Abstract
Introduction
Due to its silent clinical progression and diagnosis often occurring at advanced stages, ovarian cancer continues to be a major contributor to gynecological cancer-related mortality worldwide. Recent evidence underscores the critical therapeutic value of targeting the interplay between DNA damage response pathways, particularly poly (ADP-ribose) polymerase 1 (PARP1), and mitogen-activated protein kinase (MAPK) signaling cascades.
Methods
In this study, we investigated the anti-cancer potential of the naturally derived small molecule usnic acid in a chemotherapy-resistant epithelial ovarian cancer model (SKOV-3 cells) at the cellular and molecular levels.
Results
Our findings demonstrate for the first time that usnic acid exerts a dual-pathway apoptotic mechanism by simultaneously inhibiting PARP1 and activating the MAPK signaling pathway. Remarkably, usnic acid mimics the gene-silencing action of siRNA on PARP1, suggesting a highly specific and potent inhibitory effect at the molecular level. Although ROS involvement and pathway causality were not directly tested, this dual-action profile enhances DNA damage-induced apoptosis and highlights usnic acid as a promising therapeutic candidate.
Conclusion
The study presents a novel molecular framework in which a single small molecule can coordinate apoptosis through parallel regulatory pathways, demonstrating the potential for innovative therapeutic approaches in the treatment of aggressive and treatment-resistant ovarian cancers.
Introduction
Ovarian cancer is a leading cause of gynecological cancer-related mortality worldwide, primarily due to its asymptomatic progression and late diagnosis. Epithelial ovarian cancer (EOC) accounts for approximately 90% of ovarian cancer cases, with high-grade serous carcinoma being the most common subtype. The disease is characterized by late presentation, frequent recurrence, and resistance to standard chemotherapies such as cisplatin and adriamycin, all of which contribute to its poor prognosis.1,2
Despite significant advancements in diagnosis and therapy, aggressive, metastatic, or drug-resistant types of ovarian cancer continue to present considerable therapeutic challenges. The challenges posed by tumor heterogeneity, acquired drug resistance, and the capacity of cancer cells to evade immune surveillance frequently result in suboptimal outcomes with conventional treatment techniques, including surgery, chemotherapy, and radiation therapy. Consequently, there is an urgent need to develop novel therapeutic approaches that are more personalized, selective, and effective. These include molecularly targeted medicines, which seek to interfere with important oncogenic pathways while causing the least amount of harm to healthy tissues. 3
The potential of natural compounds, particularly secondary metabolites derived from lichens, to possess anticancer properties has garnered significant interest in recent years. These bioactive substances have demonstrated considerable potential as candidates for cancer treatment due to their multifaceted pharmacological characteristics, which encompass anti-proliferative, apoptotic, and anti-angiogenic actions. 4 The bioactive substance usnic acid is of particular interest due to its potential anticancer effects. Many studies have explained in detail the mechanisms by which usnic acid produces its anticancer effects, including DNA damage response, autophagy modification, cell cycle arrest, and apoptosis induction. 5 A number of studies have indicated a correlation between DNA damage in cancer cells and the treatment with usnic acid. Studies have shown that usnic acid induces DNA double-strand breaks in gastric and colorectal cancer cells.6,7
Poly (ADP-ribose) polymerase 1 (PARP1) plays a vital role in the repair of single-strand DNA breaks through the base excision repair pathway. The inhibition of PARP1 with drugs such as olaparib or talazoparib leads to the accumulation of unrepaired single-strand breaks. During DNA replication, these single-strand breaks can transform into double-strand breaks (DSBs). DSBs are more cytotoxic and can trigger apoptotic pathways. This mechanism has been observed to be particularly effective in cancer cells that exhibit deficiencies in homologous recombination repair, such as those with BRCA1/2 mutations. 8 Conversely, given the efficacy of the homologous recombination repair mechanism in healthy cells, these cells are uneffected. Consequently, PARP1 inhibitors present a selective and effective treatment option for cancer treatment. Inhibition of PARP1 has been demonstrated to induce DNA damage, which in turn activates the mitogen-activated protein kinase (MAPK) pathway and other critical signaling pathways. The MAPK pathway’s subfamilies, which include extracellular signal-regulated kinase (ERK), c-Jun N-terminal kinase (JNK), and p38 MAPK, regulate cell survival, death, and DNA repair activities. JNK and p38 MAPK are activated in response to DNA damage and contribute to the initiation of apoptosis. For instance, studies have demonstrated that the inhibition of MAPK4, a member of the MAPK family, potentiates the effects of radiation and elicits synergistic lethality when combined with PARP1 inhibition in cervical cancer cells. This finding indicates that MAPK4 may be involved in the DNA damage response, and its inhibition could enhance the sensitivity of cancer cells to PARP1 inhibitors. 9 ERK1/2 has been found to be associated with cell survival and proliferation. ERK1/2 can also be activated in response to DNA damage.
A recent study has demonstrated that the inhibition of ERK1/2 can lead to an augmentation in DNA damage and cell death in breast cancer cells that have been treated with PARP1 and CHK1 inhibitors. This finding suggests that ERK1/2 signaling may have a regulatory influence on the DNA damage response. 10
The interplay between PARP1 inhibition, DNA damage, and the MAPK pathway has significant therapeutic implications. The concurrent targeting of both PARP1 and specific MAPK pathways has been demonstrated to augment the cytotoxic effects on cancer cells. A recent study has demonstrated that the combination of PARP1 inhibitors and MAPK inhibitors results in enhanced efficacy in the suppression of tumor growth in models of melanoma. 8 In cancers with deficiencies in DNA repair mechanisms, such as those harboring BRCA mutations, the combination of PARP1 inhibitors with agents that modulate the MAPK pathway can result in the exploitation of synthetic lethality, leading to the selective death of cancer cells. Consequently, the inhibition of PARP1 results in DNA damage, which activates the MAPK pathway. This, in turn, regulates cell survival and apoptosis. It is imperative to comprehend the nature of this relationship to facilitate the development of efficacious combination therapies that have the potential to enhance the anticancer effects of PARP1 inhibitors.11,12
The objective of this study is to examine the therapeutic potential of usnic acid in an aggressive and chemoresistant type of epithelial ovarian cancer model (SKOV3 cell), with a specific focus on its ability to increase DNA damage and activate MAPK signaling pathways through the inhibition of PARP1. SKOV-3 cells are p53 mutants, a pivotal tumor suppressor protein that plays a critical role in DNA damage response and apoptosis. The absence of p53 has been demonstrated to be a contributing factor to resistance to DNA-damaging agents, such as cisplatin. 13 SKOV-3 cells have been found to overexpress the HER2/neu (ERBB2) receptor tyrosine kinase. HER2 activation has been shown to drive survival signaling via the PI3K/AKT and MAPK/ERK pathways. 14 SKOV-3 cells have been found to express active PARP1, a DNA repair enzyme. While they are not characterized by the presence of a specific mutation in the BRCA1 or BRCA2 gene, the administration of PARP1 inhibitors, such as olaparib, in conjunction with other therapeutic agents, has the potential to render these cells susceptible to apoptosis by impeding the process of DNA damage repair. 15 We hypothesize that usnic acid triggers apoptosis in SKOV-3 cells by increasing DNA damage and activating MAPK pathways via PARP1 inhibition in ovarian cancer. Although previous studies have demonstrated the cytotoxic effects of usnic acid, they have often lacked mechanistic depth and overlooked its potential in the treatment of treatment-resistant ovarian cancer. This study addresses a key gap in the literature by highlighting usnic acid’s ability to induce apoptosis via multiple pathways and underscoring its potential as a targeted therapeutic agent.
Materials and methods
Cell culture
This study was an in vitro investigation conducted on fully validated SKOV-3 (ATCC-HTB-77) and normal ovarian epithelial cell (OSE) cell lines (ABM, T1074) and did not involve human or animal subjects. The cell line’s source and mycoplasma-free status were verified by the manufacturer. The human ovarian adenocarcinoma cell line, SKOV-3, was cultured at 37°C in a 5% CO2-containing incubator in the presence of Dulbecco’s Modified Eagle Medium (DMEM) (Gibco, Thermo Fisher Scientific, Inc.), supplemented with 10% (v/v) FBS and 1% penicillin-streptomycin. Also, the normal ovarian epithelial cell, was maintained in RPMI 1640 medium (Gibco, Thermo Fisher Scientific, Inc.), supplemented with 10% (v/v) FBS and 1% penicillin-streptomycin.
Preparation of Usnic Acid
Usnic acid was purchased from Sigma-Aldrich (Cat. No. 329967). A stock solution of 100 μM usnic acid was prepared in DMEM medium with DMSO, ensuring that the final DMSO concentration did not exceed 0.05% (v/v). Serial dilutions of the stock solution were performed to obtain concentrations of 1.56, 3.125, 6.25, 12.5, 25, and 50 µM. Control groups received the equivalent DMSO concentration without usnic acid to ensure that any observed effects were due to usnic acid rather than DMSO.
xCELLigence real-time cell analysis (RTCA)
The analysis of usnic acid-treated and untreated SKOV-3 and OSE cells was conducted using the xCELLigence RTCA (Software Lite (2.0) DP system (ACEA Biosciences, San Diego, CA, USA). The analysis were conducted with two independent biological replicates, each with two technical replicates. The cells were seeded at a density of 1 × 104 cells/100 μl in a 16-well e-plate following a background measurement. Following the insertion of the plate into the chamber, it was subjected to an incubation period of 24 h within a CO2 incubator. Subsequently, 100 μl of varying concentrations of usnic acid (1.56–50 μM) and carboplatin, serving as a positive control, were dispensed into the e-plate wells. During the instrument’s 120 h operational cycle, readings were obtained at 15 min intervals, and the proliferation curve was monitored in real time. The xCELLigence RTCA Software Lite (2.0) was utilized to ascertain the IC50 values of usnic acid.
PARP1 siRNA transfection and gene silencing analysis
To achieve a temporary silencing of the PARP1, small interfering RNA SignalSilence® PARP siRNA I #6304 that precisely targets the PARP1-encoding mRNA was procured from Thermo Fisher Scientific. To establish a control, scramble siRNA (Scr, Silencer® Select Negative Control No. 1 siRNA, Cat. No. 4,390,843, Thermo Fisher Scientific, MA, USA) with no target in the human transcriptome was employed (obtained from Thermo Fisher Scientific). HiPerfect Transfection Reagent (Qiagen, Cat. No. 301704; Germany) was utilized in transfection studies. In order to optimize the required amount of siRNA and the transfection time, 2 × 105 SKOV-3 cells were seeded in 6-well plates. Various concentrations of PARP1 siRNA and scramble siRNAs were prepared to reach final concentrations of 1 nM, 5 nM, 10 nM, 50 nM, and 100 nM by diluting them in serum-free culture medium. The solution was then mixed with 12 μl of HiPerfect transfection reagent, manufactured by Qiagen (Germany). In order to facilitate the formation of a transfection complex, the prepared solutions were subjected to a vortex and then incubated at room temperature for a period of 15 min. Subsequently, 100 μl of the siRNA transfection mix was added dropwise into each well. The cells were then subjected to an incubation process at a temperature of 37°C, within an environment containing a specific percentage of CO2 (5%) and a specific humidity level. Following a 24 h transfection period, both cells that had been subjected to transfection and those that had not been subjected to transfection were harvested. Thereafter, the cells were washed in 2 mL of phosphate-buffered saline (PBS) and lysed in TriPure reagent for the isolation of RNA. The cDNA synthesis of usnic acid treated with the IC50 concentration of 14 µM for 32 h and PARP1 siRNA samples was carried out using the EURx cDNA synthesis kit. PARP1 expression levels were validated by SYBR Green-based quantitative real-time polymerase chain reaction (qRT-PCR) and western blot analysis.
Caspase 3/7 activity assay
To this end, a Caspase 3/7 Activity Assay Kit (Colorimetric Method) (E-CK-A383) Elabscience) was performed to determine the effects of usnic acid treatment and PARP1-siRNA transfection on the activities of caspase 3 and 7 in SKOV-3 cells. The caspase 3/7 activity assay kit is based on caspase 3/7 that can catalyze the substrate Ac-DEVD-pNA to generate yellow pNA (p-Nitroaniline). The pNA has a strong absorption near 405 nm and the activity of caspase 3/7 was calculated by determining the absorbance at 405 nm. The analysis were conducted with three independent biological replicates, each with two technical replicates. SKOV-3 cells (5 × 105 cells/well) were seeded into a 6-well plate, incubated for 24 h, and treated with an IC50 concentration of usnic acid and 25 nM PARP1 siRNA. Following the usnic acid incubation period (32 h), the wells were washed with phosphate-buffered saline (PBS), and cells were harvested using 300 µl of trypsin. The cells were then subjected to centrifugation at 600×g for 5 min. The supernatant was then discarded, and the cell pellet was resuspended in 100 μL of pre-cooled cell lysis buffer (1 × 106 cells/μL). The resuspended cell pellet was then incubated on an ice bath for 30 min. Subsequently, the lysed samples were subjected to a centrifugal process at 11,000×g for a duration of 10–15 min at a temperature of 4°C. The sample was transferred to a new eppendorf tube and stored at a low temperature. The protein concentration and caspase 3/7 activity were determined by measuring samples at OD 405 nm, following the manufacturer’s instructions.
Gene expression analysis
The RNA was extracted from three distinct cell samples: usnic asid treated (IC50 concentration of 14 µM for 32 h), PARP1-siRNA transfected, and control SKOV-3 cells. The Trizol reagent was used in the extraction process. The integrity of RNA was verified using NanoDrop spectrophotometry, and the synthesis of cDNA was carried out using the EURx cDNA synthesis kit. The Human Apoptosis Primer Library panel, which includes eight housekeeping genes and 88 apoptosis-related genes, was used to assess the expression of apoptosis-related genes. Furthermore, the expression levels of MAPK pathway genes were determined by quantitative real-time polymerase chain reaction (qRT-PCR). The analysis were conducted with two independent biological replicates, each with two technical replicates. GAPDH was utilized as the reference gene for the SYBR Green-based qRT-PCR, which was employed to assess gene expression. The 2-ΔΔCt analysis was employed to calculate the fold changes.
Western blot analysis
In the present study, total protein isolation was performed on usnic asid treated (IC50 concentration of 14 µM for 32 h), PARP1-siRNA transfected, and control SKOV-3 cells. The BCA protein was subsequently quantified, and the SDS-PAGE, PVDF membrane transfer, and chemiluminescence detection (LI-COR Odyssey CLx Imaging System) were employed for western blot analysis. The expression levels of PARP1 MAPK pathway proteins p44/42 MAPK (Erk1/2) Antibody (Cell Signaling Technology, #9102S) and CASP3 protein (Cat. No. 9662, Cell Signaling Technology) were analyzed to determine apoptosis after usnic acid and PARP1 siRNA treatment in SKOV-3 cells. It is evident that MAPK blots are representative of total proteins rather than their phosphorylated forms. β-Actin was used as a loading control and detected using a β-Actin antibody (Cat. No. 4970, Cell Signaling Technology, MA, USA) Protein quantification was performed using (ImageJ software version 1.47) with background subtraction. The analysis were conducted with two independent biological replicates, each with two technical replicates. Representative images/data from one experiment are shown, with quantification from all replicates. All blots were normalized to loading controls and quantified across independent biological replicates. Full-length, uncropped blot images are provided in Supplemental Material.
Statistical analysis
All experiments were performed with 2 independent biological replicates (3 for caspase assay), each with 2 technical replicates. Representative images from one biological replicate are shown, with quantification from all biological replicates. Due to the limited sample size (n = 2 biological replicates for most assays), formal validation of parametric test assumptions was not possible, as these tests require n ≥ 3 to provide reliable results. The consistency of trends across independent experiments and the distribution patterns of technical replicates were used to assess reproducibility. The large effect sizes observed suggest biological significance; however, these findings should be interpreted as exploratory and hypothesis-generating, requiring validation in larger, independent studies. Statistical analyses were conducted using GraphPad Prism version 9.0 (GraphPad Software, San Diego, CA, USA), and data are presented as mean ± standard deviation (SD). For pairwise comparisons, the unpaired two-tailed Student’s t-test was used. For multi-group comparisons, one-way ANOVA followed by Tukey’s multiple comparison post hoc test was applied to determine intergroup differences. Differential gene expression analyses (88-gene apoptosis panel), genes with an adjusted q-value <0.05 were considered statistically significant. Statistical significance thresholds were set at p < 0.05 or q < 0.05, as appropriate.
Results
Determination of anti-proliferative effect
The usnic acid drug candidate was found to suppress the growth of the SKOV-3 ovarian cancer cell in a time- and dose-dependent manner compared to the control, as determined by xCELLigence analysis (Figure 1). The IC50 concentration of usnic acid on SKOV-3 cells was calculated using RTCA Software Lite and was determined to be 14 μM at 32 h (p < 0.05) (Figure 1(A)). Furthermore, the xCELLigence real-time cell analysis of OSE normal ovarian epithelial cells demonstrated that usnic acid concentrations did not induce a cytotoxic effect in comparison to the control. Furthermore, the IC50 value of usnic acid for OSE cells was calculated to be 97.3 μM at 68 h (p < 0.01) (Figure 1(B)). In order to compare the therapeutic effect of usnic acid, the anti-proliferative effect of carboplatin in SKOV-3 ovarian cancer was determined. The results of the xCELLigence analysis demonstrated that the IC50 value for carboplatin in SKOV-3 cells was 10.7 µg/ml (p < 0.01) at 48 h (Figure 1(A)). A salient finding of the study was the observation that usnic acid did not induce cell death in normal OSE epithelial cells. This finding indicates that the treatment selectively targets cancer cells without significant toxicity to normal cells. Dose- and time-dependent anti-proliferative effects of usnic acid (UA) on (A) SKOV-3 and (B) OSE cells and on SKOV-3 cells using xCELLigence real-time cell analyzer (*p < 0.05; **p < 0.01). (A): Orange-control, Red-1.56 µM UA, Light green-3.125 µM UA, Dark blue-6.25 µM UA, Pink-12.5 µM UA, Light blue-25 µM UA, Purple-50 µM UA, Brown-15 µM Carboplatin, Dark green- 25 µM Carboplatin; (B): Dark green-control, Purple-1.56 µM UA, Light blue-3.125 µM UA, Brown-6.25 µM UA, Dark blue-12.5 µM UA, Pink-25 µM UA, Red-50 µM UA, Light green-Carboplatin) (n = 2 independent biological replicates, each with technical replicates, and the mean ± standard error (SE) was employed to present the data).
PARP1-siRNA transfection and validation
In this study, we investigated the therapeutic potential of usnic acid as a PARP1 inhibitor, which specifically targets the MAPK pathway, in the treatment of SKOV-3 ovarian cancer cells. To elucidate the mechanism by which usnic acid exerts its activity on the targeted pathway, we employed PARP1-siRNA transfection. In addition, the present study investigated the effect of usnic acid, the MAPK pathway, and the subsequent downstream modulation of PARP1 signaling. This study demonstrates the targetability of the MAPK pathway by usnic acid, thereby underscoring the pivotal role of this signaling axis in ovarian cancer treatment and identifying its potential as a therapeutic target. According to the results of qRT-PCR analysis, the PARP1-siRNA suppressed the expression level 3.5-fold compared to the scramble control group (p < 0.01) (Figure 2(A)). In this case, the cells exhibited an increased sensitivity to DNA damage, a compromised capacity for DNA repair, and a decreased survival capacity. In a similar manner, usnic acid treatment led to a 5.5-fold suppression in the expression level of the PARP1 gene in SKOV-3 ovarian cancer cells (p < 0.0001) (Figure 2(A)). In the context of gene expression, usnic acid demonstrated a higher capacity for suppression compared to PARP1-siRNA. Consequently, upon the application of usnic acid to SKOV-3 cells, an augmentation in DNA damage that cannot be rectified by PARP1 is observed, thereby escalating the rate of cell death. In addition, the efficacy of PARP1 silencing by RNA interference and the application of usnic acid were validated through western blot analysis. In accordance with the results of gene expression analysis, protein expression level was suppressed 2.5-fold (p < 0.01) after PARP1-siRNA transfection and 3.5-fold (p < 0.05) after usnic acid treatment in SKOV-3 cells (Figure 2(B) and (C)). The poly(ADP-ribose) polymerase 1 (PARP1) (A) gene and (B, C) protein expression levels in SKOV-3 cells treated with usnic acid and PARP1-siRNA. The expression levels of mitogen-activated protein kinase (MAPK) pathway (D) genes and (E, F) proteins in cells treated with usnic acid and PARP1-siRNA. (n = 2 independent biological replicates, each with technical replicates, and the mean ± standard error (SE) was employed to present the data) (G) Caspase 3/7 activation level in cells treated with usnic acid and PARP1-siRNA compared to control (*p < 0.05; **p < 0.01; ****p < 0.0001) (n = 3 independent biological replicates, each with technical replicates, and the mean ± standard error (SE) was employed to present the data). Full-length, uncropped blots obtained from Western blot studies are presented in the Supplemental Material and quantification was performed using densitometry with background subtraction and consistent loading controls (Scr: Scramble siRNA).
MAPK pathway analysis
The results of the MAPK pathway analysis revealed that the expression level of MAPK1 was 13.2-fold (p < 0.0001), MAPK3 was 17.2-fold (p < 0.0001), MAP2K1 was 2.32-fold (p < 0.05), and MAP2K2 was 1.6-fold. The expression level of PARP1 was suppressed by the PARP1-siRNA. Following the administration of usnic acid to SKOV-3 cells, a 28.5-fold increase in MAPK1 expression (p < 0.0001), a 29.4-fold increase in MAPK3 expression (p < 0.0001), a 4.2-fold decrease in MAP2K1 expression (p < 0.01), and a 2.3-fold decrease in MAP2K2 expression (p < 0.05) were observed. A subsequent comparison of PARP1-siRNA and usnic acid revealed that usnic acid exhibited a more pronounced inhibitory effect on all genes of the MAPK pathway compared to PARP1 silencing (p < 0.05) (Figure 2(D)). The results of the analysis performed to validate MAPK pathway activation by western blot analysis indicate that the protein expression levels of PARP1-siRNA and usnic acid samples were suppressed (p < 0.05) (Figure 2(E) and (F)).
Measurement of caspase 3/7 activity
The results of the caspase 3/7 enzymatic analysis demonstrated a 2.25-fold increase in the PARP1 siRNA-treated group and a 4.56-fold increase in the usnic acid-treated group (p < 0.0001). The activation of caspases 3/7, the presence of usnic acid, and the silencing of PARP1 suggest that DNA repair is stimulated and that cells are progressing along the apoptotic pathway (Figure 2(G)).
Gene expression analysis
In the expression level analysis of the genes belonging to the apoptosis pathway following usnic acid and PARP1-siRNA application, statistically significant changes were detected in the expression levels of 41 and 19 genes among 88 targets, respectively, as determined using the Benjamini–Hochberg false discovery rate (FDR) correction with significance set at q-value <0.05 (Figure 3(A) and (B)) (Tables 1 and 2). According to the results of qRT-PCR analysis after usnic acid and PARP1-siRNA treatment in SKOV-3 cells, the pro-apoptotic gene group promoting cell death was genes BAX, BAK1, BIK, BNIP3, BCL2L11, CIDEB, and GADD45A. Apoptosis pathway-related gene expression levels in SKOV-3 cells treated with (A) usnic acid and (B) poly(ADP-ribose) polymerase 1 (PARP1)-siRNA relative to control as determined by qRT-PCR analysis. Fold-change values were calculated using the 2-ΔΔCt method and represent relative expression levels normalized to control. Data are presented as mean ± standard error (SE) and individual data points from two independent biological replicates with technical replicates (n = 2). Statistical significance was determined using an unpaired two-tailed Student’s t-test with Benjamini–Hochberg false discovery rate (FDR) correction; genes with FDR q-value <0.05 were considered significant. Significance is indicated as *q < 0.05, **q < 0.01, ***q < 0.001. Statistically significant fold change in the expression levels of apoptosis-related genes in SKOV-3 cells after usnic acid treatment. Fold change values represent relative expression levels normalized to control and calculated using the 2-ΔΔCt method. The unpaired two-tailed Student’s t-test for qRT-PCR analysis (n = 2). Statistical significance was determined using the Benjamini–Hochberg false discovery rate (FDR) correction, with genes considered significant at FDR q-value <0.05. Statistically significant fold change in the expression levels of apoptosis-related genes in SKOV-3 cells after PARP1-siRNA treatment. Fold change values represent relative expression levels normalized to control and calculated using the 2-ΔΔCt method. The unpaired two-tailed Student's t-test for qRT-PCR analysis (n = 2). Statistical significance was determined using the Benjamini–Hochberg false discovery rate (FDR) correction, with genes considered significant at FDR q-value <0.05.
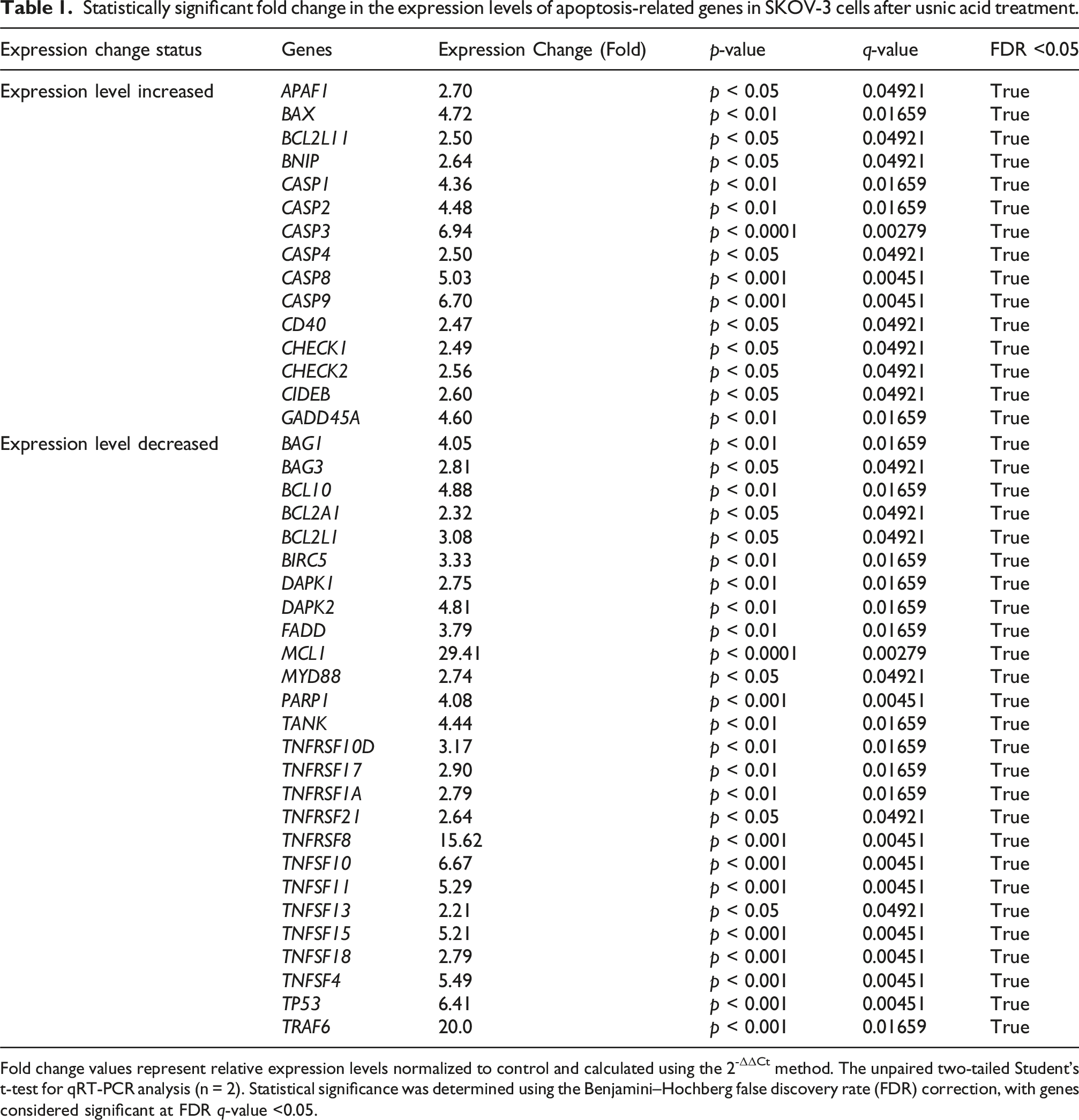

The expression levels of genes, including APAF1, CASP3, CASP4, CASP6, CASP7, CASP8, FASLG, TNF, TNFRSF10B, and TNFRSF11, were increased. The expression levels of BCL2A1, BCL2L1, BCL10, BAG1, BAG3, MCL1, BIRC1, BIRC2, TNFRSF10D, and TNFRSF17 genes in the anti-apoptotic gene group that suppress cell death were suppressed. The expression levels of FADD, MYD88, TRAF1, TRAF6, TANK, and RIPK2 genes in the signaling adaptors and modulators group, which have a decisive role in the apoptosis process, were decreased. Furthermore, the expression levels of genes such as BAG3 and TNFRSF10D, which were not included in a group, were also shown to be decreased (Figure 3(A) and (B)). The genes whose expression levels increased the most in SKOV-3 cells after usnic acid treatment were CASP9 (6.7-fold, p < 0.001), CASP3 (6.9-fold, p < 0.0001), GADD45A (4.6-fold, p < 0.01), and BAX (4.7-fold, p < 0.01). In addition, following PARP1-siRNA treatment of SKOV-3 cells, a significant increase occurred in the expression levels of 13 genes. In particular, the genes CASP9, CASP3, GADD45A, and BAX exhibited significant upregulation, with 4.4-, 3.5, 3.8-, and 3.6-fold increases, respectively, reaching statistical significance at p < 0.001 and p < 0.01.
The result of the CASP3 protein expression level after usnic acid and PARP1-siRNA application in SKOV-3 cells indicates that the CASP3 protein expression level was determined in both sample groups, thereby confirming our conclusion that apoptosis was induced. The level of CASP3 protein expression increased 4.4-fold (p < 0.01) in usnic acid-treated cells and 3.2-fold (p < 0.01) in PARP1-siRNA-treated cells compared to control (Figure 4(A) and (B)). The increase in the expression level and inhibition of the genes involved in the apoptosis pathway after inhibition of PARP1-siRNA in SKOV-3 cells were found to be at a similar level with usnic acid. Usnic acid, a small molecule, exhibits a similar inhibitory potential to that of siRNAs, as evidenced by its ability to silence PARP1. This finding suggests that usnic acid has a highly specific and potent inhibitory effect. The observation that the usnic acid molecule, which was found to have a PARP1 inhibitory effect, displays therapeutic activity by activating the MAPK pathway and apoptosis suggests that it may serve as an effective instrument in targeted cancer therapies. The expression levels of Caspase (CASP) 3 protein in SKOV-3 cells treated with usnic acid and poly(ADP-ribose) polymerase 1 (PARP1)-siRNA as determined by Western blot analysis (A) graph and (B) protein bands. (**p < 0.01) (n = 2 independent biological replicates, each with technical replicates, and the mean ± standard error (SE) and individual data points was employed to present the data). Full-length, uncropped blots obtained from Western blot studies are presented in the Supplemental Material, and quantification was performed using densitometry with background subtraction and consistent loading controls (Scr: Scramble siRNA).
Discussion
In this study, the anti-cancer potential of usnic acid on SKOV-3 ovarian cancer cells was evaluated, and the data obtained revealed that this molecule can induce bidirectional apoptosis through both PARP1 inhibition and activation of the MAPK signaling pathway. The findings suggest that usnic acid could develop a dual-pathway bidirectional apoptosis mechanism at the molecular level and may offer new approaches to cancer therapy.
In this study, the cytotoxic effect of usnic acid on normal ovarian epithelial cells (OSE) was evaluated using the xCELLigence real-time cell analysis system. According to the results obtained, usnic acid did not cause significant cytotoxicity in normal OSE cells, which revealed that the molecule exhibits selective toxicity against cancer cells. The IC50 value of usnic acid in OSE cells was determined to be 97.3 µM at 68 h (p < 0.01). The IC50 concentration obtained is significantly higher than the IC50 value determined in SKOV 3 ovarian cancer cells (14 µM- 32 h), indicating that usnic acid is approximately 7 times more selective against cancer cells. These results support the notion that usnic acid has minimal toxic effects on healthy cells in vitro and exhibits a targeted effect against SKOV-3 cancer cells.
The MAPK/ERK signaling pathway has been demonstrated to play a critical role in the regulation of fundamental cellular processes, including cell proliferation, differentiation, and survival. 16 The present study demonstrated a substantial increase in p-ERK levels following usnic acid treatment, indicating that the MAPK/ERK pathway is activated. This may be associated with signals that induce apoptosis. 17 According to current literature, the over-activation of extracellular signal-regulated kinase (ERK) has been observed to lead to cell death rather than promote cell proliferation in certain situations. 18 The present study suggests that usnic acid may shift the balance from proliferation to apoptosis by increasing ERK phosphorylation.
PARP1 expression has been observed to be elevated in a multitude of cancerous neoplasms, particularly those affecting the ovaries, breasts, and prostate glands. 19 A substantial body of research has demonstrated that PARP1 levels are elevated in ovarian cancer cells, including SKOV-3.20,21 This increase is due to tumour cells being compelled to activate more effective repair mechanisms in response to increased DNA damage. 20 PARP1 inhibitors, such as olaparib, have been shown to induce apoptosis by targeting this specific mechanism, particularly in cancers containing a mutation in the BRCA1 or BRCA2 gene. This characteristic also makes them promising candidates for drug development. 22
Furthermore, the inhibition of PARP1 appears to be a pivotal mechanism in the apoptosis-enhancing effects of usnic acid. PARP1, an enzyme, is activated in response to DNA damage, thereby initiating cellular repair processes. Nevertheless, the inhibition of PARP1 has been demonstrated to promote apoptosis by preventing the repair of DNA damage. 23 Usnic acid and its derivatives directly inhibit the activity of the PARP1 enzyme, meaning they do not indirectly cause PARP1 degradation by creating DNA damage. Zakharenko et al.’s study showed that usnic acid derivatives containing aromatic side chains directly inhibit the auto-ADP-ribosylation activity of PARP1. These derivatives can reduce the enzyme’s function by 15–50%. 24 This effect was observed directly on the enzyme, occurring in a dose-dependent manner and without DNA damage. More recent studies have also confirmed that usnic acid-based hybrid compounds directly inhibit the PARP1 enzyme, as well as effecting DNA repair proteins such as TDP1/TDP2. 25 The present study demonstrates that increased levels of cleaved PARP1 and cleaved CASP3 after usnic acid treatment support the hypothesis that usnic acid initiates an apoptotic process through DNA damage. These findings are supported by the clinical results of PARP1 inhibitors, particularly in cases of cancer involving a BRCA mutation. SKOV-3 cells carry a mutation in the p53 gene, leading to the loss of p53 protein functionality. P53 is a critical tumor suppressor that modulates the cell cycle and responds to DNA damage. It has been suggested that SKOV-3 cells may be resistant to p53-mediated apoptosis. In this instance, apoptosis should therefore be induced in an alternative, p53-independent pathway. This approach may serve as a strategic framework for the investigation of p53-independent apoptosis mechanisms 26 In this study, the application of usnic acid to SKOV-3 ovarian cancer cells resulted in an increase in the expression levels of pro-apoptotic gene family members BAX, BAK1, BIK, BNIP3, and BCL2L11, thereby indicating the initiation of apoptosis by the mitochondrial pathway. The increased expression of the CASP3, CASP7, CASP8 and CASP9 genes indicates that caspases are the key executor genes of apoptosis. Conversely, the suppression of the expression level of FASLG, TNFRSF10B, and TNF family genes, or the absence of a significant increase in expression level, indicates that the extrinsic pathway is not activated. Furthermore, the increased expression of the GADD45A, APAF1 and CIDEB genes suggests that apoptosis involving DNA damage and the mitochondrial pathway is activated. The application of usnic acid resulted in the suppression of the expression levels of BCL2A1, BCL2L2, MCL1, BAG1 and BAG3 genes, which are members of the anti-apoptotic gene family. It was determined through extensive analysis that these genes were responsible for inducing apoptosis in the cell. A limitation of this study is that PARP1 enzymatic activity was not directly evaluated, and that MAPK-mediated apoptosis was not confirmed using functional inhibition or activation approaches. Therefore, although the changes in mRNA and protein expression levels obtained in the study provide significant correlational evidence, further functional studies are required to elucidate the mechanism.
Usnic acid, a secondary metabolite derived from lichens, is a naturally occurring small molecule. As demonstrated in previous studies, usnic acid has been shown to possess both antineoplastic and pro-apoptotic properties, in addition to its ability to induce oxidative stress. 27 The presence of usnic acid has been proven to cause DNA damage. Although this study was not conducted experimentally, numerous studies in the literature have reported an occurrence of this damage associated with an increase in reactive oxygen species (ROS) within cells. 28 It should be noted that mechanistic pathways, such as the involvement of ROS, are suggested based on correlative evidence from the literature and were not directly confirmed in this study. In this context, the term ‘dual-pathway apoptosis’ indicates the combined effects of PARP1 inhibition and MAPK pathway activation in inducing apoptosis. In this case, it was determined that PARP1 and MAPK pathways were activated in SKOV-3 ovarian cancer cells due to usnic acid treatment. The study found that PARP1 is degraded by CASP3 during apoptosis, a finding that was confirmed by gene expression and western blot analysis, which is an indicator of apoptosis. 29 Consequently, usnic acid exerts a non-canonical effect, inducing PARP1 activity and subsequent apoptosis, independent of p53. The MAPK family, comprising members such as ERK, JNK, and p38, plays a pivotal role in determining cell fate (proliferation vs death). 30 It has been demonstrated that usnic acid inhibits the ERK pathway, thereby demonstrating its anti-proliferative effects. Concurrently, the activation of MAPK has been observed to trigger apoptosis by increasing BAX expression levels and decreasing BCL2 expression. The most promising result obtained as a result of the study is the determination that usnic acid triggers p53-independent apoptosis in SKOV-3 cells with p53 mutation, both through PARP1 overactivation and degradation and through ERK/MAPK activation. This finding highlights the potential effectiveness of natural molecules, such as usnic acid, in targeting cells that are resistant to conventional, p53-dependent therapeutic agents. This study aimed to evaluate the therapeutic potential of the PARP inhibitor olaparib, as well as the anti-cancer agents cisplatin and doxorubicin, in comparison with usnic acid, in SKOV-3 cells. This comparative analysis aimed to assess the efficacy of usnic acid as a potential anti-cancer agent. PARP inhibitors have been demonstrated to be effective in the context of utilizing DNA repair defects. A notable aspect of their mechanism of action is the observed synergy with BRCA mutations. 31 Cisplatin and doxorubicin are archetypal chemotherapeutic agents. Nevertheless, a certain degree of resistance may be observed as a consequence of their impact on genomic stability. 32 Cisplatin has been demonstrated to elicit apoptosis through the activation of BAX and the subsequent release of cytochrome c, even in the absence of p53. 33 Doxorubicin has been shown to induce apoptosis by increasing the production of reactive oxygen species (ROS). 34 It has been suggested that usnic acid may be beneficial for cells expressing mutant p53. This hypothesis is based on the substance’s natural structure and its ability to induce apoptosis independently of p53, as well as the mitochondrial stress resulting from this process. Recent studies have demonstrated that usnic acid exerts its biological effects by inducing apoptosis and modulating the balance between BAX and BCL2 proteins in a variety of cancer cell lines. 35 The combination of usnic acid and olaparib, either as monotherapy or in combination, has been identified as a promising treatment strategy for ovarian cancer, as it is capable of targeting distinct mechanisms involved in the disease process. According to previous studies, usnic acid has been shown to have anti-proliferative and pro-apoptotic effects on various cancer cell lines.36–38
According to current literature, usnic acid has a limited therapeutic potential and can cause serious liver damage. The final ED50/Tox value has not yet been determined in SKOV-3 or ovarian cancer in vivo models. In the interest of clinical applicability, formulation improvements, targeted delivery systems, and the development of derivative molecules will be pursued as ongoing studies.
It is known in the literature that usnic acid can exhibit hepatotoxic effects at high doses. Therefore, we also support the view that a comprehensive investigation of the dose-dependent toxicity profile is necessary in order to safely evaluate the therapeutic potential of the compound. In the continuation of our study, acute and subchronic toxicity tests will be conducted using mouse models in accordance with international standards. Systemic toxicity will be evaluated at different dose levels using liver enzymes (AST, ALT, ALP, bilirubin), kidney function indicators (BUN, creatinine), and histopathological examinations. Furthermore, the study will investigate whether the toxic effects can be reduced by combination with formulation improvements and targeted delivery systems. These studies will provide clearer data on the therapeutic potential and clinical safety of usnic acid. The objective of this study is to contribute to the preclinical development process of the molecule.
In our study, we comprehensively demonstrated the important molecular effects of usnic acid in SKOV-3 ovarian cancer cells, including PARP1 inhibition, suppression of the MAPK pathway, and increased apoptotic gene expression at the in vitro level. However, conducting in vivo animal model experiments is of great importance in terms of the clinical validity of these findings. In this regard, the next phase of our study will utilize a subcutaneous xenograft mouse model established with SKOV-3 cells. The pharmacokinetic properties, bioavailability, tissue distribution, and toxicological profile of usnic acid will be evaluated, particularly considering the risk of hepatotoxicity. These planned in vivo studies will validate the selective antitumor effect and safety profile of usnic acid in experimental animal models, thereby providing a scientific basis for the compound’s preclinical development process. In this case, usnic acid shows promise as a lead molecule for treating treatment-resistant tumors, including ovarian cancer.
However, this study has certain limitations, primarily the relatively small number of biological replicates. The use of two biological replicates for most experiments represents a significant limitation. While this sample size allowed detection of large, consistent effects, it precluded formal statistical validation of test assumptions and limited statistical power to large effect sizes only (Cohen’s d ≥ 1.0). Subtle or moderate effects may have been missed. Additionally, we measured total MAPK protein rather than phosphorylated (activated) forms, limiting direct assessment of pathway activation. ROS involvement was inferred from the literature but not directly quantified. These findings should therefore be considered preliminary and hypothesis-generating, warranting confirmation in adequately powered studies (n ≥ 3 biological replicates) with direct functional validation. Similarly, using a single ovarian cancer cell line (SKOV-3) limits the ability to generalize the findings, and validation in additional chemotherapy-resistant ovarian cancer models is required. Additionally, the study did not include toxicity assessments reflecting usnic acid’s potential therapeutic advantages, such as off-target, mitochondrial and genotoxicity assessments, nor did it include direct comparisons with clinical PARP inhibitors and other natural compounds. Potential resistance mechanisms and combination strategies with existing treatments were also not addressed. Finally, in vivo toxicity, pharmacokinetic and bioavailability studies are required to elucidate the molecular mechanism of the usnic acid molecule in greater detail. While these limitations may constrain the applicability of our findings in a clinical setting, they also provide illuminating results to support future research.
Conclusion
In conclusion, usnic acid can be considered a natural compound with potential in the treatment of ovarian cancer. Despite the therapeutic effect of usnic acid has been determined through in vitro analyses, further validation through in vivo analyses is necessary to confirm its therapeutic potential. It is suggested that usnic acid has the potential to be a promising molecule in the treatment of ovarian cancer.
Supplemental Material
Supplemental Material - Usnic Acid induces dual-pathway apoptosis in SKOV-3 ovarian cancer cells via PARP1 inhibition and MAPK pathway activation
Supplemental Material for Usnic Acid induces dual-pathway apoptosis in SKOV-3 ovarian cancer cells via PARP1 inhibition and MAPK pathway activation by Mehmet Kürşat Derici, Mine Ensoy, Pelin Mutlu, Demet Cansaran-Duman in Human & Experimental Toxicology.
Footnotes
Ethical approval
Ethical approval was not required for this study, as it involved only established and authenticated cell lines. This was confirmed by institutional exemption from the ethics committee.
Author contributions
Conceptualization and methodology were handled by MKD, ME and DCD. Experimental analyses were performed by MKD, PM and ME. Data visualization performed ME. Conceptualization data analysis and writing manuscript were performed PM and DCD.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank Ankara University, Management of Scientific Research Projects (Project no. 18B0415002), for the financial support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated or analyzed during this study are available from the corresponding author upon reasonable request.
Declarations
SKOV-3 and OSE cell lines were obtained from a certified source and authenticated using standard STR profiling. Mycoplasma testing was performed prior to experimentation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
