Abstract
Introduction
Acute lung injury (ALI) attracted attention among physicians because of its high mortality. We aimed to determine whether the phosphatidylinositol-3 kinase (PI3K)/protein kinase B (Akt) pathway is involved in the protective effects of penehyclidine hydrochloride (PHC) against lipopolysaccharide (LPS)-induced ALI.
Methods
H&E staining was used to observed pathological changes in the lung tissues. ELISA was used to evaluate the concentration of inflammatory mediators in the bronchoalveolar lavage fluid (BALF). White-light microscopy was performed to observe the TUNEL-positive nuclei. The viability of NR8383 alveolar macrophages was determined by using CCK-8. The levels of MPO, MDA, SOD, and GSH-Px were analyzed using ELISA kits. Western blotting was used to evaluate the ERS-associated protein levels and the phosphorylation of PI3K and Akt.
Results
PHC administration defended against LPS-induced histopathological deterioration and increased pulmonary edema and lung injury scores, while all of these beneficial effects were inhibited by LY. In addition, PHC administration mitigated oxidative stress as indicated by decreases in lung myeloperoxidase (MPO) and malondialdehyde (MDA) concentrations, and increases in glutathione peroxidase (GSH-Px) and superoxide dismutase (SOD) concentrations. It also alleviated LPS-induced inflammation. PHC administration attenuated apoptosis-associated protein levels, improved cell viability, and decreased the number of TdT-mediated dUTP Nick-End Labeling (TUNEL)-positive cells. Furthermore, PHC inhibited ERS-associated protein levels. Meanwhile, the protection of PHC against inflammation, oxidative stress, apoptosis, and ERS was inhibited by LY. Moreover, PHC administration increased PI3K and Akt phosphorylation, indicating that the upregulation of the PI3K/Akt pathway, while this pathway was inhibited by LY.
Conclusion
PHC significantly activates the PI3K/Akt pathway to ameliorate the extent of damage to pulmonary tissue, inflammation, oxidative stress, apoptosis, and ERS in LPS-induced ALI.
Highlights
PHC protects ALI by mitigating endoplasmic reticulum stress; PHC protects ALI by ameliorating inflammation, oxidative stress, apoptosis; and PHC protects ALI by promoting the PI3K/Akt pathway
Introduction
Acute lung injury (ALI) is one of the most common critical diseases in critical care medicine, which has attracted attention among physicians because of its high mortality. There are few effective treatments for ALI. Indeed, its mortality rate is still as high as 30%–40% despite the progress of supportive treatment techniques such as mechanical ventilation and extracorporeal membrane oxygenation (ECMO).1,2 Moreover, these techniques are invasive and involve many complications, limiting their promotion in clinical practice. At present, it is anticipated that greater stimulation of the endogenous protection of alveolar cells and enhancement of their resistance to inflammatory responses can improve the prognosis of ALI patients.
Penehyclidine hydrochloride (PHC) is a novel type of anticholinergic drug that has M receptor selectivity; it selectively acts on M1 and M3 receptors. 3 In recent years, PHC has been widely recognized as a treatment for ALI caused by pulmonary microvascular permeability and shock. 4 Notably, ALI induced by LPS was shown to be alleviated by PHC in rat models. PHC can reduce lung injury, reduce lung tissue dry/wet weight ratio and inflammation index, inhibit lung cell apoptosis, and reduce oxidative stress in rats with LPS-induced ALI. 5 PHC has attracted great interest because of its ability to protect the lung. However, the mechanism by which PHC acts against ALI is unknown and should be clarified.
The endoplasmic reticulum (ER) is the site where proteins are synthesized and folded. The ER dominates the production, modification, transport, and folding of secreted proteins while regulating intracellular Ca2+ homeostasis. ER stress generally refers to the dysfunction of the ER and may be caused by pathogenic stress signals, leading to the accumulation of misfolded and unfolded proteins. To overcome the adverse effects of ER stress and adapt to various cellular microenvironments, cells can undertake an adaptive response, the unfolded protein response. However, if misfolded and unfolded proteins continue to accumulate, or if the adaptive response fails, ER stress can be disastrous for the cell, resulting in apoptosis. 6 Studies have shown that the mechanism by which LPS induces ALI is related to ERS. 7 It was also found that reducing ERS attenuated LPS-induced ALI. 8 Our previous studies also confirmed that PHC protected against LPS-induced ALI in rats by attenuating ERS. 9 However, the mechanism by which PHC achieves remains unclear and warrants further study.
The phosphatidylinositol-3 kinase (PI3K)/protein kinase B (Akt) signaling pathway is a new therapeutic target for ALI, which is a hot topic at home and abroad. PI3K is a heterodimer, a mediator of extracellular signaling and cellular response, and a member of the phosphatidylinositol-3 kinase family with important biological properties. PI3K is composed of P110 catalytic subunit and P85 regulatory subunit and has both lipid kinase activity and protein kinase activity. It is an important signal transduction molecule in cells. When cells are stimulated by the external environment, PI3K is phosphorylated to phosphatidylinositol3–5 -triphosphate (PIP3), which is the second messenger of Akt transport to the plasma membrane. On the cell membrane, PIP3 promotes the phosphorylation of Akt to form p-Akt, which induces cell proliferation and inhibits cell apoptosis. 10 Inflammatory factors can reduce Akt phosphorylation by inhibiting PI3K. 11 The PI3K/Akt signaling pathway can promote cell survival and growth by inhibiting cell apoptosis and ERS. 12 Promotion of the PI3K/Akt pathway inhibits ERS, thereby protecting against ALI. 11 However, whether PHC defends against LPS-induced ALI via targeting the PI3K/Akt pathway mechanism has remained unclear.
In this study, we hypothesized that PHC might inhibit LPS-induced ALI. Therefore, we investigated whether PHC could inhibit inflammation, oxidative stress, apoptosis, and ER stress in vitro and in vivo, and investigated the mechanism of involvement of the PI3K/Akt pathway in the protective effects of PHC against ALI.
Materials and methods
Chemicals
List Corporation (Chengdu, China) supplied PHC. Gibco (Grand Island, NY) supplied fetal bovine serum (FBS) and Dulbecco's Modified Eagle Medium (DMEM). Roche Company (Germany) supplied TdT-mediated dUTP Nick-End Labeling (TUNEL) analysis kit and protease antagonist cocktail. Merck Millipore Technology (Darmstadt, Germany) supplied myeloperoxidase (MPO) and malondialdehyde (MDA) analysis kits. Beijing Solarbio Science & Technology Co., Ltd (Beijing, China) supplied Cell Counting Kit-8 (CCK-8). Dojindo Laboratories Co., Ltd (Shanghai, China) supplied streptomycin and penicillin. Jiancheng Bioengineering Institute (Nanjing, China) supplied bicinchoninic acid protein analysis kit. Santa Cruz Biotechnology (Santa Cruz, CA, USA) supplied primary antibodies against caspase-3, activating transcription factor 6 (ATF6), B-cell lymphoma-2 (BCL-2), and BCL-2-associated x (BAX). Abcam (Cambridge, MA, USA) supplied primary antibodies against eukaryotic translation initiation factor 2α (eIF2α), phosphorylated α subunit of eukaryotic initiation factor 2α (p-eIF2α), protein kinase R-like ER kinase (PERK), phosphoprotein kinase R-like ER kinase (p-PERK), and β-actin. Cell Signaling Technology (Danvers, MA, USA) supplied primary antibodies against PI3K, P-PI3K, Akt, P-Akt, and C/EBP-homologous protein (CHOP). Roche Molecular Biochemicals (Mannheim, Germany) supplied secondary antibodies. Sigma-Aldrich (St Louis, MO) supplied LY294002.
Rats
All animal experiments complied with the ARRIVE guidelines and were carried out in accordance with the National Research Council's Guide for the Care and Use of Laboratory Animals and approved by the Institutional Animal Care and Use Committee of the Affiliated Hospital of Putian University, China (Permit Number: 202013). All efforts were made to minimize the number of animals used and to ameliorate their suffering.
Eight-to ten-week-old male Sprague-Dawley (SD) rats weighing 250–300
Experimental design
SD rats were randomly divided into SHAM group (
Cell culture and treatment
NR8383 alveolar macrophages were cultivated in DMEM with antibiotic (1%) and FBS (10%) at 37°C with 5% CO2. The alveolar macrophages were randomly divided into SHAM group, ALI group, PHC group, and PHC+LY group. In the ALI group, LPS (100 ng/mL) was applied, followed by incubation for 24 h. In the SHAM group, the procedure was equivalent to that in the ALI group except that the same volume of normal saline was administered. In the PHC group, 2 h after LPS (100 ng/mL) administration, PHC (2.5 μg/mL) was applied, followed by incubation for 24 h. 15 Finally, in the PHC+LY group, 2 h after LPS (100 ng/mL) administration, PHC (2.5 μg/mL) and LY (10 μmol/L) were given, followed by incubation for 24 h. 16
Measurement of pulmonary apoptosis
Pulmonary cell apoptosis was observed by TUNEL staining in situ (Roche, Mannheim, Germany), in accordance with the manufacturer’s instructions. White-light microscopy was performed to observe the TUNEL-positive nuclei. In this staining context, the nuclei of apoptotic cells are brown, while those of normal cells are blue. As an index of apoptosis, the following formula was used: (the number of apoptotic cells/the total number of cells) ×100.
Lung histology
For the morphological examination, lung tissues were embedded in paraffin and cut into 5-mm sections. Hematoxylin and eosin (H&E) staining was used to observe the pathological changes in the lung tissues and evaluate the following criteria: (1) the degree of neutrophil infiltration; (2) the impairment of airway epithelial cells; (3) the degree of interstitial edema; (4) the formation of hyaline membranes; and (5) the occurrence of hemorrhage. The lung injury score was determined based on the degree of deterioration: normal = 0; minimal alteration = 1; mild alteration = 2; moderate alteration = 3; and severe alteration = 4. 13
Measurement of lung MPO and serum MDA, SOD, and GSH-Px
The levels of MPO, MDA, SOD, and GSH-Px were analyzed using ELISA kits (MPO catalog number: CSB-E08722r, MDA catalog number: A003-1-2, SOD catalog number: BC0175, GSH-Px catalog number: CSB-E12146r) following the manufacturer’s instructions.
CCK-8 analysis
The cells were seeded onto a 96-well plate containing 100 μL of culture medium. The plates were incubated in a CO2 incubator at 37°C for 24 h. Subsequently, PHC (2.5 μg/mL; 10 μL) was added and the plates were incubated for 24 h. Thereafter, 10 μL of CCK-8 solution (Beyotime, Wuhan, China) was added to each well, and the plates were incubated for 2 h. The absorbance was measured using a microplate reader at 450 nm.
Western blotting assay
A protease inhibitor cocktail supplemented with radio-immunoprecipitation assay (RIPA) buffer was used to extract protein from lung samples and NR8383 cells. A BCA analysis kit was used to determine the protein levels of the whole lysate after brief sonication and rotation (15,000 r/min, 15 min, 4°C). The protein lysates were then mixed with 2-mercaptoethanol and heated at 95°C for 5 min to denature the proteins. Following this, the denatured samples were separated over a 2-h period using 10% polyacrylamide gel electrophoresis (PAGE). The resolved proteins were then put on a polyvinylidene difluoride (PVDF) membrane (0.2 µm) and run at 100 V for 2 h in a refrigerated environment. The blots were blocked with nonfat milk (5%) in tris-buffered saline with 0.1% Tween 20 detergent (TBST) for 1 h at 25°C, and the primary antibodies were then allowed to interact with the membrane overnight at 4°C. The antibody dilution ratio of BCL-2, BAX, caspase-3, CHOP, ATF6, PEAK, PI3K, and P-PI3K is 1:2000. The antibody dilution ratio of P-PEAK, EIF2a, P- EIF2a, and P-Akt are 1:1000. The antibody dilution ratio of Akt is 1:5000. The antibody dilution ratio of β-actin is 1:10000. The membranes were then thoroughly cleaned before being incubated with HRP-conjugated secondary antibodies at 25°C for 1 h. Thermo Fisher’s iBright western blot imaging system was then utilized to produce a digital image of the blots, which was then analyzed and quantified using Image J software. 7
Ratio of wet to dry lung
The wet/dry ratio was calculated to assess tissue edema. The right lungs were weighed to determine the wet weight. Subsequently, the lung was desiccated at 80°C for 48 h and weighed to determine the dry weight.
Statistical analysis
The results are expressed as mean ± standard deviation. One-way analysis was used for comparison between groups, followed by Tukey’s post hoc test.
Results
PHC administration attenuates LPS-induced lung injury in vivo
To investigate whether PHC alleviates LPS-induced ALI in rats, we observed pathological changes at 24 h after LPS-induced ALI using H&E staining and the lung injury score. Moreover, we observed lung edema by the pulmonary wet/dry weight ratio. Compared with the rats in the SHAM group, the rats in the ALI group demonstrated diffuse edema, severe inflammatory cell infiltration, thickened alveolar walls, increased interstitial congestion, and reduced alveolar space (Figure 1(a)). Nevertheless, these histological changes were significantly decreased by the administration of PHC. PHC administration also dramatically reduced lung injury scores and lung edema compared with those of the ALI rats. Meanwhile, the protective effects of PHC against lung injury were blocked by the administration of LY294002 (Figure 1(b) and (c)). PHC administration attenuates LPS-induced lung injury in vivo. Characteristic pathological images after H&E staining (×200) (a). H&E staining scale bar = 100 um. The lung injury score (b) and the pulmonary wet/dry weight ratio (c) were evaluated 24 h after LPS administration. The findings are presented as the means ± S.D. (
PHC administration mitigates inflammation and oxidative stress in vivo
Inflammatory mediators play critical roles in ALI. Therefore, we evaluated the concentration of inflammatory mediators in the bronchoalveolar lavage fluid (BALF). The BALF concentrations of protein, IL-1β, TNF-α, and IL-6 were markedly increased after LPS treatment compared to the controls. Furthermore, PHC administration significantly diminished the inflammatory mediators when compared with those in the ALI group. Interestingly, compared with the group that received PHC administration alone, its administration simultaneously with PHC and LY294002 increased the concentrations of protein and the levels of IL-1β, TNF-α, and IL-6 in BALF (Figure 2(a)–(d)). PHC administration mitigates inflammation and oxidative stress in vivo. The pulmonary levels of IL-1β (a), IL-6 (b), TNF-α (c), and protein (d) in BALF were evaluated 24 h after LPS administration. Pulmonary MPO activity (e) and serum MDA (f), GSH-Px (g), and SOD (h) concentrations were evaluated by ELISA 24 h after LPS administration. The findings are presented as the means ± S.D. (
To evaluate whether PHC reduced the oxidative stress associated with damage due to LPS, the MPO activity and the levels of MDA, SOD, and GSH-Px were determined. PHC administration significantly decreased lung MPO and MDA concentrations, and increased GSH-Px and SOD concentrations compared with those in the ALI group. Conversely, LY294002 administration inhibited the effect of PHC on mitigating oxidative stress (Figure 2(e)–(h)).
PHC administration attenuates apoptosis in vitro and in vivo
To explore whether PHC attenuated LPS-induced apoptosis, we determined the expressions of apoptosis-associated proteins by western blotting and the expression of pulmonary apoptotic indicators by TUNEL analysis. As illustrated in Figure 3(a)–(f), compared with the levels in the control group, upregulation of caspase-3 and BAX, downregulation of BCL-2, and an increased number of apoptosis cells were observed in the ALI group, indicating apoptotic signaling activation. Furthermore, PHC administration decreased the expressions of caspase-3 and BAX, and the number of apoptotic cells, and increased the expressions of BCL-2 in rats compared with the levels in the ALI group. Conversely, LY294002 administration reversed the protective effects of PHC against apoptosis in vivo. PHC administration attenuates apoptosis in vivo. TUNEL staining was used to assess the apoptosis of rat pulmonary cells (a). White-light microscopy was performed to observe the TUNEL-positive nuclei (×200). TUNEL staining scale bar = 100 um. In TUNEL staining, the nuclei of apoptotic cells are brown, while those of normal cells are blue. The apoptotic index (%) indicates the intensity of apoptosis (b). The levels of apoptosis-associated proteins of rat pulmonary cells were determined by western blotting assay (c), as were BCL-2 levels (d), BAX levels (e), and caspase-3 levels (f). The findings are presented as mean ± S.D. (
To examine whether PHC protected alveolar macrophages, this study evaluated the expressions of apoptosis-associated proteins by western blotting and the viability of LPS-mediated NR8383 alveolar macrophages using CCK-8. LPS administration significantly reduced cell viability, upregulated the levels of caspase-3 and BAX, and downregulated the level of BCL-2. Meanwhile, PHC administration markedly increased the level of BCL-2, decreased the levels of caspase-3 and BAX, and improved cell viability. Nevertheless, LY294002 administration blocked the protective effects of PHC against apoptosis activation in NR8383 alveolar macrophages (Figure 4(a)–(e)). PHC administration attenuates apoptosis in vitro. The levels of apoptosis-associated proteins in NR8383 alveolar macrophages were evaluated by western blotting assay (a), as were BCL-2 levels (c), BAX levels (d), and caspase-3 levels (e). The viability of LPS-mediated NR8383 alveolar macrophages was determined using CCK-8 (b). The findings are presented as mean ± S.D. (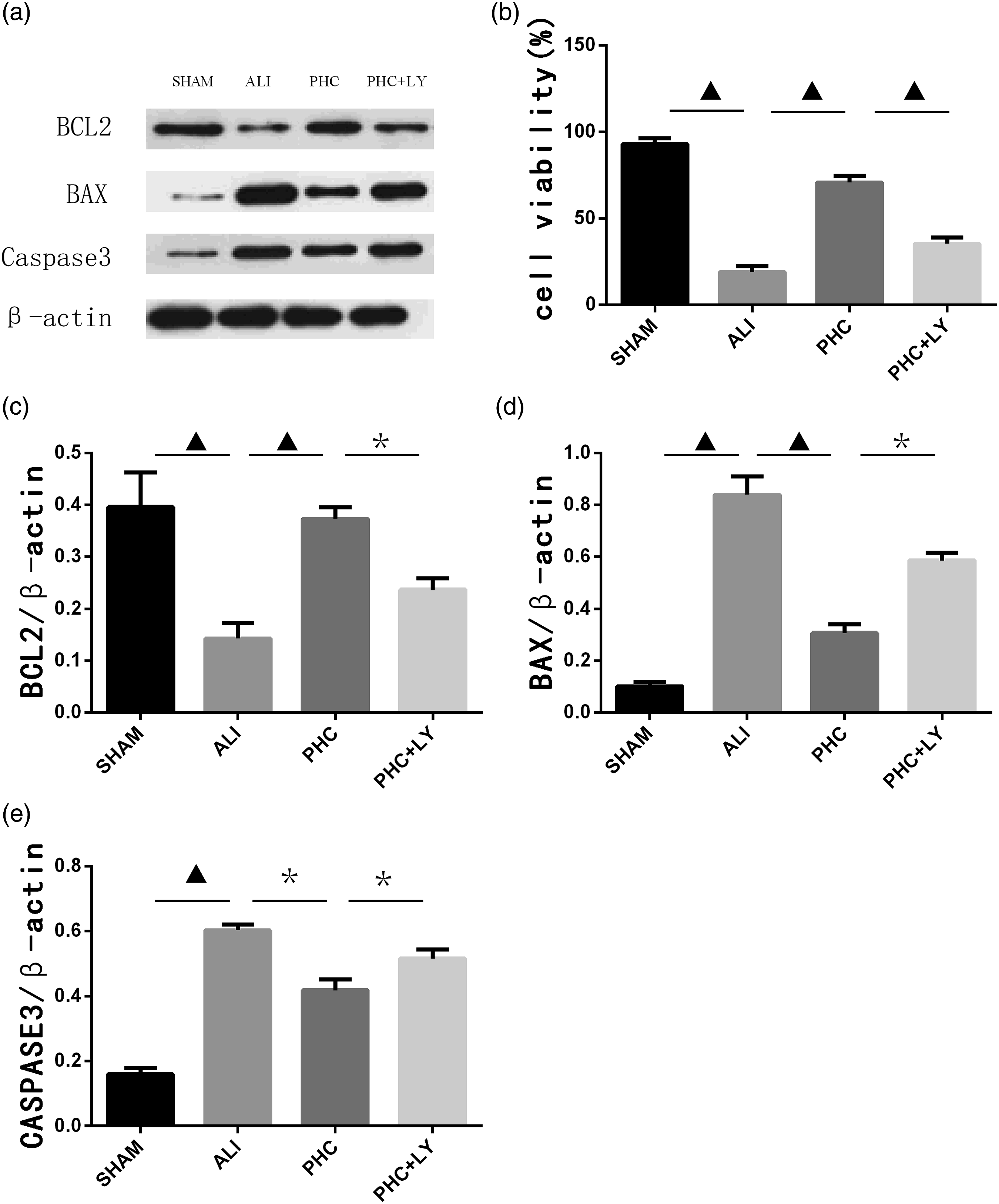
PHC administration mitigates ER stress in vitro and in vivo
To explore the role of PHC in LPS-induced ERS, we measured the expressions of eIF2α, p-eIF2α, PERK, p-PERK, CHOP, and ATF6 by western blotting in vitro and in vivo. The expression of CHOP and ATF6, and the ratios of P-PERK/PERK and P-eIF2α/eIF2α in the ALI group increased compared with those in the control group in vitro and in vivo, indicating the exacerbation of ERS. Furthermore, PHC administration dramatically decreased the expression of CHOP and ATF6, and the ratios of p-PERK/PERK and p-eIF2α/eIF2α, indicating the inhibition of ERS. However, LY294002 administration blocked the protective effects of PHC against LPS-induced ERS (Figure 5(a)–(e) and Figure 6(a)–(e)). PHC administration mitigates ER stress in vivo. (a) Western blotting assay was used to assess the levels of eIF2α, P-eIF2α, PERK, P-PERK, CHOP, and ATF6 in vivo. CHOP levels (b), ATF6 levels (c), P-PERK/PERK ratio (d), and P-eIF2α/eIF2α ratio (e). The findings are presented as the means ± S.D. ( PHC administration mitigates ER stress in vitro. (a) Western blotting assay was used to assess the levels of eIF2α, P-eIF2α, PERK, P-PERK, CHOP, and ATF6 in vitro. CHOP levels (b), ATF6 levels (c), P-PERK/PERK ratio (d), and P- eIF2α/eIF2α ratio (e). The findings are presented as mean ± S.D. (

PHC administration activates the PI3K/Akt pathway in vitro and in vivo
To investigate whether the PI3K/Akt pathway is involved in the protective effect of PHC against LPS-induced ALI, the levels of Akt, PI3K, P-Akt, and P-PI3K were assessed in vitro and in vivo. As revealed in Figure 7(a)–(c) and Figure 8(a)–(c), the ratios of P-PI3K/PI3K and P-Akt/Akt were decreased in the ALI group compared with those in the control group in vitro and in vivo, indicating that LPS inhibited the phosphorylation of PI3K and Akt. In particular, a notable enhancement of P-Akt and P-PI3K protein expressions was observed in the PHC group. Interestingly, LY294002 administration blocked the activity of PHC regarding the phosphorylation of PI3K and Akt, indicating that the PI3K/Akt pathway is involved in the protective effects of PHC against LPS-induced ALI. PHC administration activates the PI3K/Akt pathway in vivo. The expression of Akt, PI3K, P-Akt, and P-PI3K in rat pulmonary cells was assessed by western blotting assay (a). P-Akt/Akt levels (b) and P-PI3K/PI3K levels (c). The findings are presented as mean ± S.D. ( PHC administration activates the PI3K/Akt pathway in vitro. The expression of Akt, PI3K, P-Akt, and P-PI3K in NR8383 alveolar macrophages was assessed by western blotting assay (a). P-Akt/Akt levels (b) and P-PI3K/PI3K levels (c). The findings are presented as mean ± S.D. (

Discussion
In the current study, we explored the protective effects of PHC against LPS-induced ALI in vivo and in vitro. PHC administration defended against LPS-induced histopathological deterioration, increased pulmonary edema, and lung injury scores, all of which were inhibited by LY294002. In addition, PHC administration alleviated LPS-induced inflammation indicated by reducing tumor necrosis factor-alpha (TNF-α), interleukin (IL)-1β, and IL-6 levels in the lung and bronchoalveolar lavage fluid, simultaneously mitigated oxidative stress indicated by decreasing lung MPO and the concentrations of MDA, and increasing the GSH-Px and SOD concentrations. PHC administration attenuated LPS-mediated apoptosis in vitro and in vivo, as indicated by the downregulation of BAX and caspase-3, upregulation of BCL-2, improved cell viability, and decreased numbers of TUNEL-positive cells. PHC inhibited the LPS-induced upregulation of the levels of CHOP, ATF6, and the ratios of p-PERK/PERK and p-eIF2α/eIF2α, indicating ERS suppression. LY294002 could inhibit these PHC-induced protective effects against inflammation, oxidative stress, apoptosis, and ERS. Moreover, PHC administration increased PI3K and Akt phosphorylation, indicating that the PI3K/Akt pathway was upregulated. PI3K/Akt pathway was inhibited using LY294002. Our investigation revealed the mechanism by which PHC counters against LPS-induced ALI.
PHC is particularly effective and has fewer adverse effects, making it a promising drug for lung disease. 17 Several experimental studies have shown that PHC ameliorates lung injuries by suppressing apoptosis, oxidative stress, and inflammation. 18 The inflammatory response, including the release of proinflammatory cytokines, is an important feature of ALI. Indeed, inflammatory factors are closely associated with the morbidity and mortality of ALI. 19 Oxidative stress is a hallmark of redox imbalance and is thought to be associated with inflammatory responses. 19 Oxidative stress has been observed in various diseases characterized by systematic inflammatory responses and lung dysfunction. Moreover, oxidative stress can produce large amounts of oxygen free radicals, which not only induce the release of inflammatory cytokines, but also can directly damage the lung tissue. 20 Intriguingly, our study suggests that PHC ameliorates LPS-induced ALI by reducing inflammation, oxidative stress, and apoptosis, all of which were inhibited by LY294002.
ERS causes a pathological imbalance in ER homeostasis and induces physical dysfunction. Such stress is induced by ischemia (hypoxia), inflammation, and oxidative stress, and is characterized by the accumulation of unfolded proteins, which eventually leads to apoptosis and cell death. 21 The following transmembrane protein transducers act in the sensing of ERS: activating transcription factor 6 (ATF6), eukaryotic translation initiation factor 2α (EIF2a), and (PKR)-like ER kinase (PERK) in the ER. ER homeostasis can be improved by the unfolded protein response (UPR). 22 However, apoptosis is activated by the UPR as stress continues. 23 ERS-induced apoptosis has been shown to be associated with C/EBP homologous protein (CHOP). 24 The transcriptional activation of CHOP or the induction of JNK or the caspase-12 pathway is primarily associated with ERS-induced apoptosis. 25 Increasing evidence has shown the involvement of ERS in the pathogenesis of ALI. 24 ERS inhibition has been found to alleviate LPS-induced lung inflammation and apoptosis. 9 Ye et al. 26 showed that PHC protects against LPS-induced ALI by inhibiting PERK and ATF6 signals, and attenuating ERS-mediated apoptosis. This study demonstrated that LPS mediates ER stress and confirmed that PHC exerted protective effects against ALI by reducing LPS-induced ERS in vivo and in vitro. LY294002 inhibited the PHC-induced protective effects against ERS.
The PI3K/Akt signaling pathway can promote cell survival and growth in several ways, including the suppression of inflammation and the regulation of apoptosis and ERS. 27 For example, PI3K/Akt downregulates pro-apoptotic proteins including Bad, BAX, and caspase. 28 The regulation of apoptosis to protect against lung injury is also mediated by the activation of PI3K/Akt signaling. 11 Furthermore, the regulation of ERS by the PI3K/Akt signaling pathway has also been demonstrated. Activating the PI3K/Akt signaling pathway was shown to attenuate inflammation, oxidative stress, and apoptosis in alveolar epithelial cells. 29 Moreover, Liang et al. 30 demonstrated that dexmedetomidine suppresses the inflammatory response and alleviates lung ischemia-reperfusion injury in rats by activating the PI3K/Akt pathway. Our research revealed that PHC administration enhances the expression of P-PI3K and P-Akt in LPS-induced ALI, indicating that PHC promotes the PI3K/Akt pathway.
LY294002 is a specific inhibitor of the phosphatidylinositol 3-kinase (PI3K) family. 31 To determine whether PHC alleviated LPS-induced ALI in rats by promoting the PI3K/Akt pathway, we administered the PHC and LY to LPS-induced ALI in vivo and in vitro. Our results suggested that PHC administration significantly increased PI3K and Akt phosphorylation, reduced ERS and oxidative stress, and alleviated lung injury, lung inflammation and apoptosis. Interestingly, these protective effects of PHC were abolished by LY, suggesting that the PI3K/Akt signaling pathway is involved in the protective effect of PHC against LPS-induced ALI.
Although these observations are valuable, our study has some limitations. First, we claim here that PHC is an anticholinergic reagent that protects against LPS-induced ALI by promoting the PI3K/Akt pathway. However, whether PHC protects against LPS-induced ALI by antagonizing cholinergic receptors has not been confirmed in this experiment. Second, we did not perform sample size/power analysis in this experiment. We aim to confirm these in future experiments.
Conclusions
Our results demonstrated that PHC reduces the extent of damage to pulmonary tissue, ameliorates inflammation, oxidative stress, apoptosis, and ERS in LPS-induced ALI through activating the PI3K/Akt pathway.
Footnotes
Author contributions
Junting Weng, Rongjie Guo, Danjuan Liu, and Chunjin Fu: guarantor of integrity of the entire study. Junting Weng, Rongjie Guo, Danjuan Liu, and Chunjin Fu: study concepts. Junting Weng, Bingbing Shi, Min Chen, Shuoyun Weng, and Danjuan Liu: study design. Junting Weng, Rongjie Guo, Danjuan Liu, and Chunjin Fu: definition of intellectual content. Junting Weng, Shuoyun Weng, Danjuan Liu, and Chunjin Fu: literature research. Junting Weng, Rongjie Guo, Shuoyun Weng, Danjuan Liu, and Chunjin Fu: experimental studies. Junting Weng, Bingbing Shi, Shuoyun Weng, Danjuan Liu, and Chunjin Fu: data acquisition. Junting Weng, Min Chen, Shuoyun Weng, Danjuan Liu, and Chunjin Fu: data analysis. Junting Weng, Rongjie Guo, Bingbing Shi, Danjuan Liu, and Chunjin Fu: statistical analysis. Junting Weng, Rongjie Guo, Min Chen, Shuoyun Weng, and Danjuan Liu: manuscript preparation. Junting Weng, Rongjie Guo, Bingbing Shi, Min Chen, and Chunjin Fu: manuscript editing. Junting Weng, Rongjie Guo, Bingbing Shi, and Chunjin Fu: manuscript review.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Natural Science Foundation of Fujian Province of China (grant number. 2020J011253) and the Science and Technology Project of Putian City, Fujian Province (grant number. 2020S3F008).
Ethics approval and consent to participate
All animal experiments complied with the ARRIVE guidelines and were carried out in accordance with the National Research Council's ![]() and approved by the Institutional Animal Care and Use Committee of the Affiliated Hospital of Putian University, China (Permit Number: 202013).
and approved by the Institutional Animal Care and Use Committee of the Affiliated Hospital of Putian University, China (Permit Number: 202013).
Data availability statement
The data that support the findings of this study are available in the supplementary material of this article.
