Abstract
Acute lung injury (ALI) is a serious pulmonary inflammation disease with high mortality. Irigenin, an isoflavone from rhizomes of the Belamcanda chinensis, has been reported to exert anti-inflammatory, anti-oxidative, and anti-apoptotic activities in several diseases. However, it is still unclear whether irigenin can exert a beneficial effect in ALI. A network pharmacology method was utilized to predict the hub targets and potential therapeutic mechanisms of irigenin against ALI. Lipopolysaccharide (LPS) was used to establish the mice model of ALI for evaluating the effects of irigenin. According to the protein-protein interaction (PPI) network, we identified EGFR, HRAS, AKT1, SRC, and HSP90AA1 as the top five significant genes. Gene Ontology (GO) and Kyoto Encyclopaedia of Genes and Genomes (KEGG) enrichment assays showed that irigenin might affect inflammatory response, cytokine production, and cell death by the mitogen-activated protein kinase (MAPK) signaling pathway. In vivo experiment results manifested that irigenin decreased pathological changes, lung Wet/Dry weight ratio, and total protein content in bronchoalveolar lavage fluid (BALF). Irigenin also reduced the production of inflammatory cytokines, including tumor necrosis factor-a (TNF-a), interleukin-1β (IL-1β), interleukin-6 (IL-6), and interleukin-18 (IL-18), and neutrophil infiltration. Additionally, irigenin inhibited pulmonary apoptosis in LPS-treated ALI mice. Moreover, LPS-induced phosphorylation of p38, JNK, and ERK was significantly abated due to the treatment of irigenin. In summary, irigenin ameliorates LPS-induced ALI by suppressing pulmonary inflammation and apoptosis via inactivation of the MAPK signaling pathway. These findings indicated the therapeutic potential of irigenin in ALI.
Introduction
Acute lung injury (ALI) is a life-threatening respiratory illness accompanied by uncontrollable and overwhelming pulmonary inflammation. 1 ALI is usually triggered by pneumonia, sepsis, severe trauma, or acid inhalation and is likely to develop into highly lethal acute respiratory distress syndrome (ARDS) with respiratory failure and hypoxemia. 2 ALI/ARDS is featured by pneumonedema, diffuse alveolar damage, vascular hyperpermeability, inflammatory cell infiltration, proinflammatory cytokine accumulation, and alveolar epithelial cell death. 3 ARDS occupies approximately 10% of intensive care unit admissions worldwide and has a high mortality ranging from 30% to 40%. 4 ARDS is a common complication in the current pandemic coronavirus disease 2019 (COVID-19), and the fatality rate for COVID-19-associated ARDS is 45%.5,6 The lung-protective mechanical ventilation is a crucial therapeutic intervention in the management of ALI/ARDS patients, however, inappropriate ventilation can result in local and systemic inflammatory responses, further aggravating lung injury. 7 Some kinds of medicaments, such as anti-inflammatory drugs, antioxidants, anticoagulants, statins, beta-agonists, and neuromuscular blocking agents, have been used for the pharmacotherapy of ALI/ARDS. 8 Nevertheless, there are no pharmacological therapies with significant and uniform effects on disease development and mortality. Thus, searching for effective therapeutic drugs to improve the clinical prognosis of ALI patients is in great demand.
Given the complexity of ALI pathogenesis, a variety of natural products have gained researchers’ attention as pleiotropic agents to reduce inflammation and improve pulmonary function. 9 Natural compounds exert their protective effects in ALI primarily through regulating inflammatory responses, oxidative stress, and apoptosis. 10 Botanicals’-derived isoflavones are demonstrated to protect against ALI via anti-inflammatory and antioxidant activities. 11 Irigenin, an isoflavone from rhizomes of the Belamcanda chinensis, has been found to exert important biological activities in cancers, diabetic complications, inflammation, and cardiac injury. 12 However, it remains unclear whether irigenin can exert beneficial effects on ALI.
Network pharmacology has been widely utilized to investigate the molecular targets and pharmacological mechanisms of natural compounds in various human diseases. 13 Lipopolysaccharide (LPS), the main component in the outer membranes of Gram-negative bacteria, is generally used to construct the animal model of ALI. 14 In the current study, we used the network pharmacology approach to identify the key targets, biological processes, and signaling pathways of irigenin in the treatment of ALI. Subsequently, the ALI mice models were established by using LPS to confirm the protective effects and therapeutic mechanisms of irigenin in ALI.
Materials and methods
Predicting the targets of irigenin against ALI
PharmMpper, TargetNet, and SwissTargetPrediction tools were used to predict the potential targets of irigenin. ALI-related targets were obtained from GeneCards (Relevance score ≥ 20), DisGeNET, and Comparative Toxicogenomics Database (CTD) (Inference Score ≥ 50). Then, a Venn diagram was drawn to display the common targets for irigenin and ALI.
Construction and analysis of protein-protein interaction (PPI) network
The PPI network for these common genes was built using the STRING database, followed by the Cytoscape software for visualization. The key genes in this network were filtered out according to the topological parameters calculated from CytoNCA plugin in the Cytoscape.
Gene ontology (GO) and kyoto encyclopaedia of genes and genomes (KEGG) pathway enrichment
We entered these overlapping genes into the Metascape database by limiting the species to “Homo sapiens”. The screening criteria for significant biological processes and KEGG pathways as set as ‘p < 0.01’, ‘count ≥ 3’, and ‘enrichment factor > 1.5’.
Mice model of ALI
All animal experiments adhered to the guide for the care and use of laboratory animals and gained approval from the Animal Ethics Committee of the Second Affiliated Hospital of Army Medical University (approval No. AMUWEC20227500). Adult male C57BL/6 mice (aged 6–8 weeks, weighing 22 ± 2 g) were purchased from the Beijing Vital River Laboratory Animal Technology Co., Ltd. After 7 days of adaptation, mice were randomly assigned to five groups (n = 5 per group): Control, LPS, LPS + irigenin (10 mg/kg), LPS + irigenin (20 mg/kg), and LPS + irigenin (40 mg/kg) groups. Mice in the experimental groups received daily intraperitoneal administration of irigenin (10, 20, or 40 mg/kg), while mice in the control and LPS groups were intraperitoneally injected with the same amount of phosphate buffered saline (PBS). Seven days later, ALI mice model was induced as previously described.15,16 Mice in the LPS and experiment groups group were instilled intratracheally with 5 mg/kg of LPS, while mice in the control group were given equal volumes of PBS. At 6 h after LPS administration, all mice were sacrificed by 50 mg/kg pentobarbital. Then, the bronchoalveolar lavage fluid (BALF) and lung tissues were collected for further analysis.
Histopathological analysis
Lung tissues were rinsed with PBS, fixed in 4% paraformaldehyde, dehydrated with alcohol, embedded in paraffin, and then sliced into 4 μm-thick sections. After being stained with hematoxylin and eosin (H&E), the sections were put under a light microscope for morphology observation. The degree of lung injury was scored following a scale of 0–4, with 0 representing ‘non-destructive injury’, one representing ‘less than 25% injury’, two representing ‘25–50% injury’, three representing ‘50–75% injury’, and four representing ‘more than 75% injury’.
Lung wet/dry weight ratio
After being harvested, a part of the lung tissues was instantaneously taken for the measurement of wet weight. Next, the tissue samples were dried in an oven at 60°C for 72 h to obtain the dry weight. Finally, the pulmonary edema was evaluated by calculating the lung wet/dry weight ratio.
BALF collection
The mice were euthanized and the thorax was opened. Then, a small incision was made on the trachea. The lungs were gently lavaged 3 times with 0.5 mL precooled PBS to harvest the BALF. The number of total inflammatory cells in the BALF was counted using a hemocytometer. After centrifugation at 3000 g for 10 min, the protein concentration in the supernatant was determined using a BCA protein assay kit (Solarbio, Beijing, China).
Enzyme-linked immunosorbent assay (ELISA)
The levels of tumor necrosis factor-a (TNF-a), interleukin-1β (IL-1β), interleukin-6 (IL-6), and interleukin-18 (IL-18) in BALF were detected using corresponding ELISA kits (R&D systems, Minneapolis, MN, USA) as previously described. 17
Myeloperoxidase (MPO) activity assay
The pulmonary tissues were homogenized at 4°C and the homogenate was centrifuged at 10,000 r/min for 10 min. The MPO activity in lung homogenates was analyzed using the MPO kit (Jiancheng Bioengineering Institute, Nanjing, China) as previously described. 18 The absorbance at 460 nm was measured with a microplate reader.
Immunohistochemistry (IHC) assay
The paraffin-embedded lung tissues were dewaxed, rehydrated, heated in water bath for antigen retrieval, and treated with 3% H2O2 to block the endogenous peroxidase activity. After being blocked in 3% bovine serum albumin (BSA) for 20 min, the sections were incubated with the antibody against MPO (Abcam, Cambridge, UK) overnight at 4°C, followed by incubation with the appropriate secondary antibodies for 1 h at room temperature. Following treatment with 3,3′-diaminobenzidine and hematoxylin, the sections were finally analyzed under a microscope at magnification ×200.
Terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) staining
The In Situ Cell Death Detection Kit (Shanghai Ruisai Biotechnology Co., Ltd., China) was used to detect apoptosis in lung tissues. Following deparaffinization and rehydration, the lung tissue sections were treated with proteinase K for 20 min at 37°C. Then, the reaction solution (TDT:dUTP, 1:10) was added to the sections for 2 h of incubation at 37°C. After being stained with 4′,6-diamidino-2-phenylindole (DAPI) for 10 min, the slices were observed under a fluorescence microscope and the TUNEL-positive cells were calculated.
Western blot assay
Western blot assay was performed according to a previous study. 19 The lung tissues were lysed using radioimmunoprecipitation assay (RIPA) buffer containing phenylmethylsulfonyl fluoride (PMSF) and phosphatase inhibitors. A BCA protein kit was utilized to determine the protein concentraitons. The denatured protein samples (50 μg) were separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and then transferred to nitrocellulose membranes. Then, the membranes were consecutively incubated with 5% skim milk, specific primary antibody, and horseradish peroxidase (HRP)-conjugated secondary antibody. After that, the protein signals were developed using a chemiluminescence reagent and quantified with the ImageJ software. The primary antibodies including p38, p-p38, JNK, p-JNK, ERK, p-ERK, and β-actin were purchased from Cell Signaling Technology (Danvers, MA, USA).
Statistical analysis
Data are expressed as mean ± standard deviation (SD). Difference was analyzed using one-way analysis of variance (ANOVA) followed by Dunnett’s multiple comparison test. The statistical significance was set at p < 0.05.
Results
Identifying the targets of irigenin against ALI
The 2D chemical structure of irigenin is shown in Figure 1A. According to the PharmMpper, TargetNet, and SwissTargetPrediction, we identified 385 potential genes of irigenin (Supplemental Table 1). Additionally, we collected 1932 ALI-related targets from the GeneCards, DisGeNET, and CTD databases (Supplemental Table 2). Then, a Venn diagram was constructed to identify the candidate targets of irigenin in ALI treatment. As presented in Figure 1B, there were a total of 184 potential genes that might be involved in the therapeutic effects of irigenin in ALI. Potential targets of irigenin against ALI. (A) Chemical structure of irigenin. (B) Venn diagram showing the common genes of irigenin and ALI.
Identifying the key genes of irigenin acting on ALI
Then, these 184 overlapping genes were input into the STRING database to construct the PPI network. Cytoscape software was used to analyze this network based on the values of Betweenness Centrality (BC), Closeness Centrality (CC), and Degree Centrality (DC). As presented in Figure 2A, there were 176 nodes and 835 edges in this network, and the median values of DC, CC, and BC were 6, 0.354, and 56.615, respectively. According to the DC ≥ 12 (double the median DC), we acquired a network with 49 nodes and 378 edges (Figure 2B). The obtained median values of DC (14), CC (0.585), and BC (15.428) were set as the threshold for the next round of screening. Finally, a core network consisting of 23 nodes and 150 edges was received (Figure 2C and Supplemental Table 3). EGFR, HRAS, AKT1, SRC, HSP90AA1, RHOA, IL2, MAPK14, PTPN11, and PTK2 were identified as the top ten key genes for irigenin to treat ALI. PPI network construction and analysis for the overlapping genes. (A–C) The core genes in the PPI network were identified according to the topological parameters including DC, BC, and CC.
Function and pathway enrichment analysis
The 176 common genes were entered into the Metascape database to perform GO-BP and KEGG enrichment assays. As shown in Figure 3A, multiple biological processes were involved in the treatment of ALI, such as protein phosphorylation, response to lipopolysaccharide, regulation of inflammatory response, positive regulation of cytokine production, and positive regulation of cell death. According to the KEGG results, these genes were mainly enriched in pathways in cancer, lipid and atherosclerosis, MAPK signaling pathway, chemical carcinogenesis-reactive oxygen species, FoxO signaling pathway, and VEGF signaling pathway (Figure 3B). Of note, MAPK signaling was the most significant one associated with ALI. Enrichment analysis for the overlapping targets. (A) GO-BP enrichment analysis of irigenin against ALI. (B) KEGG pathway enrichment of irigenin against ALI.
Irigenin ameliorated ALI in LPS-induced mice
LPS-induced mice were used to investigate whether irigenin could exert a protective effect on ALI. The histopathologic features of lung tissues were evaluated by H&E staining. Compared with the control group, obvious alveolar damage, alveolar hemorrhage, interstitial thickening, and inflammatory cell infiltration were observed in LPS-treated mice. However, these pathological symptoms were effectively abated after treatment with irigenin (Figure 4A). The results of lung injury scores confirmed the ameliorating effect of irigenin on ALI (Figure 4B). Moreover, the wet/dry weight ratio of lung tissues in the LPS group was higher than that in the control group and was significantly reduced under the irigenin pretreatment (Figure 4C). Also, LPS administration resulted in a significant increase of total cells in BALF, which was greatly reversed to varying degrees by irigenin (Figure 4D). Irigenin ameliorated pulmonary injury in LPS-induced ALI mice model. Mice received daily intraperitoneal administration of irigenin (10, 20, and 40 mg/kg) for 7 days before intratracheal instillation of LPS. Lung tissues or BALF were harvested 8 h after LPS treatment. (A) H&E staining was used to assess the pathological alterations of lung tissues (magnification ×200). (B) A lung injury score was used to evaluate the severity of pulmonary damage. (C) Lung wet/dry weight ratio. (D) Total protein content in BALF was detected.
##
p < 0.01,
###
p < 0.001 vs. control group.*p < 0.05, **p < 0.01, ***p < 0.001 vs. LPS group.
Irigenin suppressed pulmonary inflammation in LPS-treated mice
To explore the anti-inflammatory effects of irigenin in ALI, we measured the levels of proinflammatory cytokines in BALF by using ELISA kits. Compared with the control group, the levels of TNF-α, IL-6, IL-β, and IL-18 in BALF were significantly increased in LPS-treated mice. However, irigenin treatment decreased the levels of these inflammatory markers (Figure 5A-D). The number of total cells in BALF was significantly increased in mice exposed to LPS. Compared to the LPS group, irigenin treatment lowered the number of total cells (Figure 5E). Then, we evaluated the effect of irigenin on neutrophil infiltration. As shown in Figure 5F, we found a significant increase of MPO (a neutrophil marker) activity in lung tissues, while this change was abated by irigenin treatment. Additionally, IHC analysis showed the expression of MPO was dramatically increased in the LPS group, while irigenin administration decreased MPO content (Figure 5G). Irigenin inhibited LPS-induced inflammation in ALI mice. (A–D) ELISA assays were performed to measure the levels of TNF-α, IL-6, IL-1β, and IL-18 in BALF samples. (E) A hemocytometer was used to count the number of total cells in BALF. (F) MPO activity analysis in the lung tissues. (G) Representative IHC images for MPO in the lung tissues.
##
p < 0.01,
###
p < 0.001 vs. control group. *p < 0.05, **p < 0.01, ***p < 0.001 vs. LPS group.
Irigenin repressed LPS-induced apoptosis in ALI mice
TUNEL staining was used to investigate the effects of irigenin on apoptosis in LPS-treated mice. In the LPS group, TUNEL-positive cells were significantly increased. After treatment with irigenin, TUNEL-positive cells were decreased (Figure 6A and B). Irigenin suppressed LPS-induced lung tissue apoptosis in ALI mice. (A) The apoptosis of lung tissues was examined using TUNEL staining. (B) Semiquantitative analysis of lung tissue apoptosis.
###
p < 0.001 vs. control group. **p < 0.01, ***p < 0.001 vs. LPS group.
Irigenin inactivated MAPK signaling in LPS-induced mice
The effect of irigenin on MAPK signaling was assessed by measuring the expression of p38, ERK, and JNK. Western blot assays showed that compared with the control group, the expression of phosphorylated p38, ERK, and JNK was significantly increased in the LPS group. However, irigenin administration inhibited LPS-induced increase of phosphorylated p38, ERK, and JNK (Figure 7A-D). Irigenin inactivated the MAPK signaling pathway in LPS-treated mice. (A) Western blot images of p38, JNK, ERK, and their corresponding phosphorylated proteins in the lung tissues. (B–D) Quantitative analysis of p38, JNK, ERK, and their corresponding phosphorylated proteins in the lung tissues.
##
p < 0.01,
###
p < 0.001 vs. control group. **p < 0.01, ***p < 0.001 vs. LPS group.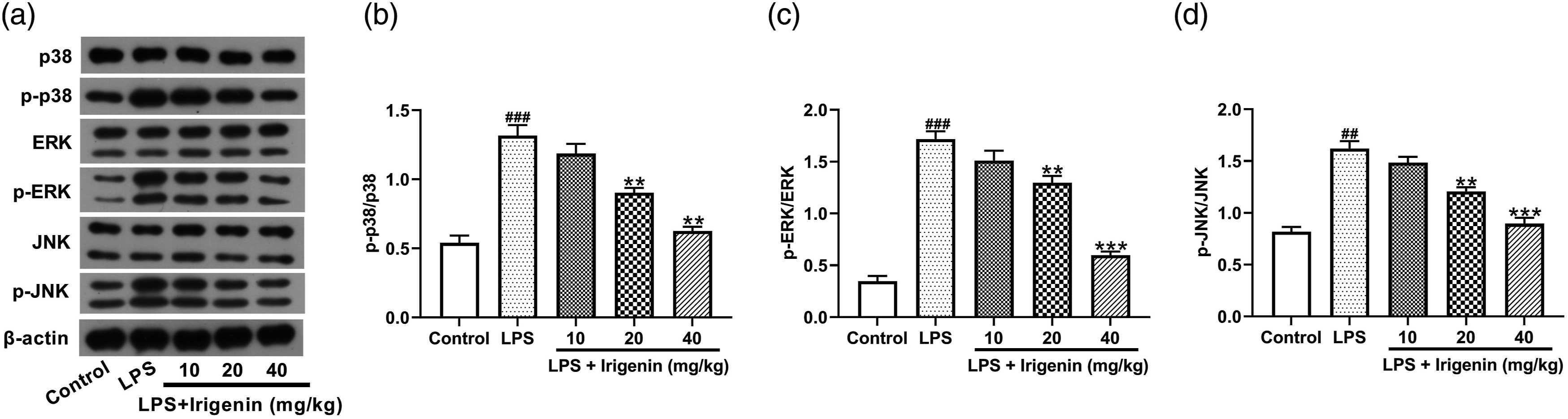
Discussion
Acute lung injury is a complicated pulmonary syndrome caused by different factors, which can progress into ARDS with high mortality. 20 Unfortunately, no effective pharmacologic treatments are approved up to now. 21 Herbal active ingredients exhibit anti-ALI activity by affecting immune cell function, preventing inflammatory response, alleviating oxidative stress, and regulating disease-related signal pathways. 22 Irigenin has been demonstrated to inhibit the inflammatory response, oxidative stress, and apoptosis in several human diseases. For instance, irigenin attenuated doxorubicin-induced cardiac dysfunction and cardiotoxicity by inhibiting apoptosis, inflammation, and oxidative stress. 23 Irigenin abated MPP+-induced neurotoxicity in BV-2 cells by repressing inflammation, oxidative stress, and apoptosis via activating the Keap1/Nrf2 pathway. 24 Irigenin protected human umbilical vein endothelial cells against angiotensin II-induced oxidative stress and apoptosis through activating the Nrf2 pathway. 25 Herein, we applied a network pharmacology method followed by in vivo experiments to confirm the biological significance and molecular mechanisms of irigenin in ALI treatment.
Based on the public databases, a total of 184 genes were filtrated as the possible therapeutic targets for irigenin to treat ALI. PPI network showed EGFR, HRAS, AKT1, SRC, and HSP90AA1 as the top five significant hub genes. GO and KEGG enrichment assays showed regulation of inflammatory response, positive regulation of cytokine production, and positive regulation of cell death as the important biological processes, and MAPK signaling as the key pathways of irigenin in ALI treatment. From the data from network pharmacology, we speculated that irigenin can exert the therapeutic effect in ALI by regulating inflammatory response, cytokine production, and cell death via MAPK signaling pathway.
Uncontrolled inflammation is a major pathological characteristic of ALI. 26 Activated inflammatory cells, such as macrophages and neutrophils, can produce a large number of proinflammatory cytokines, disrupt endothelium and alveolar epithelial barrier, and increase alveolar-capillary permeability, resulting in protein effusion and alveolar edema. 27 Plenty of inflammatory mediators, such as TNF-α, IL-1β, IL-6, and IL-18, were involved in the initiation of ALI. 28 In ALI, the early proinflammatory cytokines activate more immune cells to produce a greater number of cytokines and accumulate more neutrophils and macrophages, thereby initiating a chain reaction that further aggravates the severity of lung damage. 29 Some pro-inflammatory cytokines in the plasma and BALF are demonstrated as useful biomarkers in ALI patients, which provide help in diagnosing disease and assessing the patient’s clinical state and therapeutic efficacy. 30 MPO is a proinflammatory enzyme mainly released by neutrophils. The MPO level and activity in BALF and tissue homogenates are generally used as an index to evaluate the degree of neutrophil infiltration into tissues. 31 LPS-induced mouse is a widely accepted and repeatable ALI model for experimental research. 32 In our study, treatment with irigenin alleviated pulmonary pathological changes, wet/dry weight ratio, and total protein levels in BALF in LPS-treated mice. Moreover, irigenin treatment suppressed inflammatory cytokine secretion and inflammatory cell infiltration. These data indicated that irigenin ameliorated diffuse alveolar injury and pulmonary edema in ALI through its anti-inflammation activity.
Apoptosis occurs in multicellular organisms and maintains a homeostatic balance between the rate of cell formation and cell death. 33 Excessive apoptosis exerts an important role in the progression of ALI, and LPS can promote cell apoptosis by triggering inflammatory response and mitochondrial damage. 34 Here, TUNEL staining showed that LPS-induced apoptosis in mice was significantly attenuated after irigenin administration. These data suggested that the protective effect of irigenin in ALI may be partially attributed to its anti-apoptosis role.
MAPK pathway, activated in response to extracellular and intracellular stimuli, can regulate multiple cellular activities including proliferation, differentiation, survival, and death in different human diseases. 35 Activation of the MAPK pathway is also associated with the occurrence of inflammation in various inflammatory disorders.36-38 Inhibition of the MAPK pathway alleviated lung permeability, repressed system inflammation, and mitigated the lung injury induced by sepsis.39,40 MAPK signaling is also demonstrated to be responsible for the protective effects of several natural products on LPS-induced ALI.41,42 In this study, we found that irigenin inhibited the phosphorylated levels of p38, ERK, and JNK in LPS-induced mice, indicating that irigenin might exert a beneficial effect on ALI by inactivating the MAPK signaling. However, there still existed some limitations in the present study. Firstly, ALI is a non-selective pathophysiological process in the real world, making it difficult to prevent in advance. However, irigenin was administrated prophylactically in this study. Hence, more works are requried to clarify the clinical relevance of irigenin with post-injury treatment. Secondly, this study demonstrated only one possible molecular mechanism, while other related pathways in LPS-induced ALI might also be regulated by irigenin. Thirdly, the data from a mice study might be different in human situation especially during ALI. More prospective clinical trials are warranted to evaluate the translational utility of irigenin for preventing ALI in clinical patients.
Taken together, we preliminarily predicted the key targets and signaling pathways of irigenin in the treatment of ALI by using the network pharmacology approach. Then, we not only proved that irigenin alleviated LPS-induced ALI by suppressing inflammation and apoptosis for the first time but also showed that the anti-ALI effect of irigenin was at least partly ascribed to the inactivation of MAPK signaling.
Supplemental Material
Supplemental Material - Irigenin attenuates lipopolysaccharide-induced acute lung injury by inactivating the mitogen-activated protein kinase (MAPK) signaling pathway
Supplementary Material for Irigenin attenuates lipopolysaccharide-induced acute lung injury by inactivating the mitogen-activated protein kinase (MAPK) signaling pathway by Dan Liu, Qing Wang, Wen Yuan and Qiang Wang in International Journal of Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
