Abstract
Introduction
Intestinal ischemia/reperfusion (II/R) injury is a life-threatening situation accompanied by severe organ injury, especially acute lung injury (ALI). A great body of evidence indicates that II/R injury is usually associated with hyperlactatemia. G-protein-coupled receptor 81 (GPR81), a receptor of lactate, has been recognized as a regulatory factor in inflammation, but whether it was involved in II/R injury-induced ALI is still unknown.
Methods
To establish the II/R injury model, the superior mesenteric artery of the mice was occluded gently by a microvascular clamp for 45 min to elicit intestinal ischemia and then a 90-min reperfusion was performed. Broncho-alveolar lavage fluid (BALF) and lung tissues were obtained to evaluate the lung injury after II/R. The pulmonary histopathological alteration was evaluated by H&E staining. The concentration of proteins, the number of infiltrated cells, and the level of IL-6 were measured in BALF. The formation of neutrophil extracellular traps (NETs) was evaluated by the level of double-stranded DNA (dsDNA) and myeloperoxidase- double-stranded DNA (MPO-dsDNA) complex in BALF, and the content of citrullinated histone H3 (Cit-H3) in lung tissue. The level of HMGB1 in the BALF and plasma was measured by enzyme linked immunosorbent assay (ELISA).
Results
Administration of the GPR81 agonist 3,5-dihydroxybenzoic acid (DHBA) aggravated II/R injury-induced lung histological abnormalities, upregulated the concentration of proteins, the number of infiltrated cells, and the level of IL-6 in BALF. In addition, DHBA treatment increased the level of dsDNA and MPO-dsDNA complex in BALF, and promoted the elevation of Cit-H3 in lung tissue and the release of HMGB1 in BALF and plasma.
Conclusion
After induction of ALI by II/R, the administration of DHBA aggravated ALI through NETs formation in the lung.
Keywords
Introduction
Intestinal ischemia/reperfusion (II/R) injury is a common life-threatening complication with high mortality in the range of 60–80%, which is caused by a series of pathological conditions, such as acute mesenteric ischemia, severe burns or infection, shock, and abdominal aortic surgery.1,2 In addition to intestinal injury, ischemia/reperfusion also leads to distal organ injury and failure.3,4 A substantial body of evidence shows that the lung is the most vulnerable remote organ in II/R injury.5,6 In addition, acute lung injury (ALI) has been showed to be the main cause of death during II/R injury. 7 Despite extensive studies, the mechanism of II/R injury-induced ALI has not yet been fully elucidated.
Neutrophils are known to be key players in innate immunity. 8 In order to restricting the pathogens, neutrophils create extracellular traps, known as NETs, which is composed of chromatin filaments coated with histones and granule-derived enzymes. 9 Accumulating evidence shows that NETs is a double-edged sword because the excessive release of NETs contributes to the disease progression by triggering coagulation, inflammation, and cell death in many inflammatory diseases, including ALI.10,11 High-mobility group box 1 protein (HMGB1) is one of the major damage-associated molecular patterns (DAMPs) released from injured enterocytes during II/R.12,13 Recent studies showed that HMGB1 released from injured cells is a pivotal inducing factor for NETs formation, which is the key for organ damage and inflammatory response.14,15 Thus, increased circulating HMGB1 might trigger NETs formation during II/R, which plays essential roles in the development of II/R injury-induced ALI.
Recently, metabolic reactions have been shown to be involved in the regulation of inflammatory response.16,17 Some metabolic intermediates not only provide energy supply for inflammatory response but also function as signal molecules to regulate inflammatory response.18,19 It has been reported that II/R injury is usually associated with hyperlactatemia.20,21 Interestingly, G protein-coupled receptor 81 (GPR81), a receptor for lactate, was recently reported to be profoundly involved in the regulation of inflammation, metabolism, and cell death.22,23 Several studies also indicate that the release of HMGB1 is modulated by GPR81.19,24 We then questioned whether GPR81 would modulate HMGB1 release and NETs formation in II/R injury-induced ALI.
3,5-dihydroxybenzoic acid (DHBA) is a widely used agonist of GPR81 both
Materials and methods
Reagents
The GPR81 agonist (3,5-dihydroxybenzoic acid, DHBA) was purchased from TITAN Technology Co., Ltd (Shanghai, China). Phenobarbital sodium for anesthesia was supplied from Bopei Biotech Co., Ltd (Chongqing, China). The Quaint-iT PicoGreen dsDNA assay kit, the bicinchoninic acid (BCA) protein assay kit, and enhanced chemiluminescence (ECL) reagents were provided by Thermo Fisher Scientific (Rockford, IL, USA). The counting slides, dual chamber for cell counter was obtained from Bio-Rad Laboratories, Inc. (California, USA).
Animals’ studies
Male C57BL/6 mice (8–12 weeks, weight 22 ± 3 g) were purchased from Experimental Animal Center of Chongqing Medical University (Chongqing, China). All the mice had free access to food and water and were kept on a regular day-night cycle environment at 23 ± 2°C and relative humidity 50–60%. All animal experimental protocols were approved by the Ethics Committee of Chongqing Medical University and carried out in accordance with the National Institutes of Health guide for the care and use of laboratory animals.
Intestinal model of ischemia/reperfusion
24 h prior to experimentation, animals were fasted, but given free access to water. Forty-eight mice were used in the experiment and the mice were randomly assigned to four groups, namely, sham + PBS, sham + DHBA, II/R + PBS, and II/R + DHBA. To establish the model of II/R injury-induced ALI, the mice were anesthetized with sodium pentobarbital (50 mg/kg, intraperitoneally), and the anesthesia was maintained during the whole experiment. A midline laparotomy was performed to expose the intestine; the superior mesenteric artery was exposed and was occluded gently by a microvascular clamp for 45 min to elicit intestinal ischemia. Ischemia was confirmed based on the lack of pulse in the mesentery and pale coloration of the jejunum and ileum. Subsequently, reperfusion was initiated by removing the clamp. Reperfusion was confirmed based on the reoccurrence of pink coloration of the small intestine and intensive intestinal peristalsis, and then the incision of every mouse was sutured. A 90-min reperfusion was then performed. The mice were injected with DHBA (60 mg/kg) or PBS at the time-point of 10 min before removing the clamp. The sham-operated mice underwent the same operative protocol only without superior mesenteric artery occlusion. The mice were sacrificed after 45-min intestinal ischemia and 90-min reperfusion. Blood and BALF samples and lung tissues were obtained for the respective analysis.
Histopathological evaluation of lung tissue
The isolated left lung was immersed in 10% neutral buffered formalin for 24 h and then embedded in paraffin. Tissues were sectioned at a thickness of 5 mm and stained with H&E for histological examination. Lung injury scoring was performed to evaluate the damage degree previously described with minor modification 28 by the following pathological features: presence of exudates, inflammatory cells infiltration, intra-alveolar hemorrhage/debris, and the thickness of the alveolar septum. Each score was divided into four levels: 0, absent; 1, mild; 2, moderate; and 3, severe, and the final score was determined by adding each assessment parameter.
Collection of bronchoalveolar lavage fluid
The lungs were irrigated with .8 mL of sterile PBS (total volume 2.4 mL) three times to collect BALF. A total volume of 2.4 mL was recovered.
BALF protein concentration and cell counts
The collected BALF was centrifuged (4°C, 3000 r/min,10 min), and the supernatant was collected. The concentration of total proteins in BALF was detected by BCA protein assay kits (Thermoscientific, Rockford, IL, USA) at absorbance of 562 nm. Total cells in the BALF were counted on a cell counting chamber.
Enzyme-linked immunosorbent assay
ELISA kits were used to detected the levels of inflammatory factors, including interleukin-6 (IL-6) (NeoBioscience, Shenzhen, China) and high mobility group box 1 (HMGB1) (USCN Business, Wuhan, China), in serum and BALF, according to the manufactures’ protocols.
Measurement of NETs
The level of MPO-dsDNA was measured as described with minor modifications. 29 Briefly, BALF supernatants were incubated with anti-MPO antibody that was precoated onto the ELISA plates (NeoBioscience). After incubation 1.5 h at 37°C, the DNA bounding to MPO was measured using the Quant-iT PicoGreen kit (Invitrogen). dsDNA in BALF supernatants and serum samples was quantified using Quant-iT PicoGreen (Invitrogen) according to the manufacturer’s protocols.
Western blotting
The lung tissues were homogenized in the RIPA lysis buffer with proteinase inhibitor for extracting the total proteins, which were quantified by BCA assay kit. The proteins were electrophoresed in 12.5% SDS-PAGE gel and transferred to polyvinylidene fluoride (PVDF) membranes. Then, the membranes were blocked with 5% nonfat milk for 2 h. Subsequently, the membrane was incubated with the primary antibody Cit-H3 (1:1000 dilution, Abcam, UK) and β-actin (1:4000 dilution, 4A Biotech, China) over night at 4°C, followed by washing with TBST for 30 min and then incubation with horseradish peroxidase-linked goat anti-mouse or anti-rabbit secondary antibodies (1:4000 dilution, Proteintech, China) for 2 h at room temperature. Protein bands were detected with the enhanced chemiluminescence (ECL) detection kit and visualized with ChemiDoc Imaging Systems (Bio-Rad). The optical density of the bands was quantified by Image Lab (Bio-Rad, version 5.2) and Image J.
Statistical analyses
Statistical analysis was performed using GraphPad Prism 8.0 software (La Jolla, CA). Two-way ANOVA was followed by Tukey’s for multiple comparisons, and non-parametric tests were followed by Kruskal–Wallis tests. Statistical difference was accepted at
Results
DHBA enhanced pulmonary histopathological change induced by II/R injury
The potential damage effects of DHBA were investigated in mice with II/R. As shown in Figure 1(a) and (b), II/R induced severe histopathological alterations in the lung tissue, including leukocytes infiltration increasing in the alveolar septum and much more blood cells and exudation in the alveoli, alveoli septum edema. In addition, lung injury scoring was performed to evaluate the damage degree according to the method described in the materials and methods. These changes were aggravated after administration of DHBA. DHBA exacerbated II/R-induced lung histologic abnormalities. Mice were treated with DHBA (60 mg/kg) intraperitoneally 10 min before removing the clamp and subjected to intestinal ischemia for 45 min and reperfusion for 90 min. The histological analysis of the lung was performed with (a) H&E staining. The morphological abnormalities of the lung were observed under microscope (original magnification ×200). (b) Histological injury scores of the lungs were determined as described in the methods. The data were expressed as median and IQR *
DHBA increased vascular permeability and inflammatory response of the lung induced by II/R injury
ALI is characterized by increased vascular permeability and uncontrolled inflammatory response in the lung. In order to explore the effect of DHBA on II/R injury-induced ALI, bronchoalveolar lavage fluid (BALF) were collected to evaluate the lung injury induced by II/R injury. The results showed that II/R injury increased the content of total protein (Figure 2), the number of total cells (Figure 3), and the level of IL-6 (Figure 4) in the BALF, which were significantly promoted after treated with DHBA. DHBA elevated the level of total protein concentration in the BALF. Mice were treated with DHBA (60 mg/kg) intraperitoneally 10 min before removing the clamp and subjected to intestinal ischemia for 45 min and reperfusion for 90 min. The samples of alveolar lavage fluid were collected. BALF protein concentration was measured by BCA. All data are expressed as mean ± SD ( DHBA elevated the level of total protein concentration in the BALF. Mice were treated with DHBA (60 mg/kg) intraperitoneally 10 min before removing the clamp and subjected to intestinal ischemia for 45 min and reperfusion for 90 min. The samples of alveolar lavage fluid were collected. BALF total cells were counted a cell counting chamber. All data are expressed as mean ± SD ( DHBA elevated the level of total protein concentration in the BALF. Mice were treated with DHBA (60 mg/kg) intraperitoneally 10 min before removing the clamp and subjected to intestinal ischemia for 45 min and reperfusion for 90 min. The samples of alveolar lavage fluid were collected. The level of cytokine IL-6 was detected by ELISA. All data are expressed as mean ± SD (


DHBA enhanced NETs formation in the lung induced by II/R injury
Previous study showed that NETs formation was one of the most important factors in inflammatory disease. We wonder whether NETs play an important role in II/R injury-induced ALI. In the present study, we found that II/R upregulated the level of BALF dsDNA (DNA fibers released by NETs, Figure 5), increased the level of BALF dsDNA-MPO complex (a Marker for NETs, Figure 6), and promoted the express of Cit-H3 (a specific maker for NETs formation, Figure 7(a) and (b)). These changes were enhanced by the administration of DHBA. DHBA elevated the level of total protein concentration in the BALF. Mice were treated with DHBA (60 mg/kg) intraperitoneally 10 min before removing the clamp and subjected to intestinal ischemia for 45 min and reperfusion for 90 min. The samples of alveolar lavage fluid and plasma were collected. (a) dsDNA levels in the (b) BALF and plasma were tested, DHBA elevated the level of total protein concentration in the BALF. Mice were treated with DHBA (60 mg/kg) intraperitoneally 10 min before removing the clamp and subjected to intestinal ischemia for 45 min and reperfusion for 90 min. The samples of alveolar lavage fluid were collected. The level of BALF MPO-dsDNA complex was tested. All the data expressed as mean ± SD ( DHBA elevated the level of total protein concentration in the BALF. Mice were treated with DHBA (60 mg/kg) intraperitoneally 10 min before removing the clamp and subjected to intestinal ischemia for 45 min and reperfusion for 90 min. The samples of the lung tissues were collected. The expression of (a, b) Cit-H3 in the lung tissues were determined by immunoblotting. All the data expressed as mean ± SD (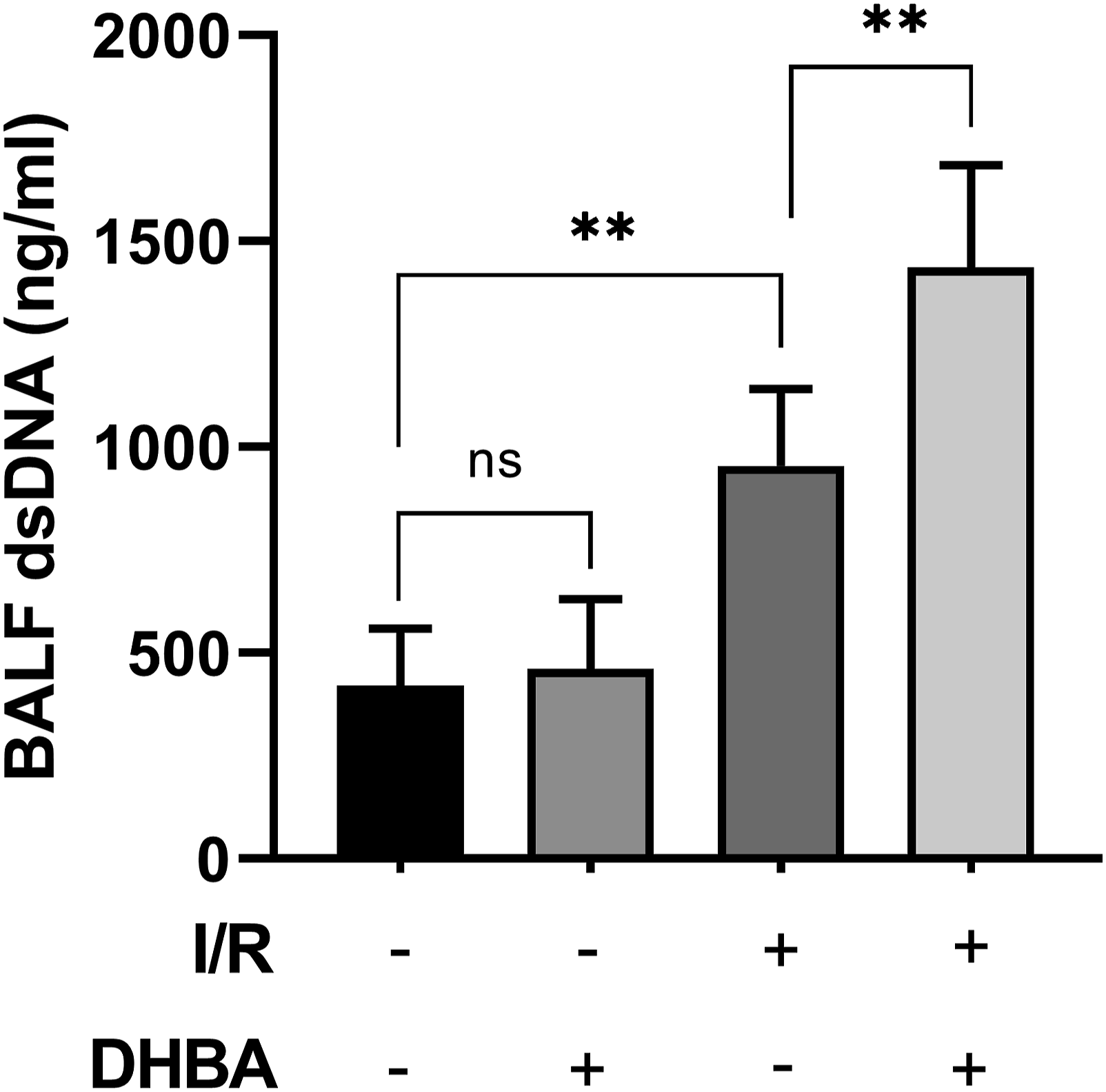


DHBA promoted the release of HMGB1 induced by II/R
As shown in Figure 8, compared to the II/R group, HMGB1 from BALF and plasma were increased in the DHBA treated group (Figure 8(a) and (b)). These results suggest that intestinal ischemia-reperfusion-induced remote lung injury might be through upregulating the levels of HMGB1 in the lung by the activation of GPR81. DHBA elevated the level of total protein concentration in the BALF. Mice were treated with DHBA (60 mg/kg) intraperitoneally 10 min before removing the clamp and subjected to intestinal ischemia for 45 min and reperfusion for 90 min. The samples of alveolar lavage fluid and plasma were collected. The levels of (a, b) HMGB-1 in the BALF and plasma were tested. The data were expressed as means ± SD (
Discussion
II/R-induced ALI is a tough problem in clinic and there are few drugs and strategy available. Lactate, a metabolite of pyruvate, is usually regarded as a metabolic by-product. But recently, lactate has been regarded as a signal molecule involved in inflammation and metabolism through its receptor GPR81. 30 Genetic depletion of lactate receptor GPR81 decreases endothelium permeability, mitigates inflammatory lung injury and improves survival outcome in sepsis mice. 31 On the contrary, inhibition of GPR81 mitigates ischemia brain injury. 26 Therefore, the activation of GPR81 may be a detrimental factor in inflammatory disease. In the present study, we found that administration of DHBA, a GPR81 agonist, aggravated the histological lesions in lung, promoted the infiltration of inflammatory cells, increased the level of exudated protein, infiltrated cells, and pro-inflammatory IL-6 in BALF. These findings suggested that treatment with DHBA exacerbated II/R injury-induced ALI. Thus, the activation of GPR81 might be a key detrimental factor in II/R-induced ALI
The activation of inflammatory cascade plays an important role in the development of II/R-induced ALI. Previous studies showed that GPR81 was widely involved in the regulation of inflammatory response both
The increasing recruitment of neutrophils to the lung and their hyper-activation are hallmarks of ALI. 36 Neutrophil-derived extracellular traps (NETs) formation is one of the most important mechanisms underlying the pathological roles of neutrophils in inflammatory diseases and organ damage. 37 Previous studies showed that the development of ALI is associated with the formation of NETs and preventing NETs formation alleviated inflammatory lung injury.38–40 In the present study, we found that the levels of MPO-dsDNA and dsDNA in BALF were both upregulated after II/R, and the immunoblotting also showed that Cit-H3, an indicator of NETs, was upregulated in the lung after II/R, and DHBA administration enhanced those changes. These data indicated that DHBA promoted NETs formation in the lung, and the activation of GPR81 may be a prompt inducible factor for NETs formation. Thus, the prevention of NETs formation in the lung is a strategy for II/R injury induced ALI.
High-mobility group box 1 (HMGB1), one of the most important damage-associated molecular patterns (DAMPs), is released by damaged cells or activated inflammatory cells. Studies show that HMGB1 induces NETs formation synergistically with other DAMPs via TLR4/9 and RAGE signaling,41,42 which in turn, leading to NETs release more DAMPs and subsequently, amplified inflammatory. 43 Previous study has found that HMGB1 is the major DAMPs released from damaged enterocytes after II/R. 12 Several studies also indicate that the release of HMGB1 would be modulated by GPR81.19,24 In the present study, the level of HMGB1 was upregulated both in the plasma and BALF after treatment with DHBA, which might subsequently enhance the formation of NETs. These results suggest that intestinal ischemia-reperfusion-induced remote lung injury might be through upregulating the levels of HMGB1. Thus, the aggravated intestinal II/R injury-induced ALI in DHBA-treated group might result from enhanced release of HMGB1 and formation of NETs.
This study has limitations. Firstly, the data from small number of samples used in each group does not adequately reveal the effect of DHBA on II/R injury-induced ALI, and a larger sample size in our research is needed. In addition, it has been shown that the formation of NETs is regulated by various DAMPs. 43 Therefore, whether the formation of NETs depends on HMGB1 requires further investigation. NETs cause inflammation and organ injury by extracellular exposure of DNA, granular proteins such as MPO, and histone.44,45 However, whether NETs directly trap and damage the lung tissue or promote the inflammatory response and lung injury through the release of histones, HMGB1, and other ingredients in the NET structure need more investigations.
Conclusion
In the present study, we found that GPR81 agonist DHBA exacerbates II/R injury-induced ALI via promoting NETs formation and upregulating the level of HMGB1 of in BALF and plasma. Thus, the aggravated ALI after intestinal II/R injury might result from enhanced release of HMGB1 and formation of NETs. In addition, this study showed that lactate and its receptor GPR81 have detrimental effects on II/R injury-induced ALI, and GPR81 might be a potential pharmacological target for the treatment of II/R injury-induced ALI.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is sponsored by Natural Science Foundation of Chongqing, China. Grant/Award Numbers: cstc2021jcyj-msxmX0197.
Ethics approval
Ethical approval for this study was obtained from Ethics Committee of Chongqing Medical University.
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
