Abstract
Background
It is well-known that ultraviolet B (UVB) causes cataracts by inducing pyroptosis and the production of reactive oxygen species (ROS) in human lens epithelial cells (HLECs). The transcription factor E2F1 (E2F1) serves as a positive regulator of disrupted pathways involved in histone modification and cell cycle regulation. However, its function in UVB-treated HLECs remains unknown.
Data Collection and/or Analysis
Collected data were statistically analyzed using one-way analysis of variance (ANOVA).
Results
Our results show that HLECs were much more susceptible to oxidative stress, pyroptosis, and E2F1 in response to UVB-irradiation, but that E2F1 down-regulation effectively counteracted these effects. E2F1 was then suggested as a potential NLRP3 transcription factor by bioinformatics studies. At the same time, luciferase and CHIP assays showed that E2F1 could bind to the NLRP3 promoter and enhance NLRP3 transcription. In addition, the protective effects of si-E2F1 against oxidative stress and pyroptosis in HLECs are counteracted by overexpressing NLRP3.
Conclusions
All of the above provided the possibility to demonstrate that E2F1 plays a crucial role in regulating oxidative stress and pyroptosis in UVB-induced HLECs through inhibiting NLRP3, and it promotes oxidative stress-induced pyroptosis by suppressing NLRP3 expression.
Introduction
Cataract is a common visual disorder in clinical practice, characterized by lens protein degeneration caused by various reasons, and cataract patients have varying degrees of vision loss. Without timely and effective treatment, it can lead to progressive vision loss or even blindness. 1 Clinical commonly used surgical treatment of the disease, surgery can significantly improve patients’ eyesight. However, the surgery has great damage to the meibomian gland, and the corneal repair is greatly affected. Postoperative dry eye syndrome is prone to occur, which is manifested as dry eyes, fatigue, and intraocular hypoxia, which seriously affects the daily life of patients.2,3 Therefore, ophthalmologists are constantly exploring new methods that can prevent cataracts from occurring at the source or delay its progression.
In recent years, a new type of programmed cell death accompanied by an inflammatory reaction, pyroptosis, has attracted people’s attention, which is characterized by the formation of pores in the cell membrane, the continuous water inflow into the cell, which makes the cell swelling and rupture, and releases the cell contents and inflammatory substances. 4 Previous research has shown that cataracts may develop when lens epithelial cells (HLECs) sustain oxidative damage due to exposure to ultraviolet B (UVB) radiation.5,6 Studies have shown that ROS can also activate pyroptosis signaling to participate in the occurrence of cataracts. 7 Nevertheless, the exact biochemical pathways via which ultraviolet light controls the progression and onset of cataracts are not yet understood. By stimulating the formation of NLRP3 inflammasomes, E2F transcription factor (E2F1), which was changed by IGF2BP1, transcriptionally increased MIF-related pyroptosis. 8 In light of the above, the current investigation set out to determine whether HLECs harm caused by UVB might be mitigated by the modulation of E2F1.
Materials and methods
Cell culture and UV irradiation
Human lens epithelial cells (SRA01/04) were obtained from Procell and cultured in DMEM with 10% FBS (Gibco) at 37°C in a humidified incubator with 5% CO2. The SRA01/04 cells were divided into two groups: a control group and an experimental group. To induce UVB cell damage, the experimental cells were exposed to UVB radiation (30 mJ/cm2). Total RNAs and proteins were extracted from the cells 12 and 24 hours after exposure.
Real-time PCR analyses
After transfection for 48 h, the waste medium solution was discarded, and 800 μL Trizol reagent was added to each well and placed on ice for 30 min for full cleavage. Total cellular RNA was isolated using 200 μL of chloroform, and the initial cDNA was generated through reverse transcription. An RT-PCR system (7500, ABI Company) was used for RT-qPCR testing.
Western blot analyses
After homogenizing the cells from each group, the total protein was extracted and subjected to electrophoresis, membrane transfer and closure, and the primary antibodies E2F1 (1:1000, Proteintech), NOX4 (1:1000, Proteintech), NOX2 (1:1000, Proteintech), SOD1 (1:200, Proteintech), SOD2 (1:1000, Proteintech), NLRP3 (1:1,000, Proteintech), GSDMD-N (1:1000, Abcam), Caspase-1 (1: 1000, Cell Signaling) and anti-GAPDH (1:5000, Proteintech) were added. The second antibody was added after membrane washing and incubated at room temperature for 1 h. Image J software analyzed the relative expression of each protein. 9
Pyroptosis was detected by flow cytometry
The cells were collected in PBS after being subjected to various treatments and then resuspended. Approximately 1 × 105 cells were treated with 10 mL Propidium iodide (PI, Beyotime) for 10 minutes at room temperature and in the dark. The prevalence of pyroptosis was measured using flow cytometry (BD).
Oxidative stress detection
To study ROS generation, we used 2′,7′-Dichlorofluorescein diacetate (Sigma) and evaluated the fluorescence using an Olympus microscope.
Luciferase assay
Two luciferase reporter plasmids, wild (WT)-NLRP3 and mutant (MUT)- NLRP3 were constructed using pGL4.4. The luciferase reporter plasmid was co-transfected with sh-E2F1 and sh-NC using Lipofectamine 3000 (Thermo Fisher). 24 h after transfection, luciferase activity in harvested cells was measured using a dual luciferase reporting assay system (Promega).
ChIP assay
The interaction between E2F1 and NLRP3 promoter region was verified by ChIP experiment. SRA01/04 cells were immobilized with 16% methyl alcohol, crosslinked and lyzed to the buffer solution, which was then subjected to ultrasonic treatment. Antibody E2F1 is then added and incubated overnight. Beads are added to obtain the protein-DNA complex. Then 5 mmol/L NaCl was added for delanking to obtain DNA molecules. The enrichment of NLRP3 promoter was detected by qRT-PCR.
Statistic processing
Mean ± standard deviation was used for all experimental data. One-way ANOVA analysis was used among multiple groups, and LSD-t test was used for pairwise comparison among groups. p < .05 was considered statistically significant.
Results
UVB-irradiation significantly induced oxidative stress, pyroptosis and E2F1 in SRA01/04 cells
ROS generation in SRA01/04 after 12 and 24 hours after exposure to UVB light was observed (Figure 1(a)). Figure 1(b) shows that the UVB group had a significant drop in SOD1 protein levels and an increase in NOX2 levels. Meanwhile, Cell pyroptosis of SRA01/04 was similarly triggered by UVB irradiation (Figure 1(E)). Pyroptosis-related proteins (active Caspase-1) and E2F1 were increased in SRA01/04 with UVB irradiation (Figure 1(c) and (d)). UVB-irradiation dramatically increased oxidative stress, pyroptosis and E2F1 in SRA01/04 cells. (a) ROS staining with DCFH-DA. (b-d) Protein expression of SOD1, NOX2, caspase 1 and E2F1 in SRA01/04 cells. (e) Flow cytometry was used to assess cell pyroptosis.
Down-regulating E2F1 dramatically reduced UVB-induced oxidative damage
E2F1 expression was inhibited in HLECs using siE2F1 to test the impact on UVB-irradiated SRA01/04 cells. E2F1 mRNA and protein expression was significantly lower in the UVB+siE2F1-1 group than in the UVB+NC group (Figure 2(a) and (b)). Figure 2(c) shows that the formation of ROS caused by UVB was reversed when E2F1 was down-regulated. At the same time, down-regulating E2F1 restored the expression of oxidative stress-related proteins, which had been altered following UVB irradiation (nox2 and NOX4 expression upregulated, SOD1 and SOD2 protein levels downregulated) (Figure 2(d)). Down-regulating E2F1 dramatically reduced UVB-induced oxidative damage. (a and b) The mRNA and protein levels of E2F1. (c) ROS level was assessed using DCFH-DA staining. (d) Protein expression of NOX4, NOX2, SOD1 and SOD2.
Reducing E2F1 effectively reduced UVB-induced pyrocytosis
Inhibiting E2F1 reverted cell pyroptosis, according to flow cytometry (Figure 3(a)). Figure 3(b) displays the findings of the TUNEL test for pyrocytosis in SRA01/04 cells. Additionally, the expression of proteins associated with pyroptosis (NLRP3, active Caspase-1, and GSDMD-N) and the levels of the inflammatory cytokine IL-1β, which had increased after UVB irradiation, were both decreased by the down-regulation of E2F1 (Figure 3(c)). Active caspase-1 was also detected by immunofluorescence staining (Figure 3(d)). Reducing E2F1 effectively reduced UVB-induced pyrocytosis. (a) Measurement of cell pyroptosis using flow cytometry. (b) ROS level was assessed using DCFH-DA staining. (c) Protein levels of NLRP3, active Caspase-1, GSDMD-N, and IL-1β were assessed. (d) Immunofluorescence staining was used to detect active Caspase-1.
Transcription factor E2F1 binds to NLRP3’s promoter
The JASPAR database demonstrates that E2F1 can control NLRP3 expression by binding to its promoter (Figure 4(a)). ChIP findings indicated that segment NLRP3-P3 and NLRP3-P4 had the highest recruitment of E2F1 binding sites on the NLRP3 gene promoter (Figure 4(b)). In Figure 4(c), the WT-E2F1 group had considerably higher luciferase activity than the control group. Figure 4(d) and (e) indicate that higher mRNA and protein levels of NLRP3 in the pcDNA3.1-E2F1 group, while lower levels in the si-E2F1 group. Taken together, these findings provided strong evidence that E2F1 may bind to the NLRP3 promoter and activate NLRP3 transcription. Transcription factor E2F1 binds to NLRP3’s promoter. (a) JASPAR software was used to analyze the E2F1 transcription start sites. (b) To identify the E2F1–NLRP3 interaction, the ChIP-qPCR test was used. (c) A luciferase reporter experiment evaluated NLRP3 promoter activity after E2F1 binding site mutation. (d and e) The mRNA and protein levels of E2F1 and NLRP3 was examined in cells after siE2F1 or pcDNA3.1-NLRP3 treatment.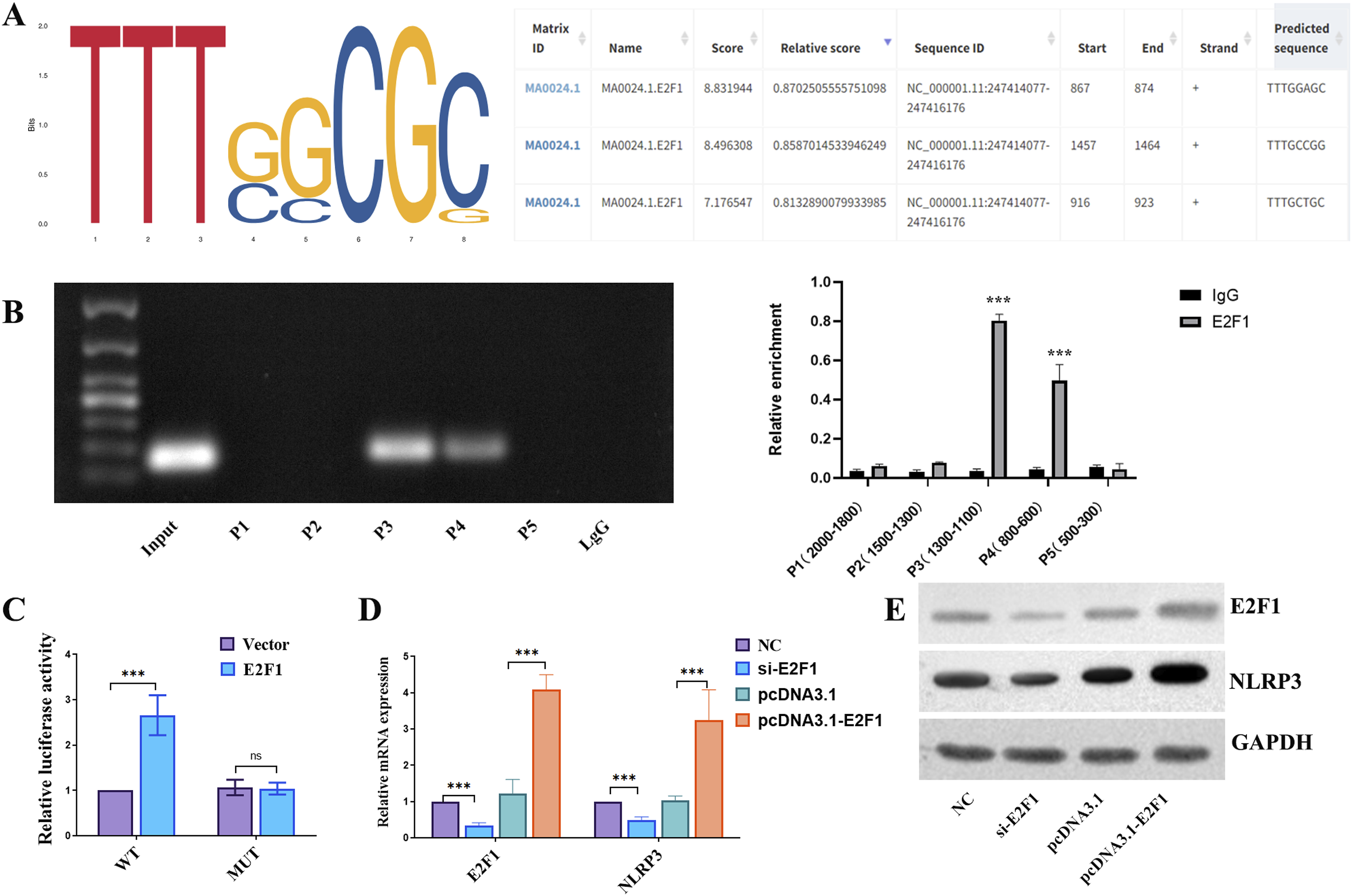
NLRP3 reverses the moderative effects of oxidative stress and pyroptosis of si-E2F1 in SRA01/04 cells
By overexpressing NLRP3 and silencing E2F1 expression in SRA01/04 cells, we were able to confirm that E2F1 affects oxidative stress and pyroptosis when exposed to UVB. First, we tested the interference efficiency of siE2F1 and pcDNA3.1-NLRP3 (Figure 5(a) and (b)). The results showed that E2F1 inhibition significantly suppressed oxidative stress (Figure 5(c) and (d)) and pyroptosis (Figure 6(a)–(c)). Meanwhile, these phenotypes were all dismissed by NLRP3 overexpression via co-transfection of siE2F1. NLRP3 reverses the moderative effects of oxidative stress of si-E2F1 in SRA01/04 cells. (a and b) The mRNA and protein levels of NLRP3. (c) ROS level was assessed using DCFH-DA staining. (d) Protein levels of SOD1 and NOX2 were measured by Western blot. NLRP3 reverses the moderative effects of pyroptosis of si-E2F1 in SRA01/04 cells. (a) Measurement of cell pyroptosis using flow cytometry. (b) Cell pyroptosis was determined by TUNEL assay. (c) Protein expression of active Caspase-1, GSDMD-N and IL-1β in SRA01/04 cells.

Discussion
Cataracts remain the leading cause of blindness globally, with their prevalence increasing year by year. Cataract development is influenced by many factors, including UVB irradiation and oxidative damage.10,11 The excessive generation of oxygen free radicals in HLEC can cause pyroptosis, mainly characterized by the release of inflammatory factors and the disruption of the cell membrane, which is thought to exacerbate cellular damage and contribute to cataract formation.12,13 Our current study showed that E2F1 is upregulated in UVB-treated SRA01/04 cells, a model of HLECs. Moreover, down-regulation of E2F1 significantly reversed UVB-induced oxidative stress and pyroptosis, suggesting a pivotal role of E2F1 in mediating these cellular processes.
Pyroptosis is a new type of programmed cell death. It has been confirmed that classical and non-classical pyroptosis pathways exist in the human body. Although the initiation signals are inconsistent, both pathways eventually activate caspase-1 by forming NLRP3 inflammatory, further shear, and activate GSDMD. Its active form, GSDMD-N, binds to lipids in the cell membrane, destroys the integrity of the membrane, and induces cell pyroptosis. 14 Recent research indicates that NLRP3 inflammasome, which is formed when cells respond to oxidative stress, accumulates some level of caspase-1 and IL-1β in the lens of cataract patients. 15 In this study, we discovered that SRA01/04 cells showed a considerable rise in the levels of the pyroptosis markers when exposed to UVB. Importantly, silencing E2F1 mitigated the UVB-induced rise in pyroptosis markers, slowed the rate of pyroptosis, and reduced oxidative stress, indicating that E2F1 plays a central role in regulating these processes in HLECs. These results underscore the contribution of pyroptosis to cataract development and suggest that E2F1-mediated regulation of pyroptosis may be an important pathway in cataractogenesis.
As an upstream transcription factor of NLRP3, E2F1 could regulate gene expression related to DNA replication, damage repair, ROS, and pyroptosis. Gong found that acetylation of E2F1, induced by oxidative stress (such as IL-6 stimulation), exacerbates cellular oxidative damage. 16 Consistent with these findings, we show that E2F1 silencing in UVB-treated SRA01/04 cells reduced the mRNA and protein levels of NLRP3, which is otherwise upregulated by UVB exposure. Flow cytometry and oxidative stress assay further proved that knocking-down of E2F1 suppressed UVB-induced SRA01/04 cells oxidative stress and pyroptosis. Meanwhile, these phenotypes were all dismissed by NLRP3 overexpression via co-transfection of siE2F1. To further substantiate this mechanism, we conducted luciferase and ChIP assays, which confirmed that E2F1 binds to the NLRP3 promoter and enhances its transcription. These experiments suggest that we can regulate the expression of NLRP3 by regulating E2F1/NLRP3 pathway. A limitation of this study is that all experiments were conducted in vitro using HLECs, and thus, the findings may not fully reflect the complexity of the in vivo response to UVB irradiation, where additional factors such as tissue-specific interactions, immune responses, and systemic influences could modulate the observed pathways.
Finally, our study proved that E2F1/NLRP3 mediated the effects of pyroptosis in HLECs and further validated the critical function of pyroptosis in cataract development. Based on these results, E2F1 may be a good option for treating and preventing cataracts since it plays a crucial role in cataract development.
Footnotes
Author contributions
FW, QZ and HL designed and performed the research; FW, FY and GY analyzed the data; FW wrote the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Sichuan Science and Technology Program (2022YFS0611).
