Abstract
Objectives:
Our work was aimed at investigating the impact and regulatory mechanism of coenzyme Q10 (
Methods:
The rats (
Key findings:
HS decreased the activity of the testicular antioxidant system, superoxide dismutase, glutathione peroxidase, and catalase, while the amount of lipid peroxidation (malondialdehyde) was increased. The index of apoptosis and mRNA expression of caspase 3 and Bax were increased, while the mRNA expression levels of Bcl-2, 3β-HSD, and 17β-HSD3 decreased after HS. Exposure to HS decreased the serum testosterone level but increased the activation of pro-inflammatory cytokines (interleukin 1 beta and tumor necrosis factor-alpha). Deleterious effects of HS on the mentioned parameters were reduced when the rats were treated with
Conclusions:
Introduction
Normal spermatogenesis occurs when the testicular temperature is 2–4°C lower than the temperature of the core body. 1 Several factors play an important role in the regulation of the testicular temperature, including scant hair distribution, thin skin, sweat glands, cremaster and dartos muscles, counter-current heat interchange between testicular veins, the pampiniform plexus, and arterial blood. 1,2
It has been reported that scrotal heat stress (HS) can produce reactive oxygen species (ROS).
2
Overproduction of ROS induced by HS can result in the disruption of seminiferous epithelium, testicular germinal atrophy, and spermatogenic arrest, although ROSs such as hypochlorite, hydroxyl (•OH), and superoxide anion radicals (
It has been demonstrated that when testicles are exposed to HS, the level of hydrogen peroxide is increased. Subsequently, lipid peroxidation is enhanced through decreasing enzymatic antioxidant activity, including catalase (CAT), superoxide dismutase (SOD), and glutathione peroxidase (GPx). 1,2
On the other hand, intrinsic (mitochondrial-dependent) and extrinsic apoptotic pathways, which are induced in response to HS, may lead to cell cycle arrest and germ cell death. 1,5 In addition, oxidative stress induced by HS could activate transcription factors such as nuclear factor κB (NF-κB), which is involved in testicular inflammation and male infertility. 6
Several studies have indicated that natural antioxidants attenuate the destructive effects of free radicals on cellular functions by binding to them.
7,8
Hence, they can possibly be used for the mitigation of the adverse effects of HS on the male reproductive system. It has been reported that coenzyme Q10 (
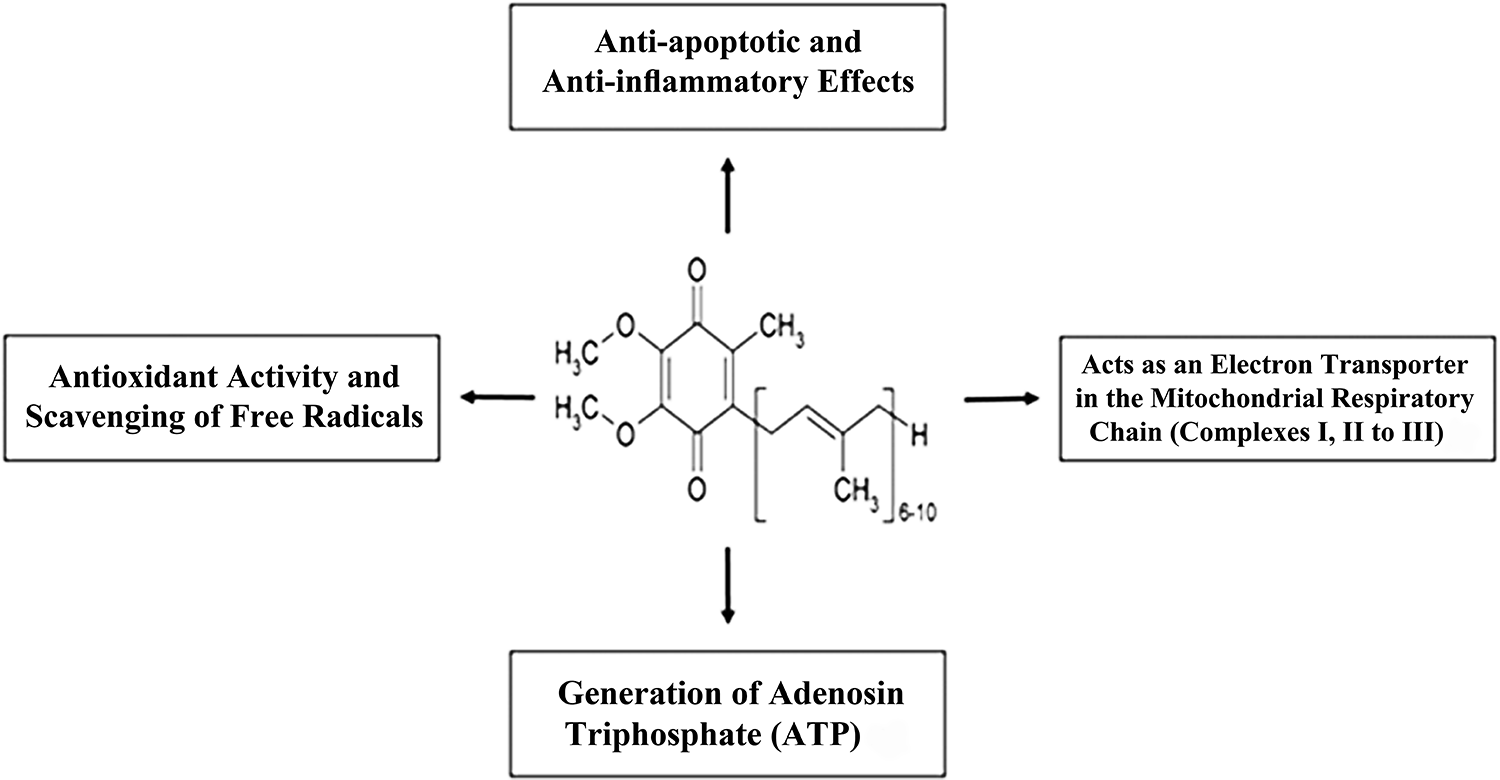
The biological activities and chemical structure of
As a component of the electron-transport chain,
In the light of these pieces of literature, no study has been performed so far to elucidate the impact of
Materials and methods
Animals and work design
In this study, 32 adult male Wistar rats (weighing 250–300 g) were housed in a room with standard conditions (22 ± 2°C temperature and 40–50% humidity) on a 12-h light/12-h dark cycle. This experimental study received an ethical code (AFG.KIMC.REC.1398.78) from the Ethical Committee of Kabul University of Medical Sciences, Afghanistan. Rats were randomly separated into four groups (
Histopathological assessments
The testis samples (right) were fixed in Bouin’s solution at 4°C (48 h), and routine histological techniques were performed. Afterward, paraffin-embedded tissues were serially sectioned at 5 µm thickness, deparaffinized, and then hematoxylin and eosin (H&E) staining was performed. The prepared sections were examined under a light optical microscope (Nikon, Japan) at 200× magnification. For each seminiferous tubule, spermatogenesis was evaluated by mean Johnson’s score (MJS) (ranging 1–10) to assess the testicular injury. 7 Moreover, 30 cross sections of round-shaped seminiferous tubules were photographed. The epithelium height (EH) and tubular diameter (TD) of each seminiferous tubule were measured by Image J 1.51 software based on our previous studies. 7,21,22
Biochemical surveys
The measurement of oxidative stress markers, including malondialdehyde (MDA), SOD, GPx, and CAT, was carried out in the testis tissues of rats according to our previous studies.
7,21
Bradford method was used to determine the concentration of protein in testicular tissues. Lipid peroxidation was evaluated using the thiobarbituric acid-reactive species and quantified as MDA levels; samples homogenized in 5.1% potassium chloride solution to obtain 1:10 (w/v) of whole homogenates, and finally, the level of MDA calculated using a thiobarbituric acid reaction based on the Uchiyama and Mihara method.
23
SOD and GPx activity was assayed by means of commercial kits (Ransod and Ransel, Randox Com, UK) based on the method of Sun et al.
24
and Paglia and Valentine,
25
respectively. Also, the activity of CAT was determined using a commercial kit (Beyotime Biotechnology, Shanghai, China) by detecting the absorbance of the colored product
TUNEL staining
Cellular apoptosis was evaluated by the TUNEL peroxidase kit (Roche, Germany). In brief, the sections were deparaffinized, dehydrated, and treated with proteinase K for 30 min at 37°C. 27 Then, the TUNEL reaction mixture was added to samples (50 µL for each sample), which were incubated for 1 h at 37°C. After incubation, the samples were three times washed with phosphate-buffered saline and incubated with converter-POD for 30 min at 37°C. Afterward, they were treated with 3,3-diaminobenzidine for 10 min and finally counterstained with hematoxylin for 1 min. The index of apoptosis was considered as the percentage of cells that were TUNEL-positive. 7,27
Measurement of testosterone level
To measure the testosterone (T) hormone, blood was centrifuged at 1000 ×
Determination of IL-1β and TNF-α levels
The concentrations of IL-1β and TNF-α in testicular tissues were evaluated using commercial kits (Sigma-Aldrich, Germany) at an absorbance of 450 nm. 6
RNA extraction, complementary DNA synthesis, and real-time quantitative reverse transcription (qRT)-PCR
Total RNA was extracted from testicles (left) using TRIzol reagent (Invitrogen, Paisley, UK, CAS Number: 9048-46-8) according to the manufacturer’s instructions. To eliminate the DNA contamination, DNase I (Invitrogen Life Technologies, Eugene, Oregon, USA, CAS Number: 7732-18-5) treatment was performed based on previous studies.
7,21
The RNA concentration was determined using the ratio of 260-nm and 280-nm absorbance, the obtained RNA was diluted, and complementary DNA (cDNA) generated using the commercial kit (Thermo Scientific, Germany, EU, CAS Number: 7732-18-5).
7,21
The sequences of specific primers for Bax, Bcl-2, caspase 3, 3β-HSD, 17β-HSD3, and glyceraldehyde-3-phosphate dehydrogenase as the endogenous control are presented in Table 1.
29,30
qRT-PCR was performed in a 20-µL reaction containing 7-mL RNase/DNase free water, 1-mL cDNA, 10-mL SYBR Green Master Mix (Sigma-Aldrich, Germany), and 2-mL forward and reverse primers.
7,21
Subsequently, the quantitative analysis of the mentioned-genes was done using the
Primers for qRT-PCR.

GAPDH: glyceraldehyde-3-phosphate dehydrogenase; qRT-PCR: real-time quantitative reverse transcription-PCR.
Results
Testicular weight
Table 2 presents the testicular weight in all the study groups. In the HS group, the weight of testicles was significantly decreased (68.32 ± 4.1%) relative to that of the control group (
Testicular weight and quantitative histological evaluation of seminiferous tubules in each group of the study.a

SD: standard deviation;
a This table shows a comparison of testicular weight and evaluating the EH, TD, and testicular MJS between all groups of the study.
b
c
Histopathological evaluations on testicles
H&E staining (Figure 2) of testicles showed normal morphology of the seminiferous tubule epithelium with dynamic spermatogenesis in the two healthy control groups, namely normal and

Photomicrographs of testicular histology stained with H&E in all the study groups. (a) The healthy control group in which normal architecture of seminiferous tubule and intact spermatogenesis are evident. (b) The HS group in which massive testicular damage, including germ cell destruction, is apparent. (c) The HS group treated with
Evaluation of CoQ10 effect on antioxidant status
Biochemical parameters including SOD, GPx, MDA, and CAT in the testicles (right) of control and experimental rats were measured to assess the efficacy of

The effect of HS and
Evaluation of CoQ10 on mRNA expression of apoptotic and steroidogenic genes
qRT-PCR was performed to investigate the impact of
Relative mRNA expression of apoptosis-related and steroidogenic genes analyzed by qRT-PCR.a
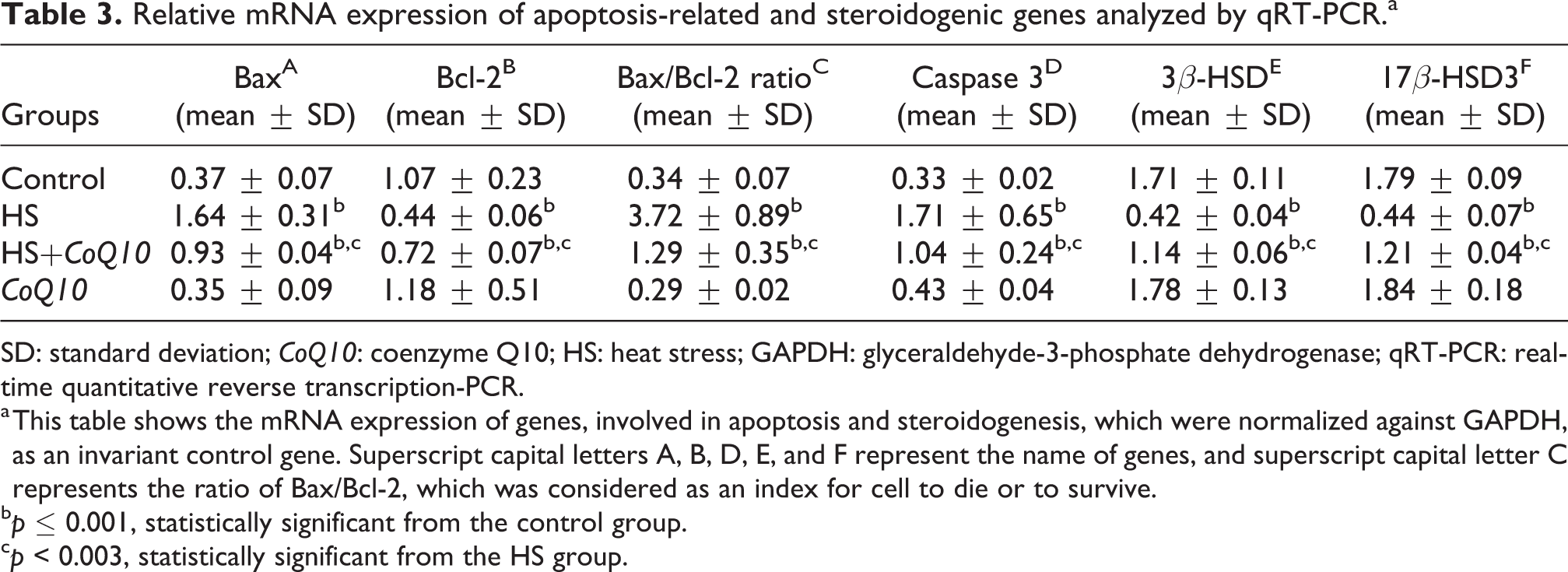
SD: standard deviation;
a This table shows the mRNA expression of genes, involved in apoptosis and steroidogenesis, which were normalized against GAPDH, as an invariant control gene. Superscript capital letters A, B, D, E, and F represent the name of genes, and superscript capital letter C represents the ratio of Bax/Bcl-2, which was considered as an index for cell to die or to survive.
b
c
The serum concentration of testosterone
HS led to a statistically significant decrease in serum testosterone concentration relative to the normal control group (

Serum testosterone (T) level (ng/mL) in all groups of the study. The graph shows a significant reduction in both HS groups (HS and HS+
Evaluation of CoQ10 on pro-inflammatory cytokines in testicles
According to Table 4, HS led to a significant increase in levels of some pro-inflammatory cytokines such as TNF-α and IL-1β relative to the normal control group (
The effects of HS and

SD: standard deviation;
a
b
Evaluation of CoQ10 on apoptosis index
TUNEL staining was directed to examine the impact of

The impact of HS and
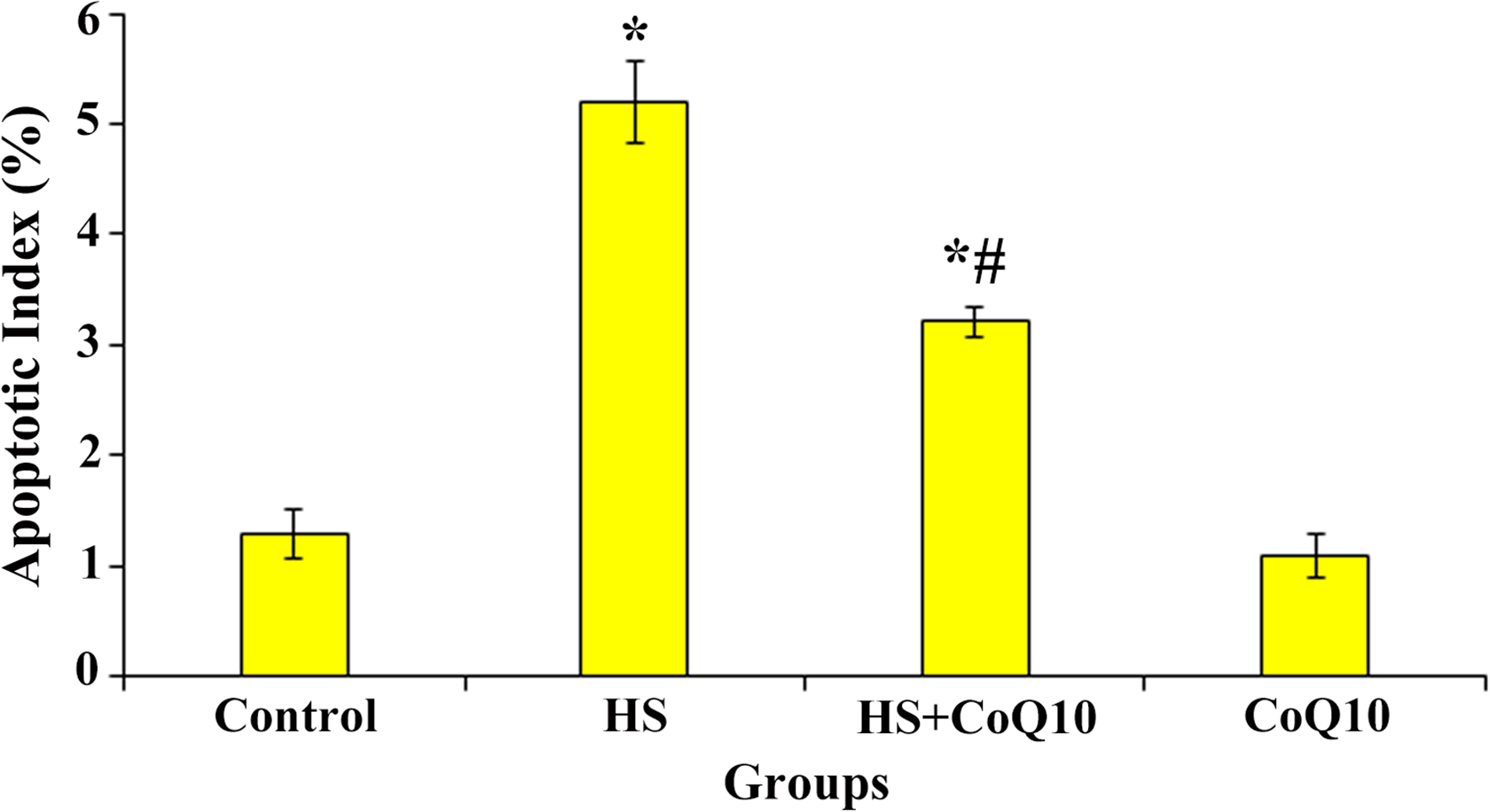
The index of apoptosis in different groups of the study. The graph showing that HS significantly increased the apoptotic index compared to the control group (*
Discussion
This study is probably the first investigation to show that the deleterious effects of hyperthermia in the testicles can be eliminated by the
In this study, there was a significant difference between the treated and untreated rats (groups 3 and 2) when the testicle weight was measured. The weight of testicles under scrotal HS decreased compared with the control rats, while in those rats treated with
Interestingly, in the HS group, histopathological images revealed that HS led to the degeneration in testicular tissue, including disturbance of the cellular arrangement in seminiferous tubules as well as germ cell loss, while the administration of
Our biochemical findings showed that scrotal HS increased the MDA level but decreased the activities of SOD, GPx, and CAT in the testicles of rats.
In our research, a high level of MDA has been considered as an index for the status of lipid peroxidation and oxidative stress. Furthermore, the determination of the grade of testicular tissue damage could be evaluated by measuring the MDA level.
7
Increased lipid peroxidation leads to the formation of free radicals, including hydroxyl (OH•), superoxide (
Another part of our results showed that the mRNA expression of genes (Bax and caspase 3) involved in apoptosis decreased compared to the healthy control group.
It has been suggested that ROS-induced oxidative stress regulates the phosphorylation of Bcl-2 family of proteins, which play significant roles in the programmed cell death pathways with individual proapoptotic (i.e. Bax and Bad) and antiapoptotic (i.e. Bcl-xL and Bcl-2) member genes.
7,21,34
ROS induced by HS can also cause protein and DNA damages in testicles of the male gender and enhance the rate of cellular injury and apoptosis (Figure 7).
31
One study showed that in the testicles under HS, the mRNA expression of Bcl-xL decreased, while the mRNA expression of caspase 3 increased, which is involved in caspase-dependent apoptosis initiating molecular pathways.
28
In this regard, our histopathological results also indicated that the number of TUNEL-positive cells was significantly higher in HS-group than normal group. Alternatively, treatment with

The impact of HS and
On the other hand, the mRNA expression of Bcl-2 as an antiapoptotic protein was significantly decreased in the testes following HS, while in HS group fed with
It has been reported that the antioxidant defense system is indirectly regulated by Bcl-2, leading to a decrease in apoptosis of germ cells.
7
Interestingly, the Bax/Bcl-2 ratio is a predictive marker of apoptosis guiding the cells to decide whether to die or to survive.
21
In the current study, the mentioned ratio was meaningfully higher in the HS-untreated group than the control group, while the administration of
HS exposure can significantly decrease the serum testosterone level and mRNA expression of 3β-HSD and 17β-HSD3, while the levels of the abovementioned parameters were significantly higher in the treated-HS group.
In the male, a large amount of testosterone is secreted by Leydig cells in testicles, while the suprarenal glands in both sexes can produce smaller amounts of it.
39
However, in Leydig cells, steroidogenesis begins when cholesterol is delivered into the inner membrane of the mitochondrial, which is mediated by the steroidogenic acute regulatory (StAR) protein.
40
Inside the mitochondria, the cytochrome P450 cholesterol side-chain cleavage enzyme (P450scc) converts the cholesterol to pregnenolone.
40
Afterward, pregnenolone is metabolized to progesterone, 17α-hydroxyprogesterone, androstenedione, and testosterone by 3β-HSD, 17α-hydroxylase/C17-20 lyase (P450c17), and 17β-hydroxysteroid dehydrogenase, respectively.
40
It has been stated that the expression of highly regulated genes, including 17β-HSD3 and 3β-HSD, is of high importance for the accurate synthesis of testosterone.
28,40,41
One study indicated that an antioxidant source (curcumin) modulated the 3β-HSD gene and prevented HS-induced damage in Leydig cells, promoting androgen biosynthesis that is essential for normal spermatogenesis.
28
Therefore, in our study, these two genes (17β-HSD3 and 3β-HSD) were used to investigate some of the testosterone synthesis pathways. We also suggest that the other pathways involved in T secretion should be investigated in the future to find how
Another part of our results concerning the levels of TNF-α and IL-1β indicated the significant increase in the group exposed to HS compared to the normal group, while in HS group fed with
While pro-inflammatory cytokines play key roles in normal homeostasis of testicles, testicular dysfunction could occur following promotion in their expression levels.
42
Elevated TNF-α and IL-1β levels can decrease the serum testosterone levels in human patients with sepsis and/or burns (Cheol Yi Hong). An in vitro study demonstrated that the treatment with TNF-α or IL-1 resulted in an inhibition of steroidogenesis in Leydig cells
43
by downregulating the expression of StAR, 3β-HSD, P450scc, and P450c17, which are involved in testosterone secretion. A number of studies have reported that pro-inflammatory cytokines, such as TNF-α, IL-1β, and IL-6, can activate NF-κB.
6,43
Activated NF-κB inhibits the transactivation of orphan nuclear receptors, including steroidogenic factor 1 as well as Nur77 (nerve growth factor IB), leading to a decrease in serum T level.
42,43
It seems that the decrease in T levels in our study may also be due to these facts. However, we suggest that
Conclusion
Our results indicate that the administration of
Footnotes
Acknowledgments
The authors would like to thank Dr Mahdi Mohaqiq from US Wake Forest University.
Author contributions
All authors participated in the design, interpretation of the studies and analysis of the data and review of the manuscript; HSH, AD, MD, MM, MAM, and VN have collected the data; HSH, FA, MMHT, and SHAR have written the paper; YKD, HSH, and MNGH have contributed data or analysis tools; HSH, AF, and MD have performed the analysis; and HSH, MD, and MMHT have conceived and designed the analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
