke, however, the blood flow restoration may exacerbate brain tissue injury as a consequence of CIRI. Hence, it is of particular importance to study drugs that mitigate CIRI and the precise molecular mechanisms of these drugs. Currently, the administration of Curculigoside alleviates CIRI in rat and affords neuroprotection as manifested by a reduction in infarct size and an improvement in the neurological function score. However, it is worth emphasizing that our findings indicate that Curculigoside exerts the anti-inflammatory effect mainly by inhibiting M1 microglia polarization, thereby reducing the production of inflammatory factors. Notably, anti-inflammatory role of Curculigoside does not mainly target neurons directly. It is noteworthy that the abovementioned studies have proposed the protective effect of Curculigoside on injured neurons, while the anti-inflammatory effect found in our work may be largely attributed to its impact on microglia polarization.
It is well accepted that microglia-mediated neuroinflammation is necessary to the innate immune response during ischemic stroke. Under pathological conditions, microglial cells polarize into two distinguishable phenotypes, including pro-inflammatory M1 and anti-inflammatory M2 phenotype. 30 Classical M1 polarized microglia are considered detrimental to CIRI recovery by producing pro-inflammatory cytokines. Conversely, M2 microglia exhibit neuroprotective properties through anti-inflammatory factor. 30 Hence, facilitating the transition of microglia from M1 to M2 phenotype emerges as a viable strategy for reducing neuroinflammation after CIRI. Moreover, growing evidence has confirmed that monomers from Chinese medicines play a role in alleviating CIRI by regulating macrophage polarization. As expected, the treatment of Curculigoside has been shown to mitigate CIRI in MCAO rat models evidenced by a decrease in infarct volume and an enhancement in the neurological function score. Besides, Curculigoside application inhibited the abnormal activation of microglia rather than that of astrocytes. Curculigoside also protected neurons from CIRI to a certain extent, however, its inhibitory effect on the abnormal activation of microglia was more significant. There are few studies that have revealed the mechanism by which Curculigoside is involved in the M1 polarization process of microglia after CIRI.
A growing number of studies have highlighted on autophagy, considering it as a fundamental intracellular process that underlies M1/M2 polarization of microglia.
31
It is reported that autophagy is a double-edged sword for CIRI, moderate autophagy contributes to pro-survival mechanism post CIRI, while the dysregulated autophagy results in secondary neurological injury.
32
Consistent with our expectations, we have found that the administration of Curculigoside attenuates CIRI by blocking mTOR signaling pathway to boost autophagy, and thereby inhibiting microglial polarization towards M1 type. TRPM2 mRNA is most abundant in the central nervous system, especially in microglial cells. The recent published work has expounded that TRPM2 is a vital regulator of macrophage activation, which is associated with multiple pathological states, such as stroke, Alzheimer’s disease and neuropathic pain.
33
In addition, knocking out TRPM2 in microglia attenuates neuroinflammation and epileptogenesis by upregulating autophagy via AMPK/mTOR pathway.
22
In the present study, we observed that TRPM2 expression was elevated after CIRI or OGD/R procedures and the treatment of Curculigoside significantly inhibited this change. Meanwhile, enhanced autophagy and decreased M1 microglial polarization were also observed following the administration of Curculigoside. To actually demonstrate whether Curculigoside is involved in restoring abnormal autophagy via downregulating TRPM2 in LPS+IFN-γ-treated BV2 cells, we overexpressed TRPM2. As expected, the inhibitory effect of Curculigoside on the polarization of BV2 cells towards the M1 phenotype was reversed, implying that TRPM2 serves as a crucial target for Curculigoside in modulating the complex polarization process triggered by LPS+IFN-γ stimulation. Taken together, we suggest that Curculigoside treatment alleviates CIRI by regulating TRPM2-mediated microglia polarization to reduce inflammation and neuronal damage (Figure 7). A key limitation is the lack of direct evidence for Curculigoside’s blood-brain barrier penetration and cerebral tissue concentration. Additionally, its metabolites, potentially the actual bioactive moieties, have not been explored. Future investigations are warranted to confirm Curculigoside’s cerebral bioavailability and bioactivity. Graphical abstract.
In conclusion, our study demonstrated that Curculigoside exerts neuroprotective effects against CIRI, probably through modulating TRPM2-mediated autophagy to reverse microglia polarization towards the pro-inflammatory M1 phenotype, providing a new insight for the clinical application of Curculigoside in CIRI treatment.
Supplemental material
Supplemental material - Curculigoside attenuates cerebral ischemia-reperfusion injury via TRPM2-mediated microglial polarization
Supplemental material for Curculigoside attenuates cerebral ischemia-reperfusion injury via TRPM2-mediated microglial polarization by Mengying Fan, Yangyang Chen, Pingping Yin, Peng Li in Human & Experimental Toxicology
Supplemental material
Supplemental material - Curculigoside attenuates cerebral ischemia-reperfusion injury via TRPM2-mediated microglial polarization
Supplemental material for Curculigoside attenuates cerebral ischemia-reperfusion injury via TRPM2-mediated microglial polarization by Mengying Fan, Yangyang Chen, Pingping Yin, Peng Li in Human & Experimental Toxicology
Supplemental material
Supplemental material - Curculigoside attenuates cerebral ischemia-reperfusion injury via TRPM2-mediated microglial polarization
Supplemental material for Curculigoside attenuates cerebral ischemia-reperfusion injury via TRPM2-mediated microglial polarization by Mengying Fan, Yangyang Chen, Pingping Yin, Peng Li in Human & Experimental Toxicology
Footnotes
Acknowledgments
The authors are grateful to the Laboratory Animal Center of China Medical University for providing the experimental facilities and technical assistance.
Ethical considerations
All animal experiments comply with the ARRIVE guidelines and should be carried out in accordance with the U.K. Animals (Scientific Procedures) Act, 1986 and associated guidelines, EU Directive 2010/63/EU for animal experiments, or the National Institutes of Health guide for the care and use of Laboratory animals (NIH Publications No. 8023, revised 1978). All experimental procedures were approved by Chinese Medical University Laboratory Animal Ethics Committee (Approval No. CMUKT2024183).
Author contributions
Mengying Fan and Yangyang Chen: Investigation, Software, Visualization, Writing-original draft. Pingping Yin: Validation, Data curation, Formal analysis, Project administration. Peng Li: Conceptualization, Methodology, Investigation, Writing-review and editing. All the authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Wuhan Municipal Health Commission Traditional Chinese Medicine Research Project (Project No. WZ24A15).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed in this study are included in this published article. RNA-seq data have been deposited in BioSample database under BioProject ID: PRJNA1414812.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
