Abstract
Background
Diabetes nephropathy (DN) is a serious diabetic problem that may progress to renal failure. The root of Curcuma longa L., often known as turmeric, provides various health benefits. Bisacurone is a bioactive terpenoid found in small amounts in turmeric that possesses anti-inflammatory and antioxidant properties. The present study focuses on the potential protective effects of bisacurone against DN via reducing renal inflammation, oxidative stress, and apoptosis.
Methods
Type 2 diabetes was created in rats by feeding them a high-fat/high-sugar diet for 8 weeks, followed by a low dose of streptozotocin and Bisacurone (50 and 100 μg/kg bisacurone) given for 4 weeks.
Results
In diabetic rats, bisacurone reduced hyperglycemia, protected against body weight (BW) loss, lowered renal markers, reduced lipid profile alterations and avoided histological abnormalities. Bisacurone treatment reduced oxidative stress by decreasing malondialdehyde (MDA) levels while enhancing antioxidant defenses through superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx) levels. Furthermore, bisacurone treatment activated the renal Nrf2/Keap1 signaling pathway but attenuated the high levels of NFκB p65, TNF-α, IL-1β, IL-6, Cox2, and iNOS. Bisacurone also reduced Bax, caspase-3, caspase-9 and cytochrome c but increased Bcl-2 in the kidneys of diabetic rats.
Conclusion
In the present study, bisacurone reduces DN by reducing hyperglycemia, oxidative stress, inflammation, and apoptosis, while also increasing Nrf2/HO-1 signaling.
Introduction
Diabetes mellitus (DM) is a metabolic condition characterized by chronic hyperglycemia caused by inadequate insulin production from pancreatic
An excess of reactive oxygen species (ROS) causes the development of DN, which is induced by hyperglycemia and oxidative stress. Excess ROS causes podocyte degeneration and death in the diabetic kidney. 7 Besides, NADPH oxidase (NOX), specifically the widely spread NOX4, is the primary source of ROS at DN. 8 NOX4-produced ROS is mainly responsible for fibrosis and podocyte death in diabetic kidneys. 9 Oxidative stress has a role in the molecular mechanism causing fibrosis in various organs, including DN. 10 Nrf2 is a transcription factor involved in antioxidant and anti-inflammatory responses, making it an important cellular defense mechanism. 11 Excess ROS may damage cellular macromolecules and activate redox-sensitive molecules like nuclear factor-kappa B (NF-kappa B), resulting in pro-inflammatory mediators and other cellular degradation. 12 Diabetes problems and DN have been linked to oxidative damage, NF-kB activation, and increased pro-inflammatory conciliators. 13 In addition, TLR4/MyD88/NF-kB signaling augments the inflammatory cascade in diabetic kidneys by regulating the production of pro-inflammatory mediators. 14 Furthermore, DN induces oxidative stress and inflammation, which leads to a series of events that results in apoptosis and cell death.
Herbal medicine has a long history of usage in the treatment of diabetes. Several studies indicate that these herbs may benefit humans and animals with DM. Because oxidative stress, inflammation, and apoptosis contribute to diabetes and its complications, medicinal herbs with antioxidant properties are utilized to alleviate these side effects, including DN. 15 Curcuma longa liter. (Zingiberaceae) is a plant that grows in both tropical and subtropical areas. Turmeric powder is widely used as a spice and medicinal throughout Asia. Turmeric extract has around 200 components, including phenolic chemicals and terpenoids. Several studies have shown that curcumin, turmeric’s major bioactive component has medicinal properties. Curcumin is responsible for anti-inflammatory, antioxidant, neuroprotective, and anticancer activities.16–18 Furthermore, bisacurone is a bioactive terpenoid found in low concentrations of turmeric. 19 Previous research has demonstrated that bisacurone can fight with free radicals, alter Nrf2/HO-1 signaling in the kidneys of diabetic rats, and lower oxidative stress, inflammation and cell death. 20 In this study, the authors looked at the protective effects of bisacurone on diabetic kidneys in Sprague Dawley rats. The authors mainly investigated the underlying molecular mechanisms via the Nrf2/NF-kB/caspase-3 pathways in diabetic rat kidneys.
Materials and methods
Chemicals and reagents
Bisacurone used in this experiment was supplied by ChemFaces Biochemical Co., Ltd. (Wuhan, China), and its overall purity was more than 99%. Glibenclamide and streptozotocin were provided by Sigma-Aldrich (St Louis, MO, USA).
Animals
This study used 40 Sprague Dawley male rats (8–10 weeks; 230–250 g weight) from the Department of Nephrology, Affiliated Hospital of Hebei University in China. The local ethics committee approved the ethics under ethics number 202203-R012. All animals acclimated for a week before the experiment. Rats were housed at a constant temperature (23 ± 2°C) and humidity (50–60%) on a 12 h light/dark cycle. All animals were given free access to food and water ad libitum.
Animal model and grouping
Thirty-two rats were given a high-fat/high-sugar diet for 8 weeks. To mimic type 2 diabetes, rats were administered 35 mg/kg of STZ dissolved in a citrate barrier (pH 4.5, 0.1 mol/L) intraperitoneally, whereas the control group (n = 8) got citric acid only.
21
Blood was drawn from the tail-vein 7 days after the STZ injection and tested for fasting blood glucose (FBG) levels using a glucometer (ACCU-CHEK, Roche, Mannheim, Germany). Diabetes in rats was specified as a blood sugar level of more than 12.5 mmol/L. Rats were divided into five groups (n = 8). Experimental rats were given the therapy orally by oral gavage for 4 weeks (Figure 1(a)). Group I (Control): The vehicle was used regularly and daily. Group II (DN): The vehicle was used regularly and daily. Group III (50BS): Diabetes rats received 50 μg/kg bisacurone daily.
22
Group IV (100BS): Diabetes rats received 100 μg/kg bisacurone daily.
22
Group V (GB): Diabetes rats received 600 μg/kg/day of glibenclamide daily.
23
Bisacurone ameliorates hyperglycemia and weight loss in diabetic rats. (a) Experimental design, (b) fasting blood glucose levels, (c) body weight loss, (d) food intake, (e) water intake, (f) HOMA-IR, (g) insulin, (h) kidney weight (KW), (i) KW/BW ratio. Data are mean ± SEM, (n = 8). *p < .05 versus control; #p < .05 versus diabetes.

Once every week, the body weight (BW), FBG levels, and food and water intake were measured. On the day before the investigation concluded, 24 h urine samples were collected. All rats were euthanized after receiving an intraperitoneal dose of 1% sodium pentobarbital, blood was collected in centrifuge tubes from the heart puncture. The kidneys were separated and then weighed to determine their weights. Consequently, kidneys were kept at −80°C or in 4% paraformaldehyde for further studies.
Biochemical analysis
The serum separated from the blood by being centrifuged at 2000 r/min for 20 min and stored at −20°C. The serum was used to estimate creatinine, albumin, urea, uric acid, urinary protein, Na+ and K+ levels using a biochemical analyzer ((BS 300, China). Blood urea nitrogen (BUN) was calculated using the following formula: BUN (mg/dl) = urea (mg/dl)/2.14. Serum insulin levels were determined using an ELISA kit (Elabscience, Wuhan, China). HOMA-IR was calculated using FBG and insulin levels. The plasma levels of triglycerides (TG), total cholesterol (TC), and high and low-density lipoprotein (HDL and LDL-L) were measured using a biochemical analyzer.
Assessment of oxidative stress and antioxidant enzymes
The kidney was homogenized in cold PBS (pH 7.4) before centrifugation at °C at 2000 r/min for 15 min. The supernatant was collected and quantified malondialdehyde (MDA), superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx) using ELISA kits (SenBejia Biotech, Nanjing, Jiangsu, China).
Determination of pro-inflammatory and inflammatory cytokine levels
Nuclear factor kappa B (NF-κB) p65, tumor necrosis factor (TNF)-α, interleukin (IL)-1β, and IL-6 levels in kidney homogenate were measured using commercial ELISA kits (R&D Systems, Minneapolis, MN, U.S.A.).
Histopathological assessment
The kidneys were fixed in 4% paraformaldehyde, dehydrated, paraffin-impregnated, and sectioned into 5 μm thick sections. Hematoxylin and eosin (H&E) were used to reveal kidney structure, Masson’s Trichrome (MT) was used to detect fibrosis, and Periodic Acid-Schiff (PAS) was used to examine mesangial matrix accumulation in glomerular and tubular diseases. Light microscopy was used to assess pathological alterations at ×400.
Immunohistochemistry and immunofluorescence
The kidneys were sectioned into 5 μm, deparaffinized, and dehydrated before antigen retrieval. After cooling, the sections were incubated for 15 min with 3% hydrogen peroxide (H2O2) to inhibit endogenous peroxidase activity, and then for 1 h with 5% bovine serum albumin (BSA) to minimize non-specific binding. Anti-NOX4 (Cat #PA5-72,816; Thermo fisher scientific) Keap-1 (Cat #PA5-99434; Thermo fisher scientific), TNF-α (Cat #PA1-40,281; Thermo fisher scientific), Caspase-9 (Cat #PA5-22252; Thermo fisher scientific), cytochrome c (Cat #BSM-33193M; Thermo fisher scientific) primary antibodies (1:100) were used to incubate sections overnight. After 3 times PBS washing, the sections were then treated for 1 h at room temperature with a second HRP-conjugated antibody (Cat # 31460; Cat # 31430; Thermo fisher scientific) and followed by one to 2 min of incubation with DAB color development and counterstain for 1 min with hematoxylin. After rehydration and cleaning, the slides were examined under a microscope (Nikon, Japan).
For immunofluorescence, after deparaffinized, and dehydrated, antigen retrieval, the kidney sections were treated overnight at −4°C with NOX2 (Cat #PA5-76034; Thermo fisher scientific), Nrf2 (Cat #PA5-27882; Thermo fisher scientific), NQO1 (Cat #39–3700; Thermo fisher scientific), NF-κB p65 (#8242, Cell signaling), TGF-β (sc-130348; Santa Cruz) primary antibodies (1:100). After PBS washing, sections were incubated with Alexa fluor 488 and 555 antibodies (Beyotime, China). After PBS washing, it was stained with DAPI (Beyotime, China). A fluorescence microscope detected fluorescence (LeciaDMI8, Germany).
Quantitative reverse-transcription PCR
Primers used for qRT-PCR.

Statistical analysis
The findings are the mean and SEM. All statistical analyses were done using one-way ANOVA followed by Tukey’s posthoc analysis using GraphPad Prism 5 (San Diego, California, USA) software. A p value of .05 was considered significant.
Results
Effect of bisacurone on hyperglycemia, weight loss and food and water intake of diabetic rats
The diabetic rats exhibited hyperglycemia as shown by a significant (p < .001) increase in FBG levels compared to the control group (p < .001; Figure 1(b)). Diabetic rats treated with 50 and 100 μg/kg bw bisacurone exhibited anti-hyperglycemic effects. Bisacurone significantly reduced blood glucose levels in diabetic rats (p < .001), although the 100 μg/kg dose had a significantly more significant impact than the 50 μg/kg dose. The BW of diabetic rats decreased significantly (p < .001), as seen in Figure 1(c). Bisacurone at 50 and 100 g/kg bw significantly delayed weight loss in diabetic rats. As shown in Figure 1(d) and (e), diabetic rats displayed increased food intake and water consumption, which decreased after bisacurone treatment at 50 and 100 μg/kg bw (p < .05).
Effect of bisacurone on HOMA-IR, insulin and kidney weights in diabetic rats
The HOMA-IR of diabetic rats was significantly greater than that of normal rats, as seen in Figure 1(f). Alternatively, HOMA-IR was reduced after 50 and 100 μg/kg bw bisacurone treatment. Furthermore, serum insulin levels (p < .001) were lower in diabetic rats compared to normal rats after treatment with 50 and 100 μg/kg bw bisacurone insulin levels higher than the diabetic group (Figure 1(g)). Diabetic rats showed significantly higher kidney weight (KW) and but not KW/BW ratios (Figures 1(h) and (i)). However, KW at 50 μg/kg bw of bisacurone did not affect diabetic rats. In diabetic rats, 100 μg/kg bw bisacurone dramatically lowered KW, but KW/BW significantly increased in both biscurone and glibenclamide treatment compared to diabetic rats.
Effect of bisacurone on serum kidney function tests in diabetic rats
Serum creatinine (Figure 2(a)), albumin (Figure 2(b)), urea (Figure 2(c)), urinary uric acid (Figure 2(d)), urine protein levels (Figure 2(e)) and BUN (Figure 2(f)) were often used to evaluate kidney function. These features were more prevalent in the diabetes group than in the normal control group showing that renal function was significantly (p < .001) compromised. Surprisingly, bisacurone treatment at 100 μg/kg bw dosages returned these values to normal levels. Electrolytes like Na+ (Figure 2(g)) and K+ (Figure 2(h)) levels were significantly (p < .001) decreased in diabetic rats. In contrast, bisacurone treatment at 50 and 100 μg/kg bw showed these levels increased compared to the diabetic rats. Bisacurone ameliorated kidney function markers in diabetic rats. (a) Serum creatinine, (b) albumin, (c) urea, (d) urinary uric acid, (e) urine protein and (f) BUN, (g) Na+, (h) K+. Data are mean ± SEM, (n = 8). *p < .05 versus control; #p < .05 versus diabetes.
Effect of bisacurone on lipid profile in diabetic rats
The diabetic group had higher levels of TG (Figure 3(a)), TC (Figure 3(b)), and LDL-L (Figure 3(d)) than the control group (p < .05) but lower levels of HDL-L (Figure 3(c)). Bisacurone treatment groups of 50 and 100 μg/kg bw reduced TG, TC, and LDL-L levels and increased HDL-L levels compared to the diabetes group. Bisacurone ameliorated lipid profile in diabetic rats. (a) TG, (b) TC, (c) HDL-L, (d) LDL-L. Data are mean ± SEM, (n = 8). *p < .05 versus control; #p < .05 versus diabetes.
Effect of bisacurone on accurate histopathology in diabetic rats
To confirm bisacurone’s protective effect on experimental rats' kidney histology. Figure 4(a) and (d) depict a control kidney with normal parenchyma, renal corpuscles, and glomeruli. Diabetic rats caused mild changes in the glomeruli and dilated proximal tubules. The kidneys of bisacurone-treated rats exhibited normal renal glomeruli, substantial improvement in the majority of proximal tubules, and renal corpuscles impairment. In addition, collagen was higher, indicates tubular atrophy and fibrosis areas were more apparent in diabetic rats than in normal rats, despite bisacurone treatment decrease the fibrosis (Figure 4(b) and (e)). The accumulation of glycogen and damage to the glomerular capillary basement membrane were more severe in the kidneys of diabetic rats, but bisacurone could also ameliorate these abnormalities (Figure 4(c) and (f)). Bisacurone ameliorated real histopathology changes in diabetic rats. (a) Hematoxylin and eosin (H&E), (b) Masson’s trichrome (MT), (c) periodic acid-schiff (PAS), (d) score of kidney damage by using H&E images, (e) Collagen area (%), (f) %PAS staining. Data are mean ± SEM, (n = 8). *p < .05 versus control; #p < .05 versus diabetes. Scale bar = 50 μm, magnification = ×40.
Bisacurone attenuates renal oxidative stress and antioxidant enzymes in diabetic rats.
Figure 5(a) demonstrates that kidney MDA (p < .01) levels were significantly higher in the diabetic group, although SOD (Figure 5(b); p < .01), CAT (Figure 5(c); p < .01), and GPx (Figure 5(d); p < .01) activity levels were lower, indicating that antioxidant capacity was diminished. In contrast, MDA levels were significantly reduced with increased SOD, CAT, and GPx activity levels in 50 and 100 μg/kg bw BW bisacurone (p < .05) treated diabetic rats kidneys. Immunohistochemistry studies demonstrated that the levels of both NOX2 (Figure 5(e)) and NOX4 (Figure 5(f)) were significantly lower in diabetic rats than in normal rats, and bisacurone treatment was able to reduce these abnormalities. Bisacurone ameliorated real oxidative stress and increase antioxidant enzymes in diabetic rats. (a) MDA, (b) SOD, (c) CAT, (d) GPx, photomicrographs showing immunostaining of (e) NOX-2 and (f) NOX-4 in the kidney of normal and diabetic rats. Scale bar = 50 μm, magnification = ×40. Data are mean ± SEM, (n = 8). *p < .05 versus control; #p < .05 versus diabetes.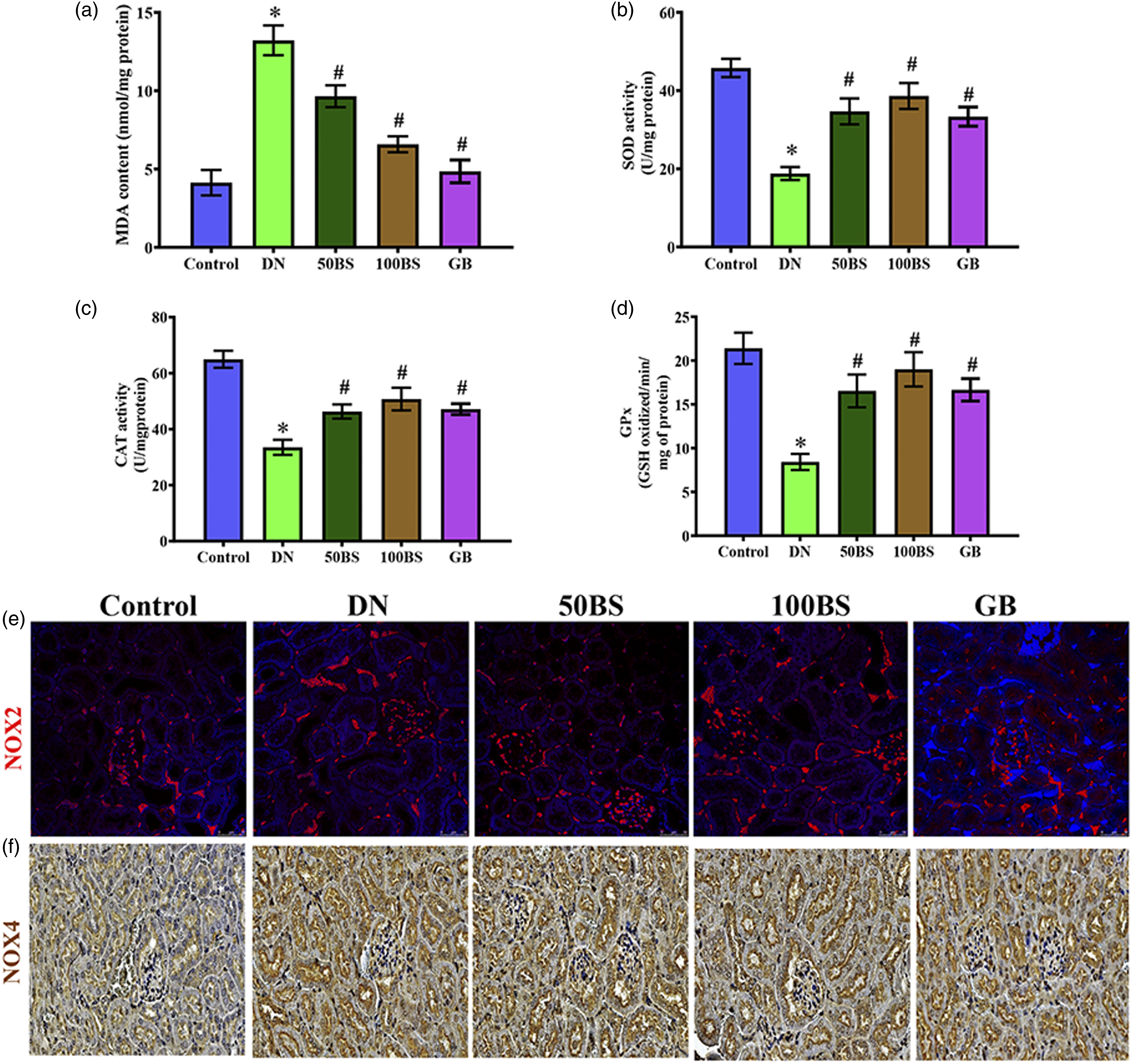
Bisacurone upregulates renal Nrf2/HO-1 in diabetic rats
We analysed the qRT-PCR/IF expression levels of Nrf2, Keap1, NQO1, and HO-1 to determine whether Nrf2/HO-1 signaling contributes to bisacurone’s kidney-protective effect in diabetic rats. The mRNA levels of Nrf2 (Figure 6(a)), NQO1 (Figure 6(c)), and HO-1 (Figure 6(d)) in diabetic rats’ kidneys were substantially lower than in the control group (p < .001), although Keap1 (Figure 6(b)) was significantly higher. Bisacurone treatment at 50 and 100 μg/kg bw enhanced Nrf2, NQO1, and HO-1 levels in diabetic kidneys while lowering Keap1 (p < .001). It was found that the expression of Nrf2 (red color, Figure 6(e)) and NQO1 (green, Figure 6(e)) in immunofluorescent results were correlated with the levels of their respective mRNAs. Furthermore, immunohistochemistry results confirmed that the distribution of Keap1 protein was higher in diabetic rats kidneys, but bisacurone treatment at 50 and 100 μg/kg bw significantly decreased Keap1 protein distribution in diabetic rats kidneys (Figure 6(f)). Bisacurone affects renal Nrf2/HO-1 pathway in kidneys. The mRNA levels of (a) Nrf2, (b) Keap1, (c) NQO1, (d) HO-1 in kidneys. Photomicrographs showing immunostaining of (e) Nrf2 (red) and NQO1 (green) (f) Keap1 in the kidney of normal and diabetic rats. Scale bar = 50 μm, magnification = ×40. Data are mean ± SEM, (n = 8). *p < .05 versus control; #p < .05 versus diabetes.
Bisacurone attenuates inflammation in the kidney of diabetic rats.
To evaluate the anti-inflammatory effects of bisacurone in the kidneys of diabetic rats, the levels of pro-inflammatory cytokines were determined (Figure 7). The diabetic rats' kidney TNF-α (Figure 7(b)), IL-6 (Figure 7(c)), IL-1β (Figure 7(d)), COX2 (Figure 7(e)) and iNOS (Figure 7(f)) level increased considerably (p < .001) compared to the control group. Bisacurone administration at 50 and 100 μg/kg bw to diabetic rats substantially decreased the levels of TNF-α (p < .001), IL-1β (p < 0=.05), IL-6 (p < .05), IL-1β (p < .01), COX2 (p < .01) and iNOS (p < .05). Additionally, glibenclamide significantly decreased pro-inflammatory cytokines in diabetic rats' kidneys. ELISA and IHC results revealed a significant increase in NF-kB-p65 (Figures 7(a) and (g); red color) expression in diabetic rats’ kidneys compared to the control group (p < .001). Besides, TGF-β (Figure 7(g); green color) is a powerful anti-inflammatory cytokine expression level that was decreased in diabetic rats kidneys, while the treatment with bisacurone and glibenclamide treatment TGF-β levels were increased as compared to diabetic rats. Bisacurone affects renal inflammatory pathway in kidneys. Levels of (a) NF-kB-p65, (b) TNF-α, (c) IL-6, (d) IL-1β, The mRNA levels of (e) COX2, (f) iNOS. Photomicrographs showing immunostaining of (g) NF-kB-p65 (red color) TGF-β (green color), (h) TNF-α in the kidney of normal and diabetic rats. Scale bar = 50 μm, magnification = ×40. Data are mean ± SEM, (n = 8). *p < .05 versus control; #p < .05 versus diabetes.
Bisacurone prevents apoptosis in the kidney of diabetic rats
Upon entering the cytoplasm, cytochrome c may activate the caspase-3 cascade. Cytochrome c released into the cytoplasm may activate caspase-9 and 3. In diabetic rats, Bcl-2 (Figure 8(a)), Bax (Figure 8(b)) caspase-3 (Figure 8(c)) caspase-9 (Figures 8(d) and (e); brown color) and cytochrome c (Figure 8(e); red color) levels were significantly (p < .001) higher than in the control group. After 4 weeks of treatment with bisacurone extracts or glibenclamide, these alterations in diabetic rat kidneys decreased considerably. Bisacurone affects renal apoptosis pathway in kidneys. The mRNA levels of (a) Bcl-2, (b), bax (c) caspase-3, (d) caspase-9. Photomicrographs showing immunostaining of (e) caspase-9 (brown color) and cytochrome c (red color) in the kidney of normal and diabetic rats. Scale bar = 50 μm, magnification = ×40. Data are mean ± SEM, (n = 8). *p < .05 versus control; #p < .05 versus diabetes.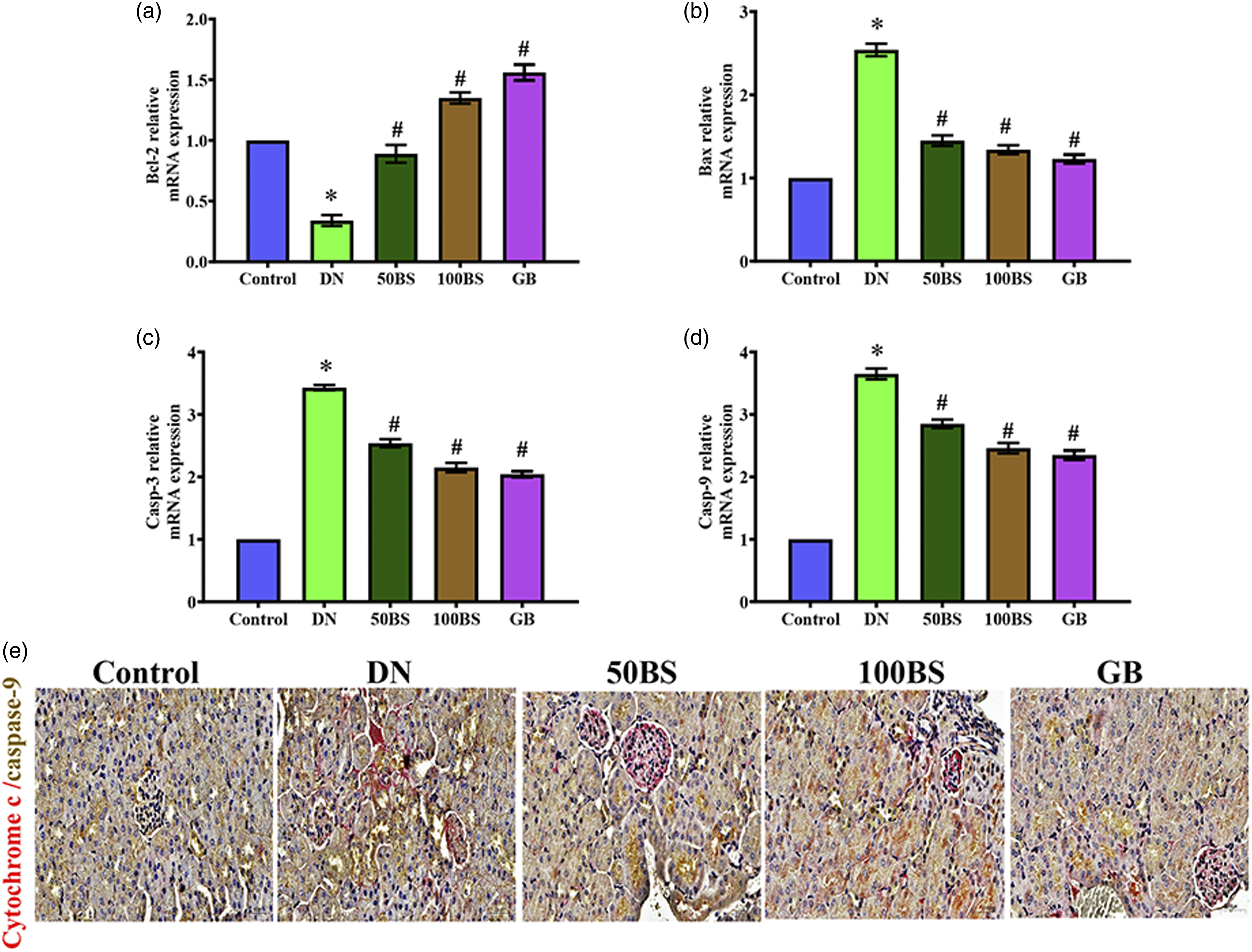
Discussion
Diabetic nephropathy (DN) is a severe diabetic complication that includes proteinuria, decreased creatinine clearance, and high creatinine, urea, and uric acid levels. In our current experiments, the type II diabetic rat model with nephropathy established by a high-fat/high-sugar diet and a modest dosage of STZ demonstrated a significant rise in blood sugar and insulin resistance, as shown by an increase in the HOMA-IR index, renal damage. 24 The current research revealed the molecular mechanism behind bisacurone’s renoprotective activity, which was employed to assess oxidative stress, inflammation, and apoptosis markers in a type II diabetic rat model. Diabetics’ metabolism and hemodynamics are affected by hyperglycemia. Excellent glycemic management is required for both preventing and treating DN. Our findings show that bisacurone reduces blood glucose levels and delays the decline in renal function in type II diabetic rats. 22 Similar to our results, turmeric extract and its principal constituent curcumin lower blood sugar levels in diabetic rats by inhibiting hepatic glucose synthesis and increasing GLUT expression.25,26
In our current study, diabetes-induced kidney damage was shown by elevated creatinine, urea, uric acid, albumin, microalbumin, and BUN levels in diabetic rats. 27 Creatinine is a glomerular function indicator and a by-product of muscle creatine. 28 The kidney eliminates the bulk of the urea generated as a consequence of protein breakdown. Diabetic kidney damage is indicated by albumin in the urine. Albuminuria is a risk factor for DN, and the 24 h urine albumin excretion rate also measures kidney damage. 29 Diabetes rats administered bisacurone showed lower creatinine, urea, uric acid, albumin, microalbumin, and BUN than diabetic rats. Our results agreed with previous reports demonstrating that turmeric extract and its major constituent curcumin protect from renal diseases by reducing creatinine, urea, uric acid, albumin, microalbumin, and BUN in diabetic rats.30,31
In our current study, diabetic rats showed much reduced Na+ and K+ levels. Hyperglycemia causes water to migrate out of cells through hyperosmolarity, resulting in dilutional hyponatremia. 32 The higher urine loss of the diabetic rats disrupted homeostasis, altering K+ concentration. The administration of bisacurone to diabetic rats increased ionic electrolyte levels and enhanced kidney purification. Lipid accumulation in podocytes, which results in podocyte degeneration and renal failure, is the primary cause of DKD. 33 In this study, diabetes rats showed greater TC, TG, and LDL-L levels, but decreased HDL-L levels. These findings imply that diabetes-related lipid changes induce kidney injury. 34 Bisacurone lowers TC and TG while increasing HDL-L. In this study, bisacurone’s renal protective actions in DN rats are associated with a reduction in circulating lipids. Our findings support previous studies that bisacurone decreases hepatic lipid accumulation by inhibiting lipogenesis and promoting lipolysis via AMPK activation. 35
Diabetic kidney damage was linked to histological changes like glomerular necrosis, interstitial hemorrhage, fibrotic and degenerative changes, inflammatory cell infiltration, and perivascular lymphocytic accumulations. 36 Bisacurone improves kidney function and reverses structural modifications caused by hyperglycemia in diabetic rats, which shows that it protects the kidneys. Based on these results, bisacurone helped diabetic rats’ kidneys work better. The previous study has demonstrated that histological alterations in rats treated with curcumin may indicate enhanced renal function, which is consistent with our findings. 37
Oxidant and antioxidant defense systems are compromised in DM. Oxidative stress has a vital role in the onset and progression of DN. In the present study, MDA was found to be increased and decreased SOD, CAT, and GPx in the diabetic kidney were indicators of antioxidant alterations. Cellular organelle damage and oxidative stress may be caused by increased MDA and decreased antioxidant defenses. Superoxide dismutase is the most critical enzymatic defense against superoxide anions. Catalase and GPx catalyze hydrogen peroxides and protect tissues from hydroxyl radicals. 38 In this research, 10 and 20 mg/kg of bisacurone increase the activity of antioxidant enzymes compared to diabetic controls. Other studies have reported similar findings, such as bisacurone reduced oxidative stress. They increased antioxidant enzymes such as SOD and GSH in the heart tissues of STZ-induced diabetic rats. 22
The nephroprotective effect of bisacurone is connected to Nrf2, an antioxidant transcription factor. Under normal conditions, Keap1 segregates Nrf2 in the cytosol. When ROS levels are elevated, Nrf2 is generated and translocated into the nucleus, where it binds to the antioxidant response element (ARE) and stimulates the transcription of antioxidant genes. 39 The activation of Nrf2 decreases oxidative stress and inhibits the development of DN. 40 Bisacurone enhanced mRNA levels of Nrf2, NQO-1, and HO-1 in diabetic rat kidneys, indicating its efficacy as an Nrf2/HO-1 signaling activator. Activation of Nrf2 by curcumin protects rats against acute liver damage produced by lipopolysaccharide, consistent with previous research. 41
Furthermore, diabetic rats showed considerable activation of NF-κB in the kidney, as demonstrated by increased IκB phosphorylation and subsequent enhanced NF-κB translocation. NF-κB is activated in diabetic kidneys in response to increased ROS production and cytokine release. 42 In our study, NF-κB activation promotes the higher expression of TNF-α, IL-1β, IL-6, COX-2, and iNOS in diabetic rats’ kidneys. TNF-α, overexpression may enhance the generation of localized oxygen free radicals, worsen oxidative stress damage in the kidneys, and promote the production of inflammatory cytokines such as IL-6, resulting in inflammatory cell leakage. 43 Bisacurone has been shown to have anti-inflammatory effects on human umbilical vein endothelial cells by inhibiting NF-κB p65 translocation into the nucleus and phosphorylation of inhibitory factor κBα (IκBα). 44 An increase in inflammatory cytokines, and extracellular matrix (ECM) deposition causes tubulointerstitial fibrosis. 45 Besides, activation of TGF-β has long been recognized as an essential mediator of renal fibrosis. 46 Furthermore, TGF-β is a potent anti-inflammatory cytokine negatively affecting kidney inflammation. 47 TGF-β with hyperglycemia may cause podocyte thickening, proteinuria, and diabetic neuropathy. 48 TGF-β protein expression was significantly increased in diabetic rats, however bisacurone treatment significantly reduced TGF-β expression in diabetic rat kidneys. In line with our findings on openness prevent prevention in previous studies, bisacurone contains TGF-β expression levels in non-alcoholic fatty liver disease.
Oxidative stress and inflammation increase apoptosis in DN. 49 Hyperglycemia-induced ROS generation in the mitochondria may increase cytochrome c release into the cytosol, resulting in an apoptosome that activates effector caspase-3. Apoptosis is a multistage process comprised of several steps. 50 Reducing the expression of the apoptosis inhibitor Bcl-2 and increasing the face of the apoptosis-promoting variable Bax is one technique for activating the apoptosis machinery. 51 After treating Bisacurone, we noticed that Bcl-2 expression was downregulated and Bax protein was significantly increased at both the protein and mRNA levels. Consequently, it increases mitochondrial membrane permeability and cytochrome C release, which activates caspase-3 and 9 and initiates the death cascade. According to other the findings, the bisacurone therapy significantly decreased the expression of caspase-3 and 9 protein levels in diabetic rats kidneys. 52
Conclusions
For the first time, our findings show that bisacurone reduces blood glucose levels significantly and improves DN-induced kidney damage. The bisacurone reduces changes in renal biochemical markers, lipid profile, oxidative stress and inflammation, fibrosis and apoptosis. Our study believes bisacurone might be applicable in treating DN. Furthermore, the current work adds to the experimental evidence o curcumin’s therapeutic potential and novel medication development.
Footnotes
Author contributions
FY: Methodology, experiments performance, data analysis; draft the manuscript; ZZ: Conceptualization, editing the manuscript, data analysis; LZ: Funding acquisition, project administration, supervision drafts the manuscript; FY, ZZ, and LZ: All authors reviewed the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
