Abstract
Background
Although ionizing radiation (IR) has been of immense value to human life due to its involvement in several fields, it doesn’t eliminate that exposure to IR results in an array of biological consequences, including oxidative stress, inflammation, and death. Thus, this study aimed to explore the curative effect of trans-Resveratrol (t-Res) on hepatic and renal injury in a rat model exposed to single and fractionated doses of γ-rays.
Methods
Rats exposed to a single dose of IR (6 Gy, as an acute effect) or a fractionated dose of IR (2 Gy/time/3 days, day after day; to imitate a chronic impact) were treated with t-Res. Then, the radio-protective effect of t-Res was investigated via biochemical and histological estimations in the liver and kidney of rats in the different groups.
Results
The data displayed a significant amelioration in biochemical and histological indices in the liver and kidney of rats exposed to IR doses and treated with t-Res. Particularly, t-Res reduced the oxidative stress milieu through decreasing HIF-1α, ROS, and MDA levels associated with increased CAT activity and Nrf-2 gene expression. Also, t-Res improved the inflammatory status via a decrease in TNF-α, NF-κB, SOCS-3, and HSP-70 genes expression linked with elevations in SIRT-1 and P53 genes expression.
Conclusion
It could be concluded that t-Res had hepatoprotective and renoprotective effects against the deleterious consequences of γ-rays exposure due to its antioxidant and anti-inflammatory properties.
Introduction
Undoubtedly, ionizing radiation (IR) has been of immense value to human life due to its involvement in several fields such as medical (diagnosis and therapy, especially treatment of cancers), agricultural, and industrial. 1 Exposure to IR results in an array of biological consequences, including oxidative stress, inflammation, carcinogenesis, and death. 2 It has been found that even sub-lethal doses of IR such as 1–4 Gy can induce short-term as well as long-term effects on healthy tissues. 1 The hazardous effect of radiation occurs through the radiolysis of water, the main component of cells and tissues ∼80%, which produce reactive oxygen species (ROS) and reactive nitrogen species (RNS). Those reactive species cause oxidative damage to macromolecules (proteins, lipids, and DNA) and activate signal transduction pathways and transcription factors. 3 Whereas, during IR exposure, hypoxia and ROS are induced.2,4 Subsequently, this induction leads to a dysregulation in the cellular antioxidant defence mechanisms and SIRT-1 expression (that regulates many cellular processes).5,6
The second-largest organ, the liver, is necessary for metabolic processes, nutrient storage, and detoxification. It also maintains homeostasis by subjecting itself to a number of risk factors, including alcohol, drugs, environmental toxins, and radiation, which can cause oxidative stress by producing free radicals, which in turn leads to a variety of serious liver diseases. 7 Also, liver is a pivotal player for immunologic and inflammatory responses. 8
The kidneys are essential organs to regulate the organisms’ fluids, electrolytes, acid-base metabolism, excrete waste metabolites, modulate the blood pressure, and activate vitamin D. Following radiation exposure, it was found that radiation induced kidney dysfunction that leads to hypertension, anaemia, osteodystrophy, toxic waste metabolites accumulate, cause uraemia and ultimately chronic renal failure. 9
However, in order to capitalize on the beneficial effects of IR, it is mandatory to prevent or reduce these side effects to the barest minimum, as, for example, during radiotherapy, the surrounding healthy tissues are at risk of being exposed, which could lead to future risk of secondary cancers. 1 Therefore, radiation countermeasure agents must reduce these hazard effects of IR. IR countermeasure agents are classified into three types based on how they are administered: (1) radioprotectors, which are used before IR exposure to protect cells and tissues from damage; (2) radiomitigators, which are applied soon after IR exposure to repair tissues before the symptoms appear; and (3) therapeutic agents, which are administered after IR exposure to improve healing of injuries through tissue regeneration. These agents act via diverse mechanisms involving scavenging of free radicals and RO/NS, improving DNA repair process, synchronizing of cells, enhancing antioxidant and redox sensitive genes, modulating cytokines and growth factors, inhibiting apoptosis, and tissue regeneration. 10 Resveratrol (RSV) (3, 4, 5-trihydroxystilbene) is a polyphenol stilbene compound that is found naturally in red wine and in skin of red fruits and vegetables such as cabbages, berries, and peanuts. 11 It has anti-inflammatory, 12 anti-oxidant, 13 and anti-carcinogenic proprieties. 14 Moreover, resveratrol is also the most widely investigated activator of SIRT-1 (Resveratrol, an agonist of SIRT1). 10
Based on the foregoing, the present study was designed to examine the modulatory effect of Sirtuin (ultra-pure trans-Resveratrol) in the liver and kidney of rats subjected to whole-body γ-irradiation, either as a single or fractionated dose of 6 Gy. This aim was achieved through monitoring: (1) the oxidative stress status indicated by hypoxia-induciblefactor-1 alpha (HIF-1α), lipid peroxidation (measured as malondialdehyde; MDA), the activity of catalase (CAT) and nuclear factor erythroid 2-related factor-2 (Nrf-2); (2) the pro-inflammatory mediators (Suppressor of cytokine signaling-3; SOCS-3, heat shock protein 70; HSP-70, Nuclear factor kappa B; NF-κB, Tumour necrosis factor-alpha; TNF-α) and anti-inflammatory mediators (P53 and SIRT-1); and (3) as well as the histological investigations of liver and kidney.
Materials and methods
Experimental design
In the present study, rats were randomly divided into six equal groups (6 rats/group) as follows: Group-1 (Control group): rats were neither treated nor irradiated, and only received saline intraperitoneally, Group-2 (t-Res): rats were treated with trans-Resveratrol at dose of 20 mg/kg (suspended in 0.5% sodium carboxy methyl cellulose in saline) intraperitoneally for 14 days 17 Group-3 (Rs-group): rats were exposed to a single dose of γ-radiation (6 Gy), Group-4 (Rs + t-Res): rats were exposed to γ-radiation (as described in group 3) and administered t-Res intraperitoneally; Group-5 (Rf): rats were exposed to fractionated doses of γ-radiation (2 Gy/fraction/3 days up to a cumulative dose of 6 Gy), and Group-6 (Rf + t-Res): rats were exposed to γ-radiation (as described in group 5) and treated with t-Res. The zero time of experiment started with γ-irradiation and terminated with the last dose of t-Res.
Collection of tissue samples for analysis
At the end of the experiment, rats were decapitated under gentle diethyl ether anaesthesia and then sacrificed. Through cardiac perforation, blood samples were drawn and then centrifuged for sera separation and biochemical assessments. The liver and kidney tissues were collected and prepared for the estimation of biochemical and histopathological examination. The remaining tissue samples were then homogenized in phosphate buffer (0.1 M, pH 7.4, ice cold) at ratio 1:10 times (w/v) and maintained for further biochemical analyses.
Biochemical assays
In the serum, liver, and kidney samples, the lipid peroxidation was measured in terms of MDA, which was measured according to the method of Yoshioka 18 through thiobarbituric acid assay to forming thiobarbituric acid reactive substances (TBARS) reading at 532 nm. Catalase (CAT) activity was estimated following the procedures described by Aebi. 19 Total protein content was determined according to the method described by Lowry et al. 20 Additionally, TNF-α was determined using ELISA kits obtained from Abcam (Cambridge, UK; CAT# ab46070). Whereas HIF-1α was estimated using rat specific antibody for HIF-1α obtained from MyBioSource (San Diego, CA, USA; CAT# MBS028091) according to the manufacturers’ instructions.
For liver function evaluation, the activities of aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were assayed in serum as described by Reitman and Frankel. 21 Also, urea and creatinine, for assessing kidney function, were measured according to the techniques of Fawcett and Soctt 22 and Bartles et al., 23 respectively.
Quantitative real-time polymerase chain reaction (qRT-PCR) expression analysis
RNA isolation and reverse transcription: RNA was extracted from liver and kidney tissue using the RNeasy plus mini kit (Qiagen, Venlo, Netherlands), according to the manufacturer’s instructions. Genomic DNA was eliminated by a DNase-on-column treatment supplied with the kit. The RNA concentration was determined spectrophotometrically at 260 nm using the Nano Drop ND-1000 spectrophotometer (Thermo Fisher scientific, Waltham, USA) and RNA purity was checked by means of the absorbance ratio at 260/280 nm. RNA integrity was assessed by electrophoresis on 2% agarose gels. RNA (1 μg) were used in the subsequent cDNA synthesis reaction, which was performed using the Reverse Transcription System (Promega, Leiden, Netherlands). Total RNA was incubated at 70°C for 10 min to prevent secondary structures. The RNA was supplemented with MgCl2 (25 mM), RTase buffer (10X), dNTP mixture (10 mM), oligod (t) primers, RNase inhibitor (20 U) and AMV reverse transcriptase (20 U/μl). This mixture was incubated at 42°C for 1 h.
Primers used for QRT-PCR.

Histopathological examination of hepatic and renal tissues
Tissue samples from the liver and kidney were fixed in 10% formalin saline for 24 h. The samples were then washed with tap water and subjected to dehydration using a sequential dilution of alcohol (ethyl, absolute and ethyl methyl). Subsequently, samples were cleared in xylene and in hot air at 56°C in an oven for 24 h and embedded in paraffin. The paraffin beeswax of tissue blocks was set and sectioned (at 4 μm thickness) using a slide microtome. Finally, on glass slides, the tissue sections were collected and de-paraffinized, then stained by haematoxylin and eosin (H&E) stain for routine examination using light microscopy according to the method of Banchroft and Stevens. 25
Statistical analysis
All statistical analysis was carried out using one-way analysis of variance (ANOVA) followed by Tukey-Kramer
Results
Impact of trans-resveratrol on the oxidative stress in γ-irradiated rats
In liver
The data illustrated in Figure 1 and Table 2 showed that there was no significant change in the levels of hepatic ROS, HIF-1α, and MDA as well as CAT activity along with the gene expression of Nrf-2 between the t-Res group and the control group. It was observed after exposure to single (Rs) and fractionated (Rf) doses of γ-radiation that the levels of ROS, HIF-1α and MDA were raised significantly ( Impact of trans-resveratrol on the oxidative stress status in liver and kidney tissue of γ-irradiated rats. Each value represents mean ± SEM ( Data summary represents the percent change in biochemical indices between groups. *denotes the percent change (%) vs. C group. **stands for the percent change (%) against Rs group. ***represents the percent change (%) compared to Rf group.

However, the administration of t-Res evidently blunted the effect of single and fractionated doses of γ-radiation exposure, as revealed by significantly lessening (
In kidney
The data exemplified in Figure 1 and Table 2 showed that the levels of HIF-1α, MDA, CAT activity, and gene expression of Nrf-2 were not significantly altered in kidney of the t-Res group when compared with normal rats. On the other hand, the Rs and Rf groups displayed a substantial increment (
In serum
In Figure 2 and Table 2, the levels of HIF-1α and MDA as well as CAT activity in showed non-significant changes in serum of the t-Res group when compared with control group. However, the exposure to γ-radiation according to the current protocol induced a significant disturbance ( Effect of trans-resveratrol on HIF-1α, ROS, MDA, CAT and TNF-α in serum of γ-irradiated rats. Each value represents mean ± SEM (
Influence of trans-resveratrol on inflammatory responses in γ-irradiated rats
In liver
When comparing the hepatic inflammatory markers in the t-Res group to the control group, the data represented in Figures 3 and 4 and Table 3 revealed no significant changes observed in the TNF-α levels and the gene expression of NF-κB, SOCS-3, HSP-70, SIRT-1, and p53. Contrarily, the data of Rs and Rf groups were showed a significant elevation ( Influence of trans-resveratrol on TNF-α level and the gene expression of NF-κB and SOCS-3 in liver and kidney tissue of γ-irradiated rats. Each value represents mean ± SEM ( Influence of trans-resveratrol on the gene expression of HSP-70, p53 and SIRT-1 in liver and kidney tissue of γ-irradiated rats. Each value represents mean ± SEM ( Data summary represents the percent change in inflammatory markers between groups. *denotes the percent change (%) vs. C group. **stands for the percent change (%) against Rs group. ***represents the percent change (%) compared to Rf group.

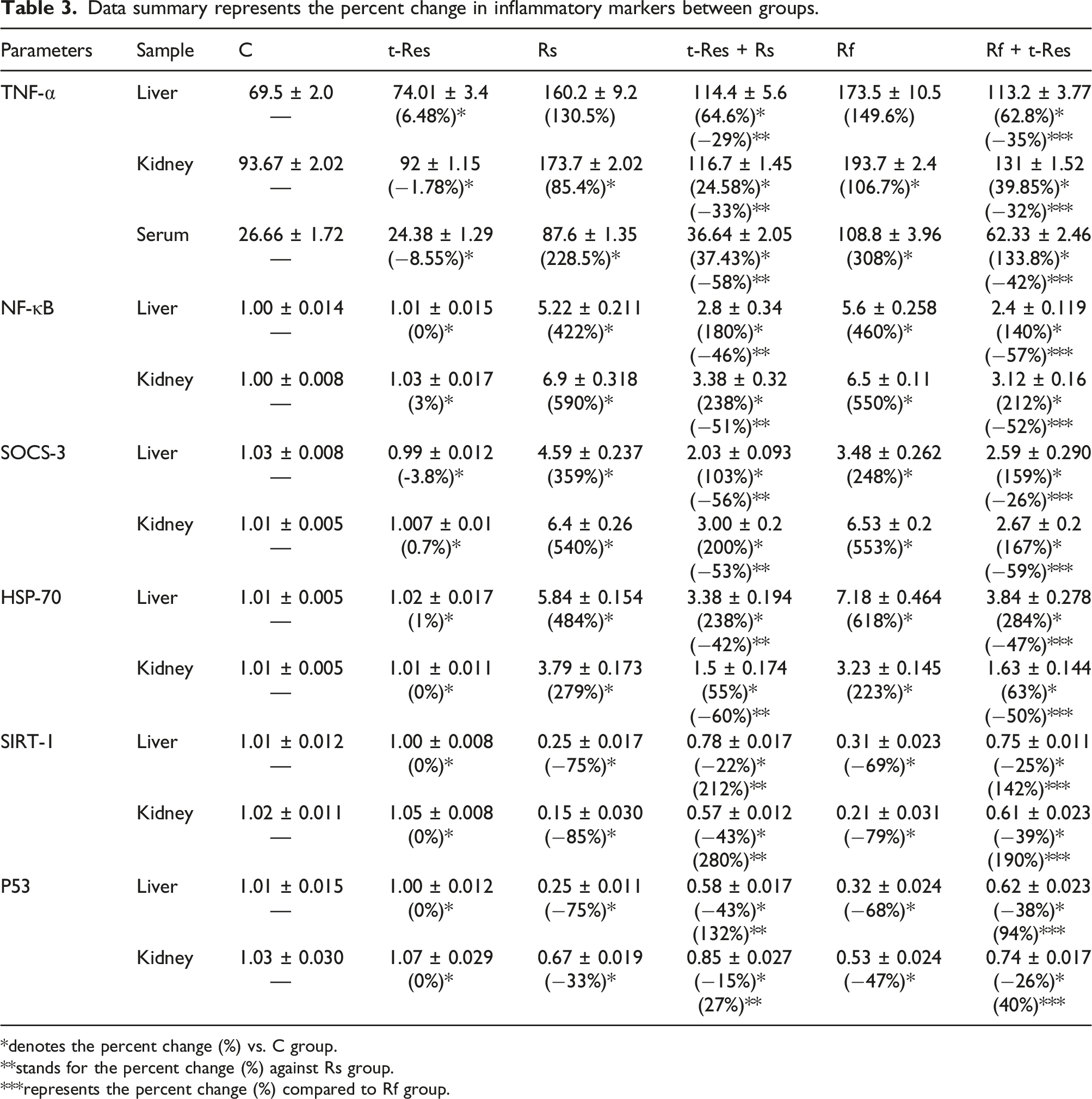
In kidney
When compared to the control group, the experimental data on the kidney inflammatory response (TNF-α level and gene expression of NF-κB, SOCS-3, HSP-70, p53, and SIRT-1) in t-Res group exhibited a non-significant difference (Figures 3 and 4, and Table 3).
In contrast, the data in Rs and Rf groups showed a significant augment (
When rats were administered t-Res after exposure to γ-radiation, the data in the Rs + t-Res and Rf + t-Res groups showed an obvious amelioration in all indicators of inflammatory response, as follows: a profound decrement (
In serum
The data obtained from the present study showed that the level of TNF-α in serum of the t-Res group was not significantly different from the control group (Figure 2 and Table 3). Oppositely, TNF-α levels in serum of irradiated groups, were significantly raised (
Effect of trans-resveratrol on the changes induced in the physiological function and histological architecture in liver and kidney of γ-irradiated rats
In serum
To evaluate the liver function, serum AST and ALT levels were monitored. There was no significant difference in the serum levels of ALT and AST enzymes of t-Res group when compared to the normal rats (Figure 5 and Table 2). Expectedly, in the rats exposed to either single (Rs) or fractionated (Rf) doses of γ-radiation, the activities of ALT and AST were boosted ( Histopathological examination and physiological function of liver and kidney in different animal groups. The tissues sections stained with hematoxylin and eosin, magnification ×400 (H&E × 400). Columns denoted with “a” significant from C, “b” significant from t-Res, “c” significant from Rs, “d” significant from Rs + t-Res, “e” significant from Rf and “f” significant from Rf + t-Res, at (
While the renal function was indicated through assessing urea and creatinine levels in serum. The data represented by Figure 5 and Table 2 showed that the concentration of urea and creatinine in the serum of t-Res group was not significantly changed (
Histological examination
The histopathological examination of the liver and kidney tissues of different animal groups was shown in Figure 5.
Discussion
Based on the data of the current study, exposure to IR led to a remarkable induction in the oxidative burden and disturbance in systemic inflammatory mediators. These findings were supported by the physiological liver and kidney function measurements and histopathological investigation, whereas the changes in the architecture and functions of these tissues that responded to the oxidative damages and inflammation were observed. These data are in agreement with the previous findings shown in several studies by El Bakary et al., 10 Moustafa and Thabet, 26 Galal et al., 27 and Qin et al. 6
These findings could be attributed to the overproduction of ROS, which induces an oxidative modification of biomolecules that is associated with the production of the pro-inflammatory mediators and initiates the inflammatory reactions through enhancing signalling cascades and activating transcription factors, subsequently causing an augmentation in the oxidative stress status and tissue damage. Whereas, during IR exposure, ROS has a negative effect on the cellular antioxidant defence mechanisms by decreasing the level of reduced glutathione and the activity of antioxidant enzymes such as catalase (CAT), superoxide dismutase (SOD), and glutathione peroxidase (GPx), which is associated with an elevation in harmful oxidative molecules such as malondialdehyde (MDA), a prominent lipid peroxidation marker. 2 Furthermore, as reported in Nakajima et al., 4 ROS is induced via hypoxia in the vascular endothelium after IR injury, which contributes to inflammation evoked in the irradiated liver. It was also realized that the hypoxia-induced upregulation of HIF-1 and NF-κB. 28 Accordingly, this might explain a case of hypoxia that was observed in the data of the current study.
Moreover, Wang et al. 29 revealed that IR disrupts the cellular junctions by activating the inflammatory NF-κB signalling pathway, which is manifested by enhanced oxidative stress and increased the levels of cytokines, including IL-6 and TNF-α. Subsequently, it was found that oxidative stress and inflammation down-regulate the expression of SIRT-1 through ROS production and NF-κB activation as reported by Kauppinen et al. 5 SIRT-1 is an NAD+-dependent class III protein deacetylase that regulates many cellular processes, including oxidative stress, metabolism, aging, and apoptosis. 6 However, it has been reported that the p53 and NF-κB responses to stress signals are diametrically opposed, with NF-κB activation generally suppressing p53 function and vice versa, suggesting that their interactions seem to be antagonistic.30,31 p53 is a tumour suppressing transcription factor and is expressed throughout the body and has a regulatory role in controlling the cell fate in response to stress. 32
Heat shock protein-70 (Hsp-70) is a molecular chaperone that is expressed in response to stress and functions in a diversity of cellular protein folding and remodelling processes. Its function almost at all stages of proteins, from synthesis to degradation, and is thus fundamental for maintaining protein homeostasis, with direct implications for human health. 33 In the current study, it could be explained that the elevation of HSP-70 expression is due to stress of IR exposure, as found by Multhoff et al 34 and Thabet et al. 35 Additionally, it was shown that the increase in HSP-70 expression in various cell types plays a crucial role in controlling NF-κB activity during inflammation.36,37 Besides that, Yong et al. 38 reported that the NF-κB activation was induced in HSP-70 over-expressing cells. Meanwhile, Sun et al. 39 discovered that elevated TNF-α level markedly enhanced SOCS-3 mRNA and protein expression and that inhibition of SOCS-3 expression is correlated with suppression of NF-κB expression. SOCS-3 is a regulator protein which can control both innate and adaptive immune system responses by regulating cytokine-mediated inflammatory signals. 40
In addition, it was shown that the elevation of NF-κB expression was associated with a reduction in Nrf-2 expression after IR exposure. 41 Nrf-2 is a transcription factor that serves as an important regulator in the antioxidant response pathways against oxidative stress in cells. 42 Several studies have demonstrated a wide variety of radio-protective roles for Nrf-2 in neutralizing ROS, repairing damaged DNA, regulating the clearance of free radicals, lipid homeostasis and cytokines expression, reducing apoptosis and promoting cell survival, indicating that Nrf-2 has become an attractive target in repairing the biological damage caused by IR.43–45
Natural radio-protectors have been recently attracting research to overcome damage occurring to normal tissue from IR-exposure due to their minimal toxicity and are easily available and inexpensive. 1 In the current study, the data showed that treatment with trans-resveratrol (t-Res; SIRTUIN) ameliorates the deleterious effects of γ-irradiation induced in liver and kidney upon biochemical and histological examination. These data are in accordance with the findings of Al-Baqami and Hamza, 46 who observed that t-Res has an antioxidant effect against oxidative damage induced by cadmium in hepatic tissue through increased activity of antioxidant enzymes and decreased MDA levels. This could be ascribed to t-Res properties as scavenger of hydroxyl, superoxide, and metal-induced radicals as well as enhancing the activities of antioxidant enzymes and the decline of lipid peroxidation (MDA levels) that have been reported in several studies.47,48 Furthermore, t-Res improved the regulation of signaling molecule Nrf-2, an important regulator in the antioxidant response pathway. 49 Besides, t-Res possesses anti-inflammatory effects, as shown in the study of Khalil et al. 50 via reducing the levels of pro-inflammatory mediators and greatly enhancing the expression level of SIRT-1 signaling pathway, which subsequently modulates p53 and NF-κB signals. 48 Also, t-Res significantly inhibited the SOCS-3. 51 Moreover, it was stated in the study of Xin et al. 52 that t-Res exerted a modulatory effect against hypoxia stress and its associated genes. According to the above, the radiomitigative potential of t-Res could explain its improving influence on the physiological function and histological examination of liver and kidney organs. This agreed with the findings of Al Dera 53 and Den Hartogh and Tsiani. 54
Taken together, it could be concluded that Sirtuin (ultra-pure trans-Resveratrol; t-Res) had a radio-protective effect via its antioxidant and anti-inflammatory effects on liver and kidney of rats subjected to a harmful whole body single or a fractionated radiation at a dose of 6 Gy. This is attributed to its ability to down-regulate the hypoxia factor (HIF-1α) and increase Nrf-2 gene expression, which is associated with a decline in lipid peroxidation (MDA) and enhancement in antioxidant enzyme (CAT). Furthermore, it has an anti-inflammatory potency on systemic inflammatory mediators (TNF-α, SOCS-3, HSP-70, NF-κB, p53, and SIRT-1), which supports its ability to curb inflammatory responses in vulnerable tissues.
Footnotes
Acknowledgements
Authors acknowledge Dr Sherein Saied Abdelgayed (Professor of Histopathology, Pathology Department, Faculty of Veterinary Medicine, Cairo University, Egypt) for accomplishing the Histopathological examination in the current study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
