Abstract
Treatment strategies encompass synchronization of more than one therapy with specific dependence on zeroing side effects of natural products that might represent a niche in the continuous struggle against cancer. Thus, this study aimed at assessing the role of
Introduction
Mounting signs reveal an increasing need to devise more effective treatment methods for many types of cancer, and at the heart of them the breast cancer, which is one of the serious threats to life of many women different ages worldwide. Although the clinic pathological signs are comparable in patients of breast cancer, they are complying differently therapies and the net results after long courses of treatments are look dissimilar. Hence, breast cancer subtypes may differ in pathway activity, progression, and response to therapy.
1
Well consideration of the underlying factors and molecular mechanisms that affect response and resistance to entrant therapeutics may be supportive in forecasting clinical response and evolving more operative treatments for different subtypes of breast cancer.
2
A lot of natural compounds have a promising property in this task. We are interested here with
The anti-proliferated action of diverse candidate cancer therapeutics is often preceded through triggering apoptosis or affecting channels finally leading to cellular death. Sirtuin 1 (SIRT1), an enzyme located primarily in the cell nucleus that deacetylates transcription factors that contribute to cellular regulation is known to obstruct apoptosis and to endorse survival of various types of cells. 6 Variations in sirtuin expression are grave in some diseases, including metabolic syndrome, cardiovascular diseases, cancer, and neuro degeneration. 7 It is imaginable that SIRT1 may exert its primary regulatory effect on cell protection and endurance of mammalian cells through the interaction with apoptosis-inducible nuclear proteins. 8 Thus, it is conceivable that SIRT1 is a key regulator of cell defense and survival of mammalian cells through the interaction with apoptosis-inducible nuclear proteins.
Tzifi et al. 9 stated that the BCL2 gene family was found to be differentially expressed in many types of cancer. The BCL2 family of proteins is the main signs of apoptosis statute. BCL2 inhibits apoptosis through the protection of mitochondrial membrane integrity. 10 It prevents BAX/BAK oligomerization and prevents the release of several apoptogenic particles from the mitochondrion. Also, BCL2 could inhibit apoptosis via binding and inactivating BAX and other pro-apoptotic proteins. 11
BCL2 might also inactivate initiator caspases like caspase-2 and caspase nine and subsequently prevent activation of executing caspases (caspase three and 7) leading to inhibition of programed cell death process. 12 Moreover, BCL2 directly might blocks cytochrome c release and therefore prevents APAF-1 and caspase-9 activation. 13
As a fact, cancer cells could develop radio resistant state and may be from the first radio therapeutic dose. 14 Jameel et al. 15 reported that, the Over-expression of Insulin like growth factor-I receptor (IGF-IR), Human Epidermal Growth factor receptors (HERS) and Vascular Endothelial growth factor (VEGF) on the cell surface and raised concentration of Epidermal Growth factor in the extracellular fluid has been connected with radio resistance. Besides, specific genes such as p53, BRCA, Bcl-2 and chromosomal characteristics like telomere lengths have been notorious as inducing substantial roles in the radiation responsiveness of a cell. 16
Unfortunately, various therapies have essential limitations that lead to therapeutic resistance and disease recurrence, which ended in therapeutic failure. The recur disease is more aggressive and it readily metastasizes to detached organs and causes death.
17
These challenges encourage scientists working in the field of anticancer to struggle to infer novel treatment strategies including the combining of more than one therapeutic method and seeking for new drugs to have the ability to circumvent cancer resistant mechanisms. Accordingly, we are intended to examine the
Materials and methods
Materials
The root extract of
Cell lines and culture media
The breast cancer cell lines have been utilized in this study were obtained from the Cell Culture Department of VACSERA (Cairo, Egypt). The human breast cancer cells (MCF-7), as well as MDA-MB-231 cells, were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with penicillin-streptomycin (100 U/ml) and 10% fetal bovine serum (FBS) in a 5% CO2 humidified chamber at 37°C. Cells at 80% confluency were used for examining different treatments in our study.
Cell Viability Assay of withania somnifera (ashwagandha)
The cytotoxic profile of
Gamma irradiation procedures
137Cs source (Gamma-cell-40 Exactor; NCRRT, AEA, Cairo, Egypt) was applicated in the irradiation procedure. All irradiated MCF-7 and MDA-MB-231 cell groups were irradiated with γ-rays at 85% confluency (single exposure to 4 Gy) at a dose rate of 0.012 Gy/Sec. A Fricke reference standard dosimeter (ISO/ASTM E 51,026) was applied in all experiments to ensure dose and dose rate uniformity. 19
Cell culture models and study protocol
To determine the anti-proliferative and radio-sensitizing efficacy of
Cell cycle and apoptosis detection by flow cytometry
After 24 h of incubation, MCF-7 and MDA-MB-231 cells (3 × 105 cells/well) were harvested with trypsinization from all groups, either treated or untreated, and washed twice in ice-cold PBS and fixed with 70% ethanol at 4°C overnight. Cells were then washed in PBS, centrifuged, and stained with propidium iodide (PI) (CAT# ab139418) (50 μg/ml) for cell cycle analysis. Staining was assessed in a FACSCanto-II flow cytometer, followed by analysis using BD Accuri-C6 Plus software (Biosciences, CA, USA). 20
Determination of MDA, ROS, SOD, and CAT
The levels of MDA (lipid peroxidation products)
Determination of IL-10, IL-6, TNF-α and COX2 by ELISA technique
The level of (Interleukin-10) (ab185986), IL-6 (ab178013), TNF alpha (ab181421) and COX2 (ab267646) was estimated by ELISA kits according to the manufacturer’s instructions, for the enzyme-linked immunosorbent assays via ELISA microplate reader (DV990 BV 416; Gio.DE VITA and CO., Rome, Italy).
Determination of NF-ΚB, BCL-2, BAX, caspase and SIRT1 by real time PCR
RNA isolation and reverse transcription: RNA was extracted from breast cancer cells (MCF-7 and MDA-MB-231 cells) using the RNase plus mini kit (Qiagen, Venlo, The Netherlands), according to the manufacturer’s instructions. Genomic DNA was eliminated by a DNase-on-column treatment supplied with the kit. The RNA concentration was determined spectrophotometrically at 260 nm using the Nano Drop ND-1000 spectrophotometer (Thermo Fisher Scientific, Waltham, USA) and RNA purity were checked by means of the absorbance ratio at 260/280 nm. RNA integrity was assessed by formaldehyde agarose gel electrophoresis on 2% agarose gels. RNA (1 μg) was used in the subsequent cDNA synthesis reaction, which was performed using the Reverse Transcription System (Promega, Leiden, The Netherlands). Total RNA was incubated at 70°C for 10 min to prevent secondary structures. The RNA was supplemented with MgCl2 (25 mM), RTase buffer (10X), dNTP mixture (10 mM), oligod (t) primers, RNase inhibitor (20 U) and AMV reverse transcriptase (20 U/μl). This mixture was incubated at 42°C for 1 h.
Quantitative real time PCR
Primers used for QRT-PCR.

Statistical analysis
All experiments were performed at least in triplicate and the results were expressed as the mean ± standard error (SEM). The data were analyzed using ANOVA followed by Tukey multiple comparison post hoc tests. Statistical analyses were carried out using Prism, version 8 (GraphPad Software, La Jolla, CA). The a, b, c (
Results
One- Anti-Proliferation effect of withania somnifera (ashwagandha) on breast cancer cell line (MCF-7 and MDA-231)
For the assessment of the possible antitumor efficacy of withania somnifera on breast cancer cell line, the MCF-7 and MDA-231cell lines were subjected to increasing concentrations of withania somnifera to evaluate withania somnifera cytotoxic activity utilizing the MTT assay. One hundred percent viable breast cancer cells cultured in RPMI complete medium are regarded as a positive control. As shown in Figure 1 (A,B,C,D,E), inhibition in breast cancer cells was observed. Twenty-4 hours of MCF-7 and MDA-231cells treatment resulted in breast cancer cells proliferation inhibition in a dose dependent manner and the half maximal inhibitory concentration (IC50) was estimated to be approximately 3801.9 μg/ml and 4897.8 μg/ml respectively. Cytotoxicity and morphology effect of withania somnifera (ashwagandha) on breast cancer cell line (MCF-7 and MDA-231).
Cell cycle arrest and apoptotic cells formation
In the present study the cell cycle of MCF-7 and MDA-231 human breast cancer cell line dispersion was estimated by flow-cytometry. In control MDA or MCF7 breast cancer cells the plurality of MDA-MB-231 or MCF7 cells (51.91% or 56.24%, respectively) are present at G1 phase and by a lesser degree at the S phase (41.75 or 38.56, respectively). A small percentage of cells are found at the pre G1 and G2/M phases. Nevertheless, the flow cytometric cell cycle analysis in both breast cancer cell lines revealed that WS inhibits cell proliferation and exhibits a significant cytostatic increase at the G2/M phase and G1 with a significant diminution at the S phase for MDA cells and at Pre G1 phase for MCF7 cell line. Besides, the exposure of both types of cells to 4 Gy of gamma resulted in no changes in MDA cells distribution compared to the control although the MCF7 cells exhibited significant increases at the G2/M phase. The combination treatment of WS and R induced cell cycle arrest at preG1 and G2/M phases for MDA cells whenever, the MCF7 displayed significant increases at preG1 only when compared to control (Figure 2(a) and (b)). The flow cytometric analysis of cell apoptosis executed 24 h. After treatments (WS, R or WS + R) showed increased apoptotic cells percentage (counting early, late and necrotic cells) in the two types of breast cancer cell lines examined as compared to control. Figures (2(c) and (d) exhibited marked increases in the apoptotic cell percentage in WS + R cells even for MDA or MCF7 cell lines over that happened in WS or R groups. Effect of 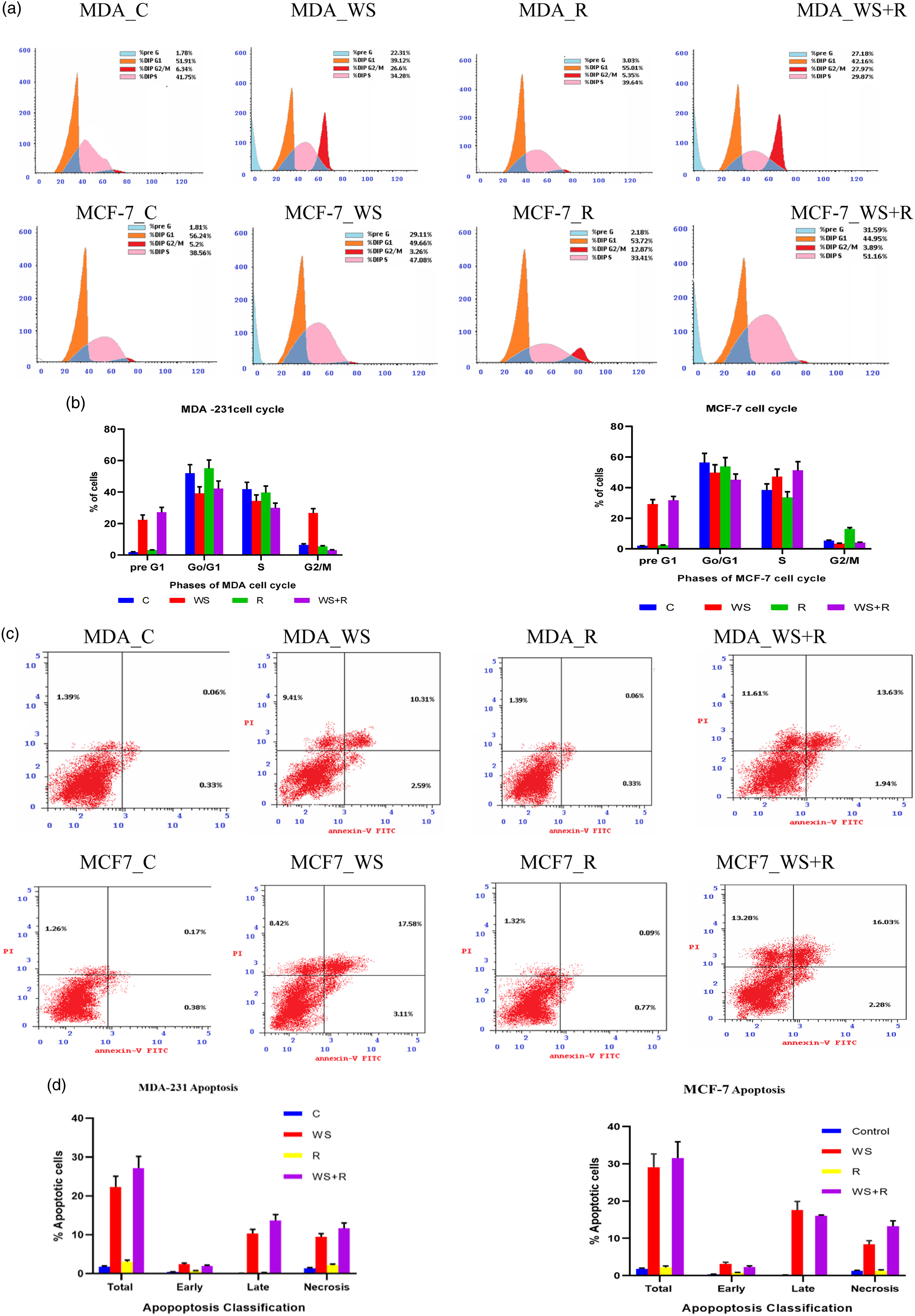
Each value represents Mean ± SEM (
Oxidative status
Statistical analysis of data from different groups of MCF-7 cells subjected to investigation in the present study revealed significant changes in oxidative status manifested by different degrees of changes in MDA and ROS concentrations and SOD and CAT activities among different cell groups. In WS-treated MCF-7 the data pointed out significant decreases ( Effect of WS and/or 4 Gy gamma irradiation on oxidative status in human MCF-7 or MDA-231.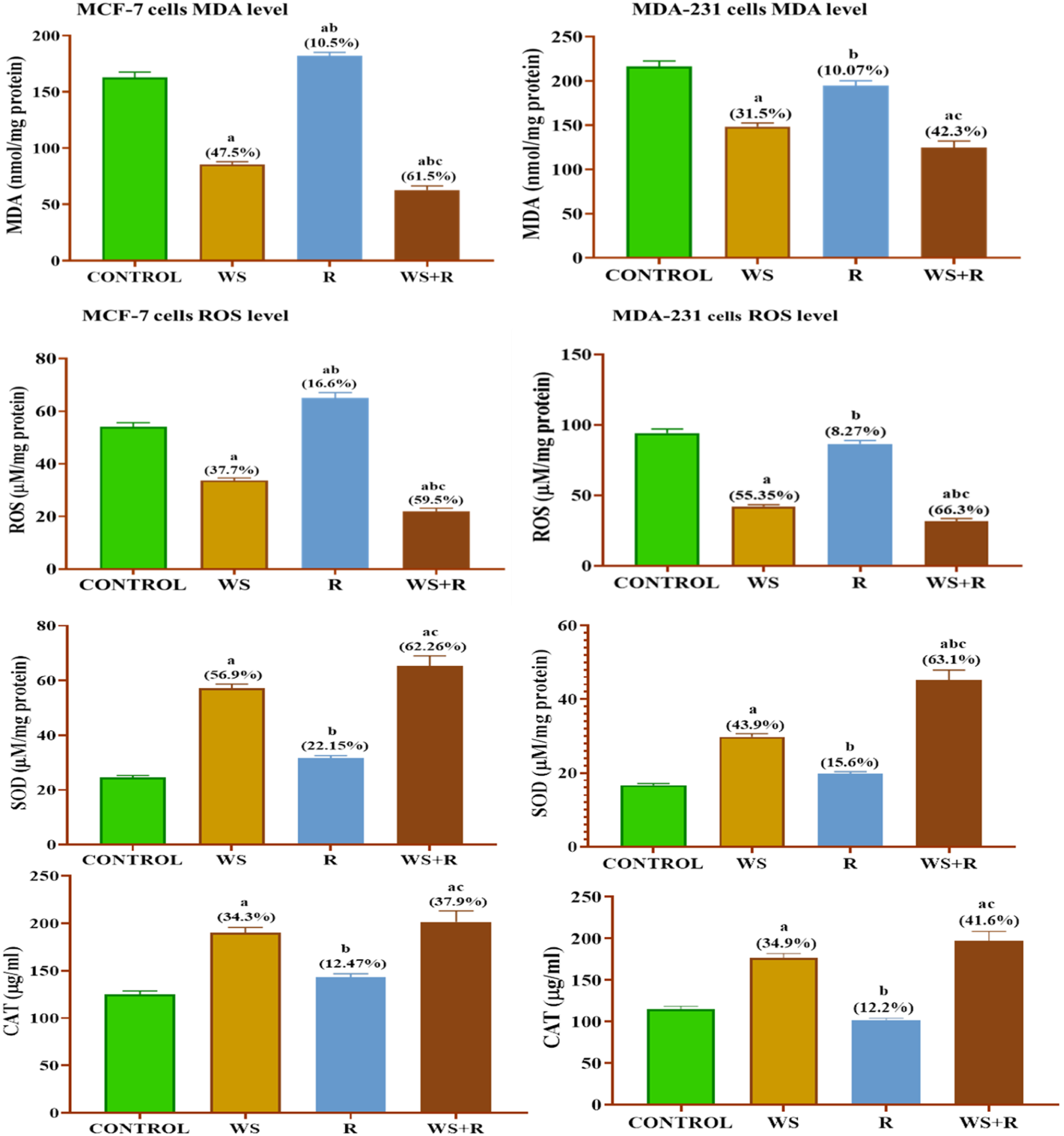
Despite, the replication of the previous experiment on MDA-MB-231 cells under the same conditions displaying a similar trend of change in oxidative status (MDA, ROS, SOD and CAT it exhibits different responding degrees among different cell groups (Figure 3). In MDA-MB -231 cells treated with WS, MDA and ROS concentrations were significantly decreased while, SOD and CAT activities were significantly increased compared with MDA-MB-231 cells. In the R group, a reversed image dominated, the MDA and ROS increased while SOD and CAT decreased compared with control cells. In addition, the analysis of data that emerged from the WS + R group pointed to significant changes in oxidative status compared with the R group (irradiated MDA-MB -231 cells).
Each value represents Mean ± SEM (
SIRT-1-BCL-2/BAX signaling pathway
Figure 4 showed the effect of WS alone or combined with gamma irradiation on the path of SIRT-1-BCL-2/BAX signaling in MCF-7 and MDA-231 breast cancer cells. In WS-treated MCF-7 the data pointed out to a significant decrease in Silent information regulator 1 (SIRT-1) and BCL-2 with a significant increase in BAX compared to control MCF-7 cells (The ratio of BCL2/BAX was decreased). Also, 4 Gy gamma Irradiation of MCF-7 cells induced -significant decrease ( Effect of WS and/or 4 Gy gamma irradiation on SIRT1-BCL-2/BAX signaling pathway in human MCF-7 and MDA-231 breast cancer cells.
Each value represents Mean ± SEM (
Apoptotic determinants NF-κB and caspase3
The data from different groups of MCF-7 cells for the apoptotic determents NF-κB and caspase3 were illustrated in Figure 5(a). In WS, R and WS + R groups, the cancer cells endure significantly decrease in NF-κB while significant increases ( Effect of WS and/or 4 Gy gamma irradiation on apoptotic determents (NF-κB and Caspase3 in human MCF-7 and MDA-231 breast cancer cells.
Each value represents Mean ± SEM (
Inflammatory determinants (TNF-α, COX2, IL-6 and IL-10)
Figure 6(a) demonstrated the results gained for C, WS, R, WS + R groups of MCF7 cell. In WS-treated MCF-7 breast cancer cells, the data pointed out to significant decreases in TNF-α and IL-6 levels and COX-2 activity as well as significant increases in IL-10 concentration, when compared to control MCF-7 cells. Also, the 4 Gy gamma irradiation of MCF-7 cells resulted in remarkable decreases ( Effect of Ashwagandha WS and/or 4 Gy gamma irradiation on inflammatory determinants (TNF-α, COX2, IL-6 and IL-10) in human MCF-7 and MDA-MB-231 breast cancer cells.
Furthermore, the results indicate that the trend of change in the determinants parameters of inflammation measured in the MDA-MB-231 cells is comparable to the changes pattern in the MCF-7 cells (Figure 6(b)). The data of the WS group of MDA-MB-231 cells revealed a significant decrease in TNF-α, IL-6 and COX-2 and a significant increase in IL-10 when compared to control MDA-MB-231 cells. In addition, in the R cell group we observe, significant decreases (
Each value represents Mean ± SEM (
Discussion
It has become almost certain that cancer cells grow and divide by holding up the pathways leading to programmed cell death (Apoptosis) in parallel with the enhancement and activation of cellular survival pathways. Also, these pathways intersect with many operative pathways (like inflammatory pathways), as well as the presence of a number of effectors molecules like Reactive oxygen species (ROS) and the silent information regulation factor 1 (sirtuin type-1, SIRT1) upon abundantly are responsible for the activation or deactivation of certain downstream or upstream molecules which give advantages to one of the two directions, whether cellular death or survival.
SIRT1 appeals great attention as an axis for the up regulation and suppression of many types of tumors. SIRT1’s promotes tumor function and potential mechanisms of cancer cell proliferation and survival. 21 Thus, it might provide valuable cancer therapeutic targets via controlling the pathways of SIRT1 expression or its upstream or downstream active relative molecules. 22
Our data recognized a significant elevation in SIRT1 expression in control MCF-7 and MDA-MB-321 cells compared to other groups (Figure 4). This increase in SIRT1 expression might reflect its vital role in the regulation and promotion of various pathways that outweigh the cell survival and inhibition of apoptosis which is confirmed by the cell cycle and annexin V results (Figure 2). The cells were present at G1 and S phases of cell cycle in the both types of positive control breast cancer cells compared to other groups. Over expression of SIRT1 significantly promoted breast cancer growth both in vitro and in vivo, whereas knockdown of SIRT1 inhibited these phenotypes. 23
The overexpression of SIRT1 endorsed colony formation, cell proliferation, and cell cycle progression in breast cancer cells in vitro and augmented tumor growth in vivo, whereas SIRT1 shut down subdued these features. 24 Several studies have demonstrated that human cancer samples showed raised levels of SIRT1 compared to their non-transformed counterparts. 25 The roles of SIRT1 in breast cancer is multifaceted depending on its substrate from upstream or downstream signaling pathway. 23
In the present study, the treatment of breast cancer cells with
Withaferin A (WA), a promising anticancer constituent of the Ayurvedic medicinal plant
Further, we detected significant increases in ROS and MDA concentrations in both types of breast cancer cells while significant decreases in SOD and CAT activities when compared to other groups (Figure 3). For the first moment it could be accredited this observation to the existence of oxidative burden inside the cancer cells and the expected providence is cell death and inhibition of cell growth. Actually, both of the MCF-7 and MDA-MB321 cells are continued division, growth and survival. Therefore, apart from ROS harmful effects, ROS have the ability to act as second messenger signaling molecules that regulate numerous pathways like, cell cycle, autophagy, apoptosis, endoplasmic reticulum (ER) stress and cellular energy metabolism. 34 Prasad et al. 35 stated that ROS can increase proliferation, inhibit proliferation, or cell apoptosis, depending on the level of ROS. Moloney and Cottor 36 stated that, mitochondrial respiratory chain is the main source of ROS production in cells and the increased ROS in cancer cells is due to the oncogenic stimulation and the increased metabolic activity. 37
The sources of intracellular ROS generation include organelles such as mitochondria which produce ROS through their enzymatic activities rather than Warburg energy production in cancer cells which are occurred predominantly through aerobic glycolytic conversion of glucose to pyruvate. 38 The ROS generation during energy production is must which could interpret the raise in ROS concentration in both types of breast cancer cells. The down regulation of SIRT1 followed by cascades leads to the formation of apoptotic bodies and cell cycle arrest could be in part occurred as a response to flaws in ROS level as well as increases in the activities of SOD and CAT in both types of cancer cells of WS and/or R groups. Obviously, some context-dependent interactions, not specific activation or inhibition is governing the relationship between SIRT1 and oxidative stress in cancer cells. ROS, at the physiological level, are crucial signaling molecules and SIRT1 protein is an important survival. 39 There is a complex interplay between the regulation of cellular activity of the SIRT1 and the presence of ROS. 40 In a context-dependent manner, these factors can control reciprocally each other’s functional activities, directly or via an integrated signaling network. It seems that there is an optimal balance between the level of ROS production and SIRT1 activity, which outbalance the cell survival over the cell death.
Also, this study identified increases in BCL2 and decreases in Bax2 proteins in association with overexpression of NFκB in control MCF-7 and MDA-MB321 breast cancer cells compared to other cell groups (Figure 4 and 5). The activation of pro inflammatory transcription factor nuclear factor-kappa B (NFκB) is a commonly observed phenomenon in breast cancer. 41 In the present study the increased in NFκB is coupled to the increases of the survival protein SIRT1. The concomitant increases in the expression of SITR1 and NFκB observed in our results could be interpreted in the view of the characteristics antagonistic crosstalk between NFκB and SIRT1 signaling pathways.
NFκB signaling can stimulate glycolytic energy flux during acute inflammation, whereas SIRT1 activation inhibits NFκB signaling and enhances oxidative metabolism and the resolution of inflammation. 42 This antagonism is crucial for the survival of the organism since the ability to respond promptly to aggressive pathogens and damages requires quick switches in energy production. On the other hand, the acute inflammatory condition needs to be resolved when that threat has disappeared, if not, this can lead to detrimental chronic inflammation. 43
NFκB is an important signaling molecule mediates tumor-cell proliferation, survival, and angiogenesis via controlling the expression of target genes, such as TNFα, IL6, BCLXL, BCL2, BCLXS, XIAP, and VEGF. 44 The increases in SIRT1 might be participating in the increases of NF-κB that exert significant roles in cell survival, signal transduction, and cell apoptosis through deacetylating vital cell signaling molecules and apoptotic related proteins. 45
On the other hand, the WS could violate the avenue to increase NF-κB concentration and spoil its afterward actions. In the present study the NF-kB concentration decreased in WS and/or WS + R group for the two kinds of cancer cells when compared with the control. Thus, the increases observed in BCL2, IL-6 and TNF-α levels besides a decrease in IL-10 concentrations (Figure 6) in response to the over expression of NF-κB that related to the magnitude of SIRT-1in both types of cancer cells could be brushed up by WS administration. It might be that NF-κB, BCL2, IL-6 and TNF-α were markedly changed as proportionated as the differences occurred between groups on the biochemical indices level, however, when these markers are compared to the other death pathways determinants, the proportional activation of these parameters will reveal that a surrogate death signaling pathways or mediators are significantly activated instead.
Influenced by the above, The BCL2; a cleavage protein of caspases was increased in positive control cells groups as compared with other groups while the expression of Caspase 3 itself as well as BAX proteins were less pronounced (Figure 4 and 5). The BCL2 family of proteins is the hallmark of apoptosis regulation.
48
New members of the
Moreover, the increased expression in TNF-α in both types of cells as reported in our data could be ascribed to the up regulation of NF-κB. TNF and NF-κB research has long been closely associated, reflecting the biological relationship between the two pathways. TNF was first shown to activate NF-κB and shortly thereafter it was shown that NF-κB regulated TNF expression. The TNF-α is a known inducer of COX-2 and the activated TNF-α leads to the up regulation of COX-2 50 (Figure 6). Since the production of pro-inflammation cytokine IL-6 has been shown to be responsible for the up regulation of COX-2 in tissue and various cell lines, 51 the relationship between COX-2 and IL-6 found to be highly expressed in MDA-MB-321 and to less extent in MCF-7 cells (Figure 6) suggesting that IL-6 is likewise could involve in COX-2 up regulation. Moreover, it was reported that IL-6 is a downstream target of NF-κB, signifying that the NF-κB pathway could be involved in IL-6 induction. 52 Besides, NF-κB could mediate the COX-2 expression. Thus, the inflammatory panel in the two types of control breast cancer cells pointed to down regulations of apoptotic pathways which endorsed by a decline in caspase3 activity (Figure 5) when compared to other cells groups. 53 The exposure to WS and/or R seems to disturb the originality status of the previously mentioned parameters in control cells even MCF-7 or MDA-MB-231cells (Figure 5). It was found that natural products as withania somnifera can sensitize cancer cells to ionizing radiation so apoptosis increase as found in groups of breast cancer cells treated with withania somnifera alone or with irradiation. 54 In addition, it was found in our recently published research paper that Ashwagandha protect normal cells from the hazardous effect of acute and chronic radiation because of its antioxidants and anti-inflammatory capabilities. 55 However, we believed that further insightful future investigations focusing on a diverse mediator involved in autophagy and necroptosis could be with benefit to decipher this discrepancy emerged and could disclose the exact mechanism of WS.
Based on our findings in the present study we could accomplish that the oxidative status as well as inflammatory responses are grave for determining the longevity, life span and reactivity of MCF7 and MDA-MB-231breast cancer cells upon exposure to WS unaccompanied or accompanied by gamma radiation. The apoptotic and survival pathways of the two kinds of cancer cells are strongly affected by changes of oxidative and inflammatory responses. The magnitude of effectiveness seemed diverse among the two kinds of cells. WS might represent a valuable key in contesting development of drug resistance of cancer cells and it could rise the radio-sensitivity of cells when WS delivered in combination with radiation exposures.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
