Abstract
High doses of gadolinium-based contrast media are reported to induce deterioration of renal function. We assessed whether prophylactic 0.9% saline hydration inhibits high-dose gadodiamide-induced renal damage in rats. Twelve Sprague-Dawley rats were randomly divided into two groups, which are given gadodiamide (5 mmol/kg) with (hydration group) or without (control group) 0.9% saline hydration. The saline (4 mL/kg) was infused as a bolus into the peritoneum every 4 h, starting 12 h before and continuing for 12 h after the gadodiamide injection. Urine was collected to calculate creatinine clearance (Ccr) 24 h before and 48 h after the gadodiamide injection. The kidneys were harvested and stained for pathologic analysis. High-dose gadodiamide induced acute kidney injury as shown by decreased Ccr and renal histology with tubular cell injuries 48 h postinjection in both the groups. However, the extent of Ccr reduction was significantly (
Introduction
Contrast medium (CM)-induced nephropathy (CIN) is a serious complication of radiological examinations that require iodinated CM in high-risk patients. 1,2 CIN is the third most common cause of acute kidney injury in hospitalized patients. 3 Gadolinium-based CM (Gd-CM) was developed for magnetic resonance imaging (MRI) and considered non-nephrotoxic. 4 –7 However, increasing evidence has shown a close association between Gd-CM and CIN. 8 –21
At the cellular level, when cultured renal tubular cells are incubated with Gd-CM or iodinated CM, the cytotoxic effects, referred to as the extent of necrosis and apoptosis, are similar. 22 Moreover, Gd-CM agents are even more nephrotoxic than iodinated CM when administered intra-arterially at equimolar doses in pigs. 9 Human studies 17,19 also report that Gd may be nephrotoxic. Clinical reports revealed a 3.5%–12% incidence of acute kidney injury associated with Gd-CM in a high-risk population with underlying renal dysfunction. In addition, the incidence of deterioration of renal function is even higher in patients who undergo digital subtraction angiography requiring high intra-arterial doses of Gd-CM. 8,23,24 The clinically recommended dose of gadodiamide is between 0.1 and 0.3 mmol/kg for an MRI examination, while in digital subtraction angiography, high intra-arterial doses (up to 220 mmol) are required, which can cause a high incidence of nephropathy. 1 It is known that the prophylactic hydration therapy effectively inhibits iodinated CM-induced nephropathy. 2 However, whether prophylactic saline hydration also inhibits high-dose gadodiamide-induced nephropathy has never been evaluated. Therefore, we assessed whether prophylactic 0.9% saline hydration inhibits high-dose gadodiamide-induced renal damage in a rat model.
Methods
Animals
Male adult Sprague-Dawley rats (weighing 250–300 g), obtained from the Animal Resource Center of the National Science Council of Taiwan, were housed at an ambient temperature of 22 ± 1°C with a 12-h light/dark cycle. Pellet rat chow and tap water were available ad libitum. All protocols were approved by the Animal Ethics Committee of the Chi-Mei Medical Center in accordance with the Guide for the Care and Use of Laboratory Animals of the USA National Institutes of Health and the guidelines of the USA Animal Welfare Act.
Inducing renal damage
After they had been intraperitoneally (i.p.) injected with an anesthetic (tiletamine hypochloride/zolazepam hypochloride (Zoletil; Virbac Laboratories, Carros, France); 5.0–7.5 mg/100 g BW), the rats underwent venous cannulation via the right femoral vein with PE-50 polyethylene tubes. Twelve rats were then randomly assigned to the hydration group (infused with gadodiamide (5 mmol/kg) and i.p. injected with saline (0.9% NaCl; 4 mL/kg; every 4 h from 12 h before to 12 h after the gadodiamide infusion)) or the control group (infused with gadodiamide (5 mmol/kg);
Evaluating renal function
Blood samples were withdrawn from the tail veins of each untreated rat to measure serum creatinine (SCr) (Jaffe reaction Creatinine kit; Wako Pure Chemical Industries, Osaka, Japan). The rats were then put into individual metabolic cages and their 24-h urine sample was collected to measure baseline urinary creatinine (UCr). The next day, the rats underwent femoral vein catheterization and were infused with gadodiamide, after which they were put into the metabolic cages to obtain a second 24-h urine sample after the gadodiamide (24–48 h after the gadodiamide infusion) infusion to measure posttreatment UCr. A second blood sample from the tail vein was obtained 48 h after the gadodiamide infusion to measure posttreatment SCr. Creatinine clearance (Ccr) was calculated using the following formula
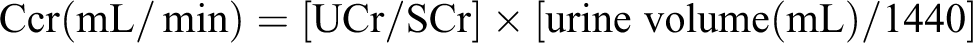

Evaluating morphological damage
To assess pathological damage to the kidney, at the end of the experiments (48 h after the gadodiamide infusion), the rats were given a lethal overdose of ethyl carbamate. Their kidneys were removed and perfused with 10% neutral-buffered formalin and then embedded in paraffin blocks. The blocks were serially sliced into 5-μm-thick sections and then stained with hematoxylin and eosin.
The sections were evaluated by a nephropathologist blinded to the treatment. The morphological changes indicating acute tubular necrosis of 100 randomly selected proximal collecting tubules (
Renal PT damage scores

PT: proximal tubule.
Statistical analysis
Continuous variables were expressed as median (25th to 75th percentile). The differences in Ccr between baseline and 48 h after the gadodiamide infusion were compared using Wilcoxon’s signed-rank test. Comparisons of control and hydration groups were done using the Wilcoxon’s rank-sum test for the percentage of Ccr reduction and pathological weighted scores for renal tubular injury. Significance was set at
Results
Effect of saline hydration on renal function
There was no significant difference in baseline Ccr between the control and hydration groups. High-dose gadodiamide induced an acute deterioration of renal function revealed by significant reductions in Ccr from baseline in both the groups (control group: from baseline = 2.11 mL/min (1.96 mL/min, 2.22 mL/min) (data are median (25th to 75th percentile)) to 1.29 mL/min (1.23 mL/min, 1.38 mL/min) 48 h postinfusion (−39%;

Ccr was lower after a gadodiamide-CM injection. Ccr was evaluated at baseline and 48 h after a gadodiamide-CM injection. (a) Gadodiamide-CM induced acute renal function deterioration in both the control and the hydration groups. (b) Prophylactic 0.9% saline hydration significantly inhibited the percentage of the reduction in Ccr. Data are means ± SD; *,†
Evaluation of renal histology
Morphological analysis of the kidney sections revealed renal proximal tubular injury in both the groups, but significant glomerular or vascular lesions were not found (Figure 2). Furthermore, the tubular cell injuries characterized by vacuolization, loss of brush borders, sloughing of tubular cells into the lumen, and flattening of the tubular epithelium were less frequently seen in the hydration group.

Pathological changes of the kidney 48 h after (a,b) a Gd-CM injection (control group) and (c,d) injection of Gd-CM plus 0.9% saline hydration (hydration group). The proximal tubules are widely vacuolated. The injured tubules (asterisks) show a loss of cell height and brush border with sloughed epithelial cells in the tubular lumen. Note that vacuolization and epithelial sloughing in the Hydration group occurred less frequently than in the control group. All figures show cells stained with hematoxylin and eosin; original magnifications are ×200 (a,c) and ×400 (b,d). Gd-CM: gadolinium-based contrast medium.
Proximal tubule vacuolization
In the control group, vacuolization was obvious and more prominent than in the hydration group. In most of the specimens, vacuoles also filled the region near the basal membrane (Figure 2(a) and (b)). Most of the vacuolization scores were 1 and 2 (Table 2). In the hydration group, sporadic vacuoles were seen, but the degree of vacuolization was significantly less than in the control group (Figure 2(c) and (d); Table 2). The weighted scores were significantly lower in the hydration group than in the control group (

Weighted scores for renal tubular injury after a Gd-CM injection: (a) vacuolization score of PT; (b) epithelial sloughing score for PT; and (c) epithelial thinning score of PT. Results are means ± SD. *
Weighted scores for cytoplasmic vacuolization, epithelial sloughing, and epithelial thinning after a Gd-CM injection in the control and hydrated groups

Gd-CM: gadolinium-based contrast medium.
Proximal tubule sloughing
Renal tubular epithelial cell damage, observed as cellular degeneration and sloughing from tubular basement membranes, was seen after a high-dose gadodiamide infusion (Figure 2). Damage was most often localized in the proximal tubular epithelia. In the control group, the lumens of numerous
Proximal tubule thinning
The injured tubules show a loss of brush borders and a reduction in tubular cell height (Figure 2). However, the difference in the extent of epithelial thinning of the proximal tubules between the two groups was not significant.
Discussion
We found that high-dose gadodiamide is nephrotoxic in rats. Morphological analysis disclosed typical CIN pathologic changes. We also found that prophylactic 0.9% saline hydration helped to protect renal function by partially inhibiting histological damage (vacuolization and epithelial sloughing) in high-dose gadodiamide-induced nephropathy rats.
The pathogenesis of iodinated CM-induced nephropathy is associated with alterations in renal hemodynamics (ischemia due to vasoconstriction), reactions to hyperosmolar agents, and direct tubular toxicity. 27 The cytotoxic effects induced by iodinated CM are osmolality-dependent. 10,22,28 Gd-based CMs are also hypertonic, with an osmolarity of two to seven times greater than that of plasma, and they are not absorbed by renal tubular epithelial cells. These characteristics of Gd chelates are similar to those of iodinated CM. The injection of high-dose of gadodiamide could result in a substantial osmotic load to the kidneys. High osmolar CM induce intense and prolonged vasoconstriction at the corticomedullary junction of the kidneys and directly impairs the autoregulatory ability of the kidneys. 1,29–35
Several agents are suggested as beneficial for preventing CIN: hydration,
Although the benefits of hydration for preventing iodinated CM-induced nephropathy are well documented, there are only limited data available on the preventive effects of hydration in patients with Gd-CM-induced nephropathy. In a prospective, randomized, controlled pilot study,
16
21 patients given Gd-CM or iodinated CM were intravenously hydrated with 1000 mL of fluid 12 h before and 12 h after CM administration. The incidence of CIN was similar in the both groups. In a retrospective study
36
of 25 patients given Gd-CM for coronary procedures and 32 patients given iodinated CM, all patients had been intravenously hydrated at a rate of 1 mL/kg/h from 12 h before to 12 h after they had been given CM and
Histological data from a limited number of animal studies 12,22,37 suggest that Gd-CM acutely injures the epithelial cells of the renal tubules. A large dose of Gd-CM induced significant cytoplasmic vacuolization in proximal convoluted tubular cells, and the pathologic change was intensified by dehydration. 37,38 Till date, there are few reports on the renal lesions associated with Gd-CM-induced nephropathy in humans. One 56-year-old patient with diabetes developed acute renal failure after being given Gd-CM in two consecutive vascular imaging procedures to explore a hypertensive crisis. 18 A kidney biopsy showed acute tubular cell injury, which included patchy tubular necrosis, tubular epithelial cell degeneration, and marked proliferation of tubular cells, together with mild interstitial edema and infiltration. In our study, renal tubular vacuolization, tubular epithelial sloughing, and tubular epithelial thinning were seen after a single high dose of intravenous gadodiamide. In the hydration group, cortical tubular cell injuries induced by high-dose gadodiamide were seen less frequently than in the control group. Vacuolization and epithelial sloughing of the proximal tubules differed significantly between the two groups. Tubular vacuolization is seen following the infusion of hypertonic solutions. Tubular epithelial sloughing reflects the death of tubular cells, which are then sloughed into the lumen. These two changes are predominant during acute kidney injury. However, proximal tubular thinning represents regenerative change, which is more apparent during the recovery phase but not during acute kidney injury. 39 Therefore, there were no changes in proximal tubule thinning in the acute stage.
Developing Gd-CM-induced nephropathy is related to preexisting risk factors such as diabetes mellitus and renal insufficiency. 17,19 Patients with diabetes mellitus are more likely to develop CIN because of their impaired baseline renal blood flow. However, an immediate decrease in renal blood flow does not occur in well-hydrated patients exposed to CM. 40 Low-dose Gd-CM seems to be well tolerated. In contrast, patients with renal insufficiency given higher doses (>0.3–0.4 mmol/kg BW) experience greater nephropathy. 8
Our study is limited by its lack of other well-documented biomarkers to support the benefits of hydration therapy, for example, checking and comparing neutrophil gelatinase-associated lipocalin in urine, cystatin C in blood, or immunochemical staining for kidney injury molecule-1 on kidney samples, which may bring more confidence. Because the study also lacks some biochemical tests—glucose, urea, total cholesterol, triglycerides, and low-density lipoprotein cholesterol in blood and urine—we do not have sufficient data for broad and firm conclusions. Second, hydration is beneficial when combined with high-dose Gd-CM, but it may not give significant benefits for lower, clinically used doses of Gd-CM.
Intravenous high-dose gadodiamide induced acute renal damage in Sprague-Dawley rats. We showed that prophylactic 0.9% saline hydration helped prevent functional and histological abnormalities in rats administered with a high dose of gadodiamide. Preventive hydration therapy is a simple and effective measure and should be considered for humans given high-dose gadodiamide, especially patients at high risk of acute kidney injury.
Footnotes
Authors’ Note
Chih-Chiang Chien and Ming-Jen Sheu contributed equally to this work.
Funding
This study was supported by grant CMFHR9766 from the Chi-Mei Medical Center.
Acknowledgments
The authors thank all the staffs in Professor Jhi-Joung Wang’s Department of Medical Research.
