Abstract
Oxidative stress appears to possess a central role in CIN pathophysiology. Resveratrol (Res) and lycopene (Lyc) are strong natural antioxidants evaluated in a limited number of CIN animal studies in vivo. The aim of the study was to evaluate the potential renoprotective effects of Res/Lyc in a CIN rabbit model. Twenty-four adult male New Zealand white rabbits were equally assigned into four groups: control (saline), CIN (intravenous iopromide; 7.5 g iodine/kg), Res + CIN (per os Res; 5 mg/kg), and Lyc + CIN (per os Lyc; 4 mg/kg). Serum Cr (sCr); symmetric/asymmetric dimethylarginine (SDMA/ADMA); oxidative stress biomarkers: malondialdehyde; total antioxidant capacity; catalase; glutathione) were evaluated in blood samples at three time points: right after (0 h); 24 h; 48 h after iopromide/saline administration. CD20+/CD3+ lymphocytes were determined (48 h). All animals were sacrificed at 48 h and both kidneys collected. Oxidative stress biomarkers were measured in renal tissue. sCr and SDMA/ADMA levels increased significantly in CIN compared to all groups. Oxidative stress secondary to CIN in blood/kidneys was suppressed by Res/Lyc. B and T lymphocytes decreased significantly in CIN compared to all groups. The present study provides emerging evidence that Res/Lyc ameliorate CIN by modulating oxidant/antioxidant balance in blood/renal tissue and by inhibiting vasoconstriction/blood cytotoxicity.
Introduction
Contrast-induced nephropathy (CIN) is a serious form of acute kidney injury (AKI) occurring 24–72 h post-exposure to iodinated contrast media (CM).1,2 The exact definition of CIN varies widely.3,4 The most recent definition according to the Acute Kidney Injury Network (AKIN) diagnostic criteria is related to an absolute or relative increase in baseline sCr levels ≥0.3 mg/dL or ≥50%, respectively or reduction in urine output <0.5 mL/kg/h for over 6 h, occurring abruptly (within 48 h) after a reno-toxic event (such as exposure to CM). 5 The incidence of CIN in individuals with a normal renal function who undergo imaging procedures is low but rises dramatically in high-risk patients such as diabetic patients; subjects with a history of congestive heart failure, chronic kidney disease, and of older age increasing morbidity, hospital stay and mortality.6,7
The pathophysiology is complex and not fully understood, however, oxidative stress appears to possess a central role.3,8 Besides, hemodynamic alterations resulting in renal medullary hypoxia and CM direct toxic effects on renal tubular epithelial cells seem to be major factors contributing to CIN.9,10 CM induces renal vasoconstriction, through induction of adenosine and endothelin, accompanied by shunting of blood flow from the medulla to the cortex. Consequently, renal blood flow to the medulla and glomerular filtration rate are reduced, followed by renal medulla ischemia. 11 The reduction in renal blood flow can also cause the release of reactive oxygen species (ROS) through oxidative stress and osmotic necrosis or vacuolization from direct toxic effects of CM on tubular cells, leading to acute tubular necrosis.12,13 ROS directly constrict the renal microcirculation and indirectly affect the renal vascular tone by mediating the effects of other vasoconstrictors, stimulating the production of vasoconstrictors, and modulating the actions of vasodilators, such as nitric oxide (NO). 14
CIN management remains supportive based mainly on intravenous hydration in high-risk patients, using saline or bicarbonate. 15 Novel biomarkers16,17 and agents with antioxidant and vasodilatory properties are attracting attention due to their ability to reduce oxidative stress/regulate vascular tone.3,4,18–20 The potential renoprotective effects of several antioxidants have been evaluated but none of them has been proven effective in human studies. 15 Resveratrol (Res) and lycopene (Lyc) are strong natural antioxidants with antimicrobial/anti-inflammatory activity found in red grapes/tomatoes.21,22 Renoprotective effects have been evaluated in a limited number of CIN animal studies in vivo23–27 and have been attributed to ROS scavenging, cell apoptosis inhibition, and alteration of regional hemodynamics through enhancement NO production.28–32 The aim of the present study was to evaluate the potential renoprotective effects of Res/Lyc in a CIN rabbit model.
Methods
Study design
Although CIN is mainly studied in rat models, this is not the ideal model, as CIN cannot be induced in normal animals. 33 Most studies have induced prerenal azotemia through 16–24-h water deprivation or even by removing a large portion of the animal’s kidneys followed by 48 h of water deprivation.34–36 Some researchers even treated rats with nephrotoxic drugs or with a combination of substances that caused nephrotoxicity. 37 Even though, morphologically rabbits resembles to rodents, protein sequence data suggest that rabbits are more closely related to primates than rodents, 38 comparative studies have shown that their renal function is more susceptible to contrast agents than the rats39,40 and CIN can be induced in normal rabbits, with a single injection of iodinated contrast media. 41 New Zealand white rabbits represent a more reliable model of nephrotoxicity, therefore, was chosen as the experimental model. 37
Twenty-four adult male New Zealand white rabbits (≈3 kg) were used. They were housed in individual metal cages and acclimatized (constant temperature 20–23°C, 12/12 h dark/light cycle). Food/water was provided ad libitum. After acclimatization animals were assigned into four equal groups: control (saline), CIN (CM [iopromide]: Ultravist® 370, Bayer Healthcare, Berlin, Germany), Res + CIN (Resveratrol, Solgar®, N.J, USA), and Lyc + CIN (Tomato Lycopene, SUPERFOODS®, Greece). Res and Lyc were administered by oral gavage, 24 h before CM exposure at a single dose of 5 mg/kg and 4 mg/kg, respectively. Prior to CM administration, animals were anesthetized using intramuscular xylazine hydrochloride (Xylapan®, Vetoquinol S.A., Lure Cedex, France) and ketamine hydrochloride (Narketan®, Vetoquinol S.A., Lure Cedex, France) at a single dose of 4 mg/kg and 40 mg/kg, respectively. An intravenous catheter was placed in a marginal ear vein for CM administration over 30 min at a single dose of 20.3 mL/kg (7.5 g iodine/kg). Blood samples were collected through the ear vein at three time points: right after CM/saline (0 h), 24 h and 48 h CM/saline administration for determining the level of sCr, symmetric dimethylarginine (SDMA)/asymmetric dimethylarginine (ADMA), oxidative stress and CD20+/CD3+ lymphocyte numbers (48 h blood samples). The animals were euthanized at 48 h post CM/Saline infusion, using sodium pentobarbital (Dolethal®, Vetoquinol S.A., Lure Cedex, France) intravenously at a single dose of 5 mL/animal and both their kidneys were removed for determining oxidative stress level. Study design is briefly illustrated in Figure 1. Study design: Twenty-four adult male New Zealand white rabbits were assigned into four equal groups: control, CIN, Res + CIN, and Lyc + CIN. Res and Lyc were administered by oral gavage, 24 h before CM exposure at a single dose. Blood samples were collected at three time points: right after CM/saline 0 h, 24 h and 48 h CM/saline administration for determining the level of sCr, SDMA/ADMA, oxidative stress and CD20+/CD3+ lymphocyte numbers. Animals were euthanized at 48 h, and both their kidneys were removed for oxidative stress evaluation.
Biochemical analysis
Blood was collected in vials without anticoagulant. Serum was isolated after centrifugation at 4000 r/min for 10 min at 21°C and stored at −20°C. sCr levels were determined using an automatic biochemical analyzer (Architect c4000, Abbott, Abbott Park, Illinois, U.S.A). CIN was defined as an absolute increase of ≥0.3 mg/dl of sCr level within 48 h after CM exposure.
SDMA and ADMA analysis
SDMA/ADMA levels were determined in blood samples using liquid chromatography-mass spectrometry (Shimadzu LC–MS 2010 EV) with electrospray ionization in positive mode and single quadrupole mass filter. Plasma was isolated by centrifugation at 4000 r/min for 10 min at 4°C and stored at −20°C.
CD20+ and CD3+ lymphocytes analysis
Blood samples (48 h) were air-dried on tiles. The immune-cytochemical method of immune-peroxidase was followed. Monoclonal mouse anti-human and polyclonal rabbit anti-human antibodies (Dako, Glostrup, Hovedstaden, Denmark) were used against CD20 and CD3 receptors to detect B and T lymphocytes, respectively. Antibodies were marked by immunoperoxidase reaction with DAB chromogen (Dako, Glostrup, Hovedstaden, Denmark). CD20+/CD3+ lymphocytes were defined under light microscopy 400x (Nikon eclipse E 400) in 100 random visual fields.
Oxidative stress biomarkers
Using colorimetric assay, oxidative stress biomarkers determined in blood and renal tissue samples. Blood samples were collected in EDTA vacutainers. Plasma was isolated by centrifugation at 4000 r/min for 10 min at 4°C and stored at −80°C, as renal tissue samples stored at −80°C until homogenization according to manufacturer’s Instructions. For malondialdehyde (MDA) analysis (Abcam, Cambridge, United Kingdom), TBA reagent was added into each sample (plasma and tissue)/standard and incubated at 95°C for 60 min. Τhe supernatant was collected and transferred to the microplate for absorbance measurement on microplate reader at optical density (OD) 532 nm. For total antioxidant capacity (TAC) assay (Abcam, Cambridge, United Kingdom), both plasma and renal tissue samples/standards were transferred to the microplate wells, Cu2+ working solution was added to the wells and incubated at room temperature for 90 min on an orbital shaker protected from light. Microplate was measured at OD 570 nm on microplate reader. For catalase (CAT) measurement (Abcam, Cambridge, United Kingdom), samples, standards and positive control, were added to the microplate wells, H2O2 solution was added to each well and incubation at room temperature for 30 min was followed. Then, stop solution was added to each well and, developer mix (Catalase Assay Buffer, OxiRed Probe and HPR Solution) was added, samples incubated at room temperature for 10°min protected from light. Microplate was measured immediately at OD 570 nm on a microplate reader. Finally, for reduced glutathione (GSH) assay (Sigma-Aldrich, St Louis, Missouri, USA), samples and sample background control were loaded to microplate wells with GSH assay buffer. Sample reaction mix was added to the plate wells. Absorbance measured at 450 nm (A450) in kinetic mode at room temperature for 40–60°min. Two time points (t1 and t2) were chosen in the linear range of the plot and the corresponding absorbance values obtained (A450(1) and A450(2)).
Statistical analysis
Sample size was calculated using G Power 3.1.9 software (significance level: 0.05, power: 0.80, analysis of variance (ANOVA)). Outcome variables (renal dysfunction biomarkers) were treated as continuous tested for normality using Shapiro-Wilk W test and compared using two-way ANOVA or Kruskal Wallis U test. Comparisons over time were performed using repeated measures ANOVA or Friedman’s test. Descriptive statistics were presented as mean (standard deviation (SD)). Data were analyzed using R: The R Project for Statistical Computing and Graphics, for Windows, R Version 4.0.4 (2021-02–15). Two-tailed p < 0.05 was considered significant.
Results
All animals in CIN group presented increase ≥0.5 mg/dL of sCr level at 48 h, confirming model validity. No differences were detected among groups at 0 h. sCr levels were significantly increased at 24 h, reaching peak at 48 h in CIN group. There was a slight decrease in sCr levels at 24 h/48 h compared to 0 h in Res + CIN/Lyc + CIN groups. Significant differences between CIN and all groups were observed at 24 h/48 h (Supplementary Table 1).
A significant elevation of SDMA/ADMA levels was observed in CIN group compared to all groups at all time points. Pick was observed at 24 h (SDMA) and 0 h (ADMA) and both decreased slightly at 48 h. SDMA/ADMA in Res + CIN/Lyc + CIN fluctuated over time at low levels without significant difference compared to control. Mean difference of both treatment groups compared to CIN was significant over time. SDMA/ADMA levels in Lyc + CIN group were slightly lower at each time point compared to Res + CIN group, without any significant difference. Results are summarized in Supplementary Tables 2 and 3.
MDA levels were significantly increased in CIN compared to all groups at 0 h/24 h, while a reduction was observed at 48 h (levels similar to control). No difference was observed in Res + CIN/Lyc + CIN groups compared to control at all time points. No difference was observed between Res + CIN/Lyc + CIN groups at all time points, although the latter group presented slightly lower levels at 0 h/24 h compared to Res + CIN/control group. MDA levels in renal tissue showed a significant increase in CIN compared to all groups. MDA levels in Res + CIN/Lyc + CIN groups fluctuated near baseline, without any difference between groups. Results are summarized in Supplementary Table 4.
Total antioxidant capacity in CIN was significantly reduced compared to all groups at 0 h/24 h. The reduction was eliminated at 48 h. No difference was observed between Res + CIN/Lyc + CIN groups at all time points; TAC levels remained near baseline over time. TAC levels in renal tissue decreased significantly in CIN compared to all groups but differences were insignificant between Res + CIN/Lyc + CIN groups. Results are summarized in Supplementary Table 5.
Reduction in catalase activity was observed in CIN compared to all groups but this reduction was eliminated at 48 h. Mean difference of CIN group at 0 h/24 h compared to all groups was significant, while at 48 h the reduced CAT activity remained significant only compared to control. CAT activity levels in Res + CIN/Lyc + CIN groups fluctuated over time within normal range. Res + CIN presented insignificantly higher CAT levels at all time points compared to Lyc + CIN group. CAT activity in renal tissue was reduced in CIN compared to all groups but this reduction was significant only compared to control and Res + CIN group. Lyc + CIN group presented insignificantly lower levels compared to control and Res + CIN group. Results are summarized in Supplementary Table 6.
GSH levels were decreased in CIN compared to all groups (trough at 24 h). This reduction was significant compared to all groups at 0 h/24 h, while at 48 h a slight increase occurred (normal values not reached) and difference was significant compared to control only. Res + CIN/Lyc + CIN groups fluctuated near baseline values, remaining stable over time. GSH levels in renal tissue were decreased in CIN compared to control group, however, this reduction was insignificant compared to Res + CIN/Lyc + CIN groups. Res + CIN/Lyc + CIN groups presented similar GSH levels, insignificantly lower and higher compared to control and CIN group, respectively. GSH level analysis results are shown in Supplementary Table 7.
CD20+/CD3+ lymphocytes were significantly decreased in CIN compared to all groups. In Res + CIN/Lyc + CIN, mean CD20+ and CD3+ lymphocytes fluctuated between CIN and control group values (Figure 2). The increase in Res + CIN and Lyc + CIN compared to CIN group was significant. Mean number of CD20+ (T lymphocytes) and CD3+ (B lymphocytes) lymphocytes in all experimental groups at 48h after CM exposure. *p < 0.050; **p < 0.01.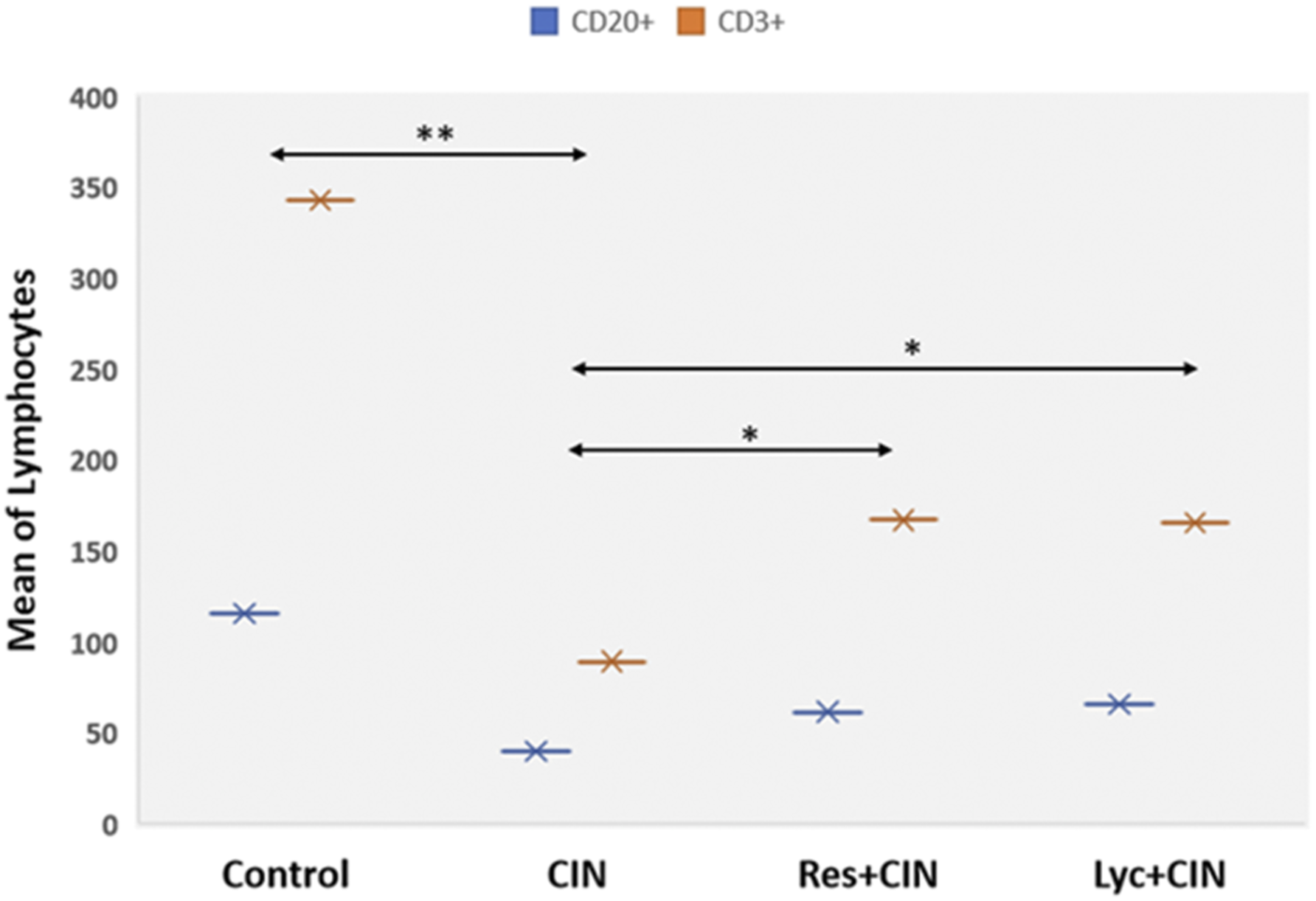
Discussion
CIN pathophysiology is complex but oxidative stress appears to possess a central role.3,8 MDA is a major biomarker of lipid peroxidation assessment. 42 TAC measures the number of free radicals scavenged by a test solution, used to evaluate antioxidant capacity of biological samples. 43 CAT is a key enzyme responsible for cell protection from oxidative damage by ROS. 44 Finally, glutathione is the main antioxidant present in cells in two forms (reduced GSH and oxidized GSSG) in equilibrium under normal conditions form. Oxidative stress results in increased levels of GSSG and measurement of intracellular GSH is a sensitive indicator of overall cell health, and its ability to resist toxic changes. 45 We showed that MDA levels were significantly increased in CIN compared to all groups at 0 h/24 h after CM administration in plasma/renal tissue. These results are supported by previous studies, showing that MDA levels increased immediately after CM exposure.46–48 Res/Lyc pre-treatment significantly decreased MDA levels in blood/renal tissue. These results indicate that both antioxidants had a positive effect due to their ability to protect mitochondrial/cell membranes from lipid peroxidation.49–52 CM administration enhance hypoxia/increase ROS production in blood/renal tissue, while production of free radicals is blocked by endogenous antioxidant systems such as TAC. 53 In our study, decreased plasma/renal tissue TAC levels in CIN group suggest a decrease in antioxidation in the systemic response. Res/Lyc was found to exert beneficial ROS scavenging properties by enhancing total antioxidant capacity despite CM exposure. Several studies reported that Res/Lyc ameliorate oxidative stress and are strongly associated with decreased TAC levels in blood/renal tissue.54–56 Changes in reduced GSH levels/CAT activity in blood/renal tissue developed due to increased generation of free radicals/reduction of antioxidant defense system secondary to CM exposure. 57 Decreased enzyme levels indicate their consumption due to increased oxidative stress. Both enzymes were significantly reduced time-independently in blood/renal tissue. Similar results were reported previously.57,58 Significantly elevated levels in blood were observed in Res + CIN/Lyc + CIN group, at 0 h/24 h compared to CIN group. Similarly, improvement was observed in reduced GSH/CAT levels in renal tissue that could be explained by the antioxidant effects of Res/Lyc, supported by previous animal studies.27,59,60
Adenosine/endothelin and free radical-induced vasoconstriction increase as well as NO and prostaglandin-induced vasodilatation decrease has been reported to contribute to ischemia in the deeper portion of outer medulla. 7 NO is a major endothelium-derived vasoactive mediator, derived from L-arginine by NO synthase. 61 SDMA/ADMA act as endogenous competitive NO synthase inhibitors and increased plasma levels represent strong risk predictor in progressive nephropathies.62–64 In our study, SDMA/ADMA levels were significantly elevated in CIN compared to all groups at all time points. SDMA/ADMA levels in Res + CIN/Lyc + CIN groups fluctuated near baseline values over time. ADMA/SDMA levels represent risk predictors in oxidative stress-related diseases, including renal dysfunction, and Res can significantly reduce blood ADMA/SDMA levels suggesting that Res effect on ADMA/SDMA metabolism may be an important mechanism for the beneficial effect on endothelial dysfunction.65–68 To our knowledge, Lyc effect on ADMA/SDMA levels has not been evaluated in any pathology before, apart from metabolic syndrome. 69
A significant decrease of CD20+ B and CD3+ T lymphocyte numbers was found in CIN compared to all groups. Lymphocytes numbers in Res + CIN/Lyc + CIN groups were near baseline. These findings indicate a generalized cytotoxic CM effect on white blood cell line, probably during cell programming and differentiation. A possible explanation could be that antigen-presenting cells are very sensitive against numerous stimuli altering their regulatory pathways, such as Nuclear Factor-κB (Nf-kB) signaling pathway. 70 Nf-kB is involved in inflammation/cell apoptosis process, activating various inflammatory factors, and stimulating cytokines expression, resulting in apoptosis and tissue damage.71,72 Several studies have shown that Res/Lyc inhibit NF-κB signaling pathway due to their strong antioxidant properties.73,74
Concluding, our model was successful based on our biochemical analysis, and CIN occurred in animals with normal kidney function without any prior intervention. Oxidative stress seemed to be one of the mechanisms implicated in CIN and oxidative status markers showed the higher ROS production in CIN group compared to the others. Intravenous administration of CM reduced the number of CD3+ B and CD20+ T lymphocytes indicating the direct cytotoxic effects of iopromide. SDMA-ADMA levels analysis revealed a statistically significant increase overtime in CIN group, which means that SDMA-ADMA are absolute indicators of renal dysfunction, and their levels are directly affected by CM administration.
Res/Lyc are strong natural antioxidants with antimicrobial/anti-inflammatory activity.21,22 They have been evaluated in a limited number of CIN animal studies in vivo with promising results (Supplementary Table 8).23–27 This is the first study evaluating Res/Lyc effect on ADMA/SDMA levels and the first to investigate their potential protective effect on white blood cell line from the cytotoxic effect of CM exposure. In accordance with previous studies, ours provides emerging evidence that Res/Lyc exert renoprotective effect, warranting further testing. The present study provides emerging evidence that Res/Lyc ameliorate CIN by modulating the oxidant/antioxidant balance in blood/renal tissue, and by inhibiting vasoconstriction/blood cytotoxicity.
Future research in this rabbit model is recommended to cover other aspects of the condition, such as molecules and signaling pathways involved in the pathophysiology, in order to establish preventive strategies. Also, the effectiveness of Res and Lyc use as a preventive strategy for CIN can be further investigated by changing the administration time point and the dosage. Future research may use more animals and conduct sampling before and after our timeline, to get a broader picture of parameter variations.
Supplemental Material
Supplemental Material - Resveratrol and lycopene ameliorate contrast-induced nephropathy in a rabbit model
Supplemental Material for Resveratrol and lycopene ameliorate contrast-induced nephropathy in a rabbit model by Eirini Fragkiadoulaki, Aristides Tsatsakis, Dragana Nikitovic, Georgios Georgiadis, Alexandra Kalogeraki, Konstantinos Kaloudis, Athanasios Alegkakis, Vasiliki Karzi and Charalampos Mamoulakis in Human & Experimental Toxicology
Footnotes
Acknowledgements
The authors would like to thank D. Pantartzi, Scientific Secretary of the Clinical Trial Office of the Department of Urology, University of Crete, Medical School, Heraklion, Crete, Greece, for the administrative and technical support.
Author contributions
Conceptualization: CM and AT. Design: CM, AT, and EF. Data acquisition: EF, KK, and VK. Data analysis: EF and AA. Interpretation of data: CM, AT, and EF. Writing––original draft: EF. Writing––review & editing: CM, AT, DN, GG, and AK.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was partially funded by the Special Account for Research Funds of University of Crete (SARF UoC No 3550) and the General Secretariat for Research and Innovation of Greece under the framework of EuroNanoMed III JTC 2018 project name:“CurcumAGE.”
Ethical approval
All animal experiments were performed at the University of Crete Medical School, in strict accordance with the National and European Union Directions/Directives/Guidelines (the American Veterinary Medical Association (AVMA) Guidelines for the Euthanasia of Animals: 2013 Edition, DIRECTIVE 2010/63/EU, The Animal Research: Reporting of In Vivo Experiments (ARRIVE) Guidelines). All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted. The entire study was approved by the Animal Welfare Committee of Medical School, University of Crete, Greece and by the Directorate of Veterinary Services of Heraklion, Crete, Greece (IRB approval number 102754).
Data availability
The data sets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
