Abstract
Introduction
For many years, 5-fluorouracil (5-FU) has been utilized as a chemotherapeutic treatment for a variety of malignancies. Unfortunately, 5-FU causes cardiotoxicity, which restricts its clinical use. Nifuroxazide (NFX) is a STAT-3 inhibitor with antioxidant and anti-inflammatory effects.
Methods
Cardiotoxicity was induced by 5-FU (30 mg/kg) once daily for 5 days. NFX was administered in two doses, 25 and 50 mg.
Results
Compared to 5-FU-control rats, NFX significantly attenuates cardiotoxicity induced by 5-FU, as indicated by decreasing creatine kinase (CK)-MB, aspartate aminotransferase (AST), alkaline phosphatase (ALP), and lactate dehydrogenase (LDH) serum levels. Histopathological examinations confirmed the protective effects of NFX against histological abrasions induced by 5-FU. NFX attenuated the oxidative damage induced by 5-FU mediated by peroxisome proliferator-activated receptor gamma (PPAR-γ) signal activation. Moreover, NFX mitigated 5-FU-induced inflammation by suppressing nucleotide-binding domain, leucine-rich repeat-containing protein 3/signal transducer and activator of transcription 3 (NLRP3/STAT3) signal activation. Notably, these protective effects are dose-dependent. Additionally, NFX mitigated 5-FU-induced apoptosis by downregulating Bax, while upregulating Bcl-2.
Conclusions
Collectively, NFX attenuated 5-FU-induced cardiac intoxication by regulating NLRP3/STAT-3, PPAR-γ, and Bax/Bcl-2 signals.
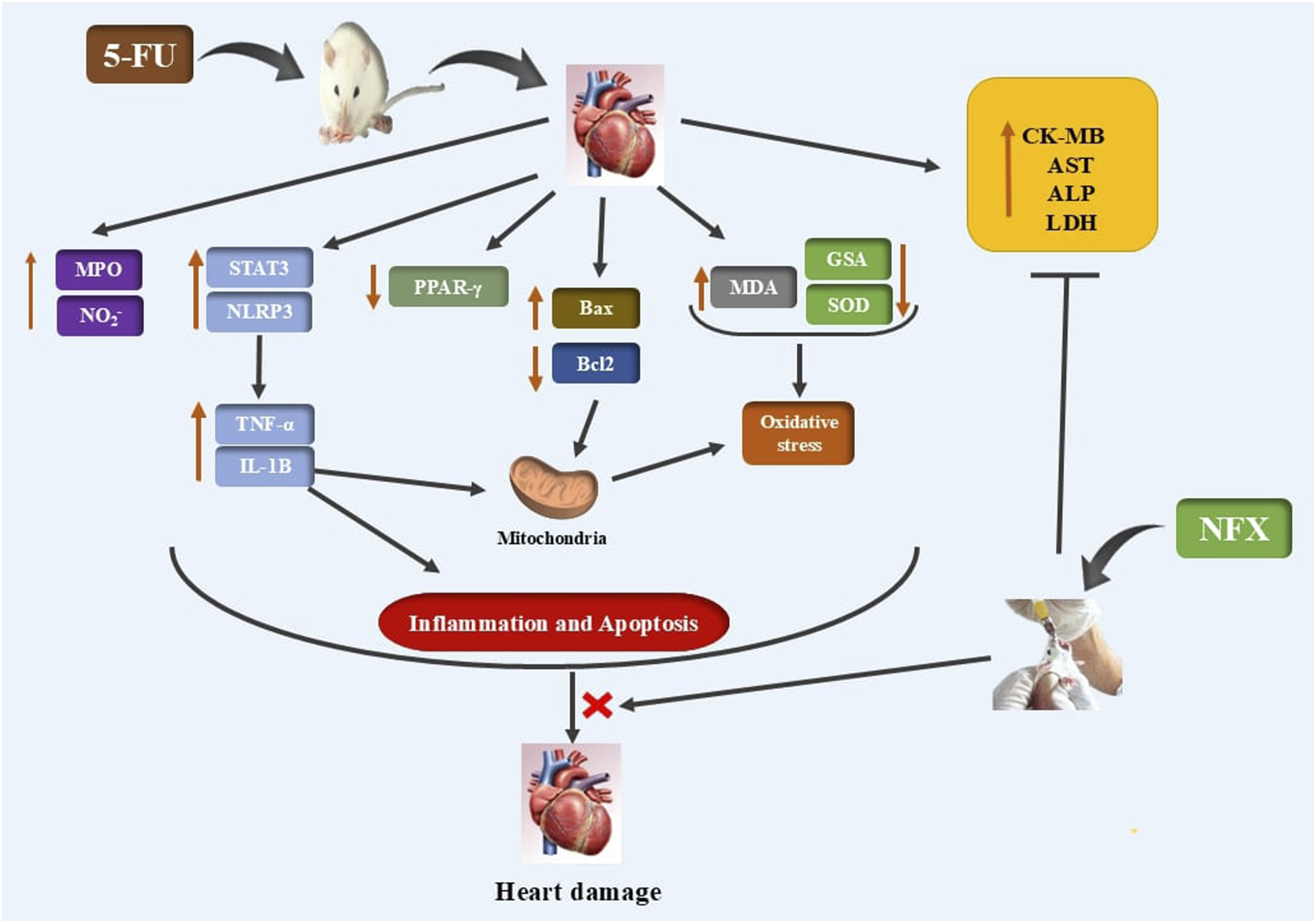
Introduction
Chemotherapy drugs have the potential to induce significant cardiotoxicity.1,2 For many years, 5-fluorouracil (5-FU) has been utilized as a chemotherapeutic treatment to treat a variety of malignancies, including head and neck, colorectal, breast, and esophageal cancers.3,4 Active (5-FU) metabolites in the cell interfere with the mitotic machinery and prevent cell proliferation by binding to DNA and RNA and inhibiting thymidylate synthase (TS). 3 The significant toxicity of 5-FU on the nervous, hematological, gastrointestinal, and cardiovascular systems limits its effectiveness. 5 5-FU is the second-most frequently used drug that causes cardiotoxicity after anthracyclines.6,7
Many mechanisms mediate cardiovascular damage caused by 5-FU. Excess production of reactive oxygen species (ROS) as a result of one of the most significant pathways being stimulated, although the heart has low levels of antioxidant enzymes. 8 Oxidative stress and inflammation are interlinked; elevated ROS levels during pathological conditions trigger biomolecular oxidation, promoting the activation of inflammatory cytokines. 9 Interestingly, the nucleotide-binding domain, leucine-rich repeat-containing protein 3 (NLRP3) inflammasome plays a significant role in innate immunity. A nod-like receptor called NLRP3 recognizes various stimuli to activate pro-caspase-1, which then cleaves into the active form of caspase-1 and causes the maturation and release of interleukin (IL)-1β. 10 On the other side, peroxisome proliferator-activated receptor gamma (PPAR-γ) is involved in a number of physiological processes, including suppressing oxidative stress. PPAR-γ activation has been widely investigated for its cardioprotective effects, making it a viable therapeutic target in illnesses linked to cardiovascular disorders. 11
Nifuroxazide (NFX), an oral nitrofuran antibiotic, is effective against various gram-positive and gram-negative pathogens as well as several types of fungi and protozoa.12,13 NFX effectively treats urinary tract infections and exhibits strong oral absorption. 14 Recent studies have demonstrated the efficiency of NFX in cancer.15–17 It’s interesting to note that recent research found NFX to be a potent inhibitor of signal transducer and activator of transcription 3 (STAT3), which promotes the expression of several genes, including pro-inflammatory genes.13,17,18 Consequently, this study aims to explore the antioxidant, anti-inflammatory, and anti-apoptotic activity of NFX against 5-FU-associated cardiac injury and explore the underlying molecular mechanism by examining the role of NLRP3/STAT-3, PPAR-γ, and Bcl-2-associated X protein (Bax)/ B-cell lymphoma 2(Bcl-2) signals.
Materials and methods
Chemicals
5-FU and NFX were purchased from Sigma (USA). CK-MB (Cat. No. 41254), ALP (Cat. No. 41240), AST (Cat. No. 41270), and LDH (Cat. No. 41220) kits were purchased from SPINREACT (Barcelona, Spain). Primary antibodies for NLRP3 (Cat. No. YPA1480) we purchased from (Biospes, China), while STAT-3 (Cat. No. sc-293151), p-STAT-3 (Cat. No. sc-81523), Bax (Cat. No. sc-20067), Bcl-2 (Cat. No. sc-7382), PPAR-γ (Cat. No. sc-81152), and β-actin (Cat. No. sc-8432) (Santa Cruz Biotechnology, USA).
Animals
In this experimental work, 30 adult male albino rats weighing about 180–210 g were used. Animals were kept in standard cages at a temperature of 25 to 27°C with a 12-h light/dark cycle, fed a regular pellet diet, and given access to water. This study’s protocol was approved by the Umm Al-Qura University Ethical Committee, under Approval No. (HAPO-02-K-012-2024-01-1947) and adhere to the ARRIVE Guidelines.
Experimental design
Thirty male adult Wistar rats were randomly allocated into five groups (n = 6): (I) Control received the vehicle only (0.5% CMC orally for 10 days); (II) NFX (50 mg/kg/day) orally 19 ; (III) group of cardiotoxicity, 5-FU (30 mg/kg) was given intraperitoneal (i.p.) once daily for 5 consecutive days at the end of 5th day pf the experiment and this dosing regimen of 5-FU was selected based on a previous report 20 ; (IV) NFX (25 mg)+5-FU group received NFX (25 mg/kg) orally for 5 days before and 5 days with 5-FU, and (V) NFX (50 mg)+5-FU group given NFX (50 mg/kg) orally for 5 days before and 5 days with 5-FU.
The experiment started with a two-week rat acclimatization period and ended after 10 days post-acclimatization. 24 h after the last treatment. Animals were anesthetized with ketamine (100 mg/kg) 21 and the heart dissected. A part of the heart was removed and fixed for histological and immunohistochemical studies on the heart. Another part was homogenized using phosphate buffer saline and centrifuged for 10 min at 1200 g to measure the levels of biochemical parameters in the heart tissue specimens. In order to conduct a Western blot, the third portion was stored in liquid nitrogen.
Measurement of serum cardiac function parameters
Using commercial kits and following the manufacturer’s instructions, serum concentrations of CK-MB, ALP, AST, and LDH were assessed.
Assessment of cardiac antioxidants and lipid peroxidation
According to the procedures outlined by Ellman 22 and Uchiyama and Mihara, 23 cardiac GSH and MDA contents were assessed. Additionally, the Marklund and Marklund techniques were used to measure the cardiac enzymatic activity of SOD. 24
Evaluation of cardiac NO2- content and MPO activity
The levels of cardiac NO2- and MPO were measured according to the methods described by Montgomery and Dymock (1961) 25 and Krawisz et al. (1984), 26 respectively.
ELISA analysis
Using a sandwich-based immunoassay approach, the Glory Science Company (USA) evaluated the cardiac levels of TNF-α and IL-1β in the heart homogenate. We followed the procedures and guidelines specified by the manufacturers for each of the previous kits utilized in this study.
Histopathological examination
After being fixed for 24 h in 10% formalin, the heart samples were washed in xylol, dehydrated in various alcohols, and embedded in paraffin. Heart tissue that had been fixed in paraffin was cut into slices that were 5-µm thick. The sections were stained with H&E and examined under a light microscope. The histopathological analysis was completed by a single independent pathologist unaware of the experiment’s design or treatment groups. 27
Immunohistochemical analysis
Heart tissue sections were immunoassayed for the primary antibodies PPAR-γ (dilution 1: 100), Bax(dilution 1: 100), and Bcl-2(dilution 1: 100). Cross-sections dewaxed were soaked and placed in an antigen retrieval solution (10 mM citrate buffer for 30 min at 121°C). 5% BSA in Tris-buffered saline is then used for blocking. Heart slices were probed with primary antibodies and left overnight at 4°C. After washing and a 30-min incubation with the secondary antibody, the sections were stained with DAB and hematoxylin counterstained. The brown colour intensity was determined in six fields on each slide using Image J® software.
Western blot
The heart samples were homogenised in a proteinase inhibitor-containing lysis solution. Protein content was calculated using the Bradford technique. 28 50 µg of total proteins were separated using SDS-PAGE and transferred onto PVDF membranes before being blocked in TBST containing 5% skimmed milk. The blocked membranes were examined using anti-STAT-3 (dilution 1: 1000), anti-p-STAT-3(dilution 1: 500), anti-NLRP3 (dilution 1: 500), and anti-β-actin (dilution 1: 3000),. The membranes were rinsed three times with TBST before an hour-long incubation with an ALP-conjugated secondary antibody. Following multiple washes, the BCIP/NPT kit was used for detection. The protein band intensity was quantified with Image J® software. Using β-actin, the data were normalised. The original blot was provided as a supplemental file.
Statistical analysis
The data were presented as mean and standard error (SEM). GraphPad Prism 7.0 statistical analysis software and the variance of one-way (ANOVA) for data were used to clarify differences between all groups. A post hoc range test (Tukey’s multiple-comparison test) was used to evaluate the differences between each pair of means. At p < 0.05, the significance test was conducted.
Results
Effect of NFX on cardiac function biomarkers
As shown in Figure 1, 5-FU injection significantly increased CK-MB, AST, ALP, and LDH serum levels as compared to the control groups. Conversely, treatment with NFX reverted these effects and decreased these markers levels significantly in a dose-dependent manner (Figure 1(a)–(d)). Effect of NFX on cardiac function biomarkers. NFX significantly decreased CK-MB (A), AST (B), ALP (C), and LDH (D) serum levels. a = significant versus control; b = significant versus 5-FU; c = significant versus 5-FU + NFX 25 mg.
Effect of NFX on histology changes induced by 5-FU
Normal cardiac histological appearance was observed in normal control and NFX-treated rats. In contrast, rats treated with 5-FU exhibited clear vacuoles between the bundles of the myocardium. Also, vacuolar degeneration of the blood vessel wall, as well as vacuoles between the muscle and loss of striation of the cross-section of myocardiocytes, were observed. Additionally, hyperemia of the blood vessels was detected. Interestingly, treatment with NFX reverted these effects and decreased these histopathological changes significantly in a dose-dependent manner, except for slight hyperemia observed in some sections of rats treated with a small dose of NFX (Figure 2). Effect of NFX on histology changes induced by 5-FU. Normal cardiac histological appearance was observed in normal control (A) and NFX-treated rats (B). In contrast, the rat heart received 5-FU, showing clear vacuoles between the bundles of the myocardium (black arrow). Also, vacuolar degeneration (green arrow) of the blood vessel wall (V-letter), as well as vacuoles in between the muscle and loss of striation of the cross-section of myocardiocytes (M-letter), were observed. Additionally, hyperemia of the blood vessels was detected (red star) (C, D, & E). Interestingly, treatment with NFX reverted these effects and decreased these histopathological changes significantly in a dose-dependent manner, except for slight hyperemia observed in some sections of rats treated with a small dose of NFX (red star). H&E(Scale bar = 50 µm; 400x magnification).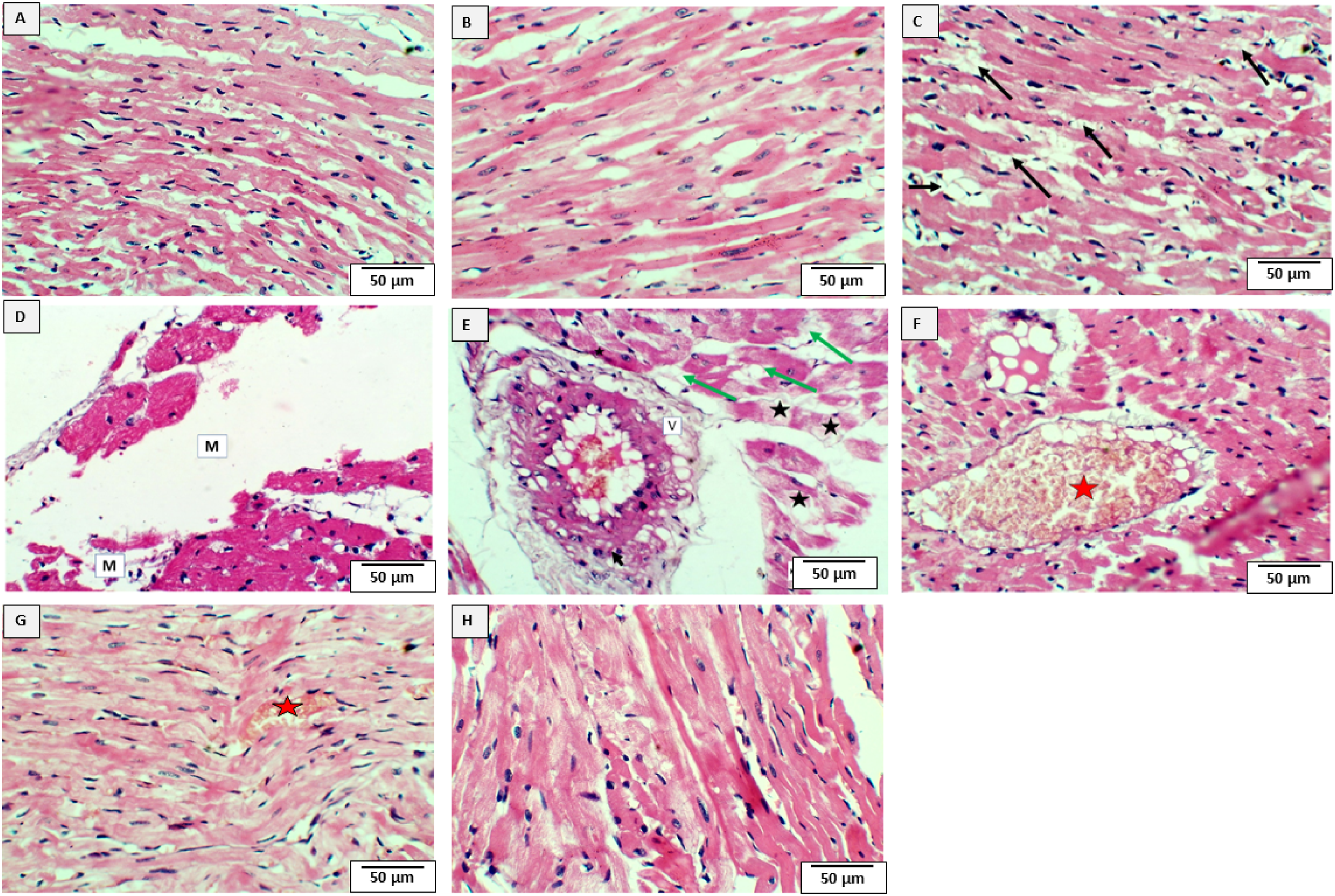
Effect of NFX on 5-FU-induced cardiac oxidative stress
5-FU remarkably increased cardiac MDA content, while co-administration of NFX caused a substantial decrease in MDA content (Figure 3(a)). Moreover, the levels of antioxidant enzymes GSH and SOD were potently declined in the 5-FU-treated, while NFX supplementation significantly restored the levels of GSH and SOD, maintaining the oxidant-antioxidant balance in a dose-dependent manner (Figure 3(b) and (c)). Effect of NFX on 5-FU-induced cardiac oxidative stress. NFX caused a substantial decrease in MDA content (a) and increased GSH (b) and SOD (c). a = significant versus control; b = significant versus 5-FU; c = significant versus 5-FU + NFX 25 mg.
Additionally, the immunohistochemistry analysis revealed that 5-FU caused a significant downregulation in PPAR-γ expression. Interestingly, NFX significantly upregulated cardiac PPAR-γ dose-dependently (Figure 4). Effect of NFX on cardiac PPAR-γ expression level in 5-FU-intoxicated rats. The immunohistochemistry analysis revealed that NFX significantly upregulated cardiac PPAR-γ dose-dependently. a = significant versus control; b = significant versus 5-FU; c = significant versus 5-FU + NFX 25 mg. (Scale bar = 50 µm; 400x magnification).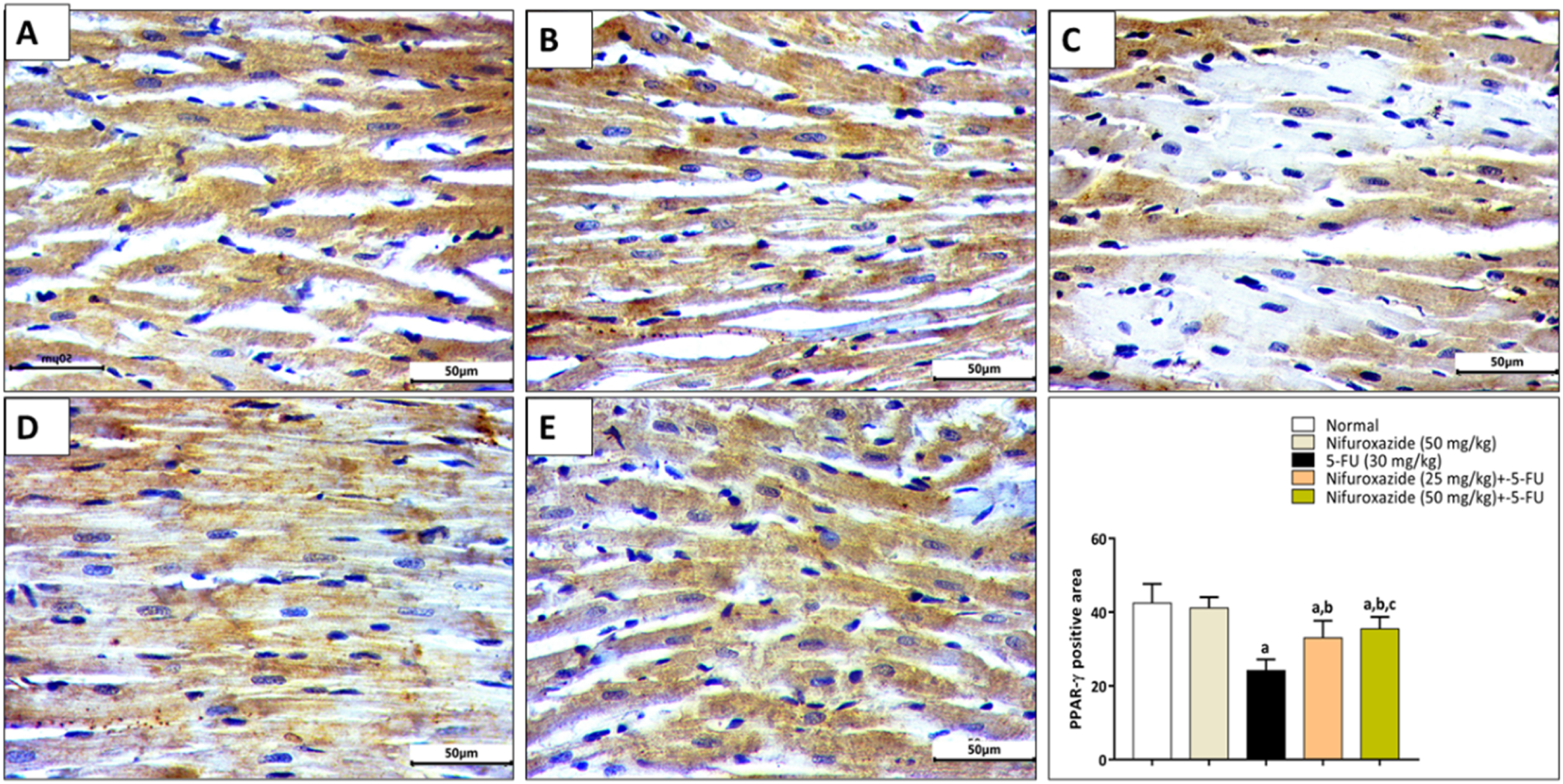
Effect of NFX on 5-FU-induced cardiac inflammation
In the current study, 5-FU increased MPO and NO2 levels. Conversely, the administration of NFX decreased the levels of MPO and NO2 (Figure 5(a) and (b)). Notably, these results were statistically significant, and the NFX effect is dose-dependent. Effect of NFX on 5-FU-induced cardiac inflammation. NFX decreased the levels of MPO (a) and NO2 (b). a = significant versus control; b = significant versus 5-FU; c = significant versus 5-FU + NFX 25 mg.
Concerning the pro-inflammatory cytokines, we found that 5-FU caused an increase in TNF-α and IL-1β concentrations. This effect was inhibited after co-treatment with NFX, which decreased their levels dose-dependently (Figure 6). Effect of NFX on inflammatory cytokines in 5-FU-induced cardiac inflammation. NFX decreased TNF-α (a) and IL-1β (b) levels. a = significant versus control; b = significant versus 5-FU; c = significant versus 5-FU + NFX 25 mg.
Mechanistically, we found out that 5-FU significantly increased the protein expression levels of NLRP3, STAT-3, and p-STAT-3 in comparison to control groups. However, the coadministration of NFX dramatically reduced the expression of NLRP3 and STAT-3 phosphorylation (Figure 7). It is worth noting that the higher the NFX dose (50 mg/kg), the higher the inflammatory activity. Effect of NFX on cardiac NLRP3 and STAT3 expression level in 5-FU-intoxicated rats. The W.B. analysis revealed that NFX significantly downregulated cardiac NLRP3 (a), STAT3 (b), and p-STAT3 (c). a = significant versus control; b = significant versus 5-FU; c = significant versus 5-FU + NFX 25 mg.
Effect of NFX on 5-FU-induced cardiac apoptosis
Furthermore, we wanted to investigate the anti-apoptotic action of NFX, so we used the immunohistochemistry technique to determine the levels of Bax and Bcl-2. Firstly, we showed that 5-FU increased the levels of Bax, while cotreatment with NFX reverted this effect and significantly decreased the Bax levels in a dose-dependent manner (Figure 8). Effect of NFX on cardiac Bax expression level in 5-FU-intoxicated rats. The immunohistochemistry analysis revealed that NFX significantly downregulated cardiac Bax dose-dependently. a = significant versus control; b = significant versus 5-FU; c = significant versus 5-FU + NFX 25 mg. (Scale bar = 50 µm; 400x magnification).
Additionally, the levels of Bcl-2 were reduced in 5-FU-injected rats. Interestingly, the coadministration with NFX restored the levels of Bcl-2 and inhibited cardiac apoptosis (Figure 9). Effect of NFX on cardiac Bcl2 expression level in 5-FU-intoxicated rats. The immunohistochemistry analysis revealed that NFX significantly upregulated cardiac Bcl2. a = significant versus control; b = significant versus 5-FU; c = significant versus 5-FU + NFX 25 mg. (Scale bar = 50 µm; 400x magnification).
Discussion
Currently, 5-FU is utilized to treat several malignant conditions. However, its clinical utility is linked to severe heart damage.29,30 Therefore, it is important to look for preventive substances to protect against these detrimental consequences induced by 5-FU. With the help of our model, we were able to investigate several damage processes and the potential contribution of NFX to their prevention.
5-FU caused myocardial damage, as evidenced by the considerable rise of serum CK-MB, AST, ALP, and LDH, and the histological signs of cardiac damage. These findings are consistent with earlier research about 5-FU-induced cardiac toxicity through the disruption of cardiac biomarkers. 31 Similar to the current study, another study found that 5-FU administration elevated AST, ALP, and CK-MB levels in rats with 5-FU-induced cardiotoxicity. 32 It is possible that myocardial cell injury, which eventually causes more serum cardiac enzyme leakage, is the cause of a rise in these enzymes’ concentrations in the blood. In contrast, when we used NFX, the serum levels of these enzymes significantly decreased. Similarly, in lipopolysaccharide (LPS)-induced ALI and myocarditis, NFX administration was associated with reduced serum cardiac function biomarkers CK-MB and LDH. 33 The outcomes of the biochemical studies are further supported by examining the histological alterations in the heart tissue.
It is unclear what explicitly the causes of 5-FU cardiotoxicity. Oxidative stress is one of the participating factors of the cardiac toxicity of 5-FU. 34 Oxidative stress is an imbalance between the elimination and production of ROS. Proteins, nucleic acids, and macromolecules may undergo alterations and permanent damage due to ROS, 35 inducing inflammatory responses and cell destruction. 36 Myocardial free radical damage has been linked to 5-FU-induced cardiotoxicity. 37 In our study, 5-FU increased lipid peroxidation by increasing MDA content and reduced the antioxidant enzymes GSH and SOD, which is similar to what was reported by Arafah et al. (2022). 38 While upon administration of NFX, MDA levels were reduced along with restoration of GSH and SOD levels, maintaining the oxidative balance. Collectively, NFX effectively mitigated oxidative stress through the upregulation of antioxidant enzyme activity.
In the same setting, we investigated the levels of PPAR-γ by Immunohistochemistry. Cardiomyocytes are known to express PPAR-γ at significant levels. 39 PPAR-γ activation contributes to maintaining many cardiac functions.40,41 It also helps to prevent inflammation. Moreover, ROS are suppressed by PPAR.41,42 We discovered that PPAR-γ levels were downregulated in the 5-FU-treated group and upregulated after NFX administration. Some researchers have shown that the direct suppression of NF-κB p65 as well as the inhibition of STAT-3 are two mechanisms of PPAR-γ anti-inflammatory effects.43,44
Nitric oxide (NO) is a signaling molecule that has been linked to inflammation.45,46 Numerous studies have demonstrated the impact of 5-FU on NO production. 47 Our data shows 5-FU increased NO levels, explaining its induced inflammatory injury. The administration of NFX counteracted this effect. Additionally, the neutrophil-related enzyme MPO has been investigated to measure neutrophil infiltration into inflamed tissue. 48 The present study’s considerable rise in myocardial MPO activity indicates neutrophil infiltration and cardiac tissue inflammation caused by 5-FU. These results are consistent with earlier studies. 49 Intriguingly, after NFX administration, the level of MPO was decreased.
Additionally, recent studies have shown that 5-FU changes the expression of pro-inflammatory cytokines. 50 Our data revealed similar findings, as after administration of 5-FU, the levels of TNF-α and IL-1β were increased. On the other hand, co-treatment with NFX significantly mitigated these effects, and findings coincided with an earlier investigation. 33 Accordingly, our data showed that NFX has an anti-inflammatory activity via regulating the NO, MPO, and pro-inflammatory cytokines.
Afterward, to study the anti-inflammatory molecular mechanism, we evaluated the levels of NLRP3, STAT-3, and p-STAT-3. It is well known that numerous inflammatory diseases’ etiology involves the NLRP3 inflammasome. 51 STAT-3 activation is key in chemotherapy-induced toxicities.52–54 Notably, NFX inhibits NLRP3/IL-1β signaling to reduce LPS-induced ALI and myocardial damage. 33 Moreover, NFX inhibits the STAT3 signaling pathway in cancer cells. 14 Earlier investigation showed that the expression of NLRP3 and IL-1β was increased by 5-FU administration. 55 According to our data, 5-FU increased NLRP3, STAT-3, and p-STAT-3 expressions. In contrast, NFX dampens these effects in a dose-dependent manner, proving its anti-inflammatory by downregulating NLRP3 and STAT-3 signals. As mentioned above, PPAR-γ has anti-inflammatory activity. Earlier findings demonstrated the anti-inflammatory function of PPAR-γ, which inhibits NLRP3 inflammasome activation by interacting with NLRP3. 56 Other findings demonstrate that, in cerulein-stimulated pancreatic acinar cells, PPAR-γ inhibits STAT-3 by directly interacting with STAT-3. 57 Collectively, NFX has anti-inflammatory effects mediated by upregulating PPAR-γ and subsequently decreasing the levels of NLRP-3 and STAT-3.
Finally, the anti-apoptotic activity of NFX is evaluated. It has been reported that 5-FU increased the proapoptotic protein, Bax, and downregulated anti-apoptotic Bcl-2 levels.58,59 Similarly, our data demonstrated that Bax is upregulated while Bcl-2 is downregulated after 5-FU injection. The opposite was achieved after NFX treatment. Supposedly, there is a cross-talk between PPAR-γ and NLRP3/STAT-3 and PPAR-γ and Bax/Bcl-2 apoptotic genes. Recent studies demonstrated that cardiomyocyte hypertrophy could be reversed by PPAR-γ pathways, which can also prevent cardiomyocyte apoptosis and change the expression of genes associated with apoptosis, such as Bcl-2/Bax. 60 In conclusion, NFX controls the PPAR-γ, NLRP3/STAT-3, and Bax/Bcl-2 signalling pathways to exert antioxidant, anti-inflammatory, and anti-apoptotic effects.
Our study’s limitations included using only male rats, and potential sex differences in cardiotoxicity were not investigated. Also, the long-term effects of NFX administration were not examined. Additionally, future studies should evaluate the efficacy of NFX in human cardiomyocytes and clinical models. Thus, these should be considered in future work.
Conclusions
NFX significantly attenuates cardiotoxicity induced by 5-FU dose-dependently. NFX attenuated the oxidative damage caused by 5-FU mediated by PPAR-γ signal activation. Moreover, NFX mitigated 5-FU-induced inflammation by suppressing NLRP3/STAT-3 signal activation. Additionally, NFX mitigated 5-FU-induced apoptosis by regulating the Bax/Bcl-2 signal. This study supports NFX’s potential as a therapeutic agent for clinical application in cardiotoxicity management.
Supplemental Material
Supplemental Material - Nifuroxazide attenuated 5-fluorouracil-induced cardiac intoxication by regulating NLRP3/STAT-3, PPAR-γ, and apoptosis signals
Supplemental Material for Nifuroxazide attenuated 5-fluorouracil-induced cardiac intoxication by regulating NLRP3/STAT-3, PPAR-γ, and apoptosis signals by Nouf S. Al-Abbas, Nehad A. Shaer in Human & Experimental Toxicology
Footnotes
Author’s note
The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
Acknowledgement
The authors express their appreciation to Umm Al-Qura University, Makkah, Saudi Arabia, and all technicians assist complete our research.
Ethical considerations
This study was approved by the Umm Al-Qura University Ethical Committee (approval no. HAPO-02-K-012-2024-01-1947) on 01/01/2024. This study was approved by the Umm Al-Qura University Ethical Committee (HAPO-02-K-012-2024-01-1947) and conducted in compliance with the ARRIVE guidelines for animal research. All procedures were performed according to the Guide for the Care and Use of Laboratory Animals, and efforts were made to minimize animal distress. Animals were housed under controlled temperature and humidity conditions with a 12-h light/dark cycle. Anesthesia was induced using ketamine (100 mg/kg), and euthanasia was performed using overdose anesthesia followed by cervical dislocation in accordance with the AVMA guidelines.
Author Contributions
“Conceptualization, N.S.A. and N.A.S.; methodology, N.S.A. and N.A.S.; software, N.S.A. and N.A.S.; validation, N.S.A. and N.A.S.; formal analysis, N.S.A. and N.A.S.; investigation, N.S.A. and N.A.S.; resources, N.S.A. and N.A.S.; data curation, N.S.A. and N.A.S.; writing—original draft preparation, N.S.A. and N.A.S.; writing—review and editing, N.S.A. and N.A.S.; visualization, N.S.A. and N.A.S.; supervision, N.S.A. and N.A.S.; project administration, N.S.A. and N.A.S.; funding acquisition, N.S.A. and N.A.S. All authors have read and agreed to the published version of the manuscript.”
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that was assessed during the duration of the study is all included in this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
