Abstract
Curculigoside has been shown to decrease oxidative stress and inflammatory reactions in many disorders, but its effects during hepatic ischemia–reperfusion injury (IRI) remain unknown. This research aims to determine the protective role and the potential mechanism of action of curculigoside in hepatic IRI. Here, a well-established rat model of partial warm IRI was constructed; serum ALT/AST and H&E staining were employed to assay the extent of liver injury; the superoxide dismutase, malondialdehyde, IL-6, and TNF-α contents were determined using the corresponding kits; the apoptosis index was evaluated by TUNEL staining; and the expression of Nrf-2, HO-1, and apoptosis-associated proteins was detected by qRT–PCR and Western blotting. The results showed that curculigoside pretreatment effectively mitigated hepatic IRI, as demonstrated by decreases in the levels of serum aminotransferases, hepatocellular necrosis and apoptosis, oxidative stress markers, infiltration of inflammatory cells, and secretion of proinflammatory cytokines. Mechanistically, the expression of Nrf-2 and HO-1 was greatly suppressed by hepatic IRI and reactivated by curculigoside. Furthermore, cotreatment with ML-385, an inhibitor of Nrf-2, counteracted the protective effect of curculigoside against hepatic IRI. The results of our study show that curculigoside plays a protective role in hepatic IRI by inhibiting oxidative stress, inflammation, and apoptosis and that its effects may be associated with activation of the Nrf-2/HO-1 pathway.
Introduction
Liver ischemia–reperfusion injury (IRI) is an inevitable pathological process that is clinically associated with liver transplantation, liver resection, and hemorrhagic shock and may lead to impaired liver function and even to irreversible liver failure.1,2 In the past few decades, basic and clinical research has been conducted with the aim of developing new strategies to reduce liver IRI. Unfortunately, there are currently no clinically feasible drugs or methods available that protect the liver from the effects of IRI. Accumulating evidence suggests that hepatic ischemia and reperfusion elevate the levels of proinflammatory cytokines and the production of reactive oxygen species (ROS), both of which lead to liver damage. 3 Therefore, it appears that anti-inflammatory and antioxidative strategies might be effective in the treatment of hepatic IRI.
As a transcription factor, nuclear factor erythroid 2-related factor 2 (Nrf-2) is expressed in many organs, where it maintains cellular defense mechanisms through its antioxidant, anti-inflammatory, and cytoprotective nature.4-6 Under normal conditions, Nrf-2 is located in the cytoplasm and is integrated with Kelch-like epichlorohydrin-1 (Keap1). When activated by ischemia, Nrf-2 rapidly detaches from Keap1 and regulates the expression of downstream genes that encode antioxidant and anti-inflammatory enzymes, especially heme oxygenase-1 (HO-1), by binding to the antioxidant response element (ARE), thereby enhancing cell function and increasing cell survival.7,8 As a major phase II detoxifying enzyme and the main mechanism through which cells protect themselves against oxidative damage, HO-1 is regulated by Nrf-2, which protects the body from ROS damage. 9 Some studies have demonstrated a protective effect of the Nrf-2/HO-1 pathway in IRI models of organs such as the brain, heart, lung, and liver, and this pathway may represent part of the mechanism of action of many ischemic therapeutics and prodrugs, including natural products and synthesized chemicals.10-13
Curculigoside exhibits protective effects against hepatic IRI. (a) Chemical structure of curculigoside. (b) Serum AST and (c) ALT levels of each group of animals. (d) Representative images of H&E-stained liver tissues. Scale bar = 200 μm (upper panels) and 100 μm (lower panels). Black arrows indicate hepatocyte necrosis, blue arrows indicate hepatocyte vacuolar degeneration, and red arrows indicate congestion. (e) Degree of hepatic injury represented as Suzuki score. All data are presented as mean ± SD (
Materials and Methods
Animals
Male Sprague–Dawley rats (200–250 g) were purchased from Wan Qian Jia Xing Bio-Technology Co., Ltd. (Hubei, China). The animals were housed under a 12-h light–dark cycle with free access to food and water in a temperature- and humidity-controlled environment. The Animal Experiment Committee of the First Affiliated Hospital of Nanchang University reviewed and approved all experimental designs (Approval No: 02520129A), and all animal experiments were conducted in accordance with the National Institutes of Health Guide for Care and Use of Laboratory Animals.
Model establishment and experimental design
A previously described nonlethal model of segmental (70%) hepatic warm ischemia and reperfusion was used in this study. 22 Briefly, rats were anesthetized via intraperitoneal injection of sodium pentobarbital (40 mg/kg), followed by placement of the animals on an electric heating pad to maintain a body temperature of 36.5–37.5°C. A midline laparotomy was performed to expose the hepatic hilum, and hepatic ischemia was induced by clamping the hepatic artery, the portal veins, and the bile ducts that supply the three upper liver lobes with a microvascular clip. After 1 h of occlusion, the clip was loosened, and 6 h of reperfusion followed. The rats were randomly divided into five groups (8 rats per group) as follows: (I) the sham group underwent laparotomy and hepatic hilum mobilization without vascular occlusion; (II) the IRI group received treatment with vehicle (5% DMSO, MCE, HY-Y0320) via intraperitoneal injection 30 min before laparotomy and then with vascular occlusion; (III) the CUR group was treated with curculigoside (20 mg/kg, MCE, HY-N0705) via intravenous injection through the lateral tail vein 30 min prior to laparotomy and then with vascular occlusion; (IV) the ML-385 group was treated with ML-385 (an Nrf-2 inhibitor, 30 mg/kg, MCE, HY-100523) via intraperitoneal injection 30 min prior to laparotomy and then with vascular occlusion; and (V) the CUR+ML-385 group was treated with curculigoside and ML-385 as described above and then with vascular occlusion. Stock solutions of curculigoside and ML-385 in 100% DMSO were prepared and diluted in PBS to 5% DMSO before application. The dose and time of injection of curculigoside and ML-385 were chosen based on previous studies.20,23,24 None of the rats died during ischemia or reperfusion. After 6 h of reperfusion, the rats were anesthetized and sacrificed by cervical dislocation according to the AVMA Guidelines for the Euthanasia of Animals (2020 Edition), and blood and three upper liver lobes were collected and stored for further tests.
Serum biochemical analysis
An automatic biochemical analyzer (Rayto, Shenzhen, China) was used to measure the serum levels of alanine aminotransferase (ALT) and aspartate aminotransferase.
Cytokine analysis
ELISA kits provided by Shanghai Enzyme-linked Biotechnology Co., Ltd. (Shanghai, China) were employed to measure serum tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6) levels according to the instructions provided by the manufacturer of the kits.
Oxidative stress analysis
A colorimetric assay kit (Nanjing Jiancheng Biology Research Institute, Nanjing, China) was used to measure the levels of superoxide dismutase (SOD) and malondialdehyde (MDA) in liver tissue according to the instructions provided with the kit.
Histopathological analysis
Paraformaldehyde (4%) was used to fix liver samples for paraffin embedding. The embedded tissues were cut into 5-μm-thick sections that were then stained with hematoxylin and eosin (H&E). Suzuki’s score was adopted to evaluate the level of hepatic tissue damage. 25
Terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling staining
Cell apoptosis was determined in fixed liver tissues using a TUNEL assay with an In Situ Cell Death Detection Kit (Roche, Basel, Switzerland). Quantification was performed by measuring the number of immunofluorescently labeled TUNEL-positive cells per mm2 in five randomly selected fields using Image-Pro Plus 6.0 software (Media Cybernetics, Rockville, MD, USA).
Immunofluorescence staining
Immunofluorescence was performed as previously described. 26 Briefly, the sections were incubated with an antibody against MPO (1:200, Servicebio, Wuhan, China), washed, and incubated with Alexa Fluor 555-conjugated secondary antibody (1:200, Servicebio, Wuhan, China). Finally, the slides were counterstained with DAPI and analyzed by fluorescence microscopy (Olympus, Japan).
Quantitative real-time polymerase chain reaction (qRT–PCR)
The primer sequences for qRT–PCR.

Western blotting analysis
Total protein was extracted from liver tissues in RIPA lysis buffer (Thermo Scientific, Rockford, USA) containing protease inhibitors, and a Bradford assay was used to determine the protein concentration. Equal amounts of total protein were separated by 10–12% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE), followed by transfer to polyvinylidene difluoride membranes. After 2 h of blocking at room temperature with 5% skimmed milk powder in TBST, the membranes were incubated overnight with primary antibodies at 4°C. The following rabbit polyclonal antibodies were used: anti-Nrf-2 (1:1000), anti-HO-1 (1:500), anti-Bcl-2 (1:1000), anti-Bax (1:5000), anti-caspase-3 (1:500) and anti-GAPDH (1:5000). After three washes in TBST, the membranes were incubated for 2 h with a horseradish peroxidase-conjugated secondary antibody (1:5000) at room temperature. All the above antibodies were purchased from Proteintech Group (Manchester, UK). An ECL chemiluminescence system (Bio–Rad, USA) was used to visualize the protein expression levels, which were then measured by densitometry using ImageJ software (NIH, Bethesda, MD, USA).
Statistical analysis
All data are presented as the mean ± SD and were analyzed using SPSS version 21.0 (SPSS, Chicago, IL, USA). The unpaired t test was employed for the comparison of two groups, and one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test was used to compare multiple groups.
Results
Curculigoside mitigates hepatic IRI
To determine whether pretreatment with curculigoside protects against hepatic IRI, the serum levels of ALT and AST were measured. As shown in Figures 1(b) and (c), the IRI group showed significantly higher ALT and AST levels than the sham group. The animals that were pretreated with curculigoside showed a marked attenuation of the increase in liver enzymes that was observed in the animals in the IRI group. Consistent with the biochemical markers, H&E staining showed that compared with the control group, the livers of the rats in the IRI group displayed severe sinusoidal congestion related to marked hepatic vacuolation and degeneration and obvious increases in Suzuki scores. Furthermore, compared to the IRI group, curculigoside pretreatment obviously reduced hepatic injury and Suzuki score (Figures 1(d) and (e)). These results indicate that curculigoside protects against hepatic IRI.
Curculigoside alleviates hepatic oxidative stress and inflammatory response induced by IRI
During hepatic IRI, oxidative stress and the inflammatory response are two primary detrimental events that contribute to hepatocyte death. We sought to assess whether curculigoside affects this process. As shown in Figures 2(a) and (b), the SOD level in the liver tissue of the animals in the IRI group was greatly decreased, and the MDA concentration was greatly increased, compared to that in the sham group. The CUR group showed a lower level of SOD and a higher MDA concentration than the IRI group. In addition, the IRI group showed greatly increased levels of expression of TNF-α and IL-6 compared with the sham group, whereas both of these proinflammatory cytokines were expressed at lower levels in the CUR group than in the IRI group (Figures 2(c) and (d)). Hepatic neutrophil infiltration was detected by MPO staining assays. As shown in Figures 2(e) and (f), rats pretreated with curculigoside showed greatly reduced hepatic infiltration by neutrophils compared with IRI controls. These outcomes indicate that oxidative stress and the inflammatory response are alleviated by curculigoside during hepatic IRI. Curculigoside alleviates hepatic inflammatory responses and oxidative stress during hepatic IRI. (a–b) Concentration of SOD (a) and MDA (b) in liver tissue. (c–d) Levels of the proinflammatory factors TNF-α (c) and IL-6 (d) in serum. (e) Representative images showing immunofluorescence staining (MPO staining) in liver sections. Scale bar=100 μm. (f) Quantitation of MPO-positive cells. All data are presented as mean ± SD (
Curculigoside protects the liver from cell apoptosis induced by IRI
Apoptosis induces hepatocyte death as a vital mechanism during liver IRI. Hepatic apoptosis was examined by measuring the levels of TUNEL, cleaved caspase-3, Bax, and Bcl-2 to investigate whether curculigoside protects against IRI-induced hepatocyte apoptosis. As shown in Figures 3(a)-(d), hepatic IRI significantly increased the expression of the pro-cell death proteins cleaved caspase-3 and Bax and decreased the expression of the anti-apoptosis protein Bcl-2 in liver tissue compared to the sham group, and these effects were sharply reversed after curculigoside pretreatment. Moreover, the number of TUNEL-positive cells was greatly increased in the IRI group compared with the sham group; nevertheless, the number of TUNEL-positive cells (Figures 3(e) and (f)) was markedly reduced after curculigoside pretreatment. These findings indicate that curculigoside reduces cell apoptosis resulting from hepatic IRI. Curculigoside reduces cell apoptosis induced by hepatic IRI. (a) Western blot analysis of Bax, Bcl-2, caspase-3, and cleaved caspase-3 expression (GAPDH served as an internal control). (b–d) Quantification of Bax (b), Bcl-2 (c), and cleaved caspase-3 (d) expression. (e) Representative images of TUNEL staining in liver sections. Scale bar=100 μm. (f) Quantitation of TUNEL-positive cells. The experiments were repeated at least three times. All data are presented as mean ± SD (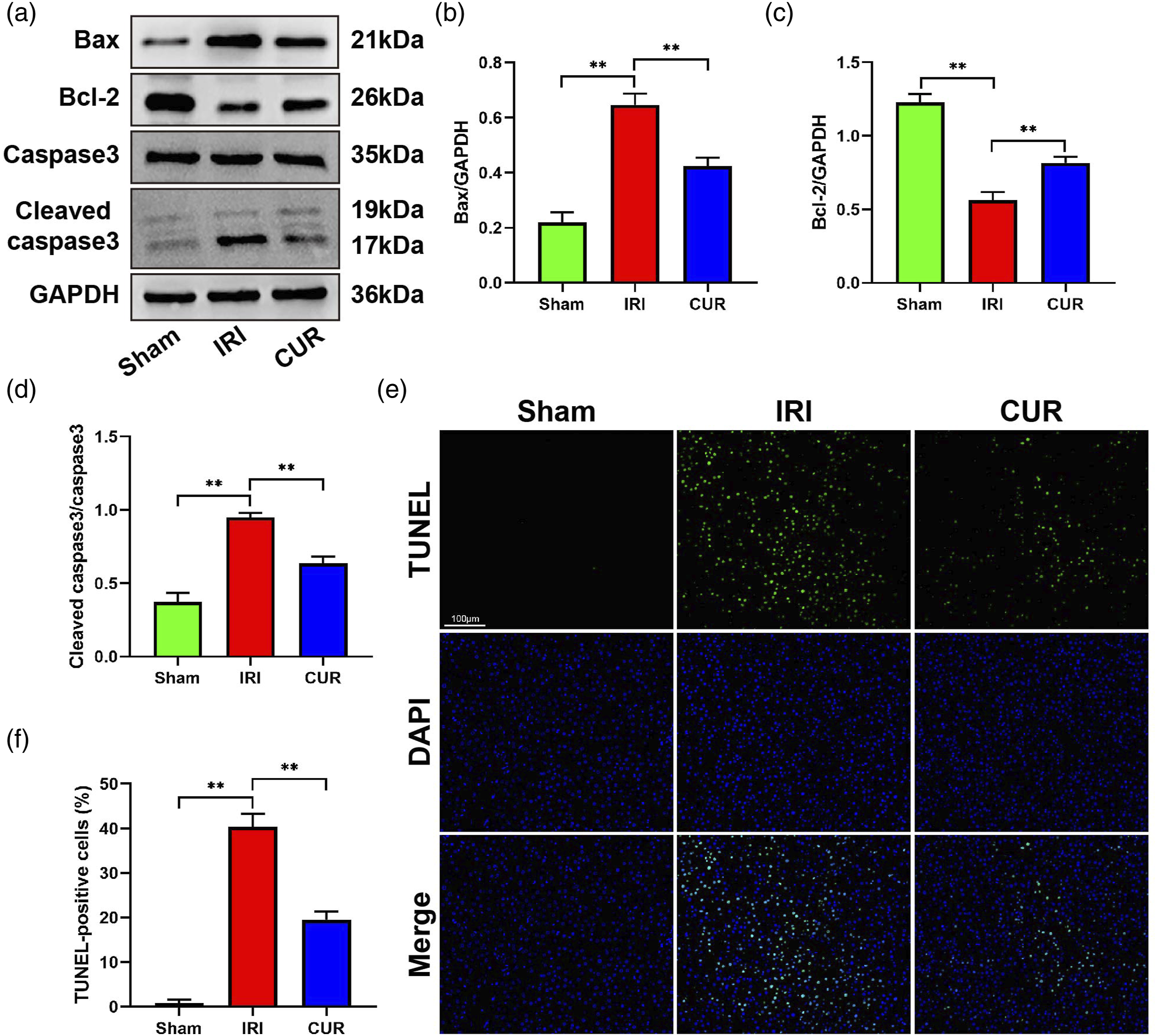
Curculigoside induces Nrf-2/HO-1 activation during hepatic IRI
The finding that curculigoside plays a significant protective role against hepatic IRI led us to investigate the mechanism underlying this effect. Past studies have shown that the Nrf-2/HO-1 signaling pathway exerts a significant regulatory effect on both inflammatory responses and cell apoptosis during hepatic IRI.
13
Therefore, we analyzed the activation of Nrf-2 and HO-1 at the protein and mRNA levels. As shown in Figures 4(a)-(e), Nrf-2 and HO-1 expression was decreased in liver tissues after hepatic IRI, and curculigoside pretreatment greatly increased the expression of Nrf-2 and HO-1. Moreover, ML-385, a specific Nrf-2 inhibitor, significantly decreased the expression of Nrf-2 and HO-1, and its inhibitory effect was partially neutralized by pretreatment with curculigoside. These results suggest that activation of the Nrf-2/HO-1 signaling pathway may be necessary for curculigoside to function. Curculigoside activates the Nrf-2/HO-1 signaling pathway during hepatic IRI. (a) Western blot analysis of Nrf-2 and HO-1 expression (GAPDH served as an internal control). (b–c) Quantification of Nrf-2 (b) and HO-1 (c) expression. (d–e) Relative mRNA expression of Nrf-2 (d) and HO-1 (e). The experiments were repeated at least three times. All data are presented as mean ± SD (
Curculigoside mitigates hepatic IRI through activation of the Nrf-2/HO-1 pathway
Based on the results presented above, it was speculated that curculigoside acts in a protective manner in hepatic IRI by activating the Nrf-2/HO-1 signaling pathway. To test this speculation, we blocked Nrf-2/HO-1 signaling using ML-385. Figures 5(a)-(f) show that liver injury was greatly aggravated in the CUR+ML-385 group compared with the CUR group, as evidenced by higher AST-, ALT-, and TUNEL-positive cell levels and Suzuki scores in the former. These results demonstrate that ML-385 pretreatment counteracts the protective effects of curculigoside against liver damage. Moreover, the curculigoside-induced increase in SOD levels and the decrease in MDA levels were reversed by ML-385 (Figures 5(g) and (h)), and inhibition of the release of TNF-α and IL-6 and neutrophil recruitment by curculigoside was nullified by ML-385 (Figures 5(i-l)). Collectively, these results indicate that curculigoside plays a protective role in hepatic IRI by activating the Nrf-2/HO-1 pathway. Curculigoside protects the liver from IRI by activating the Nrf-2/HO-1 pathway. (a–b) Serum AST (a) and ALT (b) levels in each group. (c) Typical images used in the histological analysis of H&E-stained liver tissues. Scale bar= 200 μm (upper panels) and 100 μm (lower panels). (d) The degree of hepatic injury was scored based on Suzuki scores. (e) TUNEL-positive cells were quantified. (f) Representative images showing TUNEL staining in liver sections. Scale bar=100 μm. (g–h) Concentrations of SOD (g) and MDA (h) in liver tissue. (i–j) Levels of the proinflammatory factors IL-6 (i) and TNF-α (j) in serum. (k) Representative images obtained by immunofluorescence staining (MPO staining) of liver sections. Scale bar= 100 μm. (L) Quantification of MPO-positive cells. All data are presented as mean ± SD (
Discussion
As a necessary complication during liver surgery, hepatic IRI greatly influences the prognosis of patients. Hepatic IRI induces the generation of ROS, the release of proinflammatory cytokines, and hepatocyte apoptosis, causing liver damage and organ dysfunction.
3
Research on mitigating hepatic IRI is an important topic in liver transplantation. However, the number of effective interventions that minimize this harm is still limited. The present study demonstrates that curculigoside exerts a protective effect on hepatic IRI by reducing liver necrosis, alleviating oxidative stress, and inhibiting inflammation and hepatocyte apoptosis and that the mechanism underlying these effects may be activation of the Nrf-2/HO-1 signaling pathway (Figure 6). Thus, curculigoside is expected to become a new intervention in clinical practice. Schematic diagram illustrating the protective role of curculigoside in hepatic IRI and its potential mechanism. Curculigoside attenuates hepatic IRI by inhibiting oxidative stress, inflammation, and apoptosis, possibly via the Nrf-2/HO-1 pathway.
During hepatic IRI, oxidative stress occurs as a result of a disturbed balance between the antioxidant defense system and ROS generation. 28 Lipid peroxidation and the production of extremely aggressive oxidants such as MDA are promoted by increased levels of ROS, finally resulting in hepatocyte damage and even death. 29 SOD is the major enzyme in the mitochondrial matrix that defends cells against damage caused by superoxide anions; by catalyzing the oxidation of ROS into inactive substances and oxygen, it protects cells against damage caused by IRI-induced ROS generation. 30 Protection against hepatic IRI by treatment with antioxidants might regulate the overall protective mechanism that acts to reduce the initiation and progression of hepatic IRI-induced effects. 29 Previous studies have shown that curculigoside acts as a powerful antioxidant through its ability to scavenge superoxide radicals in cell-free systems by decreasing the amount of LDH and MDA and by increasing nitrite levels and GSH-Px activity in H2O2-treated human umbilical vein endothelial cells (HUVECs). 31 Moreover, curculigoside may serve as an inhibitor of osteoclastogenesis by decreasing ROS production by reducing the activity of oxidase. 16 Importantly, curculigoside effectively induces skin flap angiogenesis and reduces IRI by alleviating oxidative stress. 32 Consistent with previous findings, the results of our experiments suggest that curculigoside pretreatment increases SOD activity and reduces MDA concentrations during hepatic IRI. Thus, curculigoside could alleviate oxidative stress-mediated injury during hepatic IRI.
The sterile inflammatory response is a significant component of the hepatic IRI process. 33 Although the mounting of a moderate inflammatory response can defend the body against alien invaders, an excessive inflammatory response may cause much more damage than the injury itself. The increased production of cytokines, including TNF-α and IL-6, is a major complication in the inflammatory cascade during hepatic IRI, and these cytokines are thought to be the main mediators of inflammation that causes liver injury by promoting the recruitment of neutrophils. 22 It has been shown that inhibition of the inflammatory response is effective in ameliorating hepatic IRI as a promising therapeutic method for protecting against hepatic IRI. 34 Studies have demonstrated that curculigoside markedly attenuates TNF-α, IL-1β, and IL-6 levels in rats with adjuvant arthritis and that it downregulates the expression of NF-κB/NLRP3. 35 Curculigoside protects against osteoporosis by inhibiting the expression of proinflammatory osteoclastogenic cytokines, including TNF-α, IL-1β, IL-6, and COX-2. 36 Moreover, curculigoside exerts a neuroprotective effect by abrogating HMGB1 release and NF-κB activation activated by TNF-α and ameliorates MCAO-induced cerebral IRI. 20 The current research demonstrates that pretreatment with curculigoside markedly decreases the release of TNF-α and IL-6 and the neutrophil recruitment induced by hepatic IRI, indicating that curculigoside attenuates the inflammatory responses that normally occur during hepatic IRI.
As a highly regulated process of programmed cell death, apoptosis is mediated by various proteins, including members of the caspase and Bcl-2 protein families. 37 During hepatic IRI, some liver parenchyma cells experience cell death in the form of apoptosis after ischemic insult, and pharmacological interventions that block apoptosis have been shown to be protective against reperfusion injury. 37 It has been reported that curculigoside protects neurons from N-methyl-D-aspartate-induced excitotoxicity by inhibiting cell death and apoptosis. 38 Moreover, curculigoside protects HUVECs from H2O2-induced apoptosis by decreasing caspase-3 activation. 31 Similarly, the results of the present work showed that treatment with curculigoside effectively reduced the expression of Bax, the activation of cleaved caspase-3, and the number of TUNEL-positive cells during hepatic IRI while increasing the expression of Bcl-2, indicating that curculigoside reduced cell apoptosis resulting from hepatic IRI.
To investigate the possible mechanisms underlying the protective effects of curculigoside against hepatic IRI, the expression levels of Nrf-2 and HO-1 in liver tissues were measured. It is generally known that the Nrf-2/HO-1 pathway enhances the cellular defense system that counteracts oxidative injury and inflammation. 13 There is increasing evidence that the Nrf-2/HO-1 pathway has a protective effect against hepatic IRI. Kudoh et al. 39 showed that inflammation, oxidative stress, and cell apoptosis during liver IRI were aggravated by depletion of Nrf-2 in mice, while activation of Nrf-2 greatly alleviated hepatic damage. Moreover, Chen et al. 24 reported that sulforaphane alleviates hepatic IRI by boosting the activation of Nrf-2/HO-1 signaling. Importantly, curculigoside mitigates oxidative stress and osteoclastogenesis by activating Nrf-2 and inhibiting the NF-κB pathway. 16 The current research shows that Nrf-2 and HO-1 were downregulated in hepatic IRI model rats and that pretreatment of the animals with curculigoside rescued the decreased levels of Nrf-2 and HO-1. To further verify the relationship between curculigoside and the Nrf-2/HO-1 signaling pathway, we used ML-385, an extensively applied inhibitor of Nrf-2, to inhibit the Nrf-2/HO-1 signaling pathway. We found that cotreatment with ML-385 abolished the protective role of curculigoside in hepatic IRI. Simultaneously, ML-385 was found to aggravate hepatic IRI; however, curculigoside counteracted the destructive effect of ML-385. These results suggest that curculigoside alleviates hepatic IRI by activating the Nrf-2/HO-1 signaling pathway.
In conclusion, this study demonstrates that curculigoside attenuates hepatic IRI by inhibiting oxidative stress, inflammation, and apoptosis through a mechanism that may be related to activation of the Nrf-2/HO-1 signaling pathway. This provides a novel potential approach to the treatment and prevention of hepatic IRI.
Footnotes
Acknowledgments
Thanks to Zhile Lv for her encouragement and support in life and study.
Authors’ contributions
Qi Xiao and Jiansheng Xiao are responsible for the conception and design of the research; Peng Du and Xingjian Zhang conducted the experiments, investigated data, and prepared the manuscript; Kaifeng Luo and Chengchao Fu produced the figures; Yong Li edited and revised the manuscript; the final version of the manuscript was approved by all authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants from Natural Science Foundation for Youth of Jiangxi Province (No. 20192BAB215012); Science and technology project of Jiangxi Provincial Health Commission (No. SKJP220203643); Science and Technology Research Project Fund of the Jiangxi Provincial Ministry of Education (No. GJJ18012, GJJ180014); and National Natural Science Foundation of China (No: 82060122).
