Abstract
The present study was designed to investigate the protective effects of carvacrol against thioacetamide (TAA)-induced oxidative stress, inflammation and apoptosis in liver of Wistar rats. In this study, rats were subjected to concomitant prophylactic oral pretreatment of carvacrol (25 and 50 mg kg−1 body weight (b.w.)) against the hepatotoxicity induced by intraperitoneal administration of TAA (300 mg kg−1 b.w.). Efficacy of carvacrol against the hepatotoxicity was evaluated in terms of biochemical estimation of antioxidant enzyme activities, histopathological changes, and expressions of inflammation and apoptosis. Carvacrol pretreatment prevented deteriorative effects induced by TAA through a protective mechanism in a dose-dependent manner that involved reduction of oxidative stress, inflammation and apoptosis. We found that the protective effect of carvacrol pretreatment is mediated by its inhibitory effect on nuclear factor kappa B activation, Bax and Bcl-2 expression, as well as by restoration of histopathological changes against TAA administration. We may suggest that carvacrol efficiently ameliorates liver injury caused by TAA.
Introduction
It has been reported that every year there has been 13% increase in the death rate from liver-related diseases.
1
Among deaths related to liver, 77% were due to viral hepatitis, 14% were associated with carvacrol abuse, and 9% were related to hepatocellular carcinoma.
2
Recent research studies and technical applications draw the interest to the utility of TAA preparation of photocatalytic nanocomposites, mesoporous nanomaterials, and metal chalcogenide thin films.
3
TAA was first used to control the decay of oranges and then as a fungicide.
4
Now, it is being used in leather, textile, and paper industries as an accelerator in the vulcanization of buna rubber and as a stabilizer for motor fuels.
3
It has been evaluated that prolonged administration of TAA cause hyperplastic liver nodules, liver cell adenomas, and hepatocarcinomas.
5
It is well understood that in the liver TAA is
There is a long history that environment has provided a remarkable number of modern drugs obtained from natural sources and are used in traditional medicine. Monoterpenes are plant products widely distributed in the plant kingdom and are present as essential oils. Monoterpenes are also known to possess antioxidant, chemotherapeutic, and chemopreventive properties in different cancer models.
8
Monoterpenoids possess different biological properties such as antioxidative, anti-inflammatory, and have preventive and therapeutic effects against various diseases.
9,10
Furthermore, monoterpenoids help in the induction of enzymes that affect carcinogen metabolism and also inhibit tumor promoter activities, which are implicated in the process of carcinogenesis.
11
Carvacrol (2-methyl-5-isopropyl phenol), which belongs to the family Labiatae, is a major monoterpenic phenol, which occurs in various essential oils among the plant species like
Materials and methods
Chemicals
Carvacrol, thioacetamide, reduced glutathione (GSH), oxidized glutathione (GSSG), nicotinamide adenine dinucleotide phosphate reduced (NADPH), flavin adenine dinucleotide (FAD), bovine serum albumin (BSA), and bisbenzamide were obtained from Sigma (St Louis, Missouri, USA). All the antibodies, chemicals, and reagents used were of the highest purity and standard commercially available.
Animals
For the present study, male Wistar rats (aged 8 weeks and weighing 150–200 g) were obtained from the Central Animal House Facility of Hamdard University, New Delhi, India, and were housed in a ventilated room at the temperature of 25 ± 5°C under a 12 h light/12-h dark cycle. The animals were acclimatized for 1 week before beginning the experimental study and had free access to standard laboratory feed (Hindustan Lever Ltd., Bombay, Maharashtra, India) and water ad libitum. The study was approved by the Committee for the Purpose of Control and Supervision of Experimental Animals (CPCSEA) with the registration number IAEC No: 173/CPCSEA and date of registration 28th January 2000.
Treatment regimen
Male Wistar rats were divided into five groups with six animals in each group. Group I was given corn oil (2.5 ml kg−1 body weight (b.w.)) Group II served as toxicant group/intraperitoneally administered TAA dose (300 mg kg−1 b.w.) freshly dissolved in saline. Carvacrol was orally administered at two doses, 25 and 50 mg kg−1 b.w. to groups III and IV, respectively, for 14 consecutive days. Groups II, III, and IV were given TAA intraperitoneally (300 mg kg−1 b.w.) on day 14. Group V was used only to ensure that the higher dose of carvacrol (50 mg kg−1 b.w.) does not produce any kind of hepatotoxicity, if given alone. After 24 h of the TAA injection, rats were killed by cervical dislocation under mild anesthesia, and blood samples were taken for various serological parameters. Liver samples were taken at the same time for immunohistochemistry and various biochemical and histological parameters.
PMS preparation
Postmitochondrial supernatant (PMS) preparation was carried out by the method described by Tahir et al. 19 Liver was removed and cleaned with ice-cold saline (0.85% sodium chloride). Liver tissues were homogenized in chilled phosphate buffer (0.1 M, pH 7.4) using a homogenizer (Remi Process Plant And Machinery Ltd, Mumbai, Maharashtra, India) and were centrifuged at 3000 r min−1 for 10 min at 4°C in a Cooling Centrifuge (Remi Process Plant And Machinery Ltd) to separate the nuclear debris. The aliquot thus obtained was centrifuged at 12,000 r min−1 for 20 min at 4°C to obtain PMS, which was used as the source of enzymes.
GSH estimation
GSH was determined as described by Ahmad et al.
20
Then, 1 ml of 10% PMS was mixed with 1 ml of 4% sulfosalicylic acid, incubated at 4°C for a minimum time period of 1 h, and then centrifuged at 4°C at 1200
GR activity
Glutathione reductase (GR) activity was measured as described by Rashid et al. 21 The reaction mixture consisted of 1.65 ml phosphate buffer (0.1 M, pH 7.6), 0.1 ml NADPH (0.1 mM), 0.05 ml GSSG (1 mM), 0.1 ml ethylenediaminetetraacetic acid (EDTA; 0.5 mM), and 0.1 ml 10% PMS in a total volume of 2 ml. Enzyme activity was assessed at 25°C by measuring the disappearance of NADPH at 340 nm and was expressed as nanomole of NADPH oxidized per minute per milligram protein using molar extinction coefficient of 6.22 × 103 M−1 cm−1.
GPx activity
Glutathione peroxidase (GPx) activity was estimated as described by Khan et al. 22 A total of 2 ml volume consisting of 0.1 ml EDTA (1 mM), 0.1 ml sodium azide (1 mM), 1.44 ml phosphate buffer (0.1 M, pH 7.4), 0.05 ml GR (1 IU ml−1), 0.05 ml reduced GSH (1 mM), 0.1 ml NADPH (0.2 mM), 0.01 ml hydrogen peroxide (H2O2; 0.25 mM), and 0.1 ml 10% PMS was used. The depletion of NADPH at 340 nm was recorded at 25°C. The activity of the enzyme was expressed as nanomole of NADPH oxidized per minute per milligram protein with the molar extinction coefficient of 6.22 × 103 M−1 cm−1.
Assay for LPO
The assay for lipid peroxidation (LPO) was carried out as described by Ahmad et al. et al.
23
The reaction mixture in a total volume of 1 ml contained 0.58 ml phosphate buffer (0.1 M, pH 7.4), 0.2 ml microsomes, 0.2 ml ascorbic acid (100 mM), and 0.02 ml ferric chloride (100 mM). The reaction mixture was incubated at 37°C in a shaking water bath for 1 h and the reaction was stopped by adding 1 ml of 10% trichloroacetic acid. Following the addition of 1 ml 0.67% thiobarbituric acid, all tubes were placed in boiling water bath for 20 min and then shifted to crushed ice-bath before centrifuging at 2500
QR activity
The QR activity was carried out as described by Khan et al. 24 The reaction mixture in a total volume of 3 ml contained 2.13 ml Tris–hydrochloric acid buffer (25 mM, pH 7.4), 0.7 ml BSA, 0.1 ml FAD, 0.02 ml NADPH (0.1 mM), and 50 μl of 10% PMS. The reduction of 2,6-dichlorophenolindophenol (DCIP) was recorded colorimetrically at 600 nm, and the enzyme activity was expressed as nanomole of DCIP reduced per minute per milligram protein using molar extinction coefficient of 2.1 × 104 M−1 cm−1.
XO activity
Xanthine oxidase (XO) activity was carried out by the method described by Khan et al. 22 The reaction mixture consisted of 0.2 ml PMS that was incubated for 5 min at 37°C with 0.8 ml phosphate buffer (0.1 M, pH 7.4). The reaction was started by adding 0.1 ml xanthine (9 mM) and kept at 37°C for 20 min. The reaction was terminated by adding of 0.5 ml ice-cold perchloric acid (10% v/v). After 10 min, 2.4 ml of distilled water was added and centrifuged at 4000 r min−1 for 10 min and milligram of uric acid formed per minute per milligram protein was recorded at 290 nm.
Measurement of liver toxicity markers: serum AST and ALT
Serum aspartate aminotransferase (AST) and alanine aminotransferase (ALT) activities were determined by the method followed by Reitman and Frankel. 25 Each substrate, 0.5 ml (2 mM α-ketoglutarate and 200 mM l-alanine or l-aspartate) was incubated for 5 min at 37°C in a water bath. To this, 0.1 ml serum was then added and the volume was adjusted to 1 ml with 0.1 M and pH 7.4 phosphate buffer. The reaction mixture was incubated for exactly 30 and 60 min at 37°C for ALT and AST, respectively. Then, to the reaction mixture, 0.5 ml of 1 mM 2,4-dinitrophenylhydrazine was added, and after another 30 min at room temperature, with the addition of 5 ml of 0.4 N NaOH the color developed was read at 505 nm.
LDH activity
Lactate dehydrogenase (LDH) activity has been estimated in serum by the method described by Khan et al. 26 The assay mixture consisted of 0.2 ml serum, 0.1 ml 0.02 M NADH, 0.1 ml 0.01 M sodium pyruvate, 1.1 ml 0.1 M and pH 7.4 phosphate buffer, and distilled water in a total volume of 3 ml. The enzyme activity was recorded at 340 nm and activity was expressed as nanomole of NADH oxidized per minute per milligram protein.
Estimation of protein concentration
The protein concentration in all the samples was determined by the method described by Nafees et al. 27
Histopathological examination
The livers were quickly removed after killing the rats and were fixed in 10% neutral-buffered formalin solution for histopathological processing. Sections were stained with hematoxylin and eosin before being observed under a microscope (Olympus, Tokyo, Japan) at 400× magnification.
Immunohistochemistry
To examine the protective effects of carvacrol on markers of inflammation and apoptosis in the liver, NFκB, Bax and Bcl-2 expression in the liver were assessed by immunohistochemical staining. Liver sections on polylysine-coated slides obtained were fixed in neutral-buffered formalin, embedded in paraffin, and were treated for NFκB, Bax and Bcl-2 antibodies for immunohistochemical analysis. The procedures were processed according to the manufacturer’s protocol recommended for the NFκB, Bax and Bcl-2 immunohistochemistry with slight modifications. Following deparaffinization and rehydration, sections were irradiated in 0.1 mol l−1 sodium citrate buffer (pH 6.0) in a microwave oven (medium low temperature) for 20 min. Then, the sections were exposed to 3% H2O2 for 10 min to bleach endogenous peroxidases, followed by rinsing 3 times in Tris buffer (pH 7.4) for 10 min. Sections were selectively incubated under humid conditions using an anti-NFκB antibody (1:100; Biolegend, San Diego, California, USA), anti-Bax antibody (1:200; Santa Cruz Biotechnology Inc., Dallas, Texas, USA) and anti-Bcl-2 antibody (1:200; Thermo Fisher Scientific, Waltham, Massachusetts, USA) for overnight at 4°C. The next day, slides were washed 3 times in Tris buffer for 10 min each. The specificity of the antibodies was tested by omission of the primary antibodies and a positive control of rat tonsil tissue. After washing in Tris buffer (pH 7.4), tissues were visualized with 3,3′-diaminobenzidine and counterstained with hematoxylin. Finally, the sections were dehydrated in xylene, mounted with a mixture of distyrene, a plasticizer and xylene (DPX) and cover slipped. Slides prepared for each case were examined using light microscopy. Positive and negative controls were conducted in parallel with NFκB and Bax- and Bcl-2-stained sections. Staining of sections with commercially available antibodies served as the positive control. Negative controls included staining tissue sections with omission of the primary antibody.
Statistical analysis
The differences between groups were analyzed using analysis of variance followed by Dunnet’s multiple comparisons test. All data points are presented as the treatment groups mean ± SEM.
Results
Pretreatment with carvacrol modulate the levels of AST, ALT, and LDH activity in the serum of Wistar rats.
TAA treatment caused significant increase in the levels of AST (96.67 ± 0.56, b
Results of pretreatment of carvacrol on serum enzymes like AST, ALT, and LDH.a

AST: aspartate aminotransferase; ALT: alanine aminotransferase; LDH: lactose dehydrogenase; TAA: thioacetamide; NADH: nicotinamide adenine dinucleotide; b.w.: body weight.
aResults represent mean ± SE of six animals per group. Carvacrol: D1 = 25 mg kg−1 b.w.; D2 = 50 mg kg−1 b. w.
b
c
d
e
f
Carvacrol pretreatment decreased MDA formation and XO activity
MDA formation was measured to demonstrate the LPO in the liver of rats against TAA-induced toxicity. A significant increase (35.83 ± 6.63, c
Results of pretreatment of carvacrol on LPO, GSH, and XO activity.

LPO: lipid peroxidation; GSH: glutathione; XO: xanthine oxidase; MDA: malondialdehyde; CDNB: 1-chloro-2,4-dinitrobenzene; TAA: thioacetamide; b.w.: body weight.
aResults represent mean ± SE of six animals per group. Carvacrol: D1 = 25 mg kg−1 b.w.; D2 = 50 mg kg−1 b. w.
b
c
d
e
f
Carvacrol pretreatment ameliorates the activities of antioxidant enzymes
Effect of carvacrol on hepatic GSH levels was evaluated. The level of GSH was decreased significantly (0.166 ± 0.01, c
A significant decrease (207.37 ± 5.53, b
Results of pretreatment of carvacrol on antioxidant enzymes like QR, GR, and GPx.

QR: quinone reductase; GR: glutathione reductase; GPx: glutathione peroxidase; TAA: thioacetamide; b.w.: body weight; NADpH: nicotinamide adenine dinucleotide phosphate reduced.
aResults represent mean ± SE of six animals per group. Carvacrol: D1 = 25 mg kg−1 b.w.; D2 = 50 mg kg−1 b. w.
b
c
d
e
Rats treated with TAA showed a significant reduction (104.91 ± 8.21, b
Rats treated with TAA showed a significant reduction (101.95 ± 7.02, b
Effect of carvacrol on histological changes
The liver architecture of control group was normal, devoid of any signs of inflammatory changes with well-preserved cytoplasm and nucleus. In group II, TAA induced centrilobular necrosis including vascular and sinusoidal congestion with infiltration of lymphocytes and macrophages, which are indicative hepatocellular injury as shown by arrows. Pretreatment with carvacrol reverted these changes in liver marked by less sinusoidal congestion and less inflammatory cell infiltration. Liver of rats treated with only higher dose of carvacrol does not show any alterations from normal liver histology as shown in Figure 1.
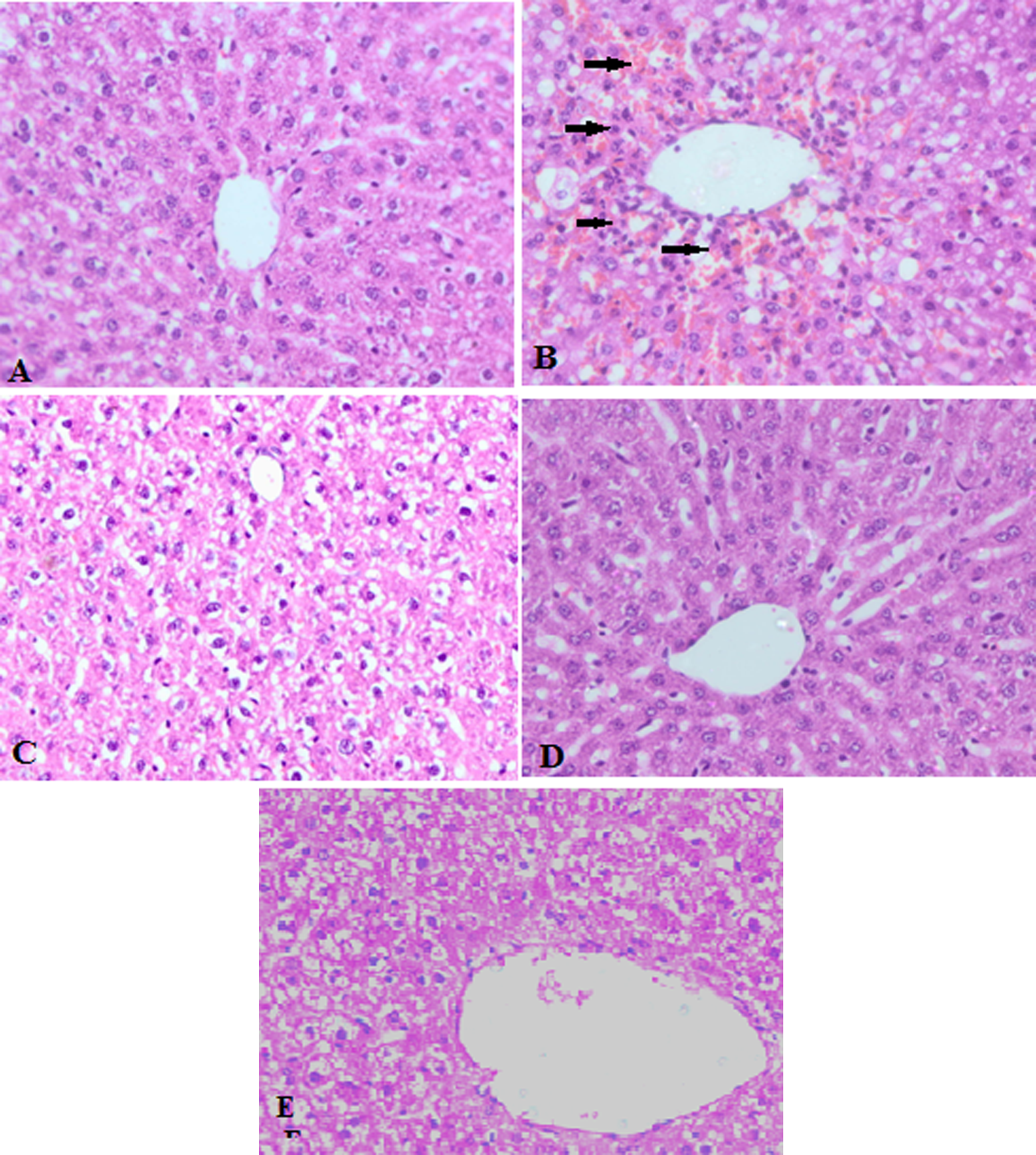
Representative photomicrographs of liver histopathology (×400). (a) Liver of control rats showing normal histology. (b) Liver of rats after TAA (300 mg kg−1 b.w.) administration showing centrilobular necrosis, infiltration of lymphocytes, and macrophages. There is sinusoidal congestion with red blood cells as shown by arrows. (c) Liver of rats fed with low dose (25 mg kg−1 b.w.) of carvacrol show mild architectural damage, less sinusoidal congestion, and less inflammatory cell infiltration. (d) Liver of rats treated with higher dose (50 mg kg−1 b.w.) of carvacrol showed negligible destruction of liver tissue. (e) Liver of rats treated with higher dose of carvacrol (50 mg kg−1 b.w.) alone does not show any alterations from normal liver histology. TAA: thioacetamide; b.w.: body weight.
Effect of carvacrol on expression of NFκB protein
The expression of NFκB protein is given in Figure 2. The immunohistochemical study revealed that there was no nuclear staining of NFκB observed in the control group, while TAA-treated group showing intense and positive stained nucleus as shown by arrows in group II (only TAA treated) when compared with the control group. There was moderate nuclear staining of NFκB protein in group III at lower dose of carvacrol and in group IV at higher dose of carvacrol showed negligible NFκB immunostaining of nucleus at high dose of carvacrol as shown by arrows. There was no significant difference in NFκB immunostaining of liver in group V as compared to group I.

Representative photomicrographs of NFκB protein expression (×400). (a) In group I NFκB protein immunostaining in control group showed no staining. (b) In group II, NFκB protein immunostaining in TAA-treated group showing intense and positive stained nucleus as shown by arrows. (c) NFκB immunostaining of liver treated with low dose of carvacrol showing very weak and diffused staining of hepatocytes as shown by arrows. (d) Almost negligible NFκB immunostaining at high dose of carvacrol was observed as shown by arrows. (e) There was no significant difference in NFκB immunostaining of liver in group V as compared to group I. NFκB: necrotic factor kappa B; TAA: thioacetamide.
Effect of carvacrol on expression of Bax and Bcl-2 protein
The expressions of Bax and Bcl-2 proteins are given in Figures 3 and 4. The immunohistochemical study revealed that in control group no detectable Bax staining was observed. In group II (TAA treated) more intense staining was observed as shown by arrows. At lower dose of carvacrol less intense staining was observed and at higher dose of carvacrol less and much diffused staining was observed as shown by arrows. The immunohistochemical study of Bcl-2 revealed moderate expression in the control group whereas pretreatment with carvacrol leads to the higher expression dose dependently when compared with only TAA-treated group as shown by arrows. There was no significant difference in Bax and Bcl-2 immunostaining of liver in group V as compared to group I.

Representative photomicrographs of Bax protein expression (×400). The immunohistochemical study revealed that in (a) group I (control group), no detectable Bax staining was observed, (b) group II, (TAA treated) more intense staining was observed as shown by arrows, (c) group III, at lower dose of carvacrol less intense staining was observed as shown by arrows, (d) group IV, at higher dose of carvacrol less and much diffused staining was observed as shown by arrows.,(e) group V, there was no significant difference in Bax protein immunostaining of liver as compared to group I. TAA: thioacetamide.
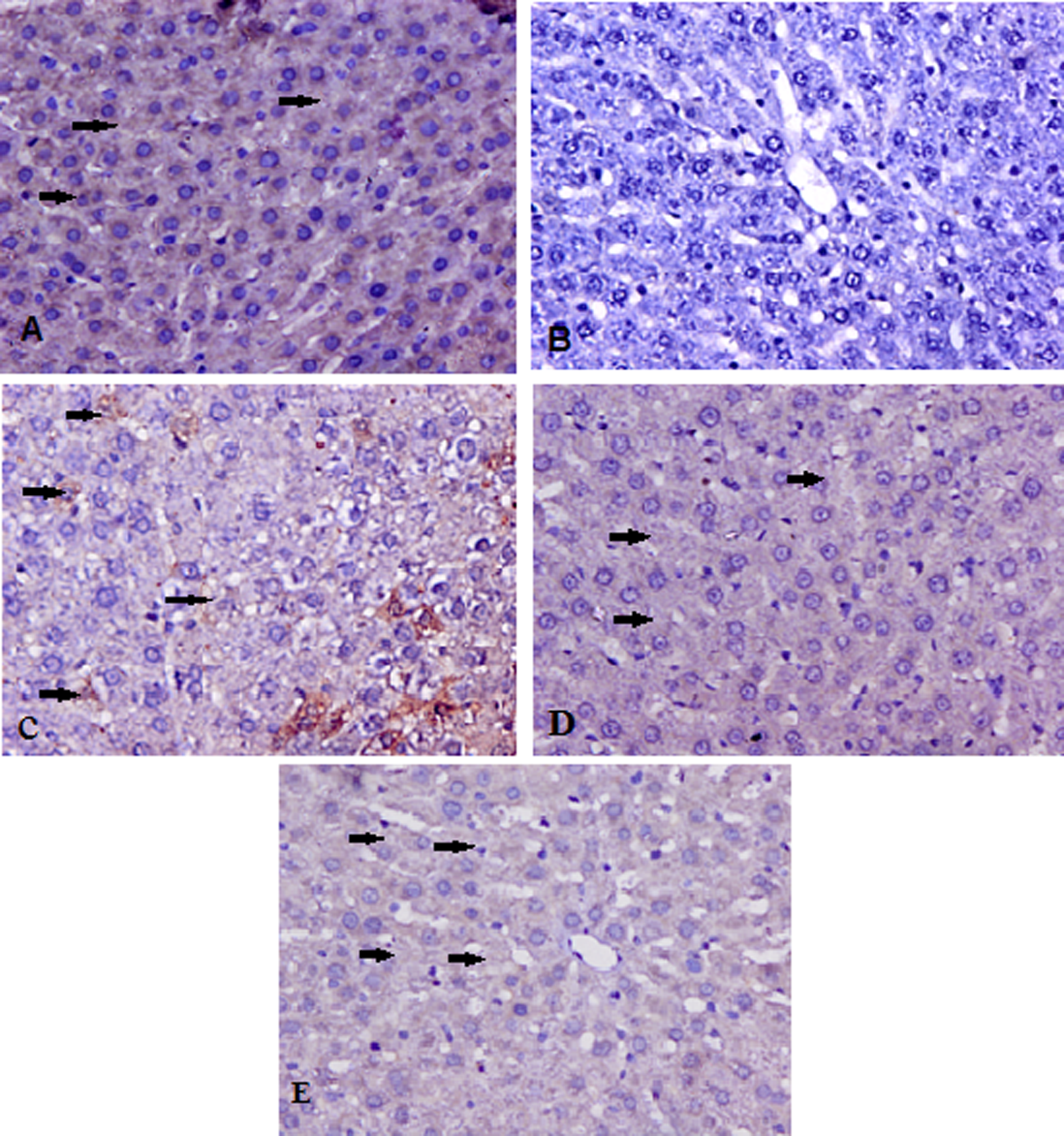
Representative photomicrographs of Bcl-2 protein expression (×400). (a) In group I, immunohistochemical study of Bcl-2 revealed moderate staining in the control group as shown by arrows. (b) In group II (TAA treated), no detectable Bcl-2 staining was observed where as in groups III and IV (c,d) pretreatment with carvacrol leads to more intense staining when compared with TAA only treated group as shown by arrows. (e) There was no significant difference in Bcl-2 protein immunostaining of liver in group V as compared to group I. TAA: thioacetamide.
Discussion
In the present study, it was observed that pretreatment with carvacrol shows protection against TAA-induced liver toxicity. Liver plays a key role in the transformation and elimination of various chemicals and is susceptible to the toxicity caused by these chemicals.
28
TAA is an effective hepatotoxicant and its toxic effects have been accredited to the results obtained from bioactivation, which produces toxic reactive metabolite thioacetamide-
In conclusion, to the best of our knowledge, our study for the first time demonstrated that carvacrol was significantly beneficial in the prevention of TAA-induced liver toxicity, possibly by scavenging reactive free radicals via boosting endogenous antioxidant system and restoration of normal histology of liver. Furthermore, we have shown that in TAA-treated liver tissue, NFκB is constitutively activated and blocking of NFκB activation with carvacrol resulted in suppression of NFκB. This is in line with the findings that many of the pathways that mediate inflammation are under the transcriptional control of NFκB. Additionally, marked decrease in protein expression of Bax and increase in the expression of Bcl-2 protein revealed that carvacrol prevents the apoptosis induced by TAA. Thus, carvacrol may have ameliorative effects against TAA-induced hepatotoxicity. But, further studies to elucidate the exact mechanism at molecular level are appreciated before its clinical application could be recommended.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
The authors are thankful to the university grants commission (UGC), New Delhi, India, for providing the funds to carry out this research work.
