Abstract
5-Fluorouracil is one of the most commonly used anticancer drugs for the treatment of various types of cancer but has potential adverse effects such as intestinal mucositis, renal, hepatic, and reproductive organ toxicity. Attention has been given to approaches to reduce the side effects and improve the therapeutic effectiveness of chemotherapeutic drugs. In this study, we have investigated the protective effect of taurine (Tau) on 5-fluorouracil (5-FU) induced adverse effects in Wistar rats. Animals were divided into four groups with six animals (n = 6) in each group. Group I received vehicle only and served as control group. Groups II, III, and IV animals were given oral gavage of 5-FU at 50 mg/kg body weight for 4 days. Tau was given to the animals of groups III and IV 30 min prior to 5-FU administration. We observed marked elevation in the myeloperoxidase (MPO) activity after 5-FU administration, which was reversed by Tau pretreatment. Histological observation of liver, kidney, intestine, testis, and prostate revealed that 5-FU administration resulted in anomalies like distortion of normal cellular architecture, infiltration of inflammatory cells, and loss of cellular integrity. These histopathological changes were markedly suppressed by Tau treatment. In conclusion, biochemical and histological findings of this study suggest that Tau has strong preventive potential against complications of anticancer drug 5-FU and hence Tau may play an important role in combinational chemotherapy to enhance the therapeutic efficacy of anticancer drugs.
Introduction
5-Fluorouracil (5-FU) is a pyrimidine analogue, and due to its broad antitumor activity as well as its synergistic action with other anticancer drugs, 5-FU is one of the most commonly used chemotherapeutic drugs for the treatment of different types of cancer like gastrointestinal, breast, colorectal, liver, skin, head and neck cancers. 1 –3
5-FU has no antitumor activity itself. The reactive metabolic products of 5-FU like 5-fluoro-uridine-5′-monophosphate inhibits the key enzyme thymidylate synthase resulting in the inhibition of DNA synthesis, 5-fluoro-uridine-5′-triphosphate interferes with RNA and results in impaired processing of RNA, and the third metabolite 5-fluoro-2′-deoxyuridine-5′-triphosphate impairs DNA synthesis. These three metabolites interferes with the central dogma of the genetic materials (DNA and RNA), which is crucial for cellular homeostasis and survival and ultimately results in cellular toxicity. 4 Further, 5-FU gets catalyzed in liver and forms dihydrouracil along with other toxic moieties resulting in toxic manifestation. 5
Oxidative stress and inflammation are the important etiological factors associated with most of the toxic manifestations and disease pathogenesis of the chemotherapeutic drugs. 6,7 Overproduction of reactive oxygen species (ROS) and inflammatory mediators by chemotherapeutic drugs causes damage or modification of cellular macromolecules resulting in up or down expression of regulators of normal cellular physiology. 8,9
Chemotherapeutic treatment of 5-FU is associated with severe toxic effects and varies dose dependently with patient to patient and sometimes leads to discontinuation of chemotherapy. 5-FU targets specifically not only cancer or tumor cells but also normal proliferating cells causing inhibition of normal cell proliferation, genomic instability, overproduction of different reactive toxic moieties or mediators, and necrosis leading to severe toxic effects and complications. Common unbearable complications are gastrointestinal, cardiac, renal, hepatic, diarrhoea, myelosuppression, dermatitis, and reproductive system anomalies. 2,3,10 –15 Therefore, it is important to find out approaches to prevent adverse effects of chemotherapy, thereby increasing the therapeutic effectiveness of anticancer drugs.
Taurine (Tau; 2-aminoethanesulfonic acid), a nonprotein and semi-essential amino acid, is ubiquitously present in most of the mammalian cells and have strong cytoprotective potential. Tau accounts for more than 50% of all free amino acids and is abundant in inflammatory cells. Main source of Tau in the body is from cysteine and methionine metabolism in the liver and dietary intake. Tau plays vital role in various cellular phenomenon such as regulation of osmolytes, antioxidant system, movement of ions, central nervous system, and retina development, reproduction, calcium ion regulation, membrane stabilization, and immunological response. 16,17 It is also used in the prevention and treatment of infectious as well as in inflammatory disorders. 17 Tau has antioxidant, anti-inflammatory, hepatorenal protective, antidiabetic, antimicrobial and antitumorigenic potential. 17,18 –22
Taking the above facts in mind, this study was designed to explore the preventive potential of Tau against 5-FU-induced toxicity in Wistar rats by targeting inflammation and histological changes.
Materials and methods
Chemicals and reagents
Potassium dihydrogen phosphate, dipotassium hydrogen phosphate, o-dianisidine dihyderochloride, hexadecyl trimethyl ammonium bromide, hydrogen peroxide (H2O2), and Tau were purchased from Sigma Aldrich (St Louis, Missouri, USA). 5-FU was supplied by ICN Biomedicals Inc. (Aurora, Ohio, USA). All other reagents used were present in the laboratory and of highest purity.
Animals
Male Wistar rats (weighing 150–200 g and aged 6–8 weeks) were obtained from animal house facility of research center, Prince Sultan Military Medical City (PSMMC), Riyadh, Saudi Arabia. All animals were housed in the animal care facility under room temperature at 22–25°C with 12-h light/12-h dark cycle in standard cages and were given free access to standard laboratory diet and water ad libitum. The animals received humane care in accordance with the Guide for the Care and Use of Laboratory Animals, published by ethical scientific research committee of PSMMC, Riyadh, Saudi Arabia.
Treatment regimen
Animals were divided into four groups with six animals in each group. Group I animals were given vehicle only and serve as control group. Animals of groups II, III, and IV were given oral gavage of 5-FU at the dose of 50 mg/kg body weight for 4 days. Tau was given to the animals of the groups III and IV at the dose of 50 mg and 100 mg/kg body weight, respectively, 30 min before 5-FU administration. All the treatments were carried out for 4 days, and 24 h after the last dose of 5-FU, animals were killed under mild anesthesia.
Tissue processing
After experimental time period, control and treated animals were killed by cervical dislocation under mild anesthesia. Liver, small intestine, kidney, testis, and prostate tissues were excised from each group and washed with ice-cold sodium chloride (0.9%). A piece of tissue from each group was preserved in 10% neutral-buffered formalin (NBF) for histological observation.
MPO activity
Myeloperoxidase (MPO) activity was assayed by the method of Bradley et al. 23 Tissue homogenate was prepared in 50 mM potassium phosphate buffer (pH 6.0) containing 0.5% hexadecyl trimethylammonium bromide. Samples were centrifuged at 14,000 r/min for 25 min at 4°C immediately after three cycles of sonication and freezing–thawing. In 0.1 ml of supernatant, MPO activity was measured, using 2.9 ml phosphate buffer (50 mM, pH 6.0) containing 0.167 mg/ml o-dianisidine dihydrochloride and 1% hydrogen peroxide at 460 nm for 3 min. MPO activity is defined as the quantity of enzyme degrading 1 µmol of H2O2/min at 25°C and is expressed as units per minute per gram tissue.
Histological analysis
For histopathological studies, rats were killed, and a portion of small intestine, kidneys, liver, testis, and prostate was removed and immediately fixed in freshly prepared 10% NBF. Then, each organ was embedded in paraffin wax and blocks were prepared. Sections of each organ (5 µm thick) were cut onto poly-
Mast cell staining
For the detection of mast cell, paraffin-embedded tissue blocks were prepared from formalin fixed solution, and sections of 5 µm were cut on poly-
Statistical analysis
The data from individual groups are presented as the means ± SEM. Differences between groups were analyzed using one-way analysis of variance, followed by Tukey–Kramer multiple comparisons test, and minimum criterion for statistical significance was set at p < 0.05 for all comparisons.
Results
Effect of Tau and 5-FU on MPO activity
MPO is one of the key abundant enzymes secreted by inflammatory cells including neutrophils, macrophages, and microglia and known as important marker of inflammation. We have found that the administration of 5-FU in group II animals lead to significant increase of liver and kidney MPO activity (Figure 1(a) and (b) respectively) as compared to group I animals (***p < 0.001, **p < 0.01). However pretreatment with Tau significantly reduced the MPO activity in groups III and IV animals as compared to group II (#p < 0.05). (Figure 1(a) and (b)).
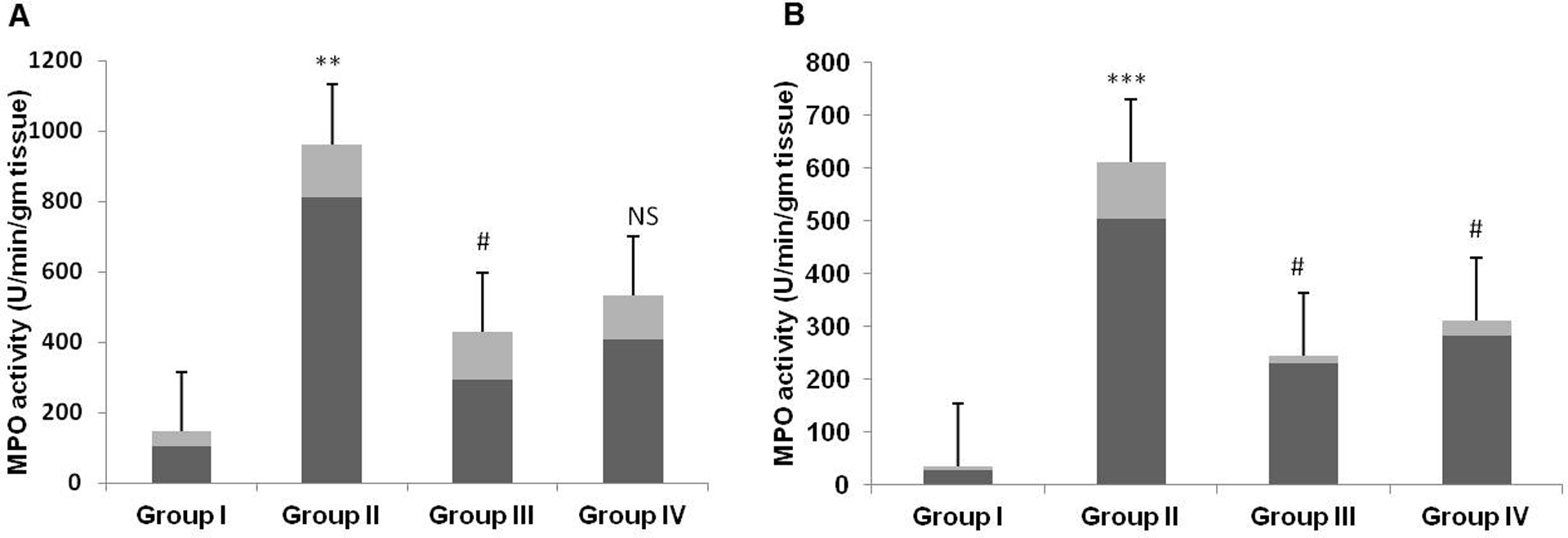
Effect of Tau and 5-FU on (a) liver and (b) kidney MPO activity. Results are represented as mean ± SE per group. MPO activity significantly increased in group II as compared to group I (p < 0.01, p < 0.001). Tau pretreatment in groups III and IV suppressed MPO activity significantly as compared to group II (p < 0.05). Tau: taurine; 5-FU: 5-fluorouracil; MPO: myeloperoxidase.
Effect of Tau and 5-FU on small intestine histoarchitecture
It is well known that chemotherapy results in various unbearable complications. Intestinal mucositis is one of the major complications associated with 5-FU-based chemotherapy. Histological analysis of small intestine shows that 5-FU administration in group II animals resulted in massive inflammatory response characterized by increased inflammatory cell infiltration in both mucosal and submucosal region. In addition, massive deformities of villi including shortening of height, loss and atrophy, marked disorganization of crypts, and accumulation of fluids were observed in group II animals as compared to group I control animals. Tau pretreatment in groups III and IV markedly suppressed the 5-FU-induced intestinal deformities as compared to group II animals (Figure 2).

Photomicrograph depicting histology of rat small intestine. The histological sections of control group showed normal histoarchitecture (a) while 5-FU treated group (group II) exhibited intense inflammatory cells infiltration, shortening, loss and atrophy of villi, marked disorganization of crypts, and accumulation of fluids (b). In groups III and IV, histological sections showed that Tau treatment suppressed 5-FU induced histological damage (c and d). (×10 magnification). Tau: taurine; 5-FU: 5-fluorouracil.
Effect of Tau and 5-FU on mast cell infiltration in intestine
Microscopic examination of small intestine revealed that in 5-FU-treated group (group II), there is marked infiltration of mast cells in the mucosal and submucosal region of the intestine. Pretreatment with Tau suppressed the infiltration of mast cells in groups III and IV as compared to group II. (Figure 3)

Photomicrograph of small intestine depicting mast cell staining. In 5-FU-treated group II animals, there is marked infiltration of mast cells (b), while no infiltration was observed in group I (a). Tau treatment in group III (c) and group IV (d) attenuated the infiltration of mast cell as compared to group II. Insets at the right panel show a magnified view (×20 magnifications) of the insets showed at the left panel (×10 magnifications). Tau: taurine; 5-FU: 5-fluorouracil.
Effect of Tau and 5-FU on liver and Kidney histology
Remarkable histological changes like massive infiltration of inflammatory cells and hepatic tissue necrosis was observed in liver sections of 5-FU only-treated group II animals, while the section of group I animals depicts normal histological architecture. Pretreatment with Tau in groups III and IV animals markedly reversed the 5-FU-induced hepatic histological changes (Figure 4). Renal histological sections of group II animals showed remarkable neutrophil infiltration, mild tubular and capsule deformities as compared to group I animals and treatment with Tau notably abrogates these changes in groups III and IV (Figure 5).
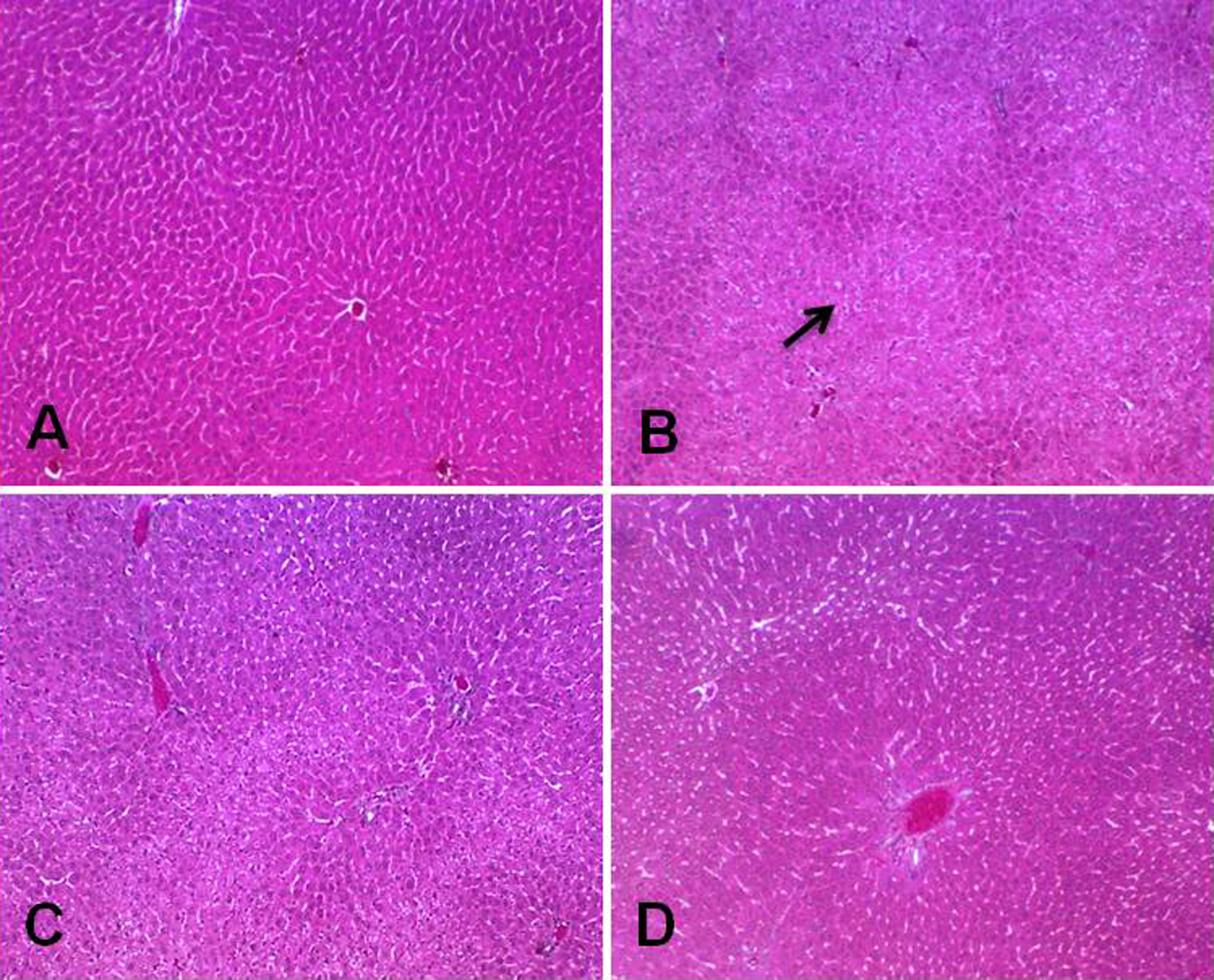
Photomicrograph depicting histology of rat liver. The histological sections of control group (a) shows normal liver histoarchitecture while 5-FU-treated group (group II) exhibited marked inflammatory cells infiltration and loss of normal cellular pattern with necrosis (b). In groups III and IV, histological sections depict that Tau treatment suppressed 5-FU-induced renal histological alterations (c and d). (×10 magnification). Tau: taurine; 5-FU: 5-fluorouracil.

Photomicrograph depicting histology of rat kidney. The histological sections of control group (a) showed normal renal histoarchitecture, while 5-FU treated group (group II) exhibited marked inflammatory cells infiltration, tubule and capsular deformities (b). In groups III and IV, histological sections showed that Tau treatment suppressed 5-FU-induced renal histological damage (c and d). (×20 magnification). Tau: taurine; 5-FU: 5-fluorouracil.
Effect of Tau and 5-FU on male reproductive organs
We found that histological analysis of testis of only 5-FU-treated group II animals depicts degeneration of seminiferous epithelium and vacuolization of sertoli cells, while atrophy of prostate acini with nuclear stratification and increased secretion was observed as compared to group I animals that showed normal testis and prostate histology. Administration of Tau in groups III and IV animals inhibited the 5-FU-induced testis and prostatic histological anomalies as compared to group II animals (Figures 6 and 7).
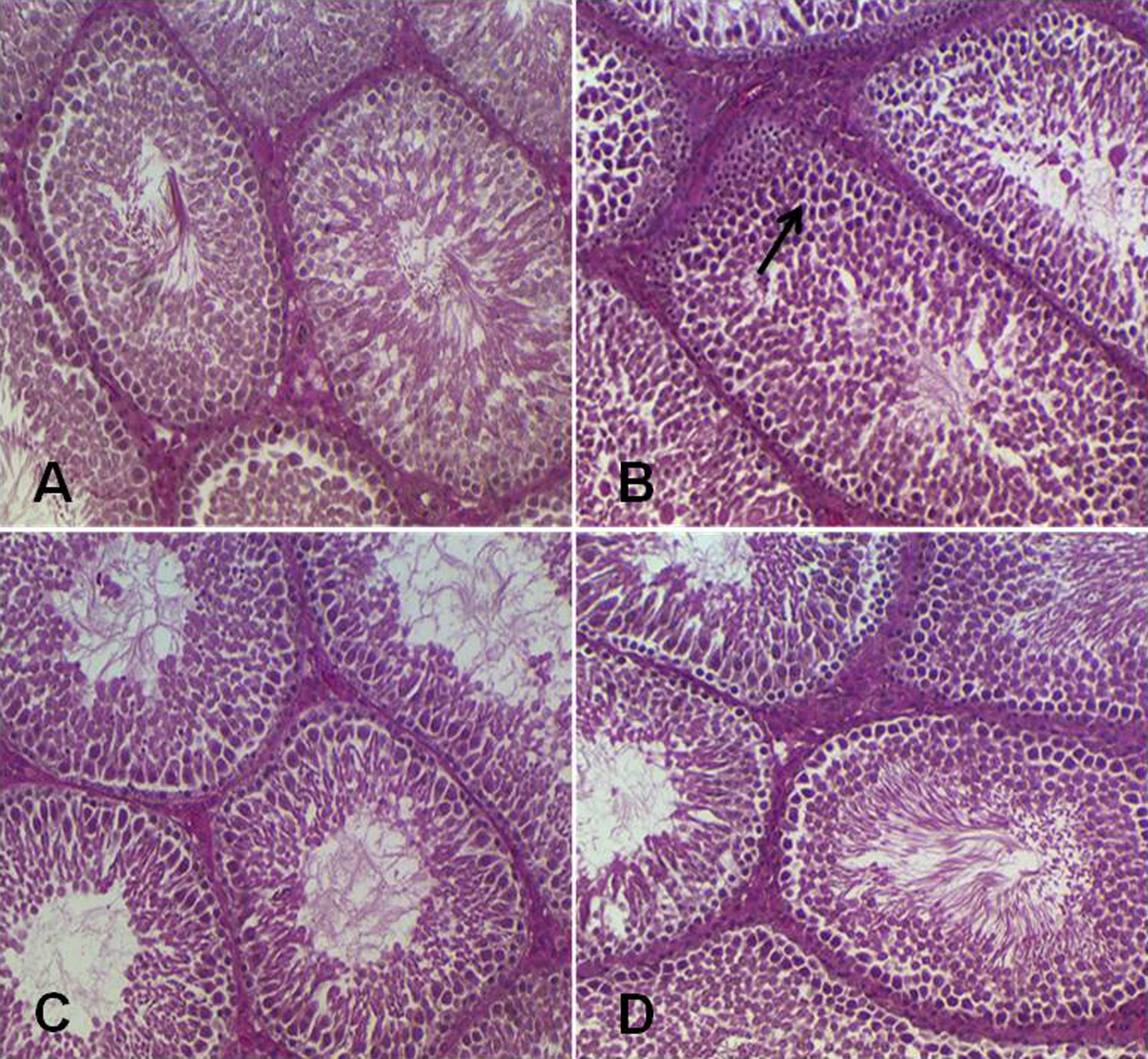
Photomicrograph depicting histology of rat testis. The histological sections of control group (a) showed normal testicular histoarchitecture, while 5-FU-treated group (group II) exhibited degeneration of seminiferous epithelium and vacuolization of sertoli cells (b). In groups III and IV, histological sections showed that Tau treatment suppressed 5-FU-induced testicular histological damage (c and d). (×20 magnification). Tau: taurine; 5-FU: 5-fluorouracil.

Photomicrograph depicting histology of rat prostate. The histological sections of control group (a) showed normal prostate histology while 5-FU-treated group (group II) exhibited marked infiltration of leukocytes and atrophy of acini (b). In groups III and IV, histological sections showed that Tau treatment suppressed 5-FU-induced prostatic histological damage (c and d). (×20 magnification). Tau: taurine; 5-FU: 5-fluorouracil.
Discussion
The findings of this study suggest that Tau has strong preventive potential against 5-FU-induced adverse effects on different organs such as liver, kidney, intestine, testis, and prostate, which was evident from biochemical and histological findings.
Chemotherapy is one of the commonly used treatment choices for cancer patients, while most of the currently used chemotherapeutic drugs are associated with substantial adverse effects on patients undergoing chemotherapy. To overcome this potential problem and to make chemotherapeutic drugs more effective for cancer patients, different approaches have been introduced in the field of cancer research. In this study, we have studied the protective effect of Tau on 5-FU-induced toxicity of different organs.
MPO is one of the key abundant enzymes secreted by inflammatory cells including neutrophils, macrophages, and microglia. 23 Although MPO plays key role in cellular protection from microbial infection by forming hypochlorous acid/hypochlorite (HOCl/OCl), it has potential role in the formation of toxic reactive moieties such as chlorine, chloramines, hydroxyl radicals, singlet oxygen, tyrosyl radicals, aldehydes, and ozone. 24,25 These reactive species may further react with host cellular macromolecules and thus contribute to the disease pathogenesis or tissue damage at the site of inflammation. 24 –26 Further, unregulated expression of MPO has been linked with the development of most of the chronic diseases including cancer, renal, cardiovascular, lung injury, and neurological disorders. 25 We have observed that 5-FU administration caused remarkable increase in MPO activity, which is in accordance with previous findings. 27 –29 Pretreatment with Tau significantly suppressed the MPO activity, which showed the anti-inflammatory potential of Tau. Previous research findings also support the inhibitory potential of Tau on MPO activity, 30 –33 which may be because of its reactivity with HOCL to form taurine cholamine that plays a vital role in the attenuation of inflammatory mediators by modulation of nuclear factor κB expression. 34 –36
Intestinal mucositis, one of the major complications associated with 5-FU-based chemotherapy, has a major impact on patients undergoing antineoplastic drug therapy. Histologically, intestinal mucositis is characterized by shortening of villi, disorganization of crypts, increased neutrophil infiltration, accumulation of fluids, increase in length of crypts, and thickness of intestinal walls. 9,13,37,38 Although the exact mechanism of chemotherapy-induced intestinal damage/mucositis is not fully explored, it is well documented that overproduction of ROS, apoptosis, hypoproliferation, abnormal expression of inflammatory mediators, and inflammation play vital role in the pathogenesis of 5-FU-induced mucositis. 9,39,40 We have observed that Tau administration ameliorated 5-FU-induced intestinal damage, which is evident from the histological analysis and this action of Tau may be due to its antioxidant, anti-inflammatory, and antiapototic potential. Another finding of this study reveal that there was marked infiltration of mast cell in only 5-FU-treated group animals, while in the control group no infiltration of mast cell was observed. Mast cell infiltration reflects an important sign of inflammation and its role has been detected in the pathogenesis of various disorders including mucositis and cancer. 41 –43 Tau supplementation has suppressed the infiltration of mast cell, which further shows the anti-inflammatory nature of Tau.
Histological analysis of liver and kidney showed marked histoarchitechtural anomalies in the 5-FU-treated animals that were suppressed by Tau. Different investigations have shown that 5-FU administration results in hepatic and renal toxicity. 5,44 –46 In liver, 5-FU gets catabolized into dihydrouracil and ultimately leads to the formation of fluoro-β-alanine, ammonia, urea, and carbon dioxide that may lead to liver and kidney toxicity. Histological observation of the current study showed that 5-FU cause remarkable histological distortion in liver and kidney, such as infiltration of inflammatory cells, necrosis, vacuolar degeneration, renal tubular degeneration, which are in agreement with previous findings. Attenuation of these histological changes by Tau is attributed to its strong antioxidant and anti-inflammatory potential.
Further, our results also demonstrate that Tau administration effectively ameliorated the morphological changes of reproductive organs induced by 5-FU administration. Previous findings also reveal that 5-FU causes male reproductive organ damage by inhibiting thymidylate synthetase and by incorporation into genetic material. 11,47 –49 Histological analysis indicate that Tau administration attenuated the 5-FU-induced degeneration of seminiferous epithelium, vacuolization of Sertoli cells in testis and atrophy of prostatic acini, nuclear stratification, and increased secretion in prostate.
In conclusion, the biochemical and histological findings of this study suggests that Tau effectively ameliorates 5-FU-induced multiple organ damage in Wistar rats, which may be because of its strong antioxidant and anti-inflammatory nature. Further studies are warranted to explore the underlying molecular mechanism of Tau.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
The authors are thankful to Prince Sultan Military Medical City, Kingdom of Saudi Arabia, to carry out this work.
