Abstract
Objectives
Sepsis, pneumonia, or trauma caused by acute lung injury (ALI) remains a high incidence and mortality worldwide.The critical ALI pathogenesis includes inflammatory cell infiltration, inflammation, oxidative stress injury, and damage to the alveolar-capillary barrier due to inflammatory apoptosis injury. Sophocarpine is reported protective against inflammatory diseases, while little is known about its effects on acute lung injury. Investigating therapeutic molecules that relieve the pathological processes might be a possible option for ALI management.
Methods
In the present study, we adopted the LPS-induced ALI model in mice to investigate the potential therapeutic effects of oral sophocarpine administration and analyze possible effects against inflammation, apoptosis, and oxidative stress.
Results
The results showed sophocarpine decreased pathological injury score of lung tissues, inflammatory cytokine level and oxidative stress indicators (H2O2, NO, and O.-2), and elevated antioxidant molecules (SOD, GSH, and CAT). Besides, sophocarpine suppressed Nrf2/Syk, NF-kB, and PI3K/AKT signaling. Sophocarpine showed beneficial effects against inflammation, apoptosis, and oxidative stress in LPS-induced ALI.
Conclusions
Our finding suggested that sophocarpine might be applied in treating septic or endotoxemia-related ALI.
Introduction
Acute lung injury (ALI) remains a significant cause of acute respiratory failure (ARF), acute respiratory distress syndrome (ARDS), and pneumonia. 1 Central pathological characteristics of ALI include inflammatory cell infiltration, exacerbated inflammation, oxidative stress injury, and damage to the alveolar-capillary barrier due to apoptosis inflammatory injury. 2 Though studies have unveiled the related molecular mechanism in ALI and targeted therapy has been applied to experimental treatment in the clinic, the incidence and mortality of ALI remain high. 2 Recent research has highlighted that apoptosis of epithelial cells, inflammation, and oxidative stress plays an essential role in ALI. Therefore, investigating more therapeutic molecules that aim to attenuate these pathological processes might be a possible option for the clinical management of ALI.
Lipopolysaccharide (LPS)-induced lung injury is a typical experimental model for the study of ALI. 3 Previous research proposed several therapeutic agents that could ameliorate the LPS-induced ALI and showed protective effects on lung injury.3,4 Investigating pharmaceutical agents that attenuate LPS-induced lung injury could indicate the possible application in the clinical management of ALI.
Sophocarpine, a pharmaceutical monomer separated from traditional Chinese herbs such as
In this article, we used the LPS-induced ALI mouse model with applications of oral sophocarpine treatment to investigate possible therapeutic effects. We also analyzed the effects of sophocarpine in molecular alterations regarding the pathological process of ALI, including the alterations in the Nrf2/Syk pathway for oxidative stress, PI3K/AKT pathway for apoptosis, and p38/JNK and NF-kB signaling for inflammation, thus investigated possible effects of sophocarpine and related signaling mechanism.
Materials and methods
Reagents and cells
The mouse alveolar epithelial cells (MLE12) were purchased from the Shanghai Institute of Biochemistry and Cell Biology, Chinese Academy of Science, and cultured in RPMI 1640 culture medium supplemented with 10% fetal bovine serum (FBS) (Gibco, USA) in cell culture incubator with 5% CO2 at 37°C. 100 U/ml penicillin with 100 mg/mL streptomycin (Gibco, USA) was added to the culture medium. For in vitro analysis, MLE12 cells were incubated with LPS (500 ng/mL) for 12 h with placebo (PBS solution) and sophocarpine (HY-N0103, MCE, USA) at 30ug/mL (SC-30) or 60ug/mL (SC-60), respectively.
Animals
C57 BL/6 male mice (20–25g) at an 8-week age were used in the present study. The mouse was obtained from the Experimental Animal Centre of Fudan University (Shanghai, China). The present study is the preliminary animal study conducted from April 2018 to April 2019 and is approved by the Ethics Committee of Experimental Animals of Huashan Hospital North, Fudan University (SCXK20130018) (Shanghai, China). The mice were raised in a specific pathogen-free (SPF) room. The temperature was at 25–26°C, and the humidity was 50% with a 12h day-night cycle. The water and food were accessible to mice. The animal allocation used in the present study is as follows: con (control), LPS-treated group, and sophocarpine group with LPS treatment (30 and 60 mg/kg body weight per day). The dosage of the sophocarpine is used as previously reported.
9
For the establishment of ALI, LPS (5 mg/kg) diluted in 50 uL PBS was injected through intranasal instillation. The control groups received equivalent PBS. All mice were anesthetized for the LPS and sophocarpine administration. Then, we collected the lung tissues and bronchoalveolar lavage fluid (BALF) at the prescribed time,
Lung wet/dry (W/D) ratio
Lung tissues in different groups were collected. The wet weights were determined and then incubated in the oven at 80°C for 3 days, and the dry weights were determined. The W/D ratio was calculated as the indicator of lung edema.
Histology analysis
Lung tissues were fixed with 4% paraformaldehyde solution for 2 days and were embedded in paraffin. The sections were stained with hematoxylin and eosin (H&E). We used a light microscope to analyze the histological alterations as previous literature proposed. 10
Analyses of ROS levels in lungs of mice
The previously reported lung levels of O.-2 were determined by the method. 9 The weighed lungs of mice were homogenized in a pH7.4-buffered lysis solution with ten mM EDTA and 20 mM HEPES. Then, tissues were centrifuged at 1000 g for 15 min. After the centrifugation, the samples were incubated with a pH=7.4, Krebs-HEPES buffer containing five mM lucigenin (Sigma, USA) for about 3 min at 37°C. Then, light emission data were measured on a microplate reader (TECAN, Switzerland). NO analysis was performed using a NO assay kit (Beyotime, China) and quantified at 540 nm on a microplate reader. Mean light unit (MLU) min/mg protein was obtained. In the present study, the levels of O.-2 were measured by adding SOD (350 U/mL) (R&D system, USA) to the medium.
Moreover, lung tissues were homogenized in normal saline and then treated with the same volume of methanol for 1 h at 4°C. The samples were centrifuged for 30min at 10,000 g, and the H2O2 evaluation was obtained using the biochemical kits from the R&D system, USA. The BCA protein quantification kit (Thermo, USA) measured the protein concentration.
Enzyme-linked immunosorbent assay (ELISA)
The weighed lung tissues were incubated in buffer (pH7.0) at 4°C which contains 10 nM EDTA, 2 mM PMSF, 0.1 mg/mL soybean trypsin inhibitor, 1.0 mg/mL BSA and 0.002% sodium acid. Samples were then homogenated and incubated for 2 h at 4°C. The supernatants were then centrifuged at 12 000 g for 10 min. Serum and pulmonary TNF-α, IL-16, and IL-1β levels were measured using the ELISA kits from the R&D system, USA, according to the manufacturer's protocol.
Quantitative real-time polymerase chain reaction (q-PCR)
The quantitative real-time PCR (Q-PCR) was performed as previously described. 11 Total RNA was obtained from lung tissues and cultured cells using TRIZOL reagent from Thermo, USA. The reverse transcription system used is 2 μg of total RNA, reverse transcription premix, and oligo (dT) in a total of 20ul solutions.
The Q-PCR used the SYBR Green PCR system (TAKARA, Japan) in ABI 7500 thermal cycler (Thermo, USA). The primers were shown here: PI3K, forward, 5′-TGACAGTAGGAGGAGGTTGG-3′, reverse, 5′-TCAGCCACATCAAGTATTGG-3′; AKT, forward, 5′-GAAGGTGATTCTGGTGAAAGAG-3′, reverse, 5′-ACACGGTTCTCAGTAAGCG-3′; Cyto-C, forward, 5′-CAGACAAGAAGAGGTTGCC-3′, reverse, 5′-CGTCATGGCAGTGTGTATTGG-3′; Bcl-xL, forward, 5′-GCAGGCGATGAGTTTGAAC-3′, reverse, 5′-TCCTTGTCTACGCTTTCCAC-3′; Caspase-9, forward, 5′-TCTTCATCTCCTGCTTAGAGG-3′, reverse, 5′-TGCTCCTTTGCTGTGAGTC-3′; Caspase-3, forward, 5′-TGGAAAGCCGAAACTCTTC-3′, reverse, 5′-AGGAATAGTAACCAGGTGCTG-3′; Caspase-6, forward, 5′-AGCGCGTACTTAAATGCAGAGG-3′, reverse, 5′-GTTGTAAGGTGGACAGGCTT-3′; Bad, forward, 5′-CAGAGTTTGAGCCGAGTGAG-3′, reverse, 5′-TCCCTGCTGATGAATGTTG-3′; GAPDH, forward, 5′-CATTCAAGACCGGACAGAGG-3′, reverse, 5′-ACATACTGCACACCAGCATCACC-3′. We analyzed the gene expression using the 2-ΔΔCt method, and GAPDH was considered the internal control.
Immunoblot analysis
The lung tissues or the cells were lysed in RIPA Buffer (Beyotime Inc, China) according to the previously proposed method. 12 Furthermore, their protein was decided by the BCA assay (Thermo, USA). SDS-PAGE separated the total protein (20 μg), then transferred to PVDF membranes (Millipore, USA), and blocked with 5% non-fat milk in PBS with a tween. The membranes were incubated with primary antibodies for 3 h or 4°C overnight. The primary antibodies were diluted at 1:1000. The information of primary antibodies were as follows: anti-mouse SOD1 (ab13498, Abcam, USA); anti-mouse Nrf2 (12721, CST, USA); anti-mouse p-Syk (ab62338, Abcam, USA); anti-mouse Syk (ab40781, Abcam, USA); anti-mouse GAPDH (5174, CST, USA); anti-mouse p-p38 (4511, CST, USA); anti-mouse P38 (8690, CST, USA); anti-mouse p-STAT3 (9145, CST, USA); anti-mouse STAT3 (9139, CST, USA); anti-mouse p-JNK (9255, CST, USA); anti-mouse JNK (9252, CST, USA); anti-mouse IkBa (5209, CST, USA); anti-mouse p-p65 (3033, CST, USA); anti-mouse P65 (8242, CST, USA); anti-mouse PI3K (4228, CST, USA); anti-mouse p-Akt (4060, CST, USA); anti-mouse Akt (2920, CST, USA); anti-mouse Bcl-xL (2764, CST, USA); anti-mouse Cyto-C (4280, CST, USA); anti-mouse C-Cas3 (9661, CST, USA); anti-mouse C-Cas9 (9509, CST, USA). Secondary antibodies, anti-mouse and anti-rabbit HRP-conjugated antibodies (7076 and 7074, CST, USA), were used in this study. The bands were determined by a chemiluminescence kit (Pierce, USA). The quantitative analysis was based on ImageJ software. 13
Statistical analysis
Data were presented as the mean ± SEM. A one-way ANOVA test for comparisons among groups by Graphpad Prism 7 (Version 7, Graphpad Software, USA). We used Dunnett’s post hoc test or Sidak's multiple comparisons test for the post hoc test.
Results
Sophocarpine ameliorates LPS-induced ALI in mice
We first analyzed the pathology of the lung after sophocarpine treatment. The results showed that sophocarpine at 30 or 60 mg/kg each day ameliorates lung injury in LPS-induced ALI (Figure 1(a)). In addition, the lung W/D ratio was increased in the LPS group. However, sophocarpine significantly reduced the lung W/D ratio (Figure 1(b)). We also demonstrated that sophocarpine decreased the level of IL-6 and TNF-alpha in BALF (Figure 1(c) and (d)) compared with the LPS group. Thus, the present results suggest sophocarpine ameliorates LPS-induced lung injury. Sophocarpine attenuates LPS-induced lung injury. a: Representatives of H&E staining of lung tissues in mice underwent LPS-induced acute lung injury with or without sophocarpine (SC) at 30 or 60 mg/kg (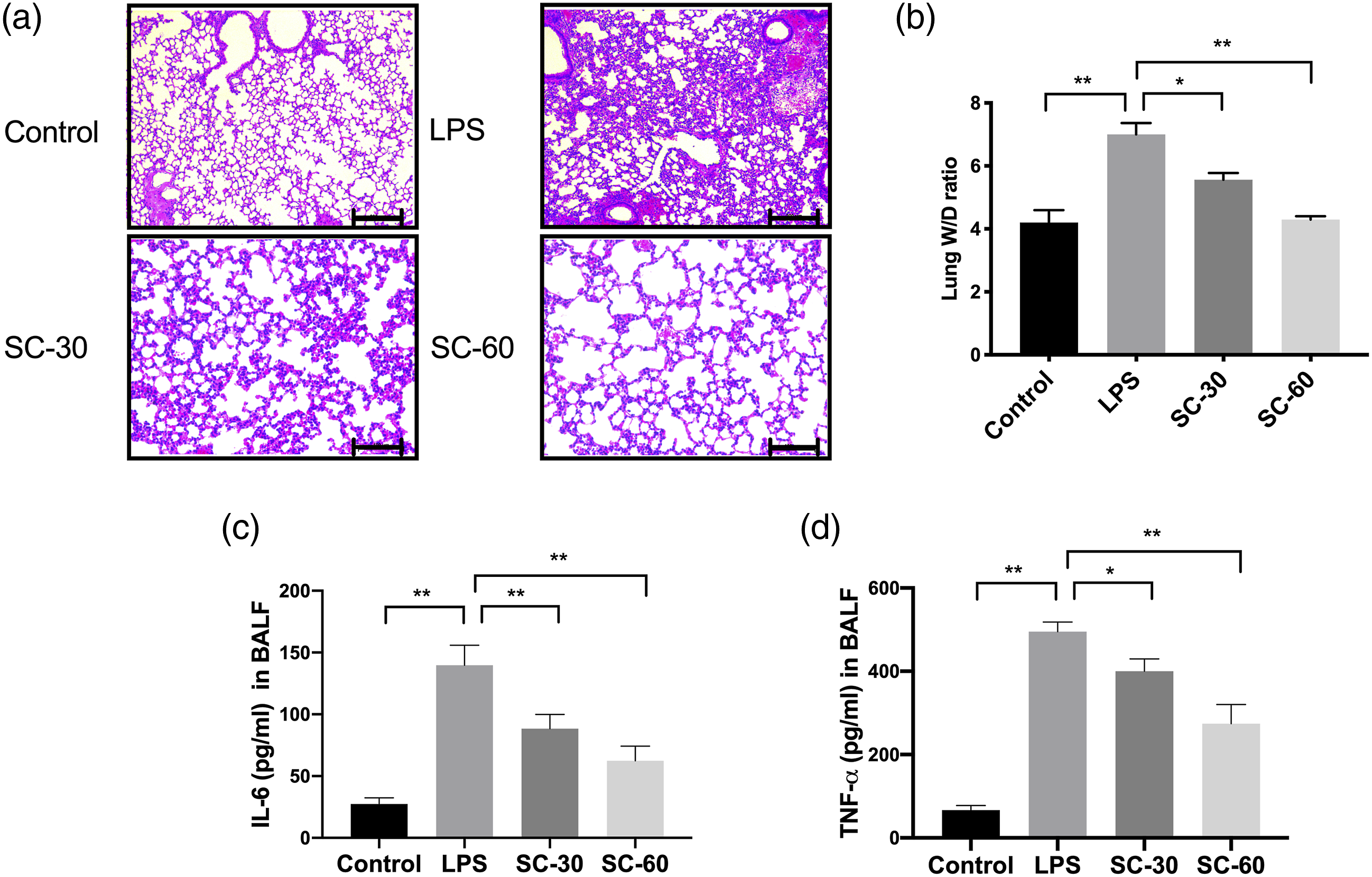
Sophocarpine decreased indicators of oxidative stress in LPS-induced ALI.
To investigate whether sophocarpine could decrease the oxidative stress in lung tissues of LPS-induced ALI mice, anti-oxidative factors including SOD, GSH and CAT were analyzed, and the level of H2O2, O.-2, and NO in the lung were measured. As the results showed, sophocarpine increased the activity of CAT and SOD and the level of GSH (Figure 2(a), (b), and (c)). Meanwhile, H2O2, NO, and O.-2 were ameliorated in mice (Figure 2(d), (e), and (f)). These results indicated that sophocarpine attenuated oxidative stress in LPS-induced ALI mice. Sophocarpine decreased indicators of oxidative stress in LPS-induced ALI. A–F: CAT activity (a), GSH level (b), SOD activity (c), H2O2 level (d), NO (e), and O-2 level (f) in the lung of mice underwent LPS-induced acute lung injury with or without sophocarpine (SC) at 30 or 60 mg/kg (
Sophocarpine influence ROS signaling in LPS-induced ALI mice
After proving the attenuation in ROS production of sophocarpine, we next investigated its inhibitory mechanism in the ROS pathway by immunoblot. As the results showed, SOD1 and Nrf2 were downregulated after LPS induction, and p-Syk was increased. After the intervention of sophocarpine, expression of SOD1 and Nrf2 were restored, and p-Syk was decreased in a dose-dependent manner (Figure 3(a) to (d)). Also, the associated protein related to oxidative stress, such as JNK, p38, and STAT3, were analyzed. The results showed that expression of STAT3, p38, and JNK was increased in LPS-induced mice while the phosphorylation level of P38, STAT3, and JNK was significantly inhibited by sophocarpine in a dose-dependent manner (Figure 3(e) to (h)). Thus, these results proved the influence of sophocarpine on oxidative stress in the lung of LPS-induced ALI mice. Sophocarpine influence the ROS pathway in LPS-induced ALI mice. A–H: Western blot (a, e), quantitative analysis of SOD1 (b), Nrf2 (c), p-Syk (d), and the ratio of phosphor-p38 (p-p38) to p38 (f), phosphor-STAT3 (p-STAT3) to STAT3 (g) and phosphor-JNK to JNK (h) in lung tissues of mice underwent LPS-induced acute lung injury with or without sophocarpine (SC) at 30 or 60 mg/kg (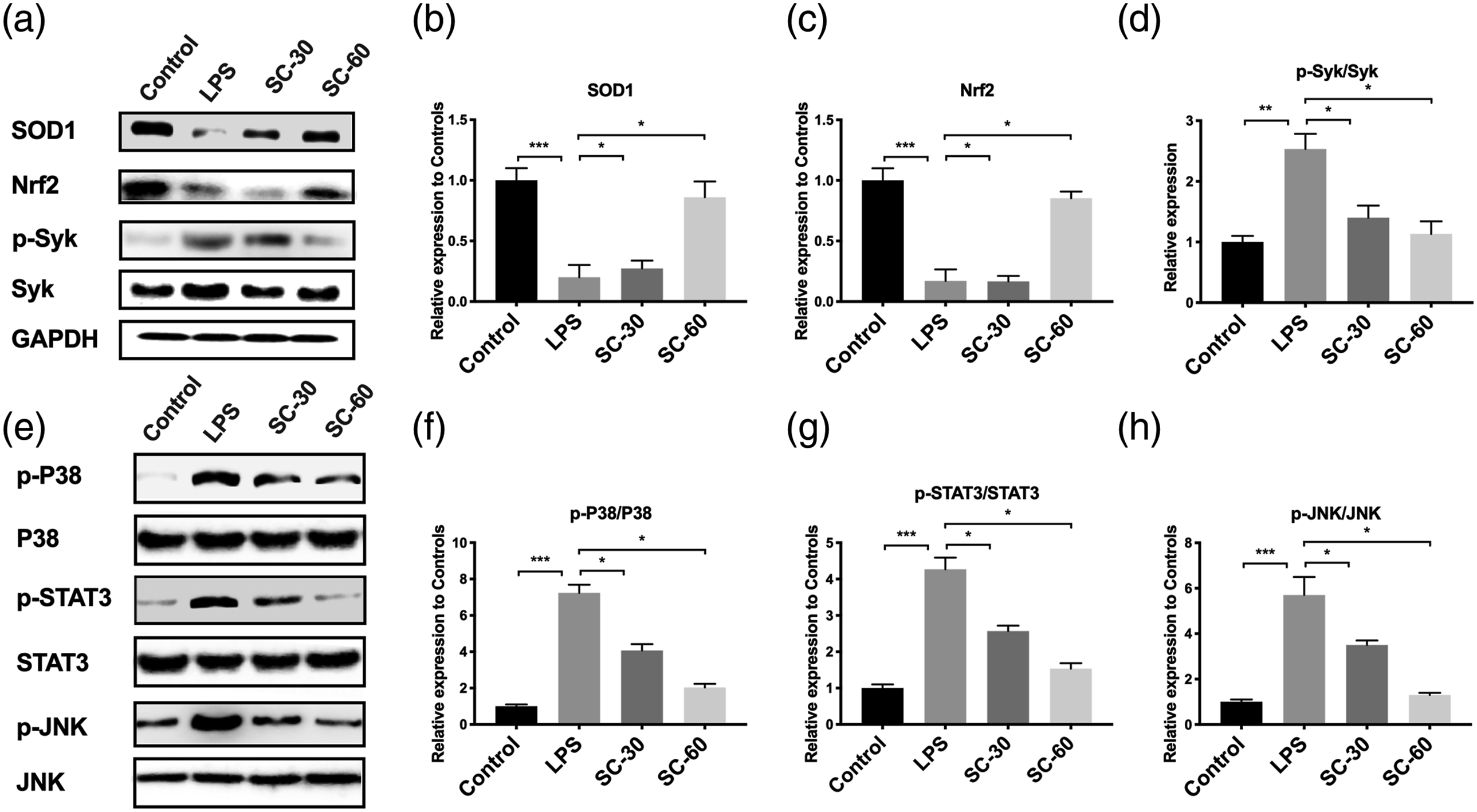
Sophocarpine ameliorated inflammation in LPS-induced ALI mice
When induced with LPS, serum levels of IL-1β, TNF-α, and IL-6 were significantly elevated compared with normal mice, while in the sophocarpine administration group, TNF-α, IL-6, and IL-1β were reduced (Figure 4(a), (b) and (c)). We further analyzed the protein level of specific molecules in lung tissues. We found that LPS elevated the expression of TNF-α and IL-1β and also regulatory protein IκBα and phosphorylated NF-κB while sophocarpine reversed these molecules in LPS-induced ALI mice (Figure 4(d) to (f)). Thus, these results indicated the anti-inflammation of sophocarpine during LPS-induced ALI. Sophocarpine ameliorated inflammation in LPS-induced ALI mice. A–C: ELISA of TNF-α (a), IL-6 (b), and IL-1β (c) in serum of mice underwent LPS-induced acute lung injury with or without sophocarpine (SC) at 30 or 60 mg/kg (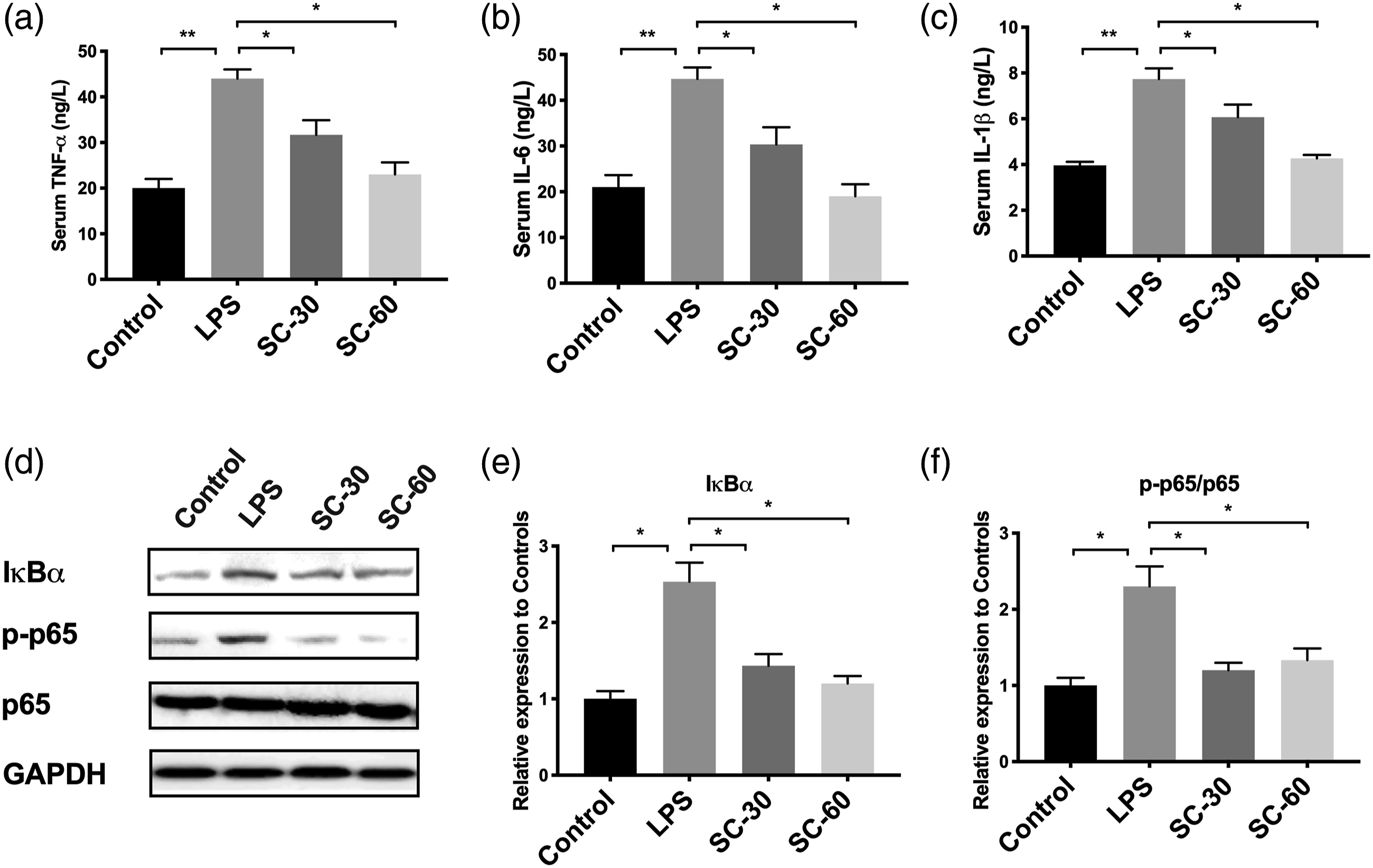
Sophocarpine suppressed apoptosis in LPS-induced ALI mice
To further investigate the alteration in apoptosis regulation, PI3K/AKT signaling was investigated. We showed that LPS increased the PI3K and AKT, and the sophocarpine administration decreased the level (Figure 5). Also, apoptosis-associated protein, Bcl-xL, Cyto-c, cleaved Caspase-3 (c-Cas3), and Caspase-9 (c-Cas9) were increased by LPS dramatically (Figure 5). After sophocarpine administration, these proteins were inhibited. These results suggested that sophocarpine suppressed these apoptosis-related genes in mRNA and protein levels. Sophocarpine suppressed apoptosis in LPS-induced ALI mice. A–G: Western blot (a) and quantitative analysis of PI3K (b), the ratio of phosphor-Akt (p-Akt) to Akt (c) Bcl-xL (d), Cyto-C (e), cleaved Caspase-9 (f) and cleaved Caspase-3 (g) in lung tissues of mice underwent LPS-induced acute lung injury with or without sophocarpine (SC) at 30 or 60 mg/kg (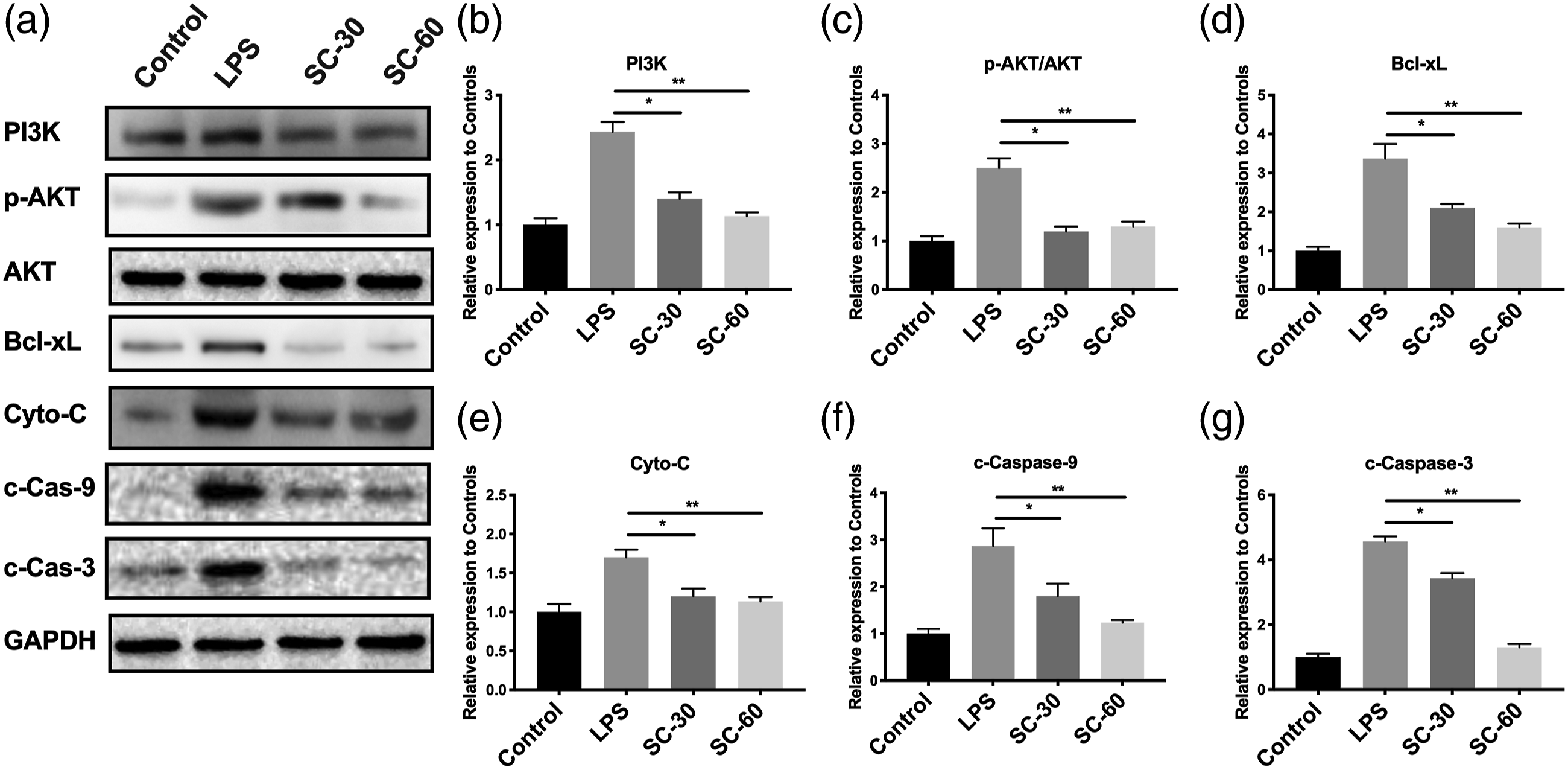
Sohpocarpine attenuates injury of LPS-treated MLE12 cells via suppressing apoptosis
To further confirm the effects of sophocarpine in MLE12 cells, we treated MLE12 cells with LPS and then subjected them to sophocarpine incubation to evaluate whether sophocarpine could reduce apoptosis of lung epithelial cells to attenuate lung injury. We found sophocarpine decreased the expression of PI3K, AKT, Bcl-xL, Bad, Apaf1, Caspase-3, Cyto-C, Caspase-6, and Caspase-9 while LPS promoted their expression (Figure 6). Thus, these results suggested that the sophocarpine could attenuate lung injury by suppressing PI3K/AKT-mediated apoptosis. Sohpocarpine attenuates the injury of LPS-treated MLE12 cells via suppressing apoptosis. A–I: Quantitative PCR of PI3K (a), Akt (b), Bad (c), Bcl-xL (d), Cyto-C (e), Apaf1 (f), Caspase 9 (g), Caspase 3 (h) and Caspase 6 (i) in MLE 12 cells treated with LPS with or without sophocarpine at 30ug/mL (SC-30) or 60ug/mL (SC-60) (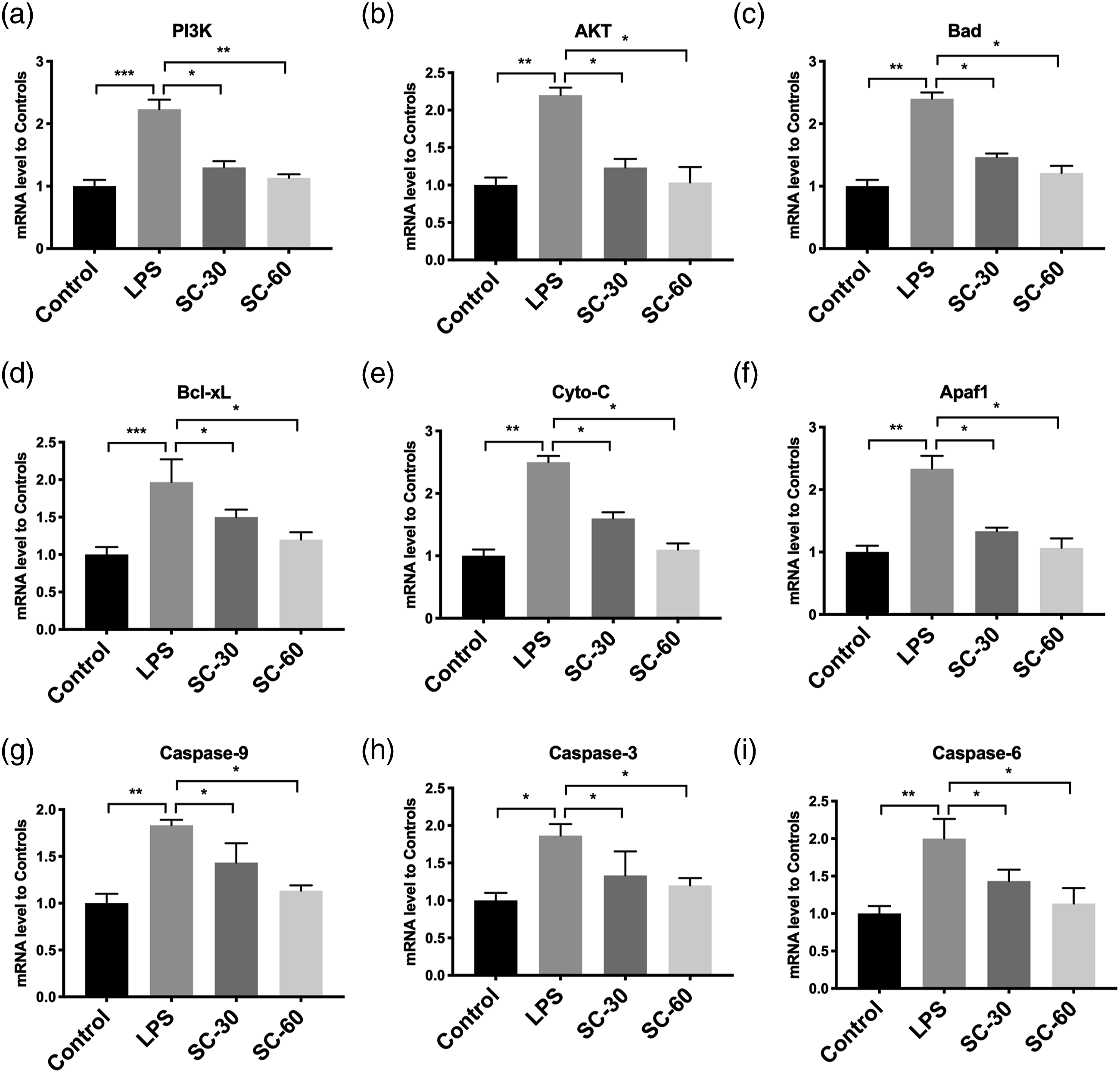
Discussion
As acute lung injury caused by Sepsis, pneumonia, or severe trauma remain a significant challenge to global intensive care, more appropriate medication and intervention should be investigated to achieve a better prognosis for the patient. The underlying pathogenesis of LPS-induced ALI has been proposed as an inflammatory injury, compromised alveolar-capillary barrier, and oxidative stress. As previous research on released inflammatory cytokine reduced the incidence and severity of ALI (12), similar results were also acquired in our experiments on the treatment of sophocarpine. In addition, in investigating anti-inflammation, substantial evidence is the alterations in molecules that drive inflammatory responses. As NF-κB and MAPK signaling play an essential role in inflammation regulation and LPS could induce p65 disassociated from IκB and further translocated to the nucleus to transcript cytokine production,14,15 we measured the mRNA and protein levels in relevant genes. These results proved the anti-inflammatory feature of sophocarpine, especially on general inflammation presented by serum cytokine level in LPS-induced ALI, and well-matched the previous research on sophocarpine.
Oxidative stress presents the balance between pro-oxidant and antioxidant. 16 ROS, such as hydrogen peroxide (H2O2) and superoxide radical (O.-2), were generated to cause regional damage.17,18 Research in ROS also found its crucial role in sepsis and organ failure, and decreased ROS level is associated with a better prognosis of organ injury. 19 Thus, by analyzing the level of H2O2, O.-2, and NO as well as the antioxidant agents, CAT, GSH, and SOD, we proved the protective effects of sophocarpine is anti-oxidative stress during ALI. Moreover, we also investigated the molecular regulation of ROS production. Previous research proposed cellular Nrf2, a binding protein of cytoskeletal-anchoring protein that senses the redox status during the ROS process. 20 Some studies discovered that p-Syk (phosphor-Syk) acts as a promoter of oxidative stress-mediated DNA damage.21,22 Therefore, we evaluated these molecules to determine the anti-oxidative effects of sophocarpine. The restored level of SOD1, Nrf2, and decreased p-Syk proved the anti-oxidative effects of sophocarpine in ALI.
We used in vivo and in vitro experiments to further prove the alteration in apoptosis. PI3K/AKT signaling and related downstream targets17,23 have been reported to play vital roles in apoptosis. In vivo and in vitro studies showed that sophocarpine suppressed the activation of PI3K/AKT signaling and further depressed the apoptosis of alveolar epithelial cells. However, the sophocarpine did not reduce the expression of Bcl-xL, according to previously published research.5,9 This may occur due to the translocation of Bcl-xL because Bcl-xL functions through binding to mitochondria. 24 The expression of Bcl-xL in the whole cell lysate may not reflex the exact process after sophocarpine treatment. Further research investigating the exact mechanism is still needed.
However, it should be noted that the present study still has some limitations. Firstly, the sample size was not determined by a power calculation. We used the whole lung tissue to analyze specific improvements after sophocarpine treatment. The improvement of specific cells or targets remains unknown. Thirdly, we only showed relevant apoptosis and oxidative stress markers when discussing the possible mechanism. The exact signaling transduction and the detailed target of sophocarpine require further investigation. Future research in therapeutic target and signal transduction of sophocarpine may help advance this molecule's knowledge in treating acute lung injury.
Conclusion
In conclusion, sophocarpine showed beneficial effects against inflammation, oxidative stress, and apoptosis in LPS-induced ALI. Molecular alterations also proved the related pathological improvements after the sophocarpine administration. Our finding suggested that sophocarpine might be a promising agent to treat septic or endotoxemia-related ALI.
Footnotes
Acknowledgements
We thank Dr. Zhengyu Jiang for the continuing instruction for our study.
Authors’ contributions
RBN, HG, and GXH performed biochemical and animal experiments. They also contributed to manuscript preparation and revision. JZ designed the project and helped with the manuscript preparation. FY and MSZ contributed to the animal experiments and helped in statistical analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Huashan Hospital Research Fund [HSBY2017013].
