Abstract
Objective
We aimed to explore the effect and potential mechanism of Sestrin 2 (SESN2) in human lens epithelial cells (HLECs).
Methods
To mimic the oxidative stress environment, SAR01/04 cells were treated with 200 μM hydrogen peroxide (H2O2) for 24 h. Cell viability and apoptosis were checked by cell counting kit-8 and flow cytometry. Western blot was taken to check the protein changes of SESN2, B-cell lymphoma-2 (Bcl-2), Bcl-2-associated X (Bax), mechanistic target of rapamycin (mTOR), phosphorylated (p)-mTOR, ribosomal protein S6 kinase B1 (p70S6K), p-p70S6K, and nuclear factor erythroid 2-related factor 2 (Nrf2). Superoxide dismutase (SOD), catalase (CAT), malondialdehyde (MDA), and reactive oxygen species (ROS) were detected via the corresponding reagent kit. The levels of interleukin (IL)-1β, IL-18, and tumor necrosis factor (TNF)-α were measured using enzyme-linked immunosorbent assay.
Results
SESN2 was down-regulated in cataract lens tissue and up-regulated in SAR01/04 cells treated with H2O2. Under treatment of H2O2, up-regulation of SESN2 improved cell viability, enhanced the activity of SOD and CAT, inhibited cell apoptosis, and reduced the levels of MDA, ROS, IL-1β, IL-18, and TNF-α, while down-regulation of SESN2 caused the contrary effects. Further bioinformatics analysis suggested that SESN2 regulated the mTOR signaling pathway. Treatment of H2O2 inhibited p-mTOR and p-p70S6K protein expression, while overexpression of SESN2 increased p-mTOR and p-p70S6K protein expression in the H2O2 group and down-regulation of SESN2 further decreased p-mTOR and p-p70S6K protein expression in the H2O2 group. Additionally, H2O2 increased Nrf2 protein expression, and overexpression of SESN2 further increased Nrf2 protein expression in the H2O2 group. Importantly, rapamycin (an inhibitor of mTOR signaling pathway) and knockdown of Nrf2 reversed the promotive effects of SESN2 on cell viability and the inhibitive effects of SESN2 on cell apoptosis, oxidative stress, and inflammatory reaction.
Conclusion
SESN2 protected HLECs damage induced by H2O2, which was related to the activation of mTOR/Nrf2 pathway.
Introduction
Cataract is a lesion in which the optical quality of the lens is reduced due to the decrease in the transparency of the lens or the color change and is one of the major eye diseases leading to blindness worldwide.1,2 Cataract can reduce the quality of life of the elderly, bring huge economic and mental burden to the patients, and has become a major global public health problem. 3 In the lens, lens epithelial cell (LEC) is the most active site of metabolism. 4 During the formation of cataract, LEC undergoes many changes, including cell viability, differentiation, DNA damage, cell cycle, and so on.5,6 Accumulating evidence indicates that a major cause of cataract is oxidative stress.7–9 Exposure of lens to oxidative stress caused by reactive oxygen species (ROS) may result in LEC apoptosis and eventually lead to the development of cataract.10,11 For the development of cataract, LEC apoptosis caused by oxidative stress is regarded as a common cellular basis.12,13 At present, surgery is still the most effective way to treat cataracts, but it is inevitable for some postoperative complications. 14 Therefore, for prevention and treatment of cataract, it is significant to further study the molecular mechanism of apoptosis, oxidative damage, and proliferation in human lens epithelial cells (HLECs).
Sestrins (SESNs) are a family of stress-induced proteins that are highly conserved in evolution. SESN2 is a member of the SESN family with a full-length cDNA of 1443bp encoding 481 amino acids. Activation of SESN2 can reduce the accumulation of ROS, maintain energy balance, reduce protein synthesis, and retard metabolic disease progression.15,16 Chen et al. found that up-regulation of SESN2 protected bovine mammary epithelial cells from hydrogen peroxide (H2O2)-induced oxidative damage through the kelch-like ECH-associated protein 1 (Keap1)-nuclear factor erythroid 2-related factor 2 (Nrf2)/antioxidant response element (ARE) pathway. 17 Fan et al. have found that SESN2 protects retinal ganglion cells from H2O2-induced oxidative stress in glaucoma. 18 However, the effect of SESN2 is not known in cataract.
In addition to antioxidant activity, SESN2 mainly participates in the regulation of the two major signal pathways of AMP-Activated Protein Kinase (AMPK) and mammalian target of rapamycin (mTOR) to maintain cell homeostasis. 19 mTOR is a serine-threonine protein kinase, originally found in yeast mutants, which forms two complexes, mTORC1 and mTORC2. 20 mTOR is involved in the regulation of energy metabolism, autophagy, and the synthesis of protein, lipid, and organelle.21,22 Meanwhile, mTOR plays a significant role in cell growth, proliferation, differentiation, and apoptosis. 23 A study has found that SESN2 regulates the growth, migration, and ROS production of endometrial cancer through mTORC1-dependent mechanism. 24 In the present study, we investigated whether SESN2 alleviated H2O2-induced oxidative stress and apoptosis. In addition, the mechanism of SESN2 was explored.
Methods
Tissue samples
The samples of cataract group (n = 25) were taken from the anterior lens capsule removed during phacoemulsification of patients with grade III cortical cataract (14 male, 11 female; aged 50–61 years). The samples of normal group (n = 15) were taken from the anterior lens capsule of patients (9 male, 6 female; aged 43–59 years) without cataracts undergoing eyeball enucleation. All patients were recruited in No. 960 Hospital of PLA Joint Logistic Support Force from October 2022 to June 2023. The Ethics Committee of No. 960 Hospital of PLA Joint Logistic Support Force (approval number: 2022-08063) approved the protocol of this study. All patients signed written informed consent.
Cell culture and transfection
In a cell incubator at 37°C and 5% CO2, Dulbecco’s modified Eagle’s medium containing 10% fetal bovine serum was applied to culture HLEC line (SRA01/04; American Type Culture Collection, USA). At 80%–90% confluence, SRA01/04 cells were exposed to different concentrations of H2O2 (0, 100, 200, and 300 µM) for 24 h. Additionally, 200 μM H2O2 was taken to treat SRA01/04 cells for 0, 12, 24, 36, and 48 h. Subsequently, SESN2 changes were tested utilizing quantitative real-time polymerase chain reaction (qRT-PCR) and Western blot.
Small interfering (si) RNA of SESN2 (si1-SESN2 and si2-SESN2) and Nrf2 (si-Nrf2) were synthesized and purified by RiboBio (Guangzhou, China). SESN2 was cloned into pCDNA eukaryotic expression vector (Invitrogen, USA). Si-NC, si-Nrf2, si1-SESN2, si2-SESN2, pcDNA-NC, and pcDNA- SESN2 were transfected to SRA01/04 cells using Lipofectamine 2000 reagent (Invitrogen). After transfection, cells were treated with 200 μM H2O2 for 24 h.
qRT-PCR
From tissues and cells, total RNA was extracted using TRIzol reagent (Invitrogen). cDNA was synthesized by the Revert Aid First Strand cDNA Synthesis Kit (Thermo Fisher scientific, USA). QRT-PCR was performed with the PowerUp SYBR Green Master Mix (Thermo Fisher Scientific). Primer sequences were listed as follows: SESN2, Forward: 5′-CTTCCGGGCCCAGGATTATAC-3′ and Reverse: 5′-AGCTGGTTCACCTCCCCATA-3’; GAPDH, Forward: 5′-TGAATGGGCAGCCGTTAGGA-3′ and Reverse: 5′-CGCCCAATACGACCAAATCAGAGA-3′. GAPDH was used as internal control, and the relative mRNA expression was calculated by 2−ΔΔCT method.
Western blot
Proteins were extracted from tissues and cells using RIPA lysis buffer (Beyotime Biotechnology, China). Next, protein concentration was measured via BCA kit (Beyotime Biotechnology). Then, proteins were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis and then transferred to the polyvinylidene difluoride membrane. After blocking in Tween 20-Tris buffer containing 5% skimmed milk for 1 h, membranes were incubated with primary antibodies (SESN2, Cell Signaling Technology, 8487, 1: 1000; Bax, Proteintech, 50599-2-Ig, 1: 8000; Bcl-2, Cell Signaling Technology, 3498, 1: 1000; p-mTOR, Proteintech, 67778-1-Ig, 1: 10000; mTOR, Proteintech, 66888-1-Ig, 1: 25000; p-p70S6K, Proteintech, 67898-1-Ig, 1: 20000; p70S6K, Proteintech, 66638-1-Ig, 1: 3000; Nrf2, Abcam, ab62352, 1: 1000; GAPDH, Proteintech, 60004-1-Ig, 1: 200000) and secondary antibody (Abcam, ab6721 and ab205719, 3: 1000). Protein expression intensity was determined by ECL kit (Beyotime Biotechnology).
Cell counting kit-8 (CCK-8)
SRA01/04 cells (2 × 105 cells per well) were seeded in a 96-well plate. After indicated treatment, 10 μL CCK-8 solution (Sigma-Aldrich, USA) was added to each well for 2 h. Absorbance value at 450 nm was measured.
Flow cytometry
After indicated treatment, SRA01/04 cells were collected. Next, the cells were resuspended in Annexin V-FITC binding buffer and then incubated with 5 μL Annexin V-FITC and 10 μL PI (Beyotime Biotechnology) for 20 min under dark. Flow cytometer (BD Biosciences) was applied to detect cell apoptosis.
Detection of superoxide dismutase (SOD), catalase (CAT), and malondialdehyde (MDA) activity
SOD activity was measured by SOD assay kit (A001-3-2, Nanjing Jiancheng Bioengineering Institute, China). CAT activity was measured by CAT assay kit (S0051, Beyotime Biotechnology). MDA level was measured by MDA assay kit (S0131S, Beyotime Biotechnology).
Detection of ROS level
After indicated treatment, SRA01/04 cells were incubated with DCFH-DA (Sigma-Aldrich) at 37°C for 30 min under dark. Then, cells were washed with PBS. ROS level was detected through flow cytometer.
Enzyme-linked immuno sorbent assay (ELISA)
After indicated treatment, the levels of IL-1β, IL-18, and TNF-α were detected by IL-1β ELISA Kit (PI305), IL-18 ELISA Kit (PI558), and TNF-α ELISA Kit (PT518), respectively, referring to manufacturer instructions. Beyotime Biotechnology provided the above kits.
Bioinformatics analysis
With “SESN2” as the key word, comparative toxicology database (CTD) website (https://ctdbase.org/) and STITCH website (https://stitch.embl.de/) were used to analyze the pathways regulated by SESN2. The bubble map showing the significantly enriched KEGG pathways was generated using ggplot2 (R package).
Statistical analysis
GraphPad Prism 7.0 software (USA) was used for analyzing the data. One-way ANOVA and Student’s t-test were used for comparisons. p < .05 was considered statistically significant.
This is an original research article.
Results
SESN2 expression was down-regulated in cataract lens tissue and up-regulated in SRA01/04 cells treated with H2O2
Figure 1 A and B showed that SESN2 expressions in the anterior lens capsule of patients with cataract were lower than those of the normal group (p < .01). After treatment with 0, 100, 200, and 300 μM H2O2, SESN2 expression was increased in a dose-dependent manner in SAR01/04 cells (p < .05, Figure 1(c)). Next, we chose 200 μM H2O2 to treat SAR01/04 cells for different time points (0, 12, 24, 36, and 48 h). Figure 1(d) indicated that SESN2 expression in SAR01/04 cells was increased at different time points (12, 24, 36, and 48 h), especially 24 h (p < .05). In subsequent experiments, 200 μM H2O2 was applied to treat cells for 24 h to mimic oxidative stress. SESN2 was down-regulated in cataract lens tissues and up-regulated in SRA01/04 cells treated with H2O2. (a) The mRNA level of SESN2 in the anterior lens capsule of patients with cataract (n = 25) and patients without cataracts (n = 15) was measured by qRT-PCR. (b) SESN2 protein level in the anterior lens capsule of patients with cataract (n = 3) and patients without cataract (n = 3) was measured by Western blot. (c) SRA01/04 cells were exposed to 0, 100, 200, and 300 µM H2O2 for 24 h and SESN2 expression was measured by qRT-PCR and Western blot. (d) SRA01/04 cells were exposed to 200 μM H2O2 for 0, 12, 24, 36, and 48 h and SESN2 expression was measured by qRT-PCR and Western blot. The mean ± standard deviation is the presentation form of the data. Compared with normal, 0 μM, or 0 h group,
*
p < .05 and
**
p < .01.
SESN2 inhibited cell apoptosis in SRA01/04 cells treated with H2O2
As displayed in Figure 2(a), compared with the si-NC group, SESN2 expression in si1-SESN2 and si2-SESN2 groups was decreased, especially the si1-SESN2 group (p < .01). H2O2 inhibited cell viability and enhanced cell apoptosis compared with the control group, and down-regulation of SESN2 further inhibited cell viability and increased cell apoptosis (p < .05, Figure 2(b) and (c)). Furthermore, H2O2 increased Bax expression and decreased Bcl-2 expression (p < .01, Figure 2(d)). When the cells were transfected with si1-SESN2, the changes of Bax and Bcl-2 expression were more obvious. We also constructed the overexpression of SESN2 cells. As shown in Figure 2(e), compared with the pcDNA-NC group, SESN2 level was increased significantly in cells transfected with pcDNA-SESN2 (p < .01). As suggested in Figure 2(f)–(h), compared with the H2O2 + pcDNA-NC group, overexpression of SESN2 improved cell viability, inhibited cell apoptosis, reduced Bax expression, and increased Bcl-2 expression (p < .01). SESN2 inhibited SRA01/04 cell apoptosis induced by H2O2. (a) SRA01/04 cells were transfected with si-NC, si1-SESN2, or si2-SESN2 and SESN2 mRNA level was measured by qRT-PCR. (b)–(d) SRA01/04 cells were transfected with si-NC or si1-SESN2 and then treated with 200 μM H2O2 for 24 h; cell viability (b), apoptosis (c), and apoptosis-related proteins (Bax and Bcl-2) (d) were detected by CCK-8 assay, flow cytometry, and Western blot, respectively. (e) SRA01/04 cells were transfected with pcDNA-NC or pcDNA-SESN2, and SESN2 mRNA level was measured by qRT-PCR. (f)–(h) SRA01/04 cells were transfected with pcDNA-NC or pcDNA-SESN2 and then treated with 200 μM H2O2 for 24 h; cell viability (f), apoptosis (g), and apoptosis-related proteins (Bax and Bcl-2) (h) were detected by CCK-8 assay, flow cytometry, and Western blot, respectively. The mean ± standard deviation is the presentation form of the data. N = 3. Compared with the control group, **p < .01; compared with the H2O2 + si-NC or H2O2 + pcDNA-NC group, #p < .05 and ##p < .01.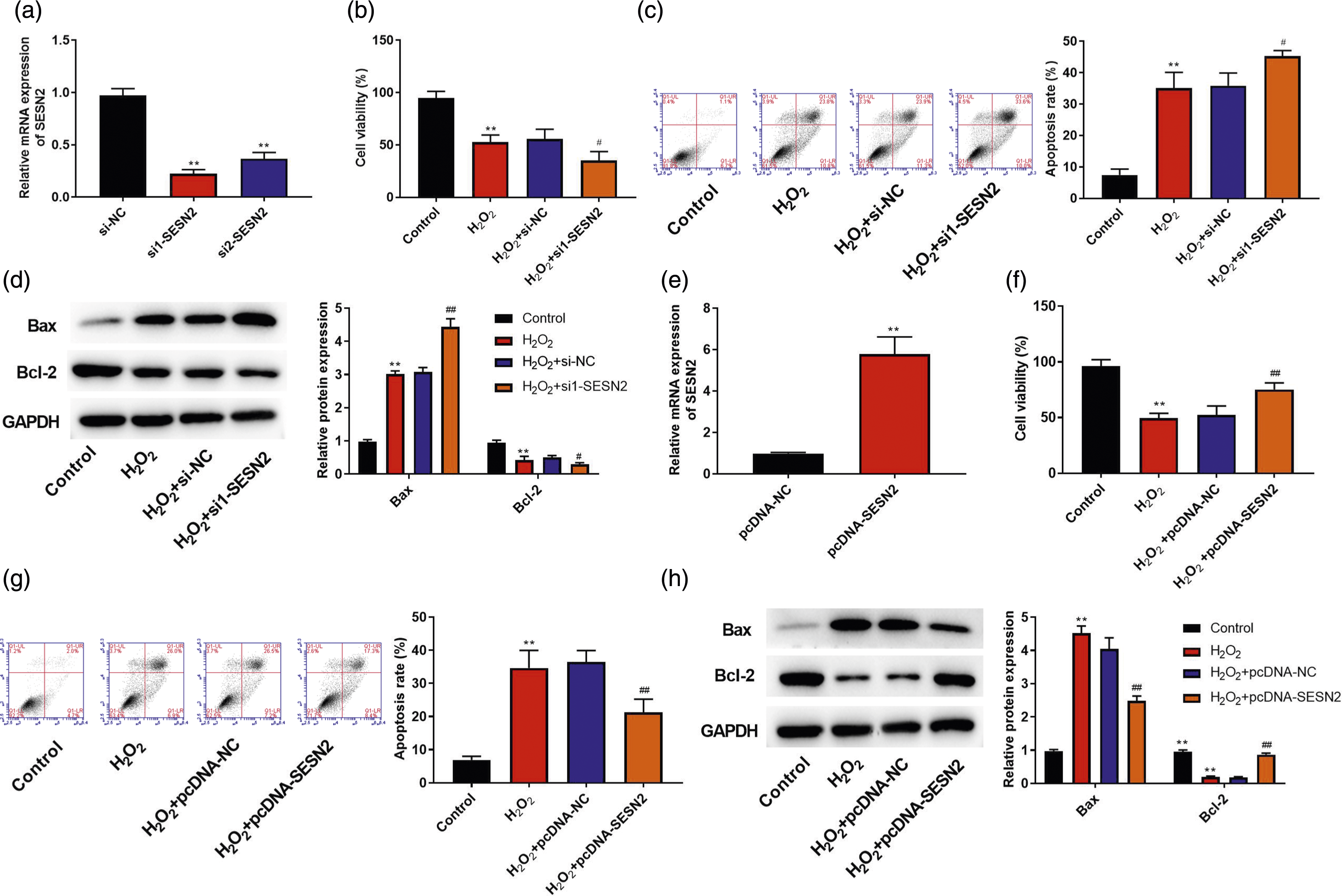
SESN2 inhibited oxidative stress in SAR01/04 cells treated with H2O2
We detected the effects of SESN2 on oxidative stress in SAR01/04 cells. Figure 3(a)–(d) showed that H2O2 reduced SOD and CAT activity and increased the levels of MAD and ROS (p < .01), but down-regulation of SESN2 further reduced SOD and CAT activity and increased the levels of MAD and ROS (p < .05). However, compared with the H2O2 + pcDNA-NC group, overexpression of SESN2 increased the activity of SOD and CAT and decreased the levels of MAD and ROS (p < .01, Figure 3(e)–(h)). SESN2 inhibited H2O2-induced oxidative stress in SAR01/04 cells. (a)–(d) SRA01/04 cells were transfected with si-NC or si1-SESN2 and then treated with 200 μM H2O2 for 24 h; SOD activity (a), CAT activity (b), MDA level (c), and ROS level (d) were measured. (e)–(h) SRA01/04 cells were transfected with pcDNA-NC or pcDNA-SESN2 and then treated with 200 μM H2O2 for 24 h; SOD activity (e), CAT activity (f), MDA level (g), and ROS level (h) were measured. The mean ± standard deviation is the presentation form of the data. N = 3. Compared with the control group, **p < .01; compared with the H2O2 + si-NC or H2O2 + pcDNA-NC group, #p < .05 and ##p < .01.
SESN2 inhibited H2O2-induced inflammatory response in SAR01/04 cells
We detected the effects of SESN2 on inflammatory response in SAR01/04 cells. Figure 4(a)–(c) showed that H2O2 increased the levels of IL-1β, IL-18, and TNF-α (p < .01), and down-regulation of SESN2 further increased the levels of IL-1β, IL-18, and TNF-α compared with the H2O2 + si-NC group (p < .01). Furthermore, compared with the H2O2 + pcDNA-NC group, overexpression of SESN2 decreased the levels of IL-1β, IL-18, and TNF-α (p < .01, Figure 4(d)–(f)). SESN2 inhibited H2O2-induced inflammatory response in SAR01/04 cells. (a)–(c) SRA01/04 cells were transfected with si-NC or si1-SESN2 and then treated with 200 μM H2O2 for 24 h; IL-1β level (a), IL-18 level (b), and TNF-α level (c) were measured. (d)–(f) SRA01/04 cells were transfected with pcDNA-NC or pcDNA-SESN2 and then treated with 200 μM H2O2 for 24 h; IL-1β level (d), IL-18 level (e), and TNF-α level (f) were measured. The mean ± standard deviation is the presentation form of the data. N = 3. Compared with the control group, **p < .01; compared with the H2O2 + si-NC or H2O2 + pcDNA-NC group, ##p < .01.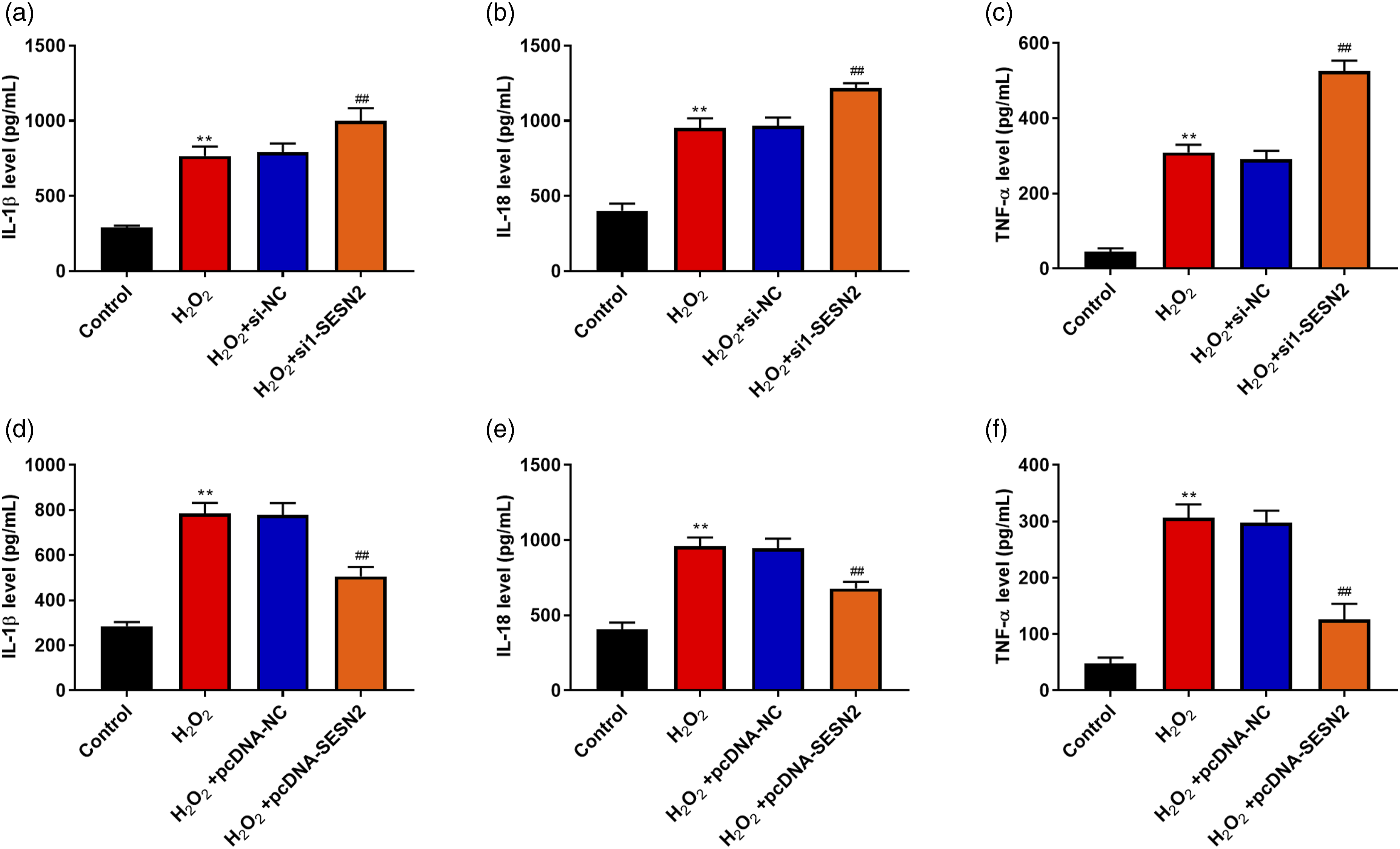
SESN2 regulated the mTOR signaling pathway
The molecular mechanism of SESN2 in SAR01/04 cells was investigated. We obtained 3 intersection pathways using the CTD and STITCH website and found that SESN2 regulated the mTOR signaling pathway (Figure 5(a) and (b)). Next, p-mTOR protein expression was detected in cataract samples. Figure 5(c) indicated that p-mTOR protein levels in the anterior lens capsule of patients with cataract were lower than those of the normal group (p < .01). In addition, mTOR pathway-related proteins were detected. Figure 5(d) showed that H2O2 inhibited the expression of p-mTOR and p-p70S6K in comparison with the control group (p < .01). Meanwhile, compared with the H2O2 + si-NC group, down-regulation of SESN2 further inhibited the expression of p-mTOR and p-p70S6K (p < .01, Figure 5(d)). However, compared with the H2O2 + pcDNA-NC group, overexpression of SESN2 increased the expression of p-mTOR and p-p70S6K (p < .01, Figure 5(e)). SESN2 regulated the mTOR signaling pathway. (a) CTD website and STITCH website were used to analyze pathway related to SESN2. (b) R package (ggplot2) was used to draw bubble chart. (c) The protein level of p-mTOR in the anterior lens capsule of cataract patients with cataract (n = 3) and patients without cataract (n = 3) was measured by Western blot. (d) SRA01/04 cells were transfected with si-NC or si1-SESN2 and then treated with 200 μM H2O2 for 24 h, and the expression levels of p-mTOR, mTOR, p-p70S6K, and p70S6K were detected by Western blot. (e) SRA01/04 cells were transfected with pcDNA-NC or pcDNA-SESN2 and then treated with 200 μM H2O2 for 24 h, and the expression levels of p-mTOR, mTOR, p-p70S6K, and p70S6K were detected by Western blot. The mean ± standard deviation is the presentation form of the data. N = 3. Compared with the control group, **p < .01; compared with the H2O2 + si-NC or H2O2 + pcDNA-NC group, ##p < .01.
Under treatment with H2O2, SESN2 protected SAR01/04 cells from oxidative stress, inflammation, and apoptosis by activating the mTOR pathway
We further explored the involvement of mTOR pathway in SAR01/04 cells treated with H2O2. SAR01/04 cells transfected with pcDNA-SESN2 were treated with rapamycin (an inhibitor of the mTOR pathway). As shown in Figure 6(a), up-regulation of SESN2 increased the expression of p-mTOR and p-p70S6K, but rapamycin inhibited the expression of p-mTOR and p-p70S6K in the H2O2 + pcDNA-SESN2 group (p < .01). As shown in Figure 6(b) and (c), after treated with rapamycin, cell viability was inhibited and apoptosis was improved in the H2O2 + pcDNA-SESN2 group (p < .05,). Furthermore, rapamycin could reverse the inhibitive effects of overexpression of SESN2 on oxidative stress, which was manifested by reducing intracellular activity of SOD and CAT and increasing the levels of MDA and ROS (p < .05, Figure 6(d)–(g)). Moreover, compared with the H2O2 + pcDNA-SESN2 group, rapamycin increased the levels of IL-1β, IL-18, and TNF-α (p < .01, Figure 6(h)–(j)). SESN2 protected HLECs from oxidative stress, inflammation, and apoptosis induced by H2O2 by activating the mTOR pathway. SRA01/04 cells were transfected with pcDNA-SESN2 and then treated with 200 μM H2O2 and 50 nM rapamycin for 24 h. (a) The expression levels of p-mTOR, mTOR, p-p70S6K, and p70S6K in SRA01/04 cells were detected by Western blot. (b) Cell viability in SRA01/04 cells was detected by CCK-8 assay. (c) Cell apoptosis in SRA01/04 cells was detected by flow cytometry. (d) SOD activity in SRA01/04 cells. (e) CAT activity in SRA01/04 cells. (f) MDA levels in SRA01/04 cells. (g) ROS levels in SRA01/04 cells. (h) IL-1β levels in SRA01/04 cells. (i) IL-18 levels in SRA01/04 cells. (j) TNF-α levels in SRA01/04 cells. The mean ± standard deviation is the presentation form of the data. N = 3. Compared with the control group, **p < .01; compared with the H2O2 group, ##p < .01; compared with the H2O2 + pcDNA-SESN2 group, &p < .05 and &&p < .01.
Under treatment with H2O2, SESN2 protected SAR01/04 cells from oxidative stress, inflammation, and apoptosis by activating Nrf2
Nrf2 plays a vital role in SESN2-mediated cytoprotection. We speculated that SESN2 might regulate H2O2-induced injury in LECs through regulating Nrf2. To test this hypothesis, the influence of SESN2 on Nrf2 was checked in SAR01/04 cells. As shown in Figure 7(a), H2O2 stimulation increased Nrf2 protein expression (p < .01); moreover, up-regulation of SESN2 further increased Nrf2 protein expression (p < .01). To further verify whether SESN2 alleviated H2O2-induced injury through activating Nrf2, pcDNA-SESN2 and si-Nrf2 were co-transfected to SAR01/04 cells, then cell viability, cell apoptosis, and the levels of SOD, MDA, IL-1β, IL-18, and TNF-α were investigated. Figure 7(b) and (c) showed that the knockdown of Nrf2 abrogated SESN2-mediated promotive effect on cell viability and inhibitory effect on cell apoptosis induced by H2O2 (p < .05). Furthermore, knockdown of Nrf2 reversed SESN2-mediated inhibitory effect on oxidative stress, which was manifested by reducing SOD activity and increasing MDA levels (p < .05, Figure 7(d) and (e)). At the same time, compared with the H2O2 + pcDNA-SESN2 + si-NC group, the knockdown of Nrf2 increased the levels of IL-1β, IL-18, and TNF-α (p < .05, Figure 7(f)–(h)). SESN2 protected HLECs from oxidative stress, inflammation, and apoptosis induced by H2O2 by activating Nrf2. (a) SRA01/04 cells were transfected with pcDNA-NC or pcDNA-SESN2 and then treated with 200 μM H2O2 for 24 h. Nrf2 protein expression was detected by Western blot. SRA01/04 cells were co-transfected with pcDNA-SESN2 and si-Nrf2 and then treated with 200 μM H2O2 for 24 h. (b) Cell viability in SRA01/04 cells was detected by CCK-8 assay. (c) Cell apoptosis in SRA01/04 cells was detected by flow cytometry. (d) SOD activity in SRA01/04 cells. (e) MDA levels in SRA01/04 cells. (f) IL-1β levels in SRA01/04 cells. (g) IL-18 levels in SRA01/04 cells. (h) TNF-α levels in SRA01/04 cells. The mean ± standard deviation is the presentation form of the data. N = 3. Compared with the control group, **p < .01; compared with the H2O2 group, ##p < .01; compared with the H2O2 + pcDNA-SESN2 + H2O2 group, &p < .05 and &&p < .01.
Discussion
The damage and apoptosis of HLECs caused by oxidative stress are the main causes of cataract. 25 Studies have found that SESN2 maintains cell growth, metabolism, and other homeostasis by regulating oxidative stress.26,27 We first studied the role of SESN2 in cataract and found that SESN2 expression was down-regulated in cataract lens tissue. This finding indicated that SESN2 downregulation might be related to the occurrence of cataract. H2O2 is a non-free radical member of the reactive oxygen species family, which can irreversibly damage LECs, leading to cell death and cataract. 28 At present, H2O2 is often used to establish in vitro cell model for the study of cataract. 29 As an anti-oxidant, the expression of SESN2 can be raised under environmental stresses, such as mitochondrial dysfunction, hypoxia, and oxidative stress.30,31 Here, we found that SESN2 was up-regulated in SAR01/04 cells treated with H2O2. Similarly, SESN2 was up-regulated in retinal ganglion cells stimulated by H2O2. 18 SESN2 may be a new target for prevention and treatment of cataract, and mechanism needs to be investigated in further experiments.
Apoptosis is a common cellular feature in the pathogenesis of cataract. 32 Studies have shown that SESN2 exerts a vital role in cell apoptosis under various stress conditions. For example, under oxidative stress, knockdown of SESN2 promoted cell apoptosis in neurological diseases. 33 It was also found that SESN2 protected dendritic cells from endoplasmic reticulum stress-related apoptosis. 34 Moreover, Hanus J, et al. found that SESN2 activated by gossypol acetic acid could prevent H2O2-induced apoptosis of retinal pigment epithelial cells. 35 In the present study, down-regulation of SESN2 inhibited cell activity, and down-regulation of SESN2 aggravated cell apoptosis induced by H2O2 via increasing Bax level and inhibiting Bcl-2 level. However, cell apoptosis induced by H2O2 was inhibited when SESN2 was up-regulated. Taken together, SESN2 inhibited HLEC apoptosis induced by H2O2.
HLEC damage induced by oxidative stress is the main pathogenesis of cataract. 36 SESN2 is an effective antioxidant protein that can accelerate the elimination of ROS induced by various stress conditions.37,38 Here, we found that the up-regulation of SESN2 inhibited the oxidative stress caused by H2O2 by increasing the activity of SOD and CAT and reducing the levels of MDA and ROS. The accumulation of pro-inflammatory cytokines in the lens may cause the expression of transforming growth factor-β and the synthesis of collagen, ultimately leading to the formation of cataract.39,40 IL-1β was found to be upregulated in human cataract samples. 41 In hereditary cataract rat model, IL-18 level was increased in lens, and IL-18 level was related to opacity of lens. 42 TNF-α level is raised in several ocular diseases, such as highly myopic cataract and primary open-angle glaucoma.43,44 In addition, TNF-α has a hand in extrinsic apoptosis pathway and contributes to cell apoptosis. 45 In the present study, up-regulation of SESN2 inhibited H2O2-induced inflammatory cytokines (IL-1β, IL-18, and TNF-α). However, down-regulation of SESN2 led to the opposite results. These results demonstrated that up-regulation of SESN2 could protect HLECs from oxidative stress and inflammation induced by H2O2.
Phosphatidylinositol 3-kinase (PI3K) activates mTOR by activating protein kinase B (AKT), which further activates the downstream factor p70S6K to exert its biological activity. 46 It has been found that the mTOR signaling pathway is inhibited during oxidative stress. 47 The mTOR signaling pathway slows down neurodegeneration by protecting cells from oxidative stress.48,49 Dong et al. found that FUNDC1 promoted cell apoptosis by inactivating PI3K/AKT/mTOR signaling in SRA01/04 cells under the condition of oxidative stress. 50 Han et al. reported that SRA01/04 cell apoptosis was inhibited by EphA2, which was related to the activation of the PI3K/AKT/mTOR pathway. 51 In the present study, p-mTOR and p-p70S6K expression was down-regulated in cells treated with H2O2, while up-regulation of SESN2 increased p-mTOR and p-p70S6K expression. Moreover, treatment with rapamycin partly reversed the inhibitive effects of SESN2 on cell apoptosis, inflammation, and oxidative stress. Recently, a growing body of research suggests that blockage of autophagy attenuates LEC apoptosis and inhibits cataract progression.52–54 Based on above findings, we speculated that SESN2 might promote the mTOR pathway, inhibit autophagy, and exhibit the protective effect in HLEC under oxidative stress.
As a key transcription factor, Nrf2 plays an important role in cell survival, differentiation, metabolic reprogramming, redox homeostasis, and cytoprotection.55,56 In cataract, Nrf2 lower levels cause oxidative stress, leading to failure of lens cell protection. 57 In diabetic rats, upregulation of the Nrf2/HO-1 axis attenuates the development and progression of cataract. 58 SESN2 enhanced Nrf2/ARE activation by down-regulating Keap1, thus protecting retinal ganglion cells from oxidative stress induced by H2O2. 18 In this study, H2O2 increased Nrf2 protein expression, and up-regulation of SESN2 further increased Nrf2 protein. Moreover, knockdown of Nrf2 partly reversed the inhibitive effects of SESN2 on cell apoptosis, inflammation, and oxidative stress. Previous studies have revealed that Nrf2 can be regulated by mTOR. Jung et al. showed that the combination of zileuton and melatonin improved kidney injury by activating the AKT/mTOR/Nrf2 pathway. 59 Zhang et al. found that vascular injury was attenuated by irisin, which was associated with the activation of the AKT/mTOR/Nrf2 pathway. 60 In our present study, we found SESN2 could activate mTOR pathway and upregulate Nrf2 expression. According to above results, we speculated that SESN2 might inhibit cell apoptosis, inflammation, and oxidative stress in HLECs treated with H2O2 through activating the mTOR/Nrf2 pathway.
Some limitations of this study should be noted. First, we only examined the changes of SESN2 expression within 48 h of H2O2 treatment. However, when SRA01/04 cells are treated with H2O2 for longer time, the changes of SESN2 expression are unclear. Second, the effect of SESN2 on HLECs in vitro was evaluated, but an in vivo animal study will contribute to validate the role and mechanism of SESN2 in cataract pathogenesis. Thus, in vivo experiments, assessing a wider range of pathological indicators, should be carried out in future. Third, we used one dose of si-SESN2 to evaluate the effect of SESN2 on HLECs; observing the effect of different doses of si-SESN2 on HLECs is also a worthwhile study direction.
Conclusion
SESN2 expression was down-regulated in cataract lens tissue and up-regulated in HLECs treated with H2O2. Moreover, up-regulation of SESN2 inhibited cell apoptosis and inflammation and alleviated oxidative stress via activating the mTOR/Nrf2 signaling pathway. Our findings provide novel theoretical basis for prevention and treatment of cataract.
Footnotes
Author contributions
Conception and design: Xiao Tian; collection and assembly of data: Xiao Tian; data analysis and interpretation: Jie Wei; final approval of manuscript: All authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Data availability statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
