Abstract
Objects
This study intends to explore the possible mechanisms of curcumin’s action after knee osteoarthritis.
Methods
Chondrocytes alone were used to mimic the cellular inflammatory response with interleukin IL-1β. Overexpressing TRPM2 chondrocytes were constructed using cell transfection technique for mechanism verification. The proliferation of chondrocytes was assessed by CCK8 assay, cellular ROS level was detected by flow cytometry, cellular inflammatory factor content was detected by ELISA kit, and molecules of cellular pyroptosis-related signaling pathway were detected by western blot and immunofluorescence. In vivo experiments, a rat knee osteoarthritis model was constructed. Cartilage integrity was assessed by histological analysis, cellular inflammatory factor content was detected by ELISA kit, and cellular pyroptosis-related signaling pathway molecules were detected by western blot and immunohistochemistry.
Results
Curcumin targeting the TRPM2/NLRP3 signaling axis significantly inhibited IL-1β induced decrease in cell viability, increase in ROS level, secretion of inflammatory factors such as TNF-α, IL-6, IL-10, etc., as well as decreased the expression of cellular scaffolding-related proteins, such as GSDMD, NLRP3 and pro-caspase-1, etc. (p < .05). Meanwhile, curcumin targeting the TRPM2/NLRP3 signaling axis also significantly improved the pathological state of cartilage tissue, maintained cartilage integrity, and reduced the secretion of inflammatory factors, and treated osteoarthritis of the knee in rats by mediating cellular pyroptosis.
Conclusions
Curcumin can effectively improve the inflammatory response of chondrocytes through the TRPM2/NLRP3 signaling axis in the treatment of osteoarthritis of the knee in rats.
Introduction
Knee osteoarthritis (KOA) is a persistent degenerative condition resulting from multiple factors. It is primarily characterized by the deterioration of articular cartilage structures and the presence of prolonged chronic inflammation. This condition involves pathological alterations in various structures, including the subchondral bone, synovium and joint capsule. 1 KOA has a prevalence in the clinic, and the incidence rate is showing an upward trend. 2 Currently, the main therapeutic options for relieving inflammation in KOA include the administration of nonsteroidal anti-inflammatory drugs (NSAIDs) and intra-articular corticosteroid injections, 3 which are effective in relieving inflammation but difficult to slow down the disease process.
Cellular pyroptosis is a controlled type of cell death that is activated by inflammatory vesicles and the activation of cysteine asparaginase. This activation leads to the production of pores in the plasma membrane, causing the cell to inflate or burst. Cellular pyroptosis is a significant factor in the development of numerous disorders. 4 Research has demonstrated a strong connection between cellular pyroptosis and the skeletal system. Specifically, the continuous activation of inflammatory vesicles leads to pyroptosis in the nucleus pulposus, which speeds up the degeneration of intervertebral discs. 5 Activation of inflammatory vesicles during the acute phase of spinal cord injury leads to neuronal cell pyroptosis thereby exacerbating secondary spinal cord injury. 6 Despite a large body of literature on the mechanisms and functions of pyroptosis, the mechanisms of how pyroptosis is involved in KOA and how it ameliorates the effects of pyroptosis after KOA have not been fully elucidated.
Natural active ingredients of plants have a wide range of applications in the field of medicine and health due to their naturalness, safety and efficacy. Curcumin is a plant polyphenol with biological functions such as anti-inflammatory, anti-apoptotic and antioxidant.7–9 Clinical studies have shown that curcumin can improve diabetes, chronic kidney disease, migraine and tumor.10–13 In vitro experiments have shown that curcumin can delay the progression of KOA lesions by scavenging ROS to achieve an anti-inflammatory effect. 14 TRPM2 (Transient Receptor Potential Cation Channel Subfamily M Member 2) is a Ca2+ non-selective cation channel, which is expressed in blood cells as well as blood vessels. 15 Studies have shown that ROS and intracellular Ca2+ activate TRPM2, which in turn stimulates the secretion of inflammatory factors from NLRP3 inflammatory vesicles, further exacerbating the body’s inflammatory response.15,16
This work aimed to examine the impact of curcumin on the activation of NLRP3 inflammatory vesicles in chondrocytes following knee osteoarthritis. To do this, we established a rat model of KOA and analyzed the potential mechanism underlying curcumin’s effects in this context.
Materials
Animals and cells
Fifty 4-week-old SPF-grade male SD rats (Jinan Pengyue Experimental Animal Breeding Co., Ltd (SCXK(L)2022 0006), CHN), weighing 210–240 g, at temperature (21 ± 2)°C, humidity 50% ± 5%, natural circadian rhythm, and free access to food and water. Human chondrocytes (Pricella, CHN, CP-H107).
Drugs and reagents
Sodium iodoacetate (MIA) (Shanghai Macklin Biochemical Technology Co., Ltd, CHN, S817623); isoflurane (Hebei Jindafu Pharmaceutical Co., Ltd, CHN, H20203325); cADPR (MCE, USA, HY-N7395); IL-1β recombinant protein (MCE, USA, HY-P701104); curcumin (Sigma, USA, 08,511); ELISA kits (Thermo Fisher, USA): TNF-α (BMS607-3), IL-1β (BMS6002), IL-6 (KMC0061), IL-10 (BMS614); BCA protein detection kit (Thermo Fisher , USA, 23235); fetal bovine serum (Gibco Invitrogen, USA, 10,099); antibodies: TRPM2 (Abcam, USA, ab69644), NLRP3 (proteintech, CHN, 68102-1-lg), Caspase-1 (Proteintech, CHN, 22915-1-AP), pro-Caspase-1 (Abcam, USA, ab238972), GSDMD (CST, USA, # 39754S), GAPDH (CST, USA, # 2118S), IL-1β (CST, USA, # 12242S), TNF-α (CST, USA, # 3707S), IL-6 (CST, USA, # 12153S), IL-10 (CST, USA, # 12163S), FITC Goat Anti-Rabbit/Mouse IgG (H + L) (Abclonal, CHN, AS011/ AS001); CCK8 kit ( DOJINDO LABORATORIES, JP, CK04); Tunel stain kit (Abcam, USA, ab206386); HE staining kit (Solarbio, CHN, G1120); Safranine o-fast green stain kit (Solarbio, CHN, G1371); ROS detection kit ( COIBO BIO, CHN, CB10450-Ra); RNAiso plus kit (Whatman, UK, 9108Q); CellROX™ green flow cytometric assay kit (Thermo Fisher, USA, C10492).
Instruments
Fully automated flow cytometer (Agilent, USA, NovoCyte), fully automated enzyme labeling instrument (Molecular Devices, USA, SpectraMax Gemini XPS), microscope (OLYMPUS, JP, CX31), multifunctional micrographic analyzer (Leica, DE, LK-5GHD), fluorescence quantitative PCR instrument (Thermo Fisher, USA, QuantStudio 1 Plus), protein gel image analysis system (Bio-rad, USA, GS-900) and so on.
Methods
Animal modeling and grouping
Following a 1-week period of acclimation feeding, the 50 rats were randomly assigned to five groups: sham group, model group, curcumin group (150 mg/kg), 17 OE-TRPM2 (cADPR, TRPM2 activator, 100 µM) group, 18 and curcumin + OE-TRPM2 group, with eight animals in each group. After anesthesia (intraperitoneal anesthesia with 1% pentobarbital sodium injection at a rate of 0.4 mL/100 g) in the model group, the medial collateral ligament and anterior cruciate ligament were cut off, the medial meniscus was removed and sutured intradermally, and penicillin was injected intraperitoneally once a day for three consecutive days after surgery to prevent infection. Starting from 5 days after surgery, the rats were driven to run for 30 min every day, and were allowed to move freely in the cage for the rest of the time, and the drugs were administered by gavage for 2 weeks of treatment after 2 weeks of consecutive driving. 19 The sham group was not resected, and the rest of the surgical procedure was the same as that of the model group. After execution of the rats, knee joint specimens were removed for subsequent processing. The animal operations were carried out in complete compliance with the ARRIVE standards, the National Institutes of Health handbook for the care and use of Laboratory animals (NIH Publications No. 8023, amended 1978), and the recommendations of the standards for Care and Use of Laboratory Animals. The tests received approval from the Medical Ethics Committee of the Affiliated Hospital of Shandong University of Traditional Chinese Medicine (Protocol Number: SDSZYYAWE20240315001).
HE staining, safranine O-fast green staining, tunel staining and evaluation
Samples were immersed in 4% paraformaldehyde before decalcification, embedding and sectioning. Tissue sections were dewaxed and hydrated followed by dropwise staining with hematoxylin-eosin solution/ safranine O-fast green solution/tunel solution, ethanol gradient dehydration, neutral gum sealing and other steps. The rat knee joint organization was observed under a microscope and photographed. TUNEL-positive cells were counted using ImageJ’s “Analyze Particles” function. Based on the number of apoptotic cells and the total number of cells, the percentage of apoptotic cells was calculated using the formula “
Immunohistochemical staining
Samples were immersed in 4% paraformaldehyde before decalcification, embedding and sectioning. After the tissue sections were dewaxed and hydrated, they were subjected to high-temperature antigen repair, followed by endogenous occlusion using 3% H2O2 and exogenous occlusion using 5% BSA. After the end of occlusion, diluted antibodies (TRPM2, 1:200; NLRP3, 1:200; GSDMD, 1:200) were added dropwise to the sections and stored in 4°C overnight, and the primary antibody incubation was completed, followed by the addition of the secondary antibody for 1 h, followed by the staining of the nuclei and dehydration of the transparent sealing film, and the protein expression was observed under the microscope.
ELISA assay
The rats were sedated and given medical treatment. Blood was extracted from the abdominal aorta and kept at room temperature for 30 min. It was then centrifuged at a speed of 3000 rpm for 10 min, with a centrifugation radius of 13.5 cm. The serum was collected after centrifugation. The serum was analyzed for IL-1β, TNF-α, ROS, IL-6 and IL-10 in accordance with the instructions of the ELISA kit.
Western blot
The lysed samples were centrifuged at 4°C for 10 min (12 000 r/min), and the supernatant was taken. The protein concentration was determined by BCA kit. Appropriate amount of sample solution was added and the protein was denatured at 100 °C × 10 min. 20 μg of the sample was taken for electrophoresis, membrane transfer and blocking. Antibodies (NLRP3, 1:1000; pro-Caspase-1, 1:500; Caspase-1, 1:2000; GSDMD, 1:2000; TRPM2, 1:500) were added overnight according to the instructions, and add secondary antibody and incubate at room temperature for 2 h. ECL color development solution was added to the samples, and the reaction was avoided from light for 1 min, then the samples were developed and fixed. The internal reference control employed in this study was GAPDH, and the grayscale values of each band were evaluated using ImageJ software.
Chondrocyte grouping and plasmid transfection
The logarithmic growth phase cells were taken, and the normal cultured cells were used as the control group. Chondrocytes were stimulated with 10 ng/mL of IL-1β to mimic chondrocyte inflammatory injury, 20 and they were divided into the model group and curcumin group (10 ng/mL). 21 The TRPM2 overexpression plasmid and negative control were transfected into chondrocytes for 6 h, and then treated with 10 ng/mL of IL-1β for 24 h. They were divided into the OE-TRPM2 group, and curcumin + OE-TRPM2 group.
CCK8 assay
The cells in the logarithmic growth phase were added to 96-well plates at a concentration of 2 × 104 cells per well. Afterwards, 100 μL of cell culture solution was added to each well. After 12 h of inoculation, the cells were divided into groups and treated with drugs, with three wells per group. Following the addition of each group with their respective medications or reagents, the plate was placed in an incubator at a temperature of 37°C with a CO2 concentration of 5% for a duration of 2 h. After a 2-h interval, CCK8 was added to start the reaction, and the optical density (OD) values of the cells in each group were measured for 3 h. The cells were then put in an incubator set at a temperature of 37°C for a period of 2 h. The optical density (OD) values of cells in each group were measured after a 3-h period.
RT-PCR assay
The RNAiso plus kit was used to extract total RNA, following the provided instructions. Subsequently, cDNA was synthesized using reverse transcription of the extracted total RNA. cDNA was then subjected to real-time polymerase chain reaction (RT-PCR), and the PCR steps and analytical methods were performed as described previously. 22 Primers were synthesized by Invitrogen (UK). Primer sequences: TRPM2 forward primer: 5′-CTACGATCCTCCCTTTTACACC-3′; reverse primer: 5′-ATCCACGACG TTGTAACTGATC-3′; GAPDH forward primer: 5′-ATGACTCTACCCACGGCAAG-3′; reverse primer: 5′-GGAAGATGGTGATGGGTTTC-3′.
Flow cytometry
The cell solution was suspended in PBS twice and subsequently utilized to quantify the quantity of cellular reactive oxygen species (ROS) utilizing the CellROX™ green kit. The analysis was conducted utilizing a completely automated flow cytometer. Following oxidative stress, the reagent attaches to DNA and produces a robust fluorescent signal, which is then quantified as the cellular quantity of ROS.
Immunofluorescence assay
Cells were fixed in 4% paraformaldehyde for 15 min, incubated in 0.1% Triton X-100 for 10 min, and closed in 1% BSA for 30 min before adding primary antibodies (TRPM2, 1:50; NLRP3, 1:100; IL-1β, 1:50) according to the reagent vendor’s instructions. Following an overnight incubation at 4°C and a 30-min wash with PBST, a secondary antibody, specifically FITC Goat Anti-Rabbit/Mouse IgG (H + L) at a dilution of 1:100, was added and agitated at room temperature for 1 h. After incubating for 5 min with DAPI at room temperature and then rinsing with PBS, the fluorescence signals were examined using a fluorescence microscope.
Statistical analysis
All data are expressed as mean ± standard error of the mean (SEM). The study utilized one-way analysis of variance (ANOVA) to compare between three or four groups. A significance level of p <.05 was used to determine statistical significance. The statistical analysis was conducted using the statistical program GraphPad Prism 8.0.
Results
Curcumin can significantly improve chondrocyte inflammatory injury
First, we successfully constructed an overexpression TRPM2 cell line (OE-TRPM2, Figure 1(A)). The results of the CCK8 assay were in agreement with the ELISA assay (Figure 1(B)). As shown Figure 1(C)–(F), the levels of inflammatory factors exhibited a notable increase in the model group in comparison to the control group. Furthermore, the OE-TRPM2 group demonstrated a considerably greater level of inflammatory factors compared to the model group. Curcumin treatment significantly reduced the levels of IL-1β, IL-6, TNF-α, and IL-10 compared to the model group, and the levels of inflammatory factors were also significantly lower in the curcumin + OE-TRPM2 group compared to the OE-TRPM2 group.We detected the cellular ROS level. The level of reactive oxygen species (ROS) in the model group was substantially greater than that in the control group, while the amount of ROS in the OE-TRPM2 group was significantly more than that in the model group. ROS levels were notably reduced in the curcumin group compared to the model group, and in the curcumin + OE-TRPM2 group compared to the OE-TRPM2 group (Figure 1(G) and (H)). These data suggest that curcumin may ameliorate chondrocyte oxidative stress and alleviate IL-1β-induced inflammatory responses by targeting TRPM2. Curcumin can significantly improve chondrocyte inflammatory injury. (A) RT-PCR to detect cell transfection efficiency. (B) CCK8 kit to detect cell viability. (C–F) ELISA kits to detect cytokine content. (G and H) Flow cytometry to detect ros levels. (I and J) Western blot detection of pathway-associated protein expression levels. OE-TRPM2, overexpressed TRPM2. TRPM2, transient receptor potential cation channel subfamily m member 2; TNF-α, tumor necrosis factor-α; IL-6, interleukin 6; IL-10, interleukin 10; IL-1β, interleukin 1 beta; NLRP3, nlr family pyrin domain containing 3; Caspase-1, cysteinyl aspartate specific proteinase-1; GSDMD, gasdermin d; GAPDH, glyceraldehyde-3-phosphate dehydrogenase. ROS, reactive oxygen species. Mean ± SEM. n = 3. **p < .01, ***p < .001.
Curcumin improves chondrocyte inflammation by regulating the TRPM2/NLRP3 signaling axis and mediating cellular pyroptosis
To better understand how curcumin works to improve inflammatory damage in chondrocytes, we analyzed the markers of cellular pyroptosis-related pathways using western blot (Figure 1(G) and (I)) and immunofluorescence assays (Figure 2). Compared with the control group, the expression of NLRP3, GSDMD, Caspase-1, NLRP3 and other genes in the model group was significantly higher, and the gene expression level of OE-TRPM2 group was significantly higher compared with than the model group. In contrast, gene expression levels were significantly lower in the curcumin group compared to the model group, and gene expression levels were also significantly lower in the curcumin + OE-TRPM2 group compared to the OE-TRPM2 group. These data suggest that curcumin may ameliorate the inflammatory response of chondrocytes by regulating the TRPM2/NLRP3 signaling axis to mediate cellular death. Curcumin significantly reduces chondrocyte inflammatory factor expression. (A and B) IL-1β immunofluorescence plots and data analysis. (C and D) NLRP3 immunofluorescence plots and data analysis. (E and F) TRPM2 immunofluorescence plots and data analysis. Mean ± SEM. n = 3. *p < .05, **p < .01, ***p < .001 (scale bar, 50 μm).
Curcumin can effectively ameliorate osteoarthritis of the knee in rats
Our in vitro experiments demonstrated that curcumin improves chondrocyte inflammatory response by mediating cellular pyroptosis through modulation of the TRPM2/NLRP3 signaling axis. Next, we verified our hypothesis by constructing a rat model of osteoarthritis of the knee. Histopathological and immunohistochemical investigations were conducted to evaluate the impact of curcumin on the inflammatory response in rats with knee osteoarthritis. The level of apoptosis was markedly higher in the model group compared to the sham group. Furthermore, it was significantly higher in the OE-TRPM2 group compared to the model group. Conversely, the curcumin group exhibited a significantly lower level of apoptosis. Additionally, the curcumin + OE-TRPM2 group demonstrated a significantly lower level of apoptosis compared to the OE-TRPM2 group (Figure 3(A) and (D)). The H&E staining revealed that both the model and OE-TRPM2 group had damaged cartilage membranes, a significant decrease in the number of chondrocytes, disorderly arrangement, and uneven staining of the matrix. However, curcumin treatment significantly improved the cartilage damage in rats, and the cartilage layer was repaired to a certain extent, and the matrix staining was more uniform (Figure 3(B)). Similarly, the results of safranine o-fast green staining showed that the model group and OE-TRPM2 group had obvious cartilage surface layer destruction, obvious reduction of saffron staining, and serious cartilage damage, while the saffron staining showed obvious after curcumin intervention treatment, which had the effect of improving joint inflammation and protecting cartilage (Figure 3(C)). To study the inflammatory changes in each group, we measured serum IL-1β, IL-6, TNF-α, IL-10 and ROS levels (Figure 3(E)–(I)). Consistent with the tunel staining results, the model and OE-TRPM2 groups showed significantly elevated levels of inflammatory factors, whereas curcumin intervention treatment significantly reduced the levels of inflammatory factors in rats. These data suggest that curcumin may ameliorate knee osteoarthritis in rats by targeting TRPM2. Curcumin can effectively ameliorate osteoarthritis of the knee in rats. (A and D) Rat bone tissue tunel staining. (B) Rat bone tissue HE (hematoxylin-eosin) staining. (C) Rat bone tissue safranin o-fast green staining. (E–I) Detection of inflammatory factors in rat serum by elisa kit. Mean ± SEM. n = 5. *p < .05, **p < .01, ***p < .001 (scale bar, 200 μm).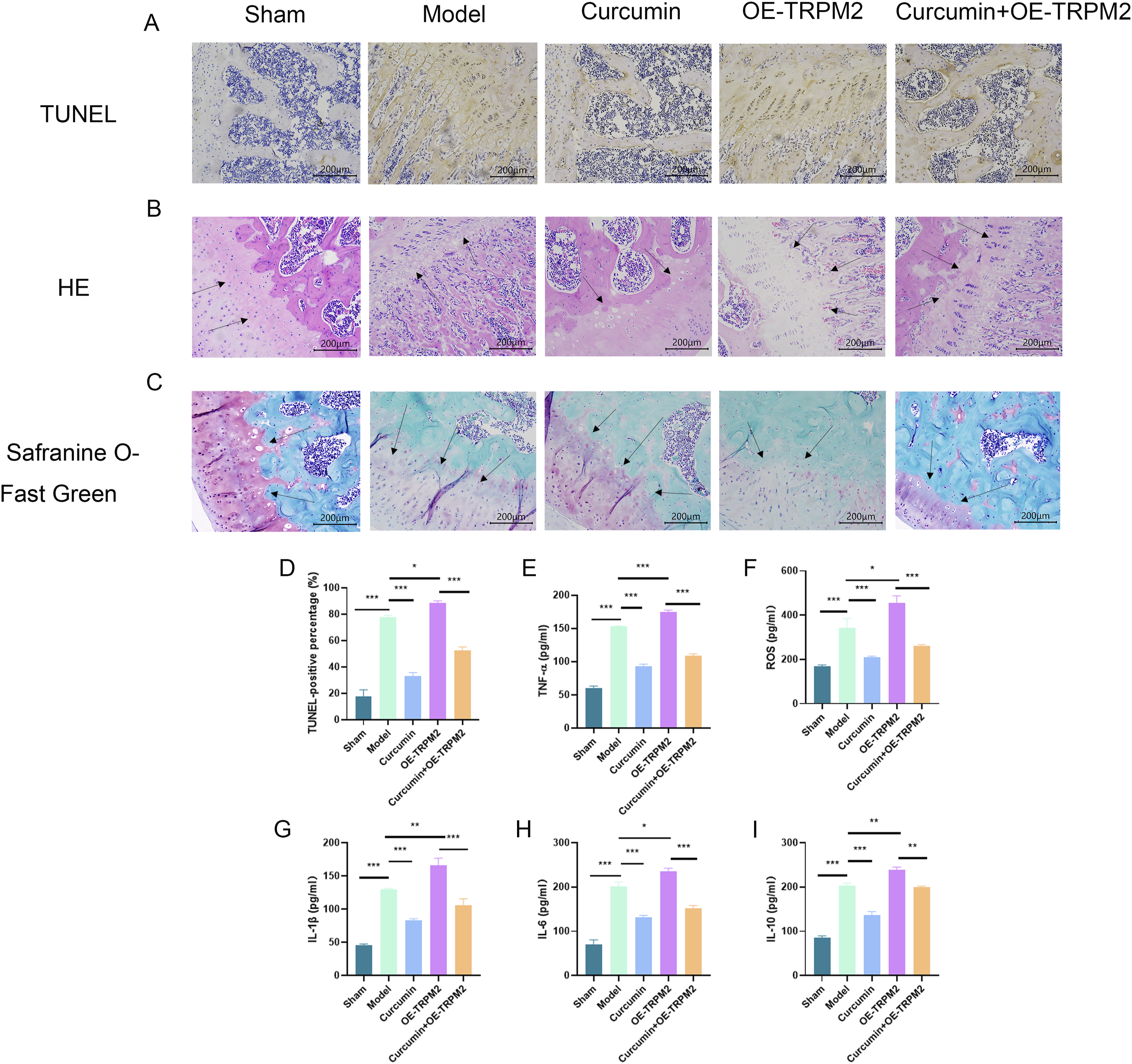
Curcumin improves knee osteoarthritis in rats by regulating TRPM2/NLRP3 signaling axis-mediated cellular death
In order to further verify the mechanism of curcumin’s action in ameliorating osteoarthritis of the knee in rats, we detected the indicators of cellular pyroptosis-related pathways by western blot and immunohistochemistry experiments. As depicted in Figure 4, the expression levels of genes including NLRP3, GSDMD, TRPM2, Pro-caspase-1, etc. were markedly elevated in the model group compared to the sham group, aligning with the findings of in vitro tests. By comparison, the curcumin group exhibited dramatically reduced gene expression levels compared to the model group. Additionally, the curcumin + OE-TRPM2 group had significantly lower gene expression levels compared to the OE-TRPM2 group. The studies indicate that curcumin may improve osteoarthritis in rat knee osteoarthritis by controlling the TRPM2/NLRP3 signaling pathway to regulate cell death. Curcumin improves knee osteoarthritis in rats by regulating TRPM2/NLRP3 signaling axis-mediated cellular death. (A–C) Immunohistochemistry of rat bone tissue and data analysis. (D and E) Western blot detection of protein expression levels of related signaling pathways in rat bone tissue and data analysis. Mean ± SEM. n = 5. *p < .05; ***p < .001; ns, no significant difference (scale bar, 200 μm).
Discussion
KOA, or knee osteoarthritis, is a prevalent type of arthritis that is distinguished by the progressive deterioration of the cartilage in the joints. 23 The constraints of existing drug therapy have prompted the advancement of innovative anti-KOA medications. Curcumin possesses excellent qualities that make it a potential frontrunner for the treatment of KOA. In the present study, we combined a rat KOA model with validation experiments on chondrocytes to reveal that curcumin inhibited chondrocyte pyroptosis by targeting the TRPM2/NLRP3 signaling axis, thereby attenuating the pathological features of KOA.
Curcumin is a naturally occurring diphenolic compound with many health biological effects, and there is growing evidence that curcumin has antioxidant, antifibrotic, and anti-inflammatory effects.24,25 Studies have shown that curcumin can inhibit cellular pyroptosis and treat a variety of diseases by regulating signaling pathways such as AKT/Nrf2/ARE and PI3K/AKT/mTOR.26,27 However curcumin has been less reported on chondrocytes and knee osteoarthritis in this aspect of cellular juxtaposition. Curcumin has been recently utilized in prospective clinical trials, demonstrating excellent outcomes in terms of the effectiveness and safety of anti-OA medications. 28 Due to the ethical and clinical problems associated with conducting histologic investigations on living patients, we opted to use rats for our histologic studies.
Cell death mechanisms and routes have been a prominent subject of investigation in recent years. Cellular pyroptosis is a mode of programmed inflammatory death in which, stimulated by signals such as bacterial viruses, intracellular pattern recognition receptors, such as NLRP3 proteins, recognize pathogenic antigenic signals and bind to the junction proteins ASC and pro-caspase-1 to form a multiprotein complex, inflammatory vesicles, and at the same time cleave pro caspase-1, producing active caspase-1. Caspase-1, in turn, has two functions during cellular pyroptosis: on the one hand, it cleaves and activates the inflammatory factors IL-1β and IL-18, which are released into the extracellular space, recruiting a large number of inflammatory cells to amplify the inflammatory response; on the other hand, caspase-1 cleaves the intermediate structural domains of the two domains of the GSDMD proteins, producing a carbon terminus-containing protein with a carbon end. On the one hand, caspase-1 can cut the connection region between the two structural domains of GSDMD protein to produce peptides containing carbon terminus and peptides containing nitrogen-terminal active domains (N-GSDMD), and multiple N-GSDMDs can form oligomers and translocate to the cell membrane for perforation, which makes the cell inside and outside of the cell communicating with each other, and destroys the osmotic balance between inside and outside of the cell membrane, resulting in the cell swelling until rupture and death, and it can also cause more inflammatory factors such as IL-1β and IL-18 to be released to the extracellular space triggering a strong inflammatory response.29,30 At the site of inflammatory injury, a large amount of ROS is generated, which induces activation of cellular TRPM2 channels and an increase in intracellular Ca2+, causing cellular damage leading to an inflammatory response. 31 TRPM2 channels, a newly discovered non-glutamate-dependent ion channel, are involved in the regulation of a number of signaling pathways in vivo through the mediation of the inward flow of cations, in particular Ca2+, and are thereby involved in the occurrence of inflammatory responses and the development of inflammatory responses in a variety of pathological processes.32,33 Processes in the onset and progression of inflammatory responses, which in turn are involved in the development of inflammation-related diseases caused by non-infectious and infectious factors. 32 Numerous studies have shown that TRPM2 channel activation is involved in the activation of NLRP3 inflammatory vesicles, secretion of inflammatory cytokines, and the onset and progression of inflammatory responses induced by a variety of factors.16,33,34 In this study, we employed immunohistochemistry, western blot, immunofluorescence, and other experimental techniques to confirm the mechanism by which curcumin targets the TRPM2/NLRP3 signaling axis to control cellular pyroptosis for the treatment of knee osteoarthritis. Further investigations should examine the intracellular distribution of TRPM2 in response to curcumin treatment and determine the presence of alternative signaling pathways.
Another noteworthy subject is the therapeutic use of curcumin. Prior studies have shown that curcumin has a limited potential to be absorbed into the bloodstream when taken orally. 23 This study utilized intraperitoneal injection as a method to prevent potential cartilage injury resulting from the restricted absorption of curcumin during arthrocentesis and oral dosing in rats. The experimental outcomes were highly favorable. Therefore, it is worthwhile to investigate other approaches for delivering curcumin, such as oral administration and injections into the subcutaneous and articular cavities.
In conclusion, we have successfully verified the chondroprotective effects of curcumin in both laboratory experiments and animal studies of knee osteoarthritis. Furthermore, curcumin might potentially preserve cartilage in knee osteoarthritis (KOA) by reducing the harmful effects of oxidative stress and improving inflammatory responses through the TRPM2/NLRP3 signaling pathway. Therefore, our findings provide new perspectives for future research on the effective management and treatment of KOA.
Conclusion
We reported that curcumin effectively ameliorated chondrocyte inflammatory response through TRPM2/NLRP3 signaling axis for the treatment of knee osteoarthritis in rats. Therefore, we propose that curcumin could be a potential therapeutic agent for knee osteoarthritis treatment.
Footnotes
Author contributions
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Shandong Province (ZR2022MH171).
