Abstract
Ulcerative colitis (UC) is a chronic inflammatory disease affecting colon. The primary symptoms of UC are diarrhoea with blood and mucus and sometimes associated with pain.
Aim:
This study aimed to evaluate the effects of diosmin (DIO) on UC.
Methods:
UC was induced in rats by rectal administration of acetic acid (AA). The degree of mucosal ulceration and damage was determined by evaluating the disease activity index (DAI) and colon damage index scores. Inflammation, oxidative stress and apoptotic responses were determined by measuring the levels of tumour necrosis factor-α (TNF-α), cyclooxygenase-2 (COX-II), malondialdehyde (MDA) and reduced glutathione (GSH) in addition to myeloperoxidase (MPO) activity and caspase-3 expression.
Results:
The results of this study indicate that AA caused increase in DAI and colon damage index scores. Also, markers of inflammation (TNF-α, COX-II and MPO) and oxidative stress (MDA and reduced GSH) were significantly elevated. These changes were associated with increases in colon caspase-3 expression. Treatment of rats with two doses of DIO produced a dose-dependent decline in DAI and colon damage index scores. Also, DIO resulted in significant reduction of inflammatory and oxidative stress markers as well as reduced the expression of caspase-3.
Conclusion:
DIO therapy decreased UC development depending on its ability to decreases inflammation, oxidative stress and apoptosis in rat colon.
Introduction
Ulcerative colitis (UC) is a chronic inflammatory bowel disease affecting the colonic mucosa. It may involve parts or all of the large intestine. Despite the aetiology of UC is not completely explained, many factors have been incorporated as possible aetiological factors such as genetic factors, infective agents, immunological factors and smoking. 1 In patients with UC, inflammation and breakdown of colonic mucosa lead to bloody diarrhoea, passage of pus and abdominal pain. 2 The activation of the intestinal immune system in UC results in generation of free radicals, pro-inflammatory cytokines, such as tumour necrosis factor-α (TNF-α) and interleukin-1β, prostaglandins and leukotrienes. 3 Many drugs are used in treatment of UC such as anti-inflammatory (aminosalicylates and glucocorticosteroids), immune modulators, antibiotics and anti-TNF-α. 4
Acetic acid (AA)-induced UC is a widely used method for induction of UC. This model mimics human intestinal inflammations through increase in generation of ulcer, inflammatory mediators, free radicals and neutrophils infiltration. 5 The primary goal of drugs used in treatment of UC is rapid remission of symptoms. Aminosalicylates are widely used drugs for treatment of mild-to-moderate UC. Sulphasalazine (SSZ) is a member of aminosalicylates and consists of sulphapyridine and 5-aminoslicylic acid moieties. The 5-aminoslicylic acid moiety of SSZ is responsible for its therapeutic effect in treatment of UC, and the majority of its side effects are related to the sulphapyridine moiety. 6
Flavonoids are natural compounds present in vegetables, fruits and other many plants. Diosmin (DIO) is a natural flavonoid usually extracted from citrus plants, and its structure was shown in Figure 1. DIO has venoprotective effect in treating some vascular diseases. Also, DIO has biological antioxidant, anti-inflammatory and anti-apoptotic activities. It is well known for boosting venous tone and lymphatic drainage with suppression of capillary hyperpermeability. 7 Several studies reported that DIO has a beneficial effect in many pathological conditions as hyperlipidaemia, diabetes mellitus and peptic ulcer. 8 –10 Since there were not enough studies conducted to investigate the effect of DIO in UC, this study is carried out to investigate the benefits of DIO administration in AA-induced UC. The protective effect of DIO may be related to its anti-inflammatory, antioxidant and anti-apoptotic features.

Structure of DIO. 7 DIO: diosmin.
Materials and methods
Drugs and chemicals
DIO is a gift from SEDICO CO (6th of October City, Egypt), while SSZ is donated by Kahira Pharma and Chem. Ind. Co. (Cairo, Egypt). Ellman’s reagent, reduced glutathione (GSH) and tetramethoxypropane were purchased from Sigma-Aldrich Company, St Louis, Missouri, USA. All other chemicals are of analytical grade.
Animals
Male Swiss albino rats, weighing 170–200 g, were kept for 1 week to acclimatize with surrounding environment in the animal house of the Faculty of Medicine, Assiut University. Food and water were given ad libitum. Experiments were conducted under accepted national guidelines for ethical care of animals. The study protocol was approved by the Animal Ethics Committee of Faculty of Medicine, Assiut University, Egypt.
Induction of colitis
Briefly, rats were fasted for 24 h with free access to water then anaesthetized with ether. A paediatric catheter was introduced intra-rectal for 8 cm then 1 ml (4% v/v) AA in normal saline was slowly instilled into colon. Next, 2 ml of air was ejected to complete distribution of AA in colon. Rats should be in a head down position for 30 s to prevent leakage of AA from the anus. Control rats were given normal saline using the same procedures. 11
Treatment design
A total of 50 rats were divided into five groups (10 rats in each).
Group I: Control group received 1 ml carboxymethyl cellulose (CMC) 0.3% one time daily for 7 days.
Group II: Colitis was induced in rats by rectal administration of 1 ml (4% v/v) AA.
Group III: Colitis was induced in rats by rectal administration of 1 ml (4% v/v) AA and treated with SSZ (100 mg/kg, orally).
Group IV: Colitis was induced in rats by rectal administration of 1 ml (4% v/v) AA and treated with DIO (25 mg/kg, orally).
Group V: Colitis was induced in rats by rectal administration of 1 ml (4% v/v) AA and treated with DIO (50 mg/kg, orally).
Both DIO and SSZ were suspended in CMC 0.3% and administrated orally for 7 days using oral tube. On the fourth day, rats were fasted for 24 h, and then UC was induced at fifth day in anaesthetized rats by rectal administration of 1 ml (4% v/v) AA in normal saline. At the eighth day, animals were anaesthetized with light ether and blood samples were collected by direct heart puncture for separation of serum. Colons were excised out from rats and washed with normal saline and then used for macroscopic scoring, histopathological examination and biochemical estimations. A portion of colonic tissues were cut into small pieces, then homogenized in ice-cold 0.1 M phosphate buffer saline pH 7.4 using tissue homogenizer and then stored at −40°C.
Determination of colon mucosal damage and disease activity index
Colonic damage was estimated according to scale of Morris et al. 12 (0: normal; 1: mucosal erythema only; 2: mild mucosal oedema, slight bleeding or erosion; 3: moderate oedema, bleeding ulcers or erosions; 4: severe ulceration, erosions, oedema and tissue necrosis and perforation).
Disease activity index (DAI) was scored by evaluating the consistency of stool and presence of rectal bleeding in all animals before and during induction of UC using the scoring system illustrated by Cooper et al. 13 The scores of stool consistency and bloody diarrhoea were as follows: stool consistency (0 and 1: normal consistency, 2 and 3: loose stool and 4: diarrhoea) and rectal bleeding (0 and 1: negative, 2 and 3: rectal occult blood and 4: gross rectal bleeding).
Determination of TNF-α and COX-II levels and MPO activity
Levels of TNF-α and cyclooxygenase-2 (COX-II) were determined in serum by utilizing enzyme-linked immunosorbent assay kit (Wkea Med Supplies Corp, Changchun Jilin, China) depending on manufacturer instructions. Results are expressed as nanograms per millilitre of serum. Myeloperoxidase (MPO) activity in colon tissue was measured spectrophotometrically depending on the steps of Krawisz et al. 14 MPO activity was expressed as unit per gram of tissue.
Estimation of lipid peroxidation and GSH
Malondialdehyde (MDA) contents in colon tissues were determined by using thiobarbituric acid reactive substance method. MDA was expressed as nanomole per gram of wet tissue. 15 While GSH content was measured in colon tissues by using Ellman’s reagent. 16
Western blots analysis of caspase-3
Total protein content of colon was determined according to the method described by Lowry et al. 17 Equal protein amount of each sample was undergo to electrophoresis on sodium dodecyl sulphate–polyacrylamide gel and then transferred to polyvinylidene fluoride membrane. This membrane was blocked with 5% fat-free milk and then incubated with the primary antibodies (caspase-3 antibody, 1:500 or β-actin antibody, 1:500) (Santa Cruz, California, USA) at 4°C overnight. The membrane was washed three times (10 min in each), then incubated with secondary antibodies (1: 5000) at 37°C for 1 h and then washed again. Immunodetection of blotting protein was done using chemiluminescence detection kit. The density of the protein bands was calculated as a ratio to β-actin.
Histopathological examination
Colon tissues were fixed in 10% formalin buffer. Paraffin tissue blocks were prepared for sectioning at 5 μm thickness. Haematoxylin and eosin (H & E) stain was used for blind microscopic examination. 18 Microscopic changes (necrosis, inflammatory cells infiltration, haemorrhage, oedema and congestion of blood vessels) were graded from 0 to 3 as follows: 0: no changes, 1: mild changes, 2: moderate changes and 3: severe changes. 19
Statistical analysis
Statistical analysis was done using GraphPad Prism 6.0 software (GraphPad, San Diego, California, USA). Data were presented as mean ± SEM. Multiple comparisons between groups were assessed by one-way ANOVA with Tukey–Kramer test as multiple comparison post-ANOVA test at level of significance 95%.
Results
Effect of DIO and SSZ on colon mucosal damage and DAI
Administration of AA induced significant increase in DAI and colon mucosal damage (CMD) scores in rats compared to control. Oral administration of SSZ or DIO significantly reduced DAI and CMD scores compared to non-protected group, but the effect of SSZ was still significantly higher than DIO. Treatment of rats with different doses of DIO resulted in significant decrease in DAI and CMD scores according to gradual dose concentration (Table 1).
Effect of DIO and SSZ on CMD and DAI.a

DIO: diosmin; SSZ: sulphasalazine; CMD: colon mucosal damage; DAI: disease activity index; AA: acetic acid.
aData were expressed as mean ± SEM (n = 10).
bSignificant difference versus control at p < 0.05.
cSignificant difference versus AA at p < 0.05.
dSignificant difference versus SSZ at p < 0.05.
Effect of SSZ and DIO on TNF-α and COX-II levels and MPO activity
Regarding the control group, administration of AA was significantly elevating the levels of TNF-α and COX-II in model rats. Both SSZ and DIO were significantly attenuating the level of TNF-α and COX-II compared to non-treated rats. TNF-α and COX-II levels of SSZ-treated rats were still significantly lower than DIO-treated animals. Animals which were given various doses of DIO (25 and 50 mg) showed a significant reduction in TNF-α and COX-II levels depending on dose concentration. The activity of MPO was significantly higher in model group, but treatment of rats with SSZ or DIO resulted in a marked decrease in MPO activity compared to non-protected rats. Protection with two doses of DIO resulted significantly in gradual inhibition of MPO activity (Table 2).
Effect of SSZ and DIO on TNF-α and COX-II levels and MPO activity.a

SSZ: sulphasalazine; DIO: diosmin; TNF-α: tumour necrosis factor-α; COX-II: cyclooxygenase-2; MPO: myeloperoxidase; AA: acetic acid.
aData were expressed as mean ± SEM (n = 10).
bSignificant difference versus control at p < 0.05.
cSignificant difference versus AA at p < 0.05.
dSignificant difference versus SSZ at p < 0.05.
Effect of SSZ and DIO on MDA and GSH contents and caspase-3 expression
Administration of AA significantly increased MDA content compared to control animals. SSZ or DIO therapy resulted in significant decrease in MDA content with respect to non-treated rats. However, MDA content of SSZ-treated rats was significantly lower compared to DIO-treated rats. The gradual dose therapies of rats with DIO produced significant dose-dependent reduction in MDA contents (Figure 2). In the model group, there was a significant reduction in GSH content compared with normal rats. SSZ- or DIO-treated groups have significant improvement in GSH level compared to the model group. But GSH levels of SSZ-treated rats were still significantly higher compared to group protected with DIO. However, GSH contents in DIO-protected groups were enhanced gradually according to dose increase (Figure 3). In this model of UC, the expression of caspase-3 in UC group was significantly higher than normal group. Treatment of rats with SSZ or DIO resulted in significant reduction in caspase-3 expression compared to model rats. But there is no difference between the expression of caspase-3 in SSZ-treated group and that of DIO-treated groups (Figure 4).
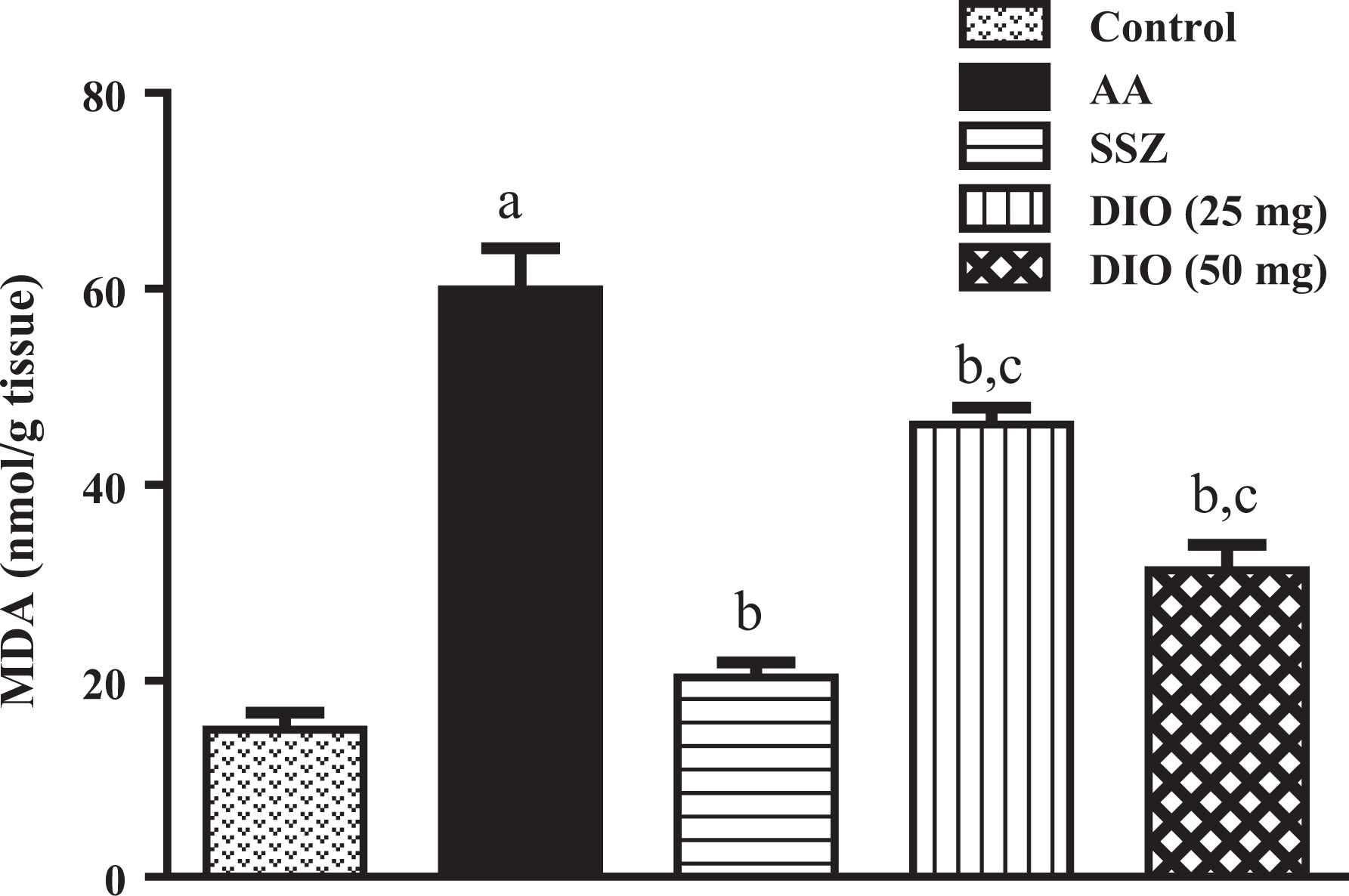
Effect of SSZ and DIO on colon MDA content. Data were expressed as mean ± SEM (n = 10). (a) Significant difference versus control at p < 0.05. (b) Significant difference versus AA at p < 0.05. (c) Significant difference versus SSZ at p < 0.05. SSZ: sulphasalazine; DIO: diosmin; MDA: malondialdehyde; AA: acetic acid.

Effect of SSZ and DIO on colon GSH content. Data were expressed as mean ± SEM (n = 10). (a) Significant difference versus control at p < 0.05. (b) Significant difference versus AA at p < 0.05. (c) Significant difference versus SSZ at p < 0.05. SSZ: sulphasalazine; DIO: diosmin; GSH: glutathione; AA: acetic acid.

Effect of SSZ and DIO on colon caspase-3 expression. Data were expressed as mean ± SEM (n = 10). (a) Significant difference versus control at p < 0.05. (b) Significant difference versus AA at p < 0.05. (c) Significant difference versus SSZ at p < 0.05. SSZ: sulphasalazine; DIO: diosmin; AA: acetic acid.
Histopathological examination of colon
Microscopically, colon of rat from control group showed normal histological layers from mucosa, submucosa and muscolosa (Figure 5(a)). Meanwhile, colon sections of rat from model group revealed UC described by severe coagulative necrosis of the mucosa (grade 3) and associated with severe (grade 3) haemorrhage, inflammatory cells infiltration in the mucosa and submucosa (Figure 5(b) and (c)). Most examined sections from SSZ-treated group revealed slight submucosal oedema (grade 1) was noticed in few sections (Figure 5(d)). However, colon of rat from (DIO 25 mg) group showed moderate changes (grade 2) as slight submucosal oedema with few inflammatory cells infiltration and congested blood vessel (Figure 5(e)). Moreover, colon sections from DIO (50 mg) treated group showed mild mucosal oedema with few inflammatory cells infiltration (Figure 5(f)).

Photomicrographs of sections of colons from rats: (a) control, ((b) and (c)) colitis model rat, (d) SSZ (100 mg/kg/day) treated rat, (e) DIO (25 mg/kg) treated rat and (f) DIO (50 mg/kg) treated rat. Sections stained with H & E, ×100. SSZ: sulphasalazine; DIO: diosmin; H & E: haematoxylin and eosin.
Discussion
UC is an inflammatory disease at the level of colonic mucosa and submucosa. Several mechanisms were discussed to understand the pathophysiology of UC such as inflammatory mediators, oxidative stress and apoptosis. In the present work, an experimental UC model was induced in male albino rats with rectal administration of AA. This model has been characterized biochemical and histological alterations that can be utilized in an evaluation of drugs efficacy against UC. Severity of UC was measured by evaluating CMD and DAI scores, whereas the degree of mucosal ulceration depends on CMD and bloody diarrhoea. Histologically, induction of UC was characterized by marked coagulative necrosis of the mucosa associated with haemorrhage and inflammatory cells infiltration. While CMD and bloody diarrhoea were decreased by the treatment of DIO that indicates its protective potential against UC. Moreover, the ability of DIO to increase ulceration healing and decrease bloody diarrhoea could relate to increasing venous tone, decreased capillary permeability and reduction of inflammation. 20
Also, treatment with DIO resulted in improvement in histopathological picture. Examined sections from colon of rats treated with DIO showed slight submucosal oedema with few inflammatory cells infiltration and congested blood vessel. Since UC is an inflammatory disease, an excessive production of pro-inflammatory mediators such as neutrophil infiltration and cytokines is considered as a cornerstone of inflammation during UC. 21 Among the family of cytokines, TNF-α counted as an inflammatory cytokine incorporated in inflammation, apoptosis, immunoregulation and stimulation of leukocyte migration. 22 TNF-α is a main cytokine involved in intestinal tract inflammation, and its expression plays a significant role in the development of UC. Therefore, its inhibition and regulation is an important target in regulation of UC development. 23 The results of the present work revealed that induction of experimental UC causes elevation in levels of TNF-α. In particular, TNF-α, which induced and secreted mainly by monocyte–macrophages, can stimulate reactive oxygen species (ROS) generation and aggravate inflammation cascade. 24 Moreover, TNF-α has involved in inflammation via inducing production of nuclear factor kappa B (NF-κB). 25 This study supposed that DIO inhibits colonic ulceration via suppression of many inflammatory mediators such as MPO, COX-II and TNF-α. Whereas the activity of TNF-α is under the control of NF-κB, 26 suppression of TNF-α is considered as a cornerstone for understanding anti-inflammatory mechanism of DIO.
COX pathway is the main pathway of inflammation raised from arachidonic acid metabolism that plays a critical role in intestinal inflammation. There are two types of COX: constitutive COX-I and inducible COX-II. COX-I is involved in production of protective prostaglandins, but COX-II has critical function in inflammation and may increase production of prostaglandins involved in UC. 27 Our study demonstrated that level of COX-II increased in AA model of UC; therefore, inhibition of COX-II activity could be valuable in the treatment of UC. Administration of DIO resulted in reduction of COX-II in comparison with non-treated rats. Many studies demonstrated that the intestinal anti-inflammatory activity of flavonoids was associated with the inhibition of the colonic expression of COX-II. 28,29 Meanwhile, neutrophil infiltration can be considered as one of the main pathological features of human UC; a neutrophil infiltration is a good marker for measuring of inflammation at injured sites. MPO is an enzyme presents highly in neutrophils and represents the degree of neutrophil infiltration. 30 Also, MPO stimulates production of hypochlorous acid, a strong reactive oxidant species, which mediates oxidative stress in UC. 31 Therefore, MPO level can be used as a marker of inflammation and oxidative stress. In the present study, induction of UC significantly increased MPO activities in colon tissues. However, administration of DIO decreased this activity indicating the anti-inflammatory and antioxidant effect of DIO in UC. Inhibition of neutrophil infiltration is evidenced by reduction of colonic MPO, thus contributing to the amelioration of the intestinal inflammation. 27 Suppression of MPO reduces neutrophil infiltration into the ulcerative colonic tissue, therefore providing the anti-inflammatory activity of DIO against intestinal inflammation and participates in alleviation of colonic ulcer caused by AA.
Production of free radicals with subsequent depletion of tissue antioxidants is still suggested aetiological factor in inflammatory bowel disease. Moreover, ROS play important role in tissue damage during inflammatory reactions and act as essential mediators of apoptosis induced by TNF-α. 32 As other studies, AA-induced UC is associated with generation of ROS. The overproduction of ROS exhausts the amount of GSH, so that it has an important role in the development of UC. GSH protects tissues from a free radicals generation that arises after induction of UC by administration of AA. 33,34 Hence, depletion of GSH contents indicates the progression of oxidative stress; treatment with DIO notably reduced the depletion of GSH and decreased the progression of oxidative stress. On the other hand, lipid peroxidation is an important factor for cellular injuries in UC. ROS attacks unsaturated fatty acids in lipids to start a free radical chain reaction and disturbance of membrane function. 35 In this study, DIO decreased the level of lipid peroxidation which indicates protective effect of DIO against UC by its antioxidant effect. Whereas treatment with DIO causes inhibition of MDA and augmentation of GSH contents, the prophylactic effect of DIO in UC is partially by reduction of oxidative stress. The antioxidant effect of DIO is related to scavenging of hydroxyl radicals and superoxide anions, therefore, decreased lipid peroxidation and enhanced the internal defence system to ameliorate oxidative tissue damage. 8
Apoptosis is a programmed cell death process involved in the normal turnover of damaged cells. Apoptosis also considered a component of a defence mechanism triggered during tissue damage and immune reactions caused by many pathological conditions. 36 Dysregulation of apoptosis recognized as pivotal key in pathogenesis of UC. Apoptosis initiation process is based on activation of a family of caspases that cleave certain proteins and initiate programmed cell death. 37 Caspases are expressed as inactive procaspases form in most cells, and when activated, they can start a series of other procaspases activation leading to degradation of intracellular proteins to drive the cells towards rapid cell death programme. 36 Beside the initiator caspases, another group of executioner caspases, which includes caspases-3, is responsible for cell death during apoptosis. 37 In the present work, administration of AA resulted in an increase in expression of caspase-3, which is specified as an apoptotic marker, and predicted the role of apoptosis in pathogenesis of UC. Results of our study explained the effect of DIO on apoptotic state in UC. DIO treatment produced a reduction in expression of caspase-3, revealing that DIO suppresses colonic ulceration through inhibition of apoptosis. This result is agreed with other studies that evaluate the anti-apoptotic effect of DIO in experimental diseases models. 10,38 After administration for long period, the most available therapeutic agents for UC such as glucocorticosteroids and 5-aminosalicylic acid have side effects. Therefore, the development of additional safe and efficient drug for UC such as DIO could reduce side effects of anti-inflammatory drugs.
Since ROS and pro-inflammatory mediators play important role in induction of apoptosis, 39 the potential effect of DIO in suppression of apoptosis could be related to a reduction of lipid peroxidation and inflammation.
Conclusion
In conclusion, this study found that DIO has a good activity against development of UC. Therefore, using DIO could be a useful strategy for preventing progression of UC and may be one among the drugs used for UC. The efficacy of DIO in treatment of UC depends on its antioxidant, anti-inflammatory and anti-apoptotic actions.
Footnotes
Acknowledgement
The authors are extremely thankful to Dr Kawkab A Ahmed, professor of pathology, Faculty of Veterinary Medicine, Cairo University, for his kind support in performing the histopathological studies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
