Abstract
Objective:
Coronary microembolization (CME)-induced cardiomyocyte apoptosis is the primary factor in causing cardiac dysfunction. Resveratrol (RES) is known to play a protective role in a variety of cardiovascular diseases, yet it is not known whether RES has a protective role in CME. Therefore, the effect of RES on cardiomyocyte apoptosis and cardiac function damage which are induced by CME in rats was investigated in this study.
Methods:
Fifty Sprague-Dawley rats were separated into 5 groups randomly (10 rats were included in each): sham group, CME group, RES+CME group, RES+CME+Sirtuin-1 (SIRT-1) inhibitor EX527 (RES+CME+EX) group, and CME+EX group. Cardiac function, serum c-troponin I (cTnI) level, apoptotic index, and microinfarct were measured by cardiac ultrasound, myocardial enzyme assessment, TdT-mediated dUTP Nick-end labeling and hematoxylin-basic fuchsin-picric acid staining. The levels of p53, p53 acetylation, SIRT-1, Bax, Bcl-2, and cleaved caspase-3 were detected by Western blot.
Results:
Myocardial dysfunction, enhanced apoptotic index as well as cTnI were caused after the operation of CME. Coronary microembolization induced increased expression of p53 acetylation and cleaved caspase-3, while the SIRT-1 and Bcl-2/Bax ratio was reduced. The CME effect was reversed by RES while EX527 attenuated this protective effect.
Conclusions:
Resveratrol can improve cardiac function, in the sense that it attenuates CME-induced cardiomyocyte apoptosis, which is perhaps associated with its inhibition pro-apoptotic pathway of p53 which is transcription-independent.
Introduction
Coronary microembolization (CME), which often occurs in percutaneous coronary intervention or thrombolytic therapy, is usually induced when atherosclerotic unstable plaques rupture spontaneously or plaque debris or small thrombus blocks the distal end of the coronary artery. 1 Coronary microembolization can induce slow flow or no reflow in the myocardium and consequently causes local myocardial contractile dysfunction and fatal arrhythmia, which seriously affects the cardiac functions and prognosis of patients. 2,3 Considerable research suggests that, in the acute phase of CME, microinfarctions occur in the local myocardium, and necrosis and apoptosis occur in cardiomyocytes. Cardiomyocyte apoptosis is a key mechanism underlying the systolic dysfunction which is induced by CME. Inhibition of cardiomyocyte apoptosis can reduce myocardial injury caused by CME. 4 –6 Sun et al 7 suggest that p53 is involved in the cardiomyocyte apoptosis following the operation of CME in rats. Wherefore, inhibition of p53 expression can reduce cardiomyocyte apoptosis and consequently improves myocardial contractile function, which is primarily achieved by regulating the Bbc3/BCL-2/caspase-3 pathway.
Resveratrol (RES), a polyphenolic compound widely found in natural plants, has multiple pharmacological activities, with high safety yet few side effects. 8 Resveratrol exerts multiple cardiovascular protective effects according to recent studies on cardiovascular diseases, such as antioxidative stress, 9 inhibition of platelet aggregation, 10 regulation of blood lipids, 11 anti-apoptosis, 12 and anti-myocardial ischemia-reperfusion injury. 13 However, there is little research into whether RES can regulate CME-induced myocardial apoptosis and whether RES reduces myocardial injury and consequently improves cardiac function. In this condition, a better understanding of the relationship between RES and cardiomyocyte apoptosis is required to inform the therapy and prevention of CME. Therefore, this investigation aimed to identify and clarify the effect of RES on cardiomyocyte apoptosis as well as the relationship between SIRT-1 and its downstream anti-apoptotic pathway in CME rats, to reveal the possible mechanism of the anti-apoptotic effect of RES in CME-induced myocardial injury.
Materials and Methods
Animals Preparation
The Institutional Animal Care and Use Committees at the Guangxi Medical University approved all the procedures, which were then carried out as per a protocol on Use of Laboratory Animals which is released from the National Institute of Health Guidelines. Fifty Sprague-Dawley (SD) rats (male, weighted 250-300 g) were provided by Guangxi Medical University. The rats in the experiment were maintained under standard conditions and with a controlled 12 h/12 h-light–dark cycle at 25°C temperature.
Coronary Microembolization Modeling and Experimental Grouping
Firstly, 30 to 40 mg/kg pentobarbital was injected intraperitoneally into the rats to keep them under anesthesia, with the method described by Su et al. 14 Then tracheal intubation was performed, with a ventilator to assist breathing. After this step, thoracotomy was then performed on the left sternal border between the third and fourth intercostal spaces. After this step was finished, the ascending aorta was then clamped for a time of 10 seconds by a vascular clamp after being separated. Approximately 3000 microspheres (suspended in normal saline of 0.1 mL) with 42 μm diameter (Biosphere Medical Inc, Rockland, Maine) were rapidly injected using a microinjector from the apex of the left ventricle. After breathing is stabilized, the chest was closed in layers and tracheal intubation was removed. Postoperatively, 800 000 units of penicillin were intraperitoneally injected in the rats. Similarly, saline of 0.1 mL was injected into each rat in the sham group. The 50 SD rats were divided in a random and equal manner into Sham, CME, RES+CME, RES+CME+EX, and CME+EX group. Each rat in the RES+CME group was injected with 20 mg/kg RES (Sigma Chemical Co, St Louis, Missouri) via tail vein 30 minutes prior to building the CME model; while in the RES+CME+EX group, each rat was injected with RES and EX527 (Sigma Chemical Co, St Louis, Missouri), at a dose of 20 mg/kg and 1 μg/kg, respectively. Each rat in the CME+EX group was injected with 1 μg/kg EX527 via tail vein 30 minutes prior to building the CME model.
Detection of Cardiac Function
Here a Hewlett Packard Sonos 7500 Ultrasound instrument, with a 12 MHz frequency probe (Philips Technologies, Amsterdam), was applied to measure cardiac output (CO), left ventricle fractional shortening (LVFS), left ventricular ejection fraction (LVEF), and left ventricular end-diastolic diameter (LVEDd). In all the measurements, averaged values of triple cardiac cycles were adopted. An expert in echocardiography examined all the echocardiograms.
Measurement of Serum c-Troponin I
At 12 hours following CME or sham operation and before killing, 1.0 mL blood was collected at the position of femoral vein, and then serum c-troponin I (cTnI) was determined in line with the Kit instructions (Roche, Inc, Basel, Switzerland).
Tissue Extraction and Sample Processing
After the detection of cardiac function in the former step, potassium chloride (2 mL, 10%) was injected into each rat at the position of the tail vein, for the purpose that the heart of each rat can be harvested immediately while in the ventricular diastolic phase. Atrial appendage was excluded in the experiment. The ventricle was separated into heart base and the apex at the midpoint along the long axis of the left ventricle, in a fashion of parallel to the atrioventricular groove. After being processed in liquid nitrogen, the apex was immediately transferred to and preserved at a −80°C refrigerator for the following Western blot detection. The base of the heart was embedded using paraffin and then sliced continuously (4 μm for each slice) after being fixed for 12 hours with 4% paraformaldehyde. The slices were used for the following staining with hematoxylin-basic fuchsin-picric acid (HBFP), TdT-mediated dUTP Nick-End Labeling (TUNEL), and hematoxylin-eosin.
Detection of Cardiomyocyte Apoptosis by TUNEL
TdT-mediated dUTP Nick-end labeling assays were performed to detect myocardial apoptosis using a commercial kit (Roche,Massachusetts, Boston) based on the manufacture’s protocol. The apoptotic cell nuclei was stained green and normal nuclei stained blue. Five visual fields in each slice were randomly chosen, and the average number of apoptotic cells per 200 cells was determined. The apoptosis index (AI) was determined as follows: AI = (the number of positive cells/the total number of counted cells) × 100%. 15 Image-Pro Plus version 6.0 sofware was employed in our data analysis.
Measurement of Myocardial Microinfarct Areas
In diagnosing early myocardial ischemia, HBFP staining can stain nucleus in blue color, normal myocardial cytoplasm in yellow, as well as ischemic myocardium and red blood cells in red. A DMR+Q550 pathological image analyzer (Leica, Wetzlar, Germany) was used to observe (×200 magnification) each HBFP-stained slice. For each slice, 5 random visual fields were selected. The Leica Qwin analysis software plane method was applied in this study to determine the infarct area, which was then divided by gross observed area to calculate the infarct percentage. 16
Western Blot Assay
About 10% to 15% sodium dodecyl sulfate-polyacrylamide gel electrophoresis was used to separate the total protein collected from the cardiac tissue before they were transferred to PVDF membranes (Millipore, Atlanta, Georgia), which were blocked with nonfat milk or 5% bovine serum albumin for 1.5 hours at room temperatures before they were incubated at 4°C overnight using media of primary antibodies against p53 (Santa Cruz Biotechnology, Santa Cruz, California), p53 with acetylated K183 (Abcam, Cambridge, United Kingdom), SIRT-1 (Millipore, Billerica, Massachusetts), Bcl-2, Bax, cleaved caspase-3, or GAPDH. The antibodies Bcl-2, Bax, cleaved caspase-3, or GAPDH were provided by Cell Signaling Technology (Danvers, Massachusetts).
The secondary antibodies conjugated with horseradish peroxidase were used to incubate the membranes in tris buffered saline with tween 20 (TBST) for 2 hours at room temperatures after TBS containing 0.1% Tween 20 (TBST) was used to wash the membranes for 5 times. A chemiluminescence detecting equipment (enhanced version, Pierce, Rockford, Illinois) was used to detect the signals. Image Lab software of Bio-Rad , Hercules, CA was used to assess and quantify the bands for protein amounts.
Statistical Analysis
Quantitative data were presented in a format of mean value (standard deviation). Student t test was used for comparison between 2 groups. While for the analysis of differences among 3 or more groups, we applied one-way analysis of variance. Statistical significance was defined as P < .05.
Results
Changes in Heart Function Indicators in Rats
Table 1 shows that cardiac dysfunction was induced by CME, which was characterized by increased LVEDd and decreased CO, LVFS, and LVEF. The cardiac dysfunction caused by CME was improved by RES pretreatment, while EX527 attenuated these protective effects.
Changes in Cardiac Function
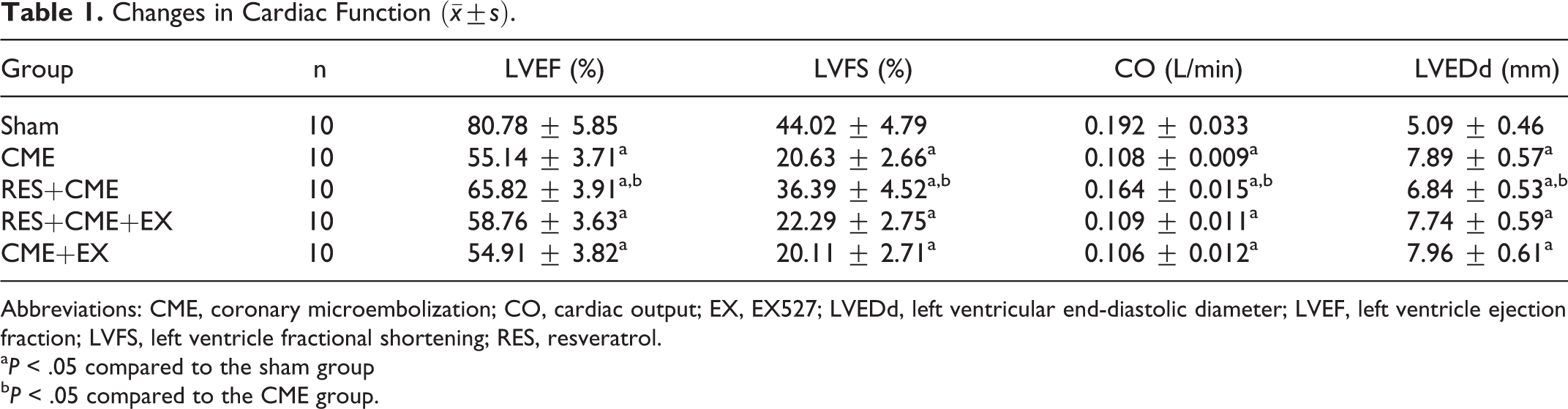
Abbreviations: CME, coronary microembolization; CO, cardiac output; EX, EX527; LVEDd, left ventricular end-diastolic diameter; LVEF, left ventricle ejection fraction; LVFS, left ventricle fractional shortening; RES, resveratrol.
a P < .05 compared to the sham group
b P < .05 compared to the CME group.
Resveratrol-Reduced Serum Troponin I Levels After CME
Figure 1 shows that cTnI was increased significantly in CME group compared to sham group. On the contrary, RES significantly inhibited this increase albeit its protective effects were attenuated by EX527.

Parameters of cTnI in rats of each group. a P < .05 compared to sham group. b P < .05 compared to CME group, n = 10. CME indicates coronary microembolization; EX, EX527; RES, resveratrol.
Pathological Observation of CME
Hematoxylin-eosin and HBFP staining results
Albeit no obvious infarcts were noted, subcutaneous ischemia occurred occasionally in sham group. In the other 3 groups, however, multiple microinfarctions were observed. These lesions were mostly wedge-shaped with a focal distribution and were more common in the subendocardial and left ventricle, as shown in Figure 2. Hematoxylin-eosin staining showed that myocardial cell nucleus dissolved or disappeared in the microinfarction, cytoplasmic red staining, degeneration, peripheral myocardial edema, red blood cell exudation and peripheral inflammatory cell infiltration, and microembolism in arteriole (Figure 3). The infarct size of CME group, RES+CME group, RES+CME+EX group and CME+EX group was (9.83% ± 3.16%), (6.51% ± 3.05%), and (9.46% ± 3.29%), (10.25% ± 2.89%), respectively, (Figure 2). The infarct size of the CME group, RES+CME group, RES+CME+EX group, and CME+EX group was increased significantly in comparison with the sham group. The infarct size of the RES+CME group was decreased, but the RES+CME+EX group and CME+EX group did not reduce infarct size, compared to the CME group.
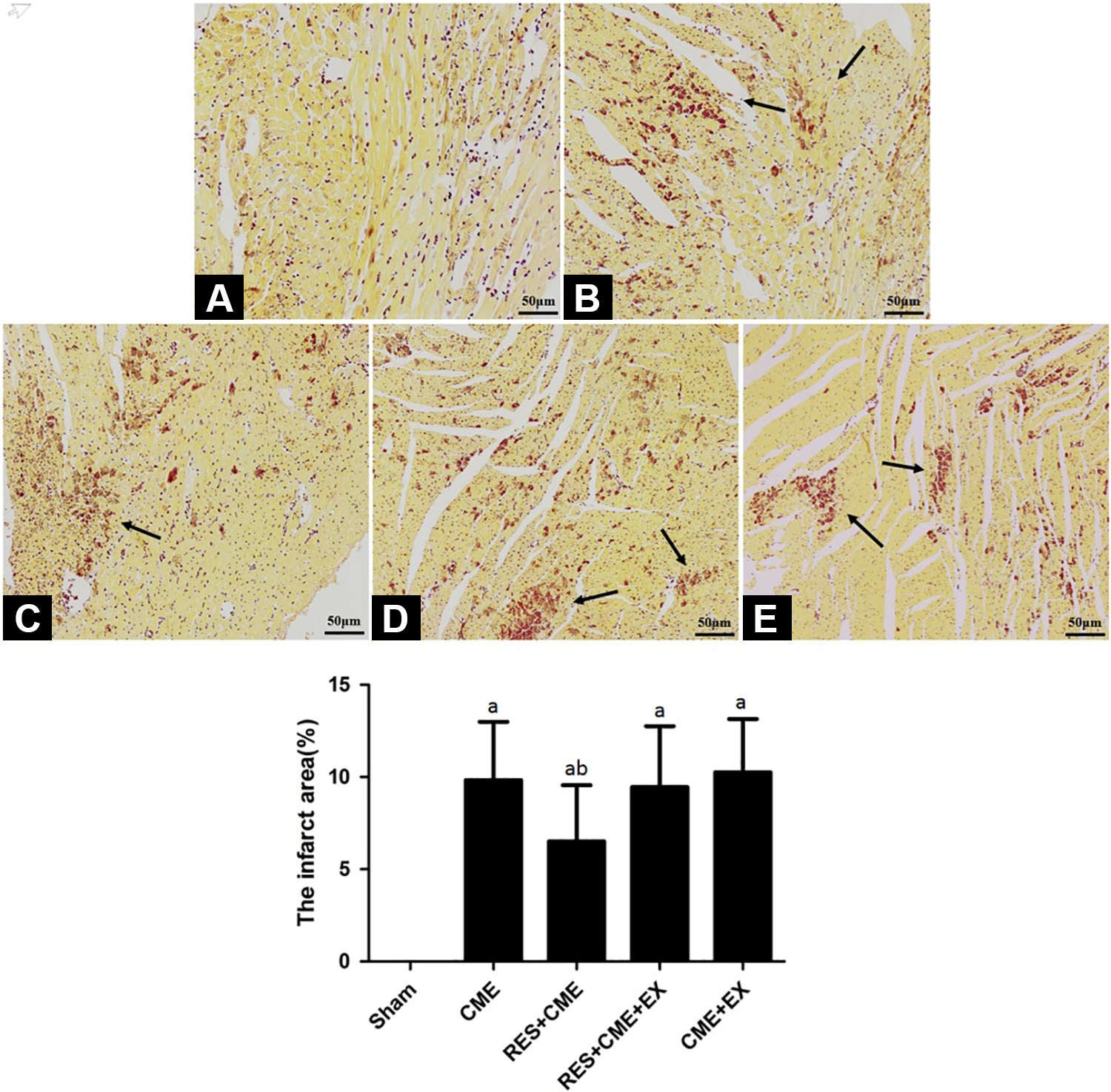
Pathohistological examination by HBFP staining (magnification, ×200; bar = 50 μm). A to E, Sham group, CME group, RES+CME group, RES+CME+EX group, and CME+EX group, respectively. Ischemic myocardium in red, arrow indicates microinfarct areas. a P < .05 compared to sham group; b P < .05 compared to CME group, n = 10. CME indicates coronary microembolization; EX, EX527; HBFP, hematoxylin-basic fuchsin-picric acid; RES, resveratrol.

HE staining in pathohistological examination at 12 hours after CME (magnification, ×400; bar = 50 μm). HE staining showed microspheres with inflammatory cell infiltration. Arrow indicates microspheres. CME indicates coronary microembolization; HE, hematoxylin and eosin.
TdT-mediated dUTP Nick-end labeling staining results
The signal was localized in the myocardial cells nucleus, showing a green color, compared to the blue in the normal cell nucleus. In the groups of sham, CME, RES+CME, RES+CME+EX, and CME+EX, the apoptotic index of cardiomyocytes was (0.37% ± 0.074%), (7.05% ± 1.68%), (3.41% ± 0.57%), (7.26% ± 1.83%), and (7.83% ± 1.91%), respectively (Figure 4). This index in the groups of CME, RES+CME, RES+CME+EX, and CME+EX was increased markedly compared to sham group. While this index in the RES+CME group was decreased significantly, it was not decreased in the RES+CME+EX group and CME+EX group, compared to the group of CME.
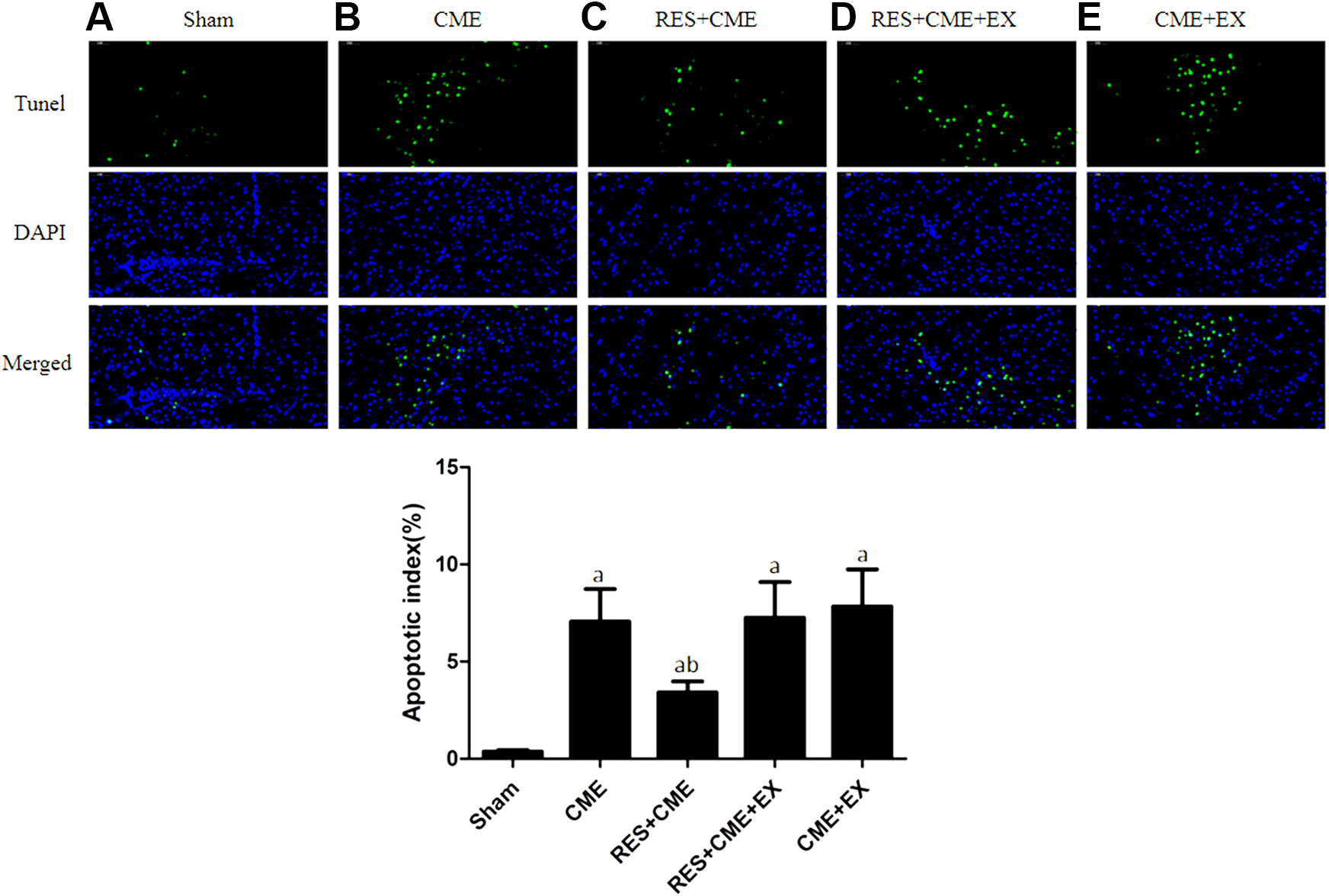
Cardiomyocyte apoptosis by TUNEL staining (magnification, ×400; bar = 50 μm). A to E, Sham group, CME group, RES+CME group RES+CME+EX group, and CME+EX group, respectively. Apoptotic nuclei in green, normal cardiomyocytes in blue. Arrow indicates apoptotic cardiomyocytes nuclei. a P < .05 compared to sham group; b P < .05 compared to CME group. n = 10. CME indicates coronary microembolization; EX, EX527; RES, resveratrol; TUNEL, TdT-mediated dUTP Nick-end labeling.
Resveratrol Effect on Myocardial Apoptosis
Cleaved caspase-3, Bcl-2, and Bax protein expression was detected to confirm cardiomyocyte apoptosis after CME. Cleaved caspase-3 protein expression in the CME group demonstrated a significant increase compared to the sham group (Figure 5), and the Bcl-2/Bax ratio was downregulated markedly (Figure 6). After the pretreatment with RES, cleaved caspase-3 expression showed significant decrease; Bcl-2/Bax ratio showed significant increase; while EX527 attenuated these anti-apoptotic effects.

RES effects on SIRT1 and caspase-3 expression. a P < .05 compared to sham group; b P < .05 compared to CME group, n = 10. CME indicates coronary microembolization; EX, EX527; RES, resveratrol.

RES upregulates Bcl-2 and inhibits Bax expression. a P < .05 compared to sham group; b P < .05 compared to CME group, n = 10. CME indicates coronary microembolization; EX, EX527; RES, resveratrol; SIRT-1, Sirtuin-1.
Overexpressed SIRT1 Protein and Weakened p53 Protein Acetylation Were Induced by RES
The SIRT1 protein level was compared among the 4 groups, to determine in the CME-induced myocardial apoptosis, whether SIRT1 is involved (Figure 5). The SIRT1 protein level was detected at a moderate level in the sham group, while at a level slightly increased after treatment with CME; however, this level was detected at a markedly increased level in the RES+CME group, and was attenuated by EX527 in the RES+CME+EX group. Further study in acetylation assay showed that (Figure 7), in the sham group, the p53 protein was acetylated weakly while in the CME group it was noticeably acetylated. The p53 protein acetylation was weakened by RES in the RES+CME group; while in the RES+CME+EX group, this weakened acetylation was attenuated by EX527.
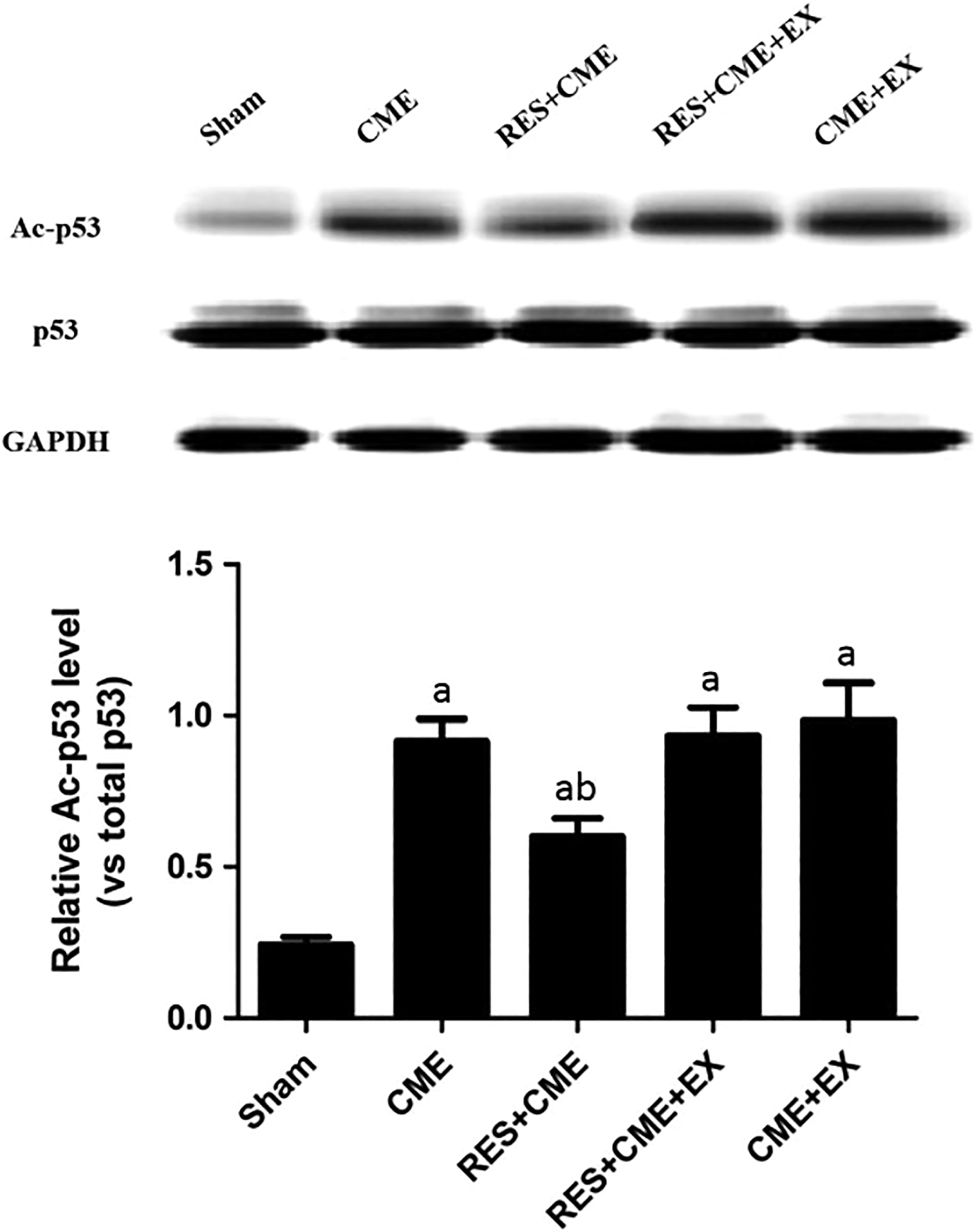
RES inhibits p53 acetylation. a P < .05 compared to sham group; b P < .05 compared to CME group. n = 10. CME indicates coronary microembolization; EX, EX527; RES, resveratrol; SIRT-1, Sirtuin-1.
Discussion
Resveratrol is widely found in many plants, such as grapes, peanuts, and Polygonum cuspidatum. It has strong biological properties, such as antioxidation, anti-aging, anti-inflammatory, and anti-apoptosis. 17 To determine the potential protection of CME-induced cardiomyocyte apoptosis from this material, a rat model of CME-induced cardiomyocyte apoptosis in vivo was developed and tested here. Coronary microembolization resulted in cardiac dysfunction, induced a notable rise in cTnI, and also increased the myocardial microinfarct size and the cardiomyocyte AI in rats. However, the CME-induced cardiomyocyte apoptosis and myocardial injury were effectively reduced by injecting 20 mg/kg RES in the tail vein 30 minutes before CME, which are solid evidence that RES can attenuate CME-induced cardiomyocyte apoptosis and consequently improves cardiac function.
Resveratrol is a potent agonist of the NAD-dependent deacetylase SIRT1 (Figure 4). It is reported that RES can protect the heart by inducing an anti-apoptotic signal. 18 Additionally, RES can also increase the activity of SIRT1, which is a common anti-aging gene. 19 In a study by Zhang et al, RES increased the content of SIRT1 in myocardial tissue damaged by doxorubicin and consequently inhibited the cardiomyocyte apoptosis thereof. 20 Chen et al found that RES increased the content of SIRT1 in H9c2 cardiomyocytes under hypoxic conditions, thereby reducing the apoptosis of cardiomyocytes induced by hypoxia. 21 In addition, Hsu et al found that high expression of SIRT1 increased the expression of Bcl-xL in myocardial tissue and decreased expression of the proapoptotic protein Bax and cleaved caspase-3, thereby inhibiting cardiomyocyte apoptosis induced by ischemia-reperfusion. 22 This study showed that the expression of SIRT1 in myocardial tissue after CME was increased by RES, accompanied by a decrease in the cardiomyocyte AI following CME. In addition, we also found that when RES and the SIRT1 activity inhibitor, EX527, were simultaneously applied, the anti-cardiomyocyte apoptosis effect of RES was significantly inhibited. Therefore, it can be proved that RES exerts SIRT1-dependent anticardiomyocyte apoptosis effect by increasing the expression level of SIRT1 in myocardial tissue after CME.
This study further explored the downstream molecular pathways by which SIRT1 inhibits apoptosis. The tumor suppressor p53 positively regulates the proapoptotic protein Bax as an important transcription factor. 23 Studies have shown that acetylation of multiple lysine sites in p53 is essential for its maintenance of transcriptional activity. 24 Studies in rats, mice, and humans have found that SIRT1 can inhibit the binding of p53 to DNA fragments by deacetylating p53 lysines, thereby inhibiting the activity of p53 transcriptional pro-apoptotic proteins. 25 –27 Zhang et al found that increase in expression of SIRT1 reduced the level of p53 acetylation in myocardial tissue and thus inhibited the apoptosis of cardiomyocytes induced by doxorubicin, which is realized by inhibiting the expression of the proapoptotic protein Bax. 20 Nadtochiy et al also found that high expression of SIRT1-reduced p53 acetylation level in myocardial tissue, thereby inhibiting cardiomyocyte apoptosis induced by ischemia-reperfusion. 28 This investigation addressed the hypothesis that RES protection of CME-induced cardiomyocyte apoptosis is associated with the activation of SIRT1 as well as the following deacetylation and inactivation of p53. The results of this study show that when SIRT1 expression level is increased, the level of p53 acetylation in myocardial tissue is significantly decreased, and that Bcl-2 expression is increased as expression of Bax and cleaved caspase-3 is decreased. When SIRT1 activity is inhibited by EX527, the level of p53 acetylation in myocardial tissue is increased significantly; on the other hand, expression of the Bcl-2 protein is decreased as the expression of Bax and that of cleaved caspase-3 protein are increased. This indicates that the RES cardioprotection is associated with the inhibition of the pro-apoptotic pathway of p53 which is transcription-independent.
In summary, this study demonstrates that RES plays a crucial role in myocardial protection in myocardial injury caused by CME. The mechanism underlying this protection is primarily by increasing the expression of SIRT1 in myocardial tissue, so that the level of p53 acetylation in myocardial tissue is significantly reduced, thereby inhibiting the pro-apoptotic activity of p53 and ultimately reducing apoptosis of cardiomyocytes induced by CME.
Footnotes
Author Contribution
Q.M. contributed to conception and design. X.L. contributed to design. Q.M. and Y.L. drafted the manuscript. Y.W. contributed to acquisition and interpretation. Y.L. contributed to analysis and interpretation. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Guangxi Natural Science Foundation (No.2017GXNSFAA198214).
