Abstract
Despite being one of the most frequently used chemotherapy agents, cisplatin exhibits substantial hepatorenal injury by triggering oxidative stress, inflammation, and apoptosis pathways. The current investigation studied the possible protective effects of calcitriol on cisplatin-induced hepatorenal toxicity. Mice were divided randomly as follows: control group, calcitriol group (received calcitriol 5 µg/kg, p.o. for 14 days), cisplatin group (received a single i.p. injection of cisplatin 10 mg/kg on the 10th day), and calcitriol + cisplatin group (received calcitriol 5 µg/kg, p.o. for 14 days and cisplatin 10 mg/kg, i.p. on the 10th day). The possible interaction between calcitriol and cisplatin on cell viability was tested in HepG2 cells by MTT assay. Hepatorenal toxicity induced by cisplatin was reversed by calcitriol, as evidenced by improved histological examinations and liver and kidney function tests. In addition, calcitriol counteracted oxidative stress and enhanced Nrf2 and Mrp2 expression in the liver and kidney while suppressing levels of p38 MAPK in cisplatin-treated mice. Calcitriol also inhibited cisplatin-induced hepatic and renal inflammation, as determined by suppressing TNF-α and enhancing IL-10 levels. By downregulating caspase-3, calcitriol also promoted liver and kidney tissue survival in mice treated with cisplatin. Moreover, cisplatin’s cytotoxic effects were significantly potentiated when calcitriol was combined with cisplatin. The current study showed that calcitriol protects against cisplatin-induced hepatorenal injury by suppressing oxidative stress, inflammation, and apoptosis, which the Nrf2-Mrp2/p38 MAPK pathway might regulate.
Introduction
The vast prevalence of cancer and the adverse effects of chemotherapy pose major challenges for developing novel interventions to minimize the toxicities induced by chemotherapy. 1 As one of the most well-known chemotherapeutic agents, cisplatin has been widely used in treating many different types of malignancies, such as cell carcinoma and testicular and ovarian cancer. 2 There are, however, some adverse effects associated with cisplatin treatment that limit its effectiveness and result in the prolongation of treatment cycles. 3 Cisplatin accumulates most frequently in the kidneys and liver, causing nephrotoxicity and hepatotoxicity, respectively, in 36 and 28% of cancer patients. 4 Additionally, cisplatin has been shown to reduce patient survival and quality of life significantly. 3
The exact mechanism of cisplatin-induced hepatorenal toxicity is not fully understood. It is believed that increased levels of free radicals and inflammatory cytokines may contribute to oxidative stress and inflammation, which are key factors in cisplatin toxicity. 5 However, the exact mechanisms by which these effects are mediated are still unclear. 6 Several signaling pathways, mainly those controlled by the nuclear factor erythroid 2-related factor 2 (Nrf2) transcription factor, are activated to regulate oxidative and inflammatory responses via the activation of antioxidant response elements-dependent genes transcription. 7 Nrf2 transcription targets proteins involved in efflux transporters (such as multidrug resistance-associated proteins, Mrps), antioxidant enzymes, anti-inflammatory responses, and proteasomal degradation.8,9 The activation of the Nrf2-dependent cellular defense system has been suggested as a possible therapeutic target for cisplatin-induced oxidative damage. 10 p38 mitogen-activated protein kinase (MAPK) is another key mediator suggested to be crucial in mediating cisplatin-induced oxidative stress and inflammation. 11 It was reported that p38α MAPK signaling serves anti-inflammatory roles, contingent on the production of interleukin-10 (IL-10). 12 Furthermore, prior studies have suggested that p38 MAPK is involved in tumor necrosis factor-alpha (TNF-α) production and contributes to cisplatin-induced cell death.13,14 This suggests that p38 MAPK may be an attractive target in preventing cisplatin-induced toxicity.
Calcitriol, also known as 1,25-dihydroxy vitamin D3, is the active form of vitamin D that is produced in the kidneys as a result of vitamin D metabolism. 15 Calcitriol regulates calcium and phosphorus metabolism, which is essential to bone remodeling. 16 As calcitriol regulates the transcription of different genes through vitamin D receptors of most cells, it plays a crucial role in regulating cell metabolism, including antiproliferation, proapoptotic, anti-cell-migration, and antiangiogenic effects. 17 Interestingly, calcitriol showed antineoplastic activity in vitro and in vivo and modulated chemotherapeutic agents’ antitumor activities.18,19 Therefore, the current study investigated whether calcitriol can support cisplatin-based chemotherapy. Considering calcitriol’s antioxidant potential,20,21 we further investigated its possible protective effect on alleviating cisplatin-induced hepatorenal toxicity. Consequently, calcitriol was tested regarding modulation of oxidative stress, inflammation, and apoptosis in hepatic and renal tissues of mice challenged with cisplatin, focusing on the effect of calcitriol on Nrf2, Mrp2, and p38 MAPK as potential mechanisms.
Materials and methods
Chemicals and drugs
Cisplatin was purchased from Sigma-Aldrich (St. Louis, MO, USA). Calcitriol was procured from GlpBio Technology (Montclair, CA, USA). Calcitriol was dissolved in concentrated dimethyl sulfoxide (DMSO) and stored at –20°C, then diluted in normal saline as described by Tukaj et al. 22 The mouse IL-10 ELISA kit was purchased from Abcam (Cambridge, UK).
Animal model and experimental design
The present study used 24 male Swiss mice (30–35 g) from the National Research Center (Giza, Egypt). In a room with a temperature of 24 ± 2°C and an artificially lit cycle (12 h dark/light) set free of chemicals, animals were housed in cages and given access to a standard laboratory diet and tap water ad libitum. Per the National Institutes of Health Guide for the Care and Use of Laboratory Animals, all animal procedures were approved (Approval No: 320/4/2022) by the Faculty of Medicine Ethics Committee at Minia University, Egypt.
Animals were randomly divided into four groups of six mice each. The first group served as a control and received a normal saline solution as a vehicle. Animals in the second group received a daily p.o. administration of calcitriol (5 µg/kg) by gavage for 14 consecutive days. Animals in the third group received a single i.p. injection of cisplatin (10 mg/kg body weight) on the 10th day. In the fourth group, mice were given oral calcitriol (5 µg/kg/day) for 14 days, and on the 10th day, a single i.p. injection of cisplatin (10 mg/kg body weight) was administered. The dose of calcitriol and cisplatin treatments was selected according to previous studies.22,23
Sample preparation
Following the experiment, all animals were decapitated. Sera were separated from blood samples using centrifugation at 700 g for 15 min at 4°C, then stored at –20°C. The liver and kidney of each mouse were quickly excised and washed with saline. In 10% neutral-buffered formalin, liver tissue and the left kidney were fixed immediately for histological and immunohistochemical examinations. Other liver and kidney tissues were snap-frozen in liquid nitrogen and stored at –80°C for further analysis. The liver and kidney tissues were homogenized in 10% w/v ice-cold phosphate buffer (0.01 M, pH 7.4). Following centrifugation at 700 g for 20 min, the resulting supernatant was utilized to assess liver and kidney functions, perform western blotting analysis, measure IL-10 colorimetrically, and determine the expression of Nrf2 and p38 MAPK genes using real-time polymerase chain reaction (PCR).
Assessment of liver and kidney functions
The activities of serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were measured using kits purchased from Biodiagnostic (Giza, Egypt). The method used was based on the one described by Reitman and Frankel. 24 Whereas serum urea and creatinine levels were determined spectrophotometrically using methods described by Fawcett and Scott 25 and Schirmeister et al., 26 respectively, by kits that were purchased from Biodiagnostic.
Assessment of oxidative stress parameters in liver and kidney tissues
Liver and kidney tissue homogenates were utilized to assess total antioxidant capacity (TAC), malondialdehyde (MDA), and nitric oxide (NO). TAC was measured spectrophotometrically using commercial TAC kits (Biodiagnostic) at 505 nm. Based on Fenton’s reaction, the assay measures the suppression of sodium benzoate conversion to thiobarbituric acid via reactive oxygen species. 27
Lipid peroxidation in terms of MDA formation was estimated according to the method of Ruiz-Larrea et al. 28 As a result of MDA’s reaction with thiobarbituric acid, a pink-colored complex of thiobarbituric acid reactive substance is formed, which can be spectrophotometrically measured at 532 nm. NO was estimated spectrophotometrically using the method described previously. 29 In this method, nitrite concentration is measured as a proxy for NO production by adding a Griess reagent, which converts nitrite into a deep purple azo compound with a 540 nm absorbance.
Histological evaluation of hepatic and renal toxicities
Hepatic and renal tissues were dehydrated using increasing concentrations of alcohol, cleared with xylene, embedded in paraffin wax, and then sectioned to a thickness of 5 µm. Following staining with hematoxylin-eosin (H&E), the sections were examined under light microscopy (Olympus CX23LEDRFS1, Olympus, Tokyo, Japan) to evaluate histopathological changes.
In hepatic tissues, cytoplasmic vacuolization and sinusoidal congestion were assessed histochemically and scored from 0 to 4 as previously described: 30 0 = none, 1 = mild, 2 = moderate, 3 = marked, and 4 = severe to diffuse. As per Kocoglu et al. 31 and Ahmed et al., 32 histochemical analysis of renal tissues was performed as follows: score (−): assigned normal, score (+): between normal and mild levels, score (++): less than 25% of the total fields examined had histopathological changes. Score (+++): is moderate; less than 50% of the total fields examined show histopathological changes. Score (++++): indicates that less than 75% of the examined fields revealed histopathological changes.
Immunohistochemical analysis
A paraffin-embedded mice liver and kidney sections were deparaffinized and rehydrated. Endogenous peroxidase activity was blocked by pretreatment with 0.01% hydrogen peroxide. A citrate buffer solution of 0.01 M (pH 6) was used for 10 min to unmask the antigenic site. Following antigen retrieval, sections were incubated overnight with anti-Mrp2 monoclonal antibody (sc-59608, 1:200; Santa Cruz Biotechnology, Dallas, TX, USA), anti-TNF-α polyclonal antibody (A0277, 1:100; ABclonal, Port Talbot, UK), and anti-active caspase-3 monoclonal antibody (bsm-33199M, 1:200; Bioss Inc, Woburn, MA, USA). After 1 h of incubation in avidin-biotin solution, the sections were washed and incubated for 10 min in peroxidase substrate (DAB) solution. Lastly, sections were counterstained with hematoxylin, dehydrated in absolute alcohol, and cleared of xylene before mounting. 33
Real-time PCR
Following the manufacturer’s directions, TRI Reagent (Zymo Research, Irvine, CA, USA) was used to extract total RNA from liver and kidney homogenates. In the StepOne Real-Time PCR Detection System (Applied Biosystems, Foster City, CA, USA), a 250 ng RNA template was used for real-time PCR with the HERA SYBR Green qPCR kit (Willowfort, Birmingham, UK) and specific primers. As a reference gene, GAPDH was used to quantify the SYBR Green data. The primers of the Nrf2 gene were sense: 5′-CCCAGCACATCCAGACAGAC-3′ and anti-sense: 5′-TATCCAGGGCAAGCGACTC-3′, 34 and the primers of the p38 MAPK gene were sense: 5′-CGAAATGACCGGCTACGTGG-3′ and anti-sense: 5′-CACTTCATCGTAGGTCAGGC-3′, 35 and the primers of GAPDH were sense: 5′-GTCGGTGTGAACGGATTTG-3′ and anti-sense: 5′-CTTGCCGTGGGTAGAGTCAT-3′. 36 The relative expression levels of Nrf2 and p38 MAPK genes were calculated using VanGuilder et al. 37 All experimental samples were graphed as relative expressions compared with the control samples, where controls were set at 1 as a reference.
Cell viability assay
The HepG2 liver cancer cell line was used for the cell viability assay, as described previously. 38 The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) method was used to assess cell survival. HepG2 liver cancer cells were subcultured at 104 cells/well for 1 day in DMEM. The cells were treated with cisplatin alone at 1, 10, and 100 µM or combined with calcitriol at 1, 10, and 100 µM for another 24 h. 15 μL of MTT reagent (5 mg/mL PBS) was added to each well and incubated at 37°C for another 4 h, followed by the addition of DMSO (100 µL) to dissolve the formazan crystals. The absorbance at 540 nm is then recorded using a microplate reader.
Statistical analysis
In both hepatic and renal tissues, mean values and standard errors of the mean (SEM) were presented and tested for significance, using a one-way analysis of variance (ANOVA) followed by a Tukey’s post-analysis test. Additionally, two-way ANOVA followed by the Bonferroni test was used to compare the effect between hepatic and renal tissues in each treated group. Tests were considered significant at
Results
Effect on liver and kidney function parameters
Cisplatin significantly increased ALT and AST activities after a single i.p. injection compared to the control group. Moreover, urea (120%) and creatinine (234%) levels increased significantly compared to the control group. A 14-day treatment with calcitriol (5 µg/kg) prevented increased liver and kidney function indexes caused by cisplatin (Table 1).
Effect of calcitriol treatments on serum liver and kidney function parameters in cisplatin-treated mice.

Data are represented as mean ± SEM (
ALT: alanine aminotransferase; AST: aspartate aminotransferase.
Significant differences from control and CIS groups, respectively, at
Histopathological results
Normal control liver sections demonstrated regular hepatic cord architecture separated by blood sinusoids and normal central veins. Hepatocytes had acidophilic cytoplasm and rounded vesicular nuclei. The portal tracts containing the portal veins and bile ducts were seen. A significant increase in hepatic cytoplasmic vacuolation and sinusoidal congestion was observed in liver sections of mice treated with cisplatin, resulting in disorganization of the hepatic cords and presence of pyknotic cells, inflammatory cells’ infiltration, dilated congested central and portal veins. The adverse effects of cisplatin on hepatic tissue were markedly reduced by pretreatment with calcitriol as evidenced by apparently normal hepatocytes, central and portal veins with minimal congestion, slightly dilated blood sinusoids, cells with vacuolated cytoplasm, minimal inflammatory cells’ infiltration (Figure 1, Tables 2 and 3).
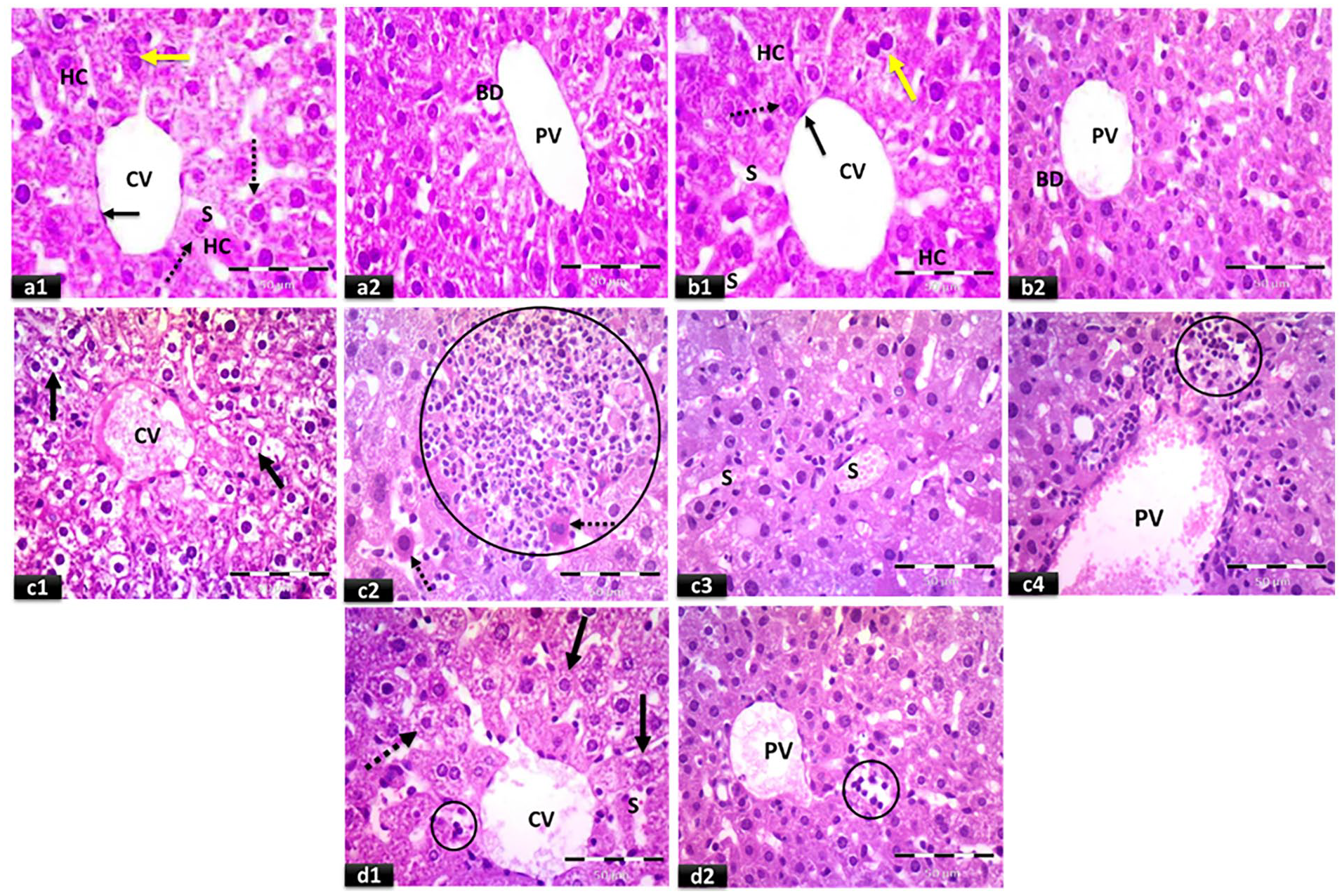
Representative photomicrographs of light microscopic examination of H&E-stained liver sections: (a) control group, (b) calcitriol group, (c) cisplatin group, and (d) calcitriol + cisplatin group (×400, scale bar = 50 μm). Normal central vein (CV) and strands of hepatocytes (HC) can be seen in (a1 & b1). The central vein is outlined by thin endothelium with flattened nuclei (black arrows). Hepatocytes with acidophilic cytoplasm and rounded vesicular nuclei (dotted arrows), binucleated cells (yellow arrows), and narrow blood sinusoids (S) are observed. (a2 & b2) showing the portal tract containing the portal vein (PV) and bile duct (BD) lined with single cuboidal cells. (c1–c4) showing congested central vein (CV), vacuolated hepatocytes (black arrows), cells with strongly acidophilic cytoplasm and darkly stained nuclei (dotted arrows), inflammatory cells’ infiltration (circles), dilated congested blood sinusoids (S), and dilated congested portal vein (PV). (d1 & d2) showing apparently normal central vein (CV) with minimal congestion, apparently normal hepatocytes with acidophilic cytoplasm and vesicular nuclei (black arrows), cells with vacuolated cytoplasm (dotted arrow), slightly dilated blood sinusoids (S), apparently minimal inflammatory cells’ infiltration (circles), and slightly congested portal vein (PV).
Histopathological scoring of hepatic cytoplasmic vacuolization.

0: none; 1: mild; 2: moderate; 3: marked; 4: severe to diffuse.
Significant differences from control and cisplatin groups, respectively, at
Histopathological scoring of hepatic sinusoidal congestion.

0: none; 1: mild; 2: moderate; 3: marked; 4: severe to diffuse.
Significant differences from control and cisplatin groups, respectively, at
Histological examination of kidney sections from normal control mice showed a regular structure of renal corpuscles and convoluted tubules with a relatively normal distinct lumen. The cisplatin-treated group showed a marked increase in interstitial inflammatory cells along with distorted renal corpuscles, as well as significant tubular necrosis manifested with tubular cell vacuolation and cells with ghosts of nuclei and disappearance of cytoplasm, and lumen distortion. Vascular and glomerular capillaries congestion and extravasated RBCs were also noticed. After calcitriol protection, cisplatin’s adverse effects on renal tissue were markedly reduced. However, hyaline casts in some tubular lumina and the presence of cells with deeply stained nuclei can be noticed (Figure 2, Table 4).

Representative photomicrographs of light microscopic examination of H&E-stained renal cortex sections: (a) control group, (b) calcitriol group, (c) cisplatin group, and (d) calcitriol + cisplatin group. (a1 & b1) Show normal organization of the renal cortex consisting of renal corpuscles (RC), proximal convoluted tubules (P), and distal convoluted tubules (D). (a2 & b2) reveal normal structure of renal corpuscles (RC), Bowman’s space (BW) lined with simple squamous epithelium (arrows). The convoluted tubules (P & D) have a relatively regular distinct lumen. (c1–c4) showing shrunken, distorted corpuscle (S) with a widening of Bowman’s space (dotted arrows), desquamation of some cells with cellular debris in the lumen of tubules (squares), vascular congestion (C), and congested glomerular capillaries (G). Notice vacuolation of some tubular cells (thin arrows) and other cells with ghosts of the nuclei and disappearance of cytoplasm (triangle). Notice also extravasated RBCs with inflammatory cells’ infiltration (circle). Some tubular cells with apoptotic figures (deeply acidophilic cytoplasm with dark pyknotic nuclei (thick arrows) were also noticed. (d1) shows restoration of the normal appearance of the renal cortex, renal corpuscles (RC), and tubules. Notice the hyaline casts in some tubular lumina (arrows). (d2) reveals the normal structure of renal corpuscles (RC), Bowman’s space (BW) with lining simple squamous epithelium (arrows), proximal convoluted tubules (P), and distal convoluted tubules (D). Notice cells with deeply stained nuclei (dotted arrow) and some hyaline casts in tubular lumen (green arrows). a1, b1, c1, d1 (×100, scale bar = 200 μm) and a2, b2, c2, c3, c4, d2 (×400, scale bar = 50 μm).
Histopathological scoring of kidney tissues.

Score (−): assigned normal; score (+): between normal and mild levels; score (++): less than 25% of the total fields examined had histopathological changes; score (+++): is moderate; less than 50% of the total fields examined show histopathological changes; score (++++): indicates that less than 75% of the examined fields revealed histopathological changes.
Effect on oxidative stress parameters
Data presented in Figure 3 showed that a single i.p. injection of cisplatin significantly increased the levels of MDA and NO in the hepatic (196 and 174%) and renal (177 and 159%) tissues, respectively, compared to control values. Serum TAC levels were significantly reduced, recording 19.6% below control values. In mice treated with calcitriol and cisplatin, the liver and kidney tissues showed comparable MDA and NO levels to control ones. Similarly, serum TAC level was restored to normal levels with concomitant calcitriol administration and cisplatin. The liver tissues of mice treated with cisplatin showed significantly higher MDA and NO levels than those recorded in their renal tissues. Furthermore, calcitriol protection of cisplatin intoxication resulted in a significantly lower NO level in renal tissues than in hepatic tissues.

Effect of calcitriol (CAL) on hepatic and renal levels of (a) malondialdehyde (MDA) and (b) nitric oxide (NO; as nitrate/nitrite) levels in cisplatin (CIS)-treated rats. (c) shows the effect of CAL on the serum levels of TAC in CIS-treated rats. Data are represented as mean ± SEM (
Effect on mRNA of Nrf2 and p38 MAPK
In liver and kidney tissues, gene expression of both Nrf2 and p38 MAPK exhibited a significant increase in the cisplatin group compared to the control group. Compared to the cisplatin group, hepatic and renal Nrf2 levels were significantly elevated by 1.73 and 1.56 folds, respectively, with calcitriol protection. On the other hand, the upregulation in p38 MAPK gene expression in both liver and kidney tissues of the cisplatin group was completely prevented by calcitriol coadministration. Of note, challenges with cisplatin caused a significant elevation of p38 MAPK gene expression in renal tissue compared to hepatic tissue (Figure 4).

Effect of calcitriol (CAL) on hepatic and renal levels of (a) nuclear factor erythroid 2-related factor 2 (Nrf2) mRNA and (b) p38 mitogen-activated protein kinase (MAPK) mRNA in cisplatin (CIS)-treated rats. Data are represented as mean ± SEM (
Effect on Mrp2 protein expression
Figure 5 shows that administration of calcitriol induced a significant increase in hepatic and renal Mrp2 expressions in the canalicular part of the hepatocytes and the brush border of the proximal convoluted tubules compared to the control group (9.85 ± 0.17 and 11.2 ± 0.13 vs 6.31 ± 0.09 and 7.8 ± 0.11 and). Similarly, cisplatin administration significantly increased Mrp2 expression in both liver and kidney tissues compared to the control group. Pretreatment with calcitriol caused a significant increase in Mrp2 expression by 96.8 and 63.2% in both hepatic and renal tissues compared to the cisplatin group. Interestingly, the recorded increase in Mrp2 expression with calcitriol protection was significant in renal tissues compared to hepatic tissues.

Panel I: Representative immunohistochemical photomicrographs of liver (a–d) and renal cortex (e–h) stained for multidrug resistance-associated protein 2 (Mrp2) (×400, scale bar = 50 μm), where (a, e) control groups, (b, f) calcitriol (CAL) groups, (c, g) cisplatin (CIS) groups, and (d, h) CAL + CIS groups. For control groups, (a) shows positive expression in the membrane of hepatocytes facing the canaliculi (yellow arrow), while (e) shows positive expression in the apical brush border of proximal convoluted tubules (arrows). For CAL groups, (b) shows positive expression in hepatocyte membranes facing the canaliculi (yellow arrows) and positive cytoplasmic expression in some hepatocytes (dotted arrow), while (f) shows positive expression in the apical brush border of proximal convoluted tubules (arrow) and faint cytoplasmic expression in some tubular cells (dotted arrows). For CIS groups, (c) shows scattered cells with positive cytoplasmic expression (dotted arrow) and positive expression in hepatocyte membranes facing the canaliculi (yellow arrow), while (g) shows tubular cells with positive cytoplasmic expression (dotted arrows). For CIS + CAL groups, (d) shows many hepatocytes with strong positive cytoplasmic expression (dotted arrows), while (h) shows strong cytoplasmic expression in tubular cells (dotted arrows). Panel II: A semi-quantitative analysis of Mrp2 mean area fraction. Data are represented as mean ± SEM (
Effect on TNF-α and IL-10 levels
Figure 6 depicts a profound spike in hepatic and renal TNF-α protein expression (16 and 15 folds, respectively) in mice treated with cisplatin compared to the control group. Similarly, the hepatic and renal IL-10 levels were significantly elevated in the cisplatin group compared to the control group. Protection with calcitriol in cisplatin-challenged mice caused a dramatic depletion of TNF-α protein expression in both liver (37.5%) and kidney (42.8%) tissues compared to the cisplatin group. On the other hand, a significant elevation in hepatic and renal IL-10 levels was recorded in the calcitriol + cisplatin group compared to the cisplatin group. It has been observed that the protective effects of calcitriol on the proinflammatory marker TNF-α and the anti-inflammatory marker IL-10 were more significant in hepatic tissues than in renal tissues.
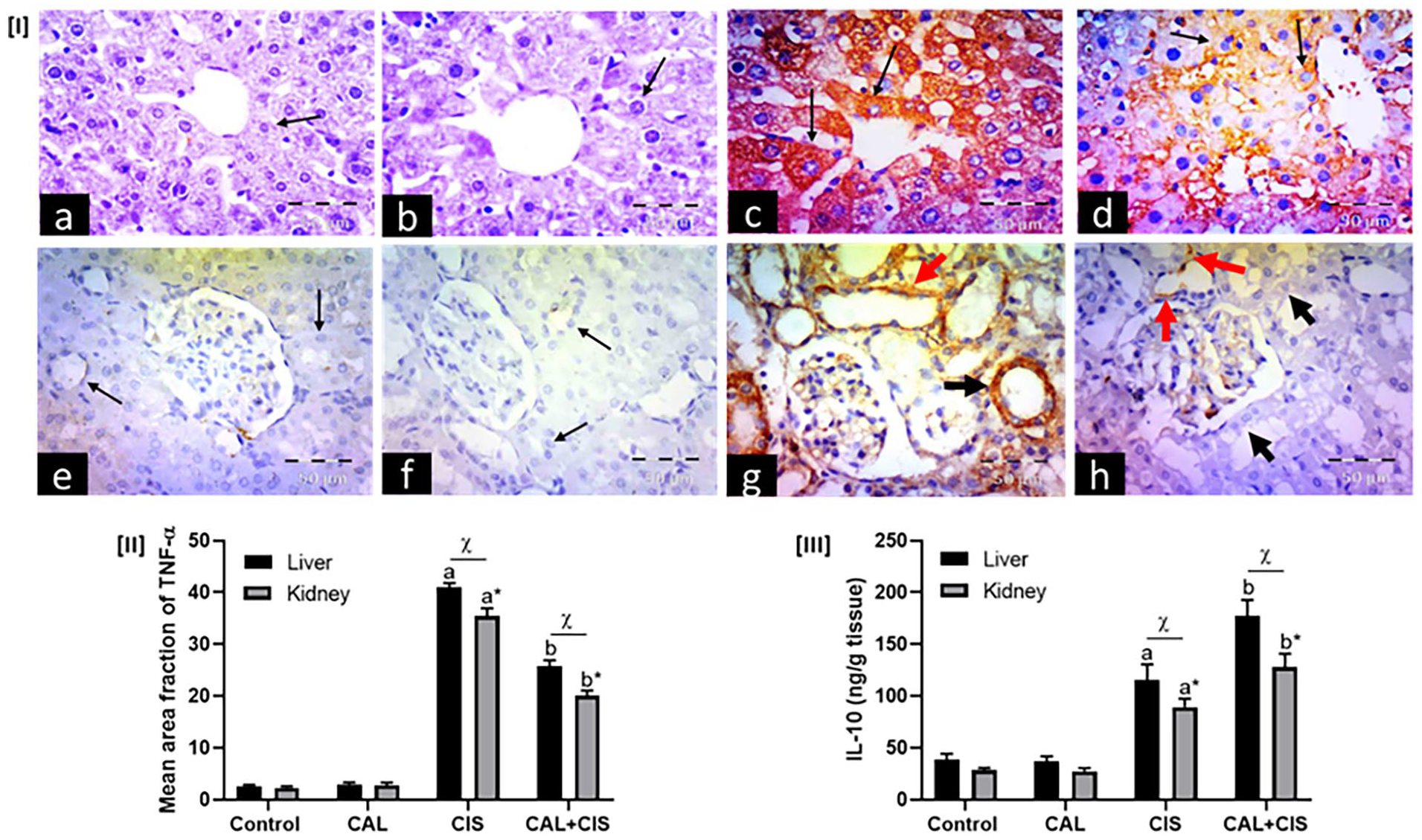
Panel I: Representative immunohistochemical photomicrographs of liver (a–d) and renal cortex (e–h) stained for tumor necrosis factor-alpha (TNF-α) (×400, scale bar = 50 μm), where (a, e) control groups, (b, f) calcitriol (CAL) groups, (c, g) cisplatin (CIS) groups, and (d, h) CAL + CIS groups. (a, b) show negative expression in hepatocytes (arrows). (c) shows many hepatocytes with strong positive cytoplasmic expression (arrows). (d) shows scattered cells with faint positive cytoplasmic expression (arrows). On the other hand, (e, f) show negative expression in the tubular cells (arrows). (g) shows strong cytoplasmic expression in proximal convoluted tubules (black arrow) and distal convoluted tubules (red arrow). (h) shows scattered tubular cells with minimal positive cytoplasmic expression (red arrows), while many cells have negative expression (black arrows). Panel II: A semi-quantitative analysis of TNF-α mean area fraction. Panel III Effect of CAL on hepatic and renal IL-10 levels in CIS-treated rats. Data are represented as mean ± SEM (
Effect on caspase-3 protein expression
Single i.p. injection of cisplatin significantly increased hepatic and renal caspase-3 protein expression, recording 7.25 and 4.75 folds more than the control group. However, treatment with calcitriol significantly prevented the recorded elevation in both liver and kidney tissues compared to the cisplatin group (Figure 7).
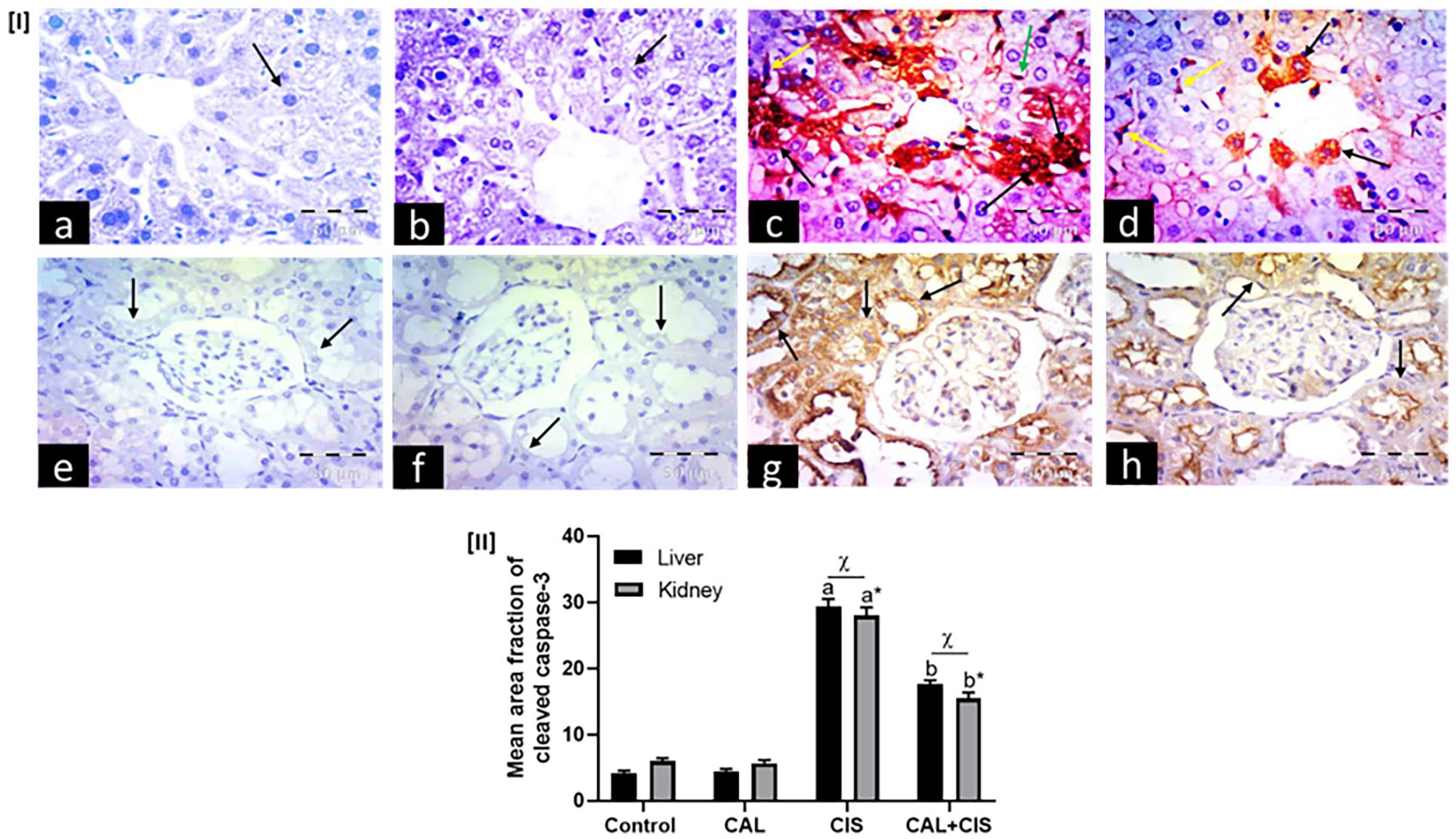
Panel I: Representative immunohistochemical photomicrographs of liver (a–d) and renal cortex (e–h) stained for cleaved caspase-3 (×400, scale bar = 50 μm), where (a, e) control groups, (b, f) calcitriol (CAL) groups, (c, g) cisplatin (CIS) groups, and (d, h) CAL + CIS groups. (a, b) show negative expression in hepatocytes (arrows), while (e, f) show negative expression in renal tubular cells (arrows). (c) shows abundant hepatocytes with positive nuclear and cytoplasmic expression (black arrows) and positive expression in Kupffer cells (yellow arrow) and endothelial cells (green arrow), while (g) reveals strong cytoplasmic expression in tubular cells (arrows). (d) shows scattered cells with positive cytoplasmic expression (black arrows) and positive expression in Kupffer cells (yellow arrows), while (h) reveals faint cytoplasmic expression in tubular cells, mostly in the apex (arrows). Panel II: A semi-quantitative analysis of caspase-3 mean area fraction. Data are represented as mean ± SEM (
Effect on cisplatin-induced cytotoxicity in vitro
HepG2 cells were cultured with cisplatin alone at 1, 10, and 100 µM or combined with calcitriol at 1, 10, and 100 µM (Figure 8). Cisplatin alone significantly reduced HepG2 survival in a concentration-dependent manner. Cisplatin’s cytotoxic effects were significantly potentiated when calcitriol was combined with cisplatin. The best cytotoxic effects were achieved in cells treated with calcitriol at 100 µM and cisplatin at 100 µM.

Effect of cisplatin (CIS; 1, 10, 100 µM) alone and in combinations with calcitriol (CAL; 1, 10, 100 µM) on the cell viability of HepG2 cells. Data are represented as mean ± SEM. “a,” “b,” “c,” and “d” are significant differences from control, 1, 10, and 100 μm CIS, respectively, at
Discussion
Cisplatin is a platinum-based therapy found to be very effective against a range of malignancies. 39 Cisplatin chemotherapy causes multiple organ toxicities, including hepatic and renal toxicities, which are among the most burdensome side effects. This may result in early discontinuation of the treatment. 40 In the current study, we examined calcitriol’s possible anti-inflammatory, antioxidant, and anti-apoptotic properties in relation to cisplatin’s deleterious effects on mice liver and kidney tissues. A single cisplatin dose caused significant liver and kidney injury, as indicated by histopathological changes in both hepatic and renal tissues, as well as an increase in serum ALT and AST activity. Serum concentrations of creatinine and urea were also elevated. These results were previously confirmed by Bishr et al. 11 and Hassan et al. 41
Several studies have demonstrated that cisplatin causes intracellular reactive oxygen and nitrogen species in hepatic and renal tissues.42,43 During oxidative stress, pro-oxidants dominate antioxidants, disrupting redox signaling and damaging cells. The present data showed that cisplatin increased the hepatic and renal MDA and NO levels along with decreased serum TAC. The current findings suggest that cisplatin-induced toxic effects on the liver and kidney are thought to be mediated through oxidative stress and lipid peroxidation, as confirmed by previous studies. 11 The current results showed that administration of calcitriol (5 µg/kg) for 14 consecutive days resulted in beneficial effects in protection against cisplatin hepatotoxicity, manifested by improved hepatic histopathological structure and liver function tests. Moreover, the findings showed nephroprotective effect by preventing elevated levels of urea and creatinine and renal histopathological alterations. Calcitriol’s protective action against cisplatin-induced hepatorenal toxicities may be attributed to its ability to suppress inflammatory cytokines 44 and the enhancement of the endogenous antioxidant pathways. 45 In the present study, calcitriol provides antioxidant capability in cisplatin-treated mice as it lowers lipid peroxidation secondary to its effect on TAC levels.
Through the induction of numerous antioxidant genes, Nrf2 serves as a stress-responsive signaling pathway for oxidative stress modulation. 46 Here, we demonstrated that calcitriol remarkably upregulates Nrf2 mRNA expression, which could upregulate serum antioxidant activity in cisplatin-treated mice. These findings suggested that calcitriol might extinguish cisplatin-induced oxidative stress via activation of Nrf2-antioxidant signaling. Our data were consistent with the results obtained by Vahidinia et al., 47 who reported Nrf2 as a cytoprotective signaling pathway that drives the antioxidant effects of calcitriol in ischemia-induced brain injury. In another independent study by Cui et al., 48 Nrf2 was reported to maintain redox balance, essential to calcitriol’s antioxidant and anti-apoptotic properties. The study showed that Nrf2 genetic knockout abolished the anti-apoptotic activity of calcitriol following traumatic brain injury. In agreement with that finding, the current study reported a downregulation of hepatic and renal caspase-3 protein expression in cisplatin-challenged mice treated with calcitriol, and that downregulation could be intricately connected with the Nrf-2 signaling pathway.
As an efflux drug transporter, Mrp2 transports cisplatin-glutathione conjugates, resulting in excreting of the insulting toxicant.49,50 Thus, upregulation of this transporter may enhance hepatic and renal elimination of subsequent exposure to cisplatin. Interestingly, the present study reported an upregulation of Mrp2 in liver and kidney tissues along with calcitriol treatment in cisplatin-challenged mice. The upregulation of hepatic and renal Mrp2 expression levels in the calcitriol + cisplatin group might be related to increased Nrf2 levels in the same group. According to various reports, Nrf2 is the most important transcription factor regulating Mrp2, 51 and Mrp2 was found to be one of the direct target genes of Nrf2, and thus, it could induced by Nrf2 activators. 52 Furthermore, it is noteworthy that cisplatin treatment alone resulted in the upregulation of Nrf2, IL-10, and Mrp2 compared to the control group. This is consistent with cisplatin’s known mechanism of inducing oxidative stress and inflammation, which can activate these pathways as a cellular defense response. 53 However, the degree of upregulation observed in the calcitriol-treated group was more pronounced, emphasizing the role of calcitriol in further augmenting these protective responses.
Cisplatin-induced hepatotoxicity and nephrotoxicity have been reported to be associated with the p38 MAPK signaling pathway, which mediates cellular inflammation and apoptosis.14,54 In agreement, our results revealed the ability of cisplatin to upregulate p38 MAPK mRNA expression in liver and kidney tissues. Our results also showed that cisplatin-treated mice exhibited elevated levels of hepatic and renal TNF-α and caspase-3 protein expressions and reduced IL-10 levels. In agreement with earlier studies, p38 MAPK activation is strongly linked to inflammation and apoptosis as significant biological outcomes. 55 Post-transcriptional regulation of inflammatory gene expression has also been associated with the p38 pathway. 56 Notably, in various disease models, p38α MAPK signaling has been shown to exhibit both pro-inflammatory and anti-inflammatory roles, relying on the production of TNF-α and IL-10, respectively.12,55 Furthermore, the involvement of p38 MAPK in apoptosis is proposed to be linked to the activation of signaling cascade. 55 In the current study, calcitriol pretreatment reversed cisplatin-induced changes in hepatic and renal TNF-α, IL-10, and caspase-3 levels. This suggests the modulation of the p38 MAPK signaling pathway as a possible mechanism for preventing cisplatin damage. This action may be attributed to the antioxidant effect exhibited by calcitriol in the animal model of cisplatin-induced hepatorenal toxicity. Various reports indicated that reactive oxygen species mediate activation of the p38 MAPK-related downstream proteins that contribute to apoptosis and necrosis.14,57 A significant body of literature suggests that p38 MAPK promotes apoptotic cell death. 58 Moreover, p38 MAPK is also involved in TNF-α-induced apoptosis. 59 In support, the results of the current study showed that calcitriol as an antioxidant significantly protects hepatic and renal tissues against cisplatin-mediated inflammation and apoptosis by abrogating oxidative stress and p38 MAPK activation.
In light of calcitriol’s cytoprotective effects against cisplatin-induced hepatorenal toxicity, which are partly through Mrp2 upregulation, we wondered if this upregulation could also disrupt the anticancer efficacy of cisplatin. As a proof of concept, we examined the viability of the HepG2 cells, a liver cancer cell line, in the absence and presence of calcitriol in conjunction with cisplatin. The results of the current study indicated that calcitriol potentiated cisplatin antiproliferative effects in a concentration-dependent manner. These findings align with those of Light et al., 60 who demonstrated that calcitriol may potentiate cisplatin cytotoxicity in vitro and in vivo by affecting cell cycle progression.
Study limitation
While the current study elucidates the protective effects of calcitriol against cisplatin-induced hepatorenal toxicity, several limitations warrant consideration. Firstly, the employment of a murine model, while beneficial, may not fully replicate human physiological responses, thereby constraining the extrapolation of findings to clinical contexts. Additionally, the relatively short duration of the study and the administration of a single dose of cisplatin might not accurately reflect the chronic exposure effects or the nuanced interactions between calcitriol and cisplatin over extended periods. While the histopathological and biochemical analyses provide significant insights, they may not encompass all underlying mechanisms of toxicity or protective action; thus, supplementary molecular and cellular investigations are recommended for a more comprehensive understanding. Lastly, the study observed a potentiation of cisplatin’s cytotoxic effects in conjunction with calcitriol, necessitating further scrutiny to preclude potential adverse outcomes.
Conclusion
The present work highlights the protective effects of calcitriol against cisplatin-induced hepatorenal toxicities in mice. We have demonstrated that calcitriol attenuated apoptosis and inflammatory response in cisplatin-treated mice through a mechanism involving upregulating Nrf2-Mrp2 expressions and the subsequent suppression of oxidative stress. The antioxidant capacity of calcitriol was associated with inhibition of p38 MAPK in both liver and kidney tissues of cisplatin-treated mice. These favorable actions suggest that calcitriol could be used as an adjunct therapy to mitigate cisplatin-induced hepatorenal toxicity. The translational implication is that calcitriol could be explored in clinical settings to reduce the side effects of cisplatin in patients undergoing chemotherapy, potentially improving their overall treatment tolerance and outcomes.
Footnotes
Acknowledgements
We acknowledge King Abdulaziz City of Science and Technology (KACST), Saudi Arabia, for their financial support.
Authors’ contributions
MAM, RA, and SAA designed the experimental project. MAM, RA, MFGI, HM, and SAA contributed to acquiring, analyzing, and interpreting the data. MAM, RA, MFGI, HM, and SAA wrote, reviewed, and edited the manuscript. MAM supervised and provided funding. Finally, all contributing authors have read and approved the submitted manuscript.
Data availability
Data are contained within the article or available upon reasonable request from the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by King Abdulaziz City of Science and Technology (KACST), Saudi Arabia (Grant No. 12-MED3156-06).
Ethical approval
All animal procedures were approved (Approval No: 320/4/2022) by the Faculty of Medicine Ethics Committee at Minia University, Egypt.
Animal welfare
The present study followed the National Institutes of Health Guide for the Care and Use of Laboratory Animals for humane animal treatment and complied with relevant legislation.
