Abstract
Liver fibrosis is a wound-healing response and the activation of the hepatic stellate cell (HSC) is the core of hepatic fibrosis. MicroRNAs (miRNAs) participate in the development of fibrosis. It is reported that histone deacetylases (HDAC2) tyrosine phosphorylation by cellular-Abelsongene (c-Abl) induces malignant growth of cells. Here, we investigated the effect of miR-122-5p on the proliferation and apoptosis of HSCs. Normal human HSC line LX-2 and LX-2 cells stimulated by transforming growth factor (TGF)-β1 for 24 h were cultured and assigned into the blank group and the TGF-β1 group. The miR-122-5p inhibitor and its negative control were transfected into LX-2 cells and miR-122-5p mimic and its negative control were transfected into LX-2 cells stimulated by TGF-β1. The result showed that miR-122-5p expression was decreased in TGF-β1-stimulated LX-2 cells. miR-122-5p overexpression reduced the mRNA and protein levels of collagen I and α-smooth muscle actin, inhibited cell proliferation, and accelerated cell apoptosis. miR-122-5p targeted c-Abl. Meanwhile, miR-122-5p overexpression inhibited HSC activation by suppressing the c-Abl/HDAC2 pathway. In summary, miR-122-5p overexpression exerted anti-fibrosis effect and inhibited HSC activation by suppressing the c-Abl/HDAC2 pathway.
Keywords
Introduction
Liver fibrosis is a response caused by chronic liver injury as a result of various factors which generate chronic inflammation and then lead to abnormal wound healing. 1 Excessive extracellular matrix deposition and fibrous scar formation are typical characteristics of liver fibrosis. 2 The main risk factors of liver fibrosis include chronic hepatitis C virus infection, alcohol abuse, and non-alcoholic steatohepatitis. 3 The most advanced stage of liver fibrosis is liver cirrhosis that contributes to the high morbidity and mortality in patients with chronic liver disease, which affects about 1–2% of global population and leads to more than one million deaths each year around the world.4–6 Modern diet and changes of food habits are also key contributors to the development of hepatic disorders. 7 Nowadays, regular consumption of flavor-enhancing high-lipid diet induces non-alcoholic fatty liver disease-mediated hepatic fibrosis and cardiovascular anomalies by generating reactive oxygen species and altering the redox equilibrium.8,9 Hepatic stellate cells (HSCs) are the main producing cells of extracellular matrix and exist in a quiescent non-proliferative state in normal liver.10–11 The activation of HSCs is the known mechanism leading to liver fibrosis. 12 Therefore, it is important to understand the molecular mechanism of HSC activation in the management of liver fibrosis.
MicroRNAs (miRNAs) are short RNA molecules 19–25 nucleotides in size which play crucial roles in post-transcriptional gene regulation. 13 miRNAs have been documented in the pathogenesis of liver fibrosis, 14 which is an inevitable pathological process for the progression of chronic hepatitis to liver cirrhosis. miR-122 expression shows negative correlation with the development of liver fibrosis in patients with chronic hepatitis C. 15 Meanwhile, miRNAs have been found to play a pivotal role in fibrosis and cirrhosis, including those in HSC activation and proliferation as well as production of extracellular matrix proteins.16–17 miRNAs might affect liver fibrosis by participating in various biological processes like cell proliferation and apoptosis. 18 What is more, the elevation of vascular endothelial growth factors and transforming growth factor-β stimulates liver fibrosis by activating the NF-kB-mediated inflammatory response and cell apoptosis evoked by mitochondrial caspase.9,19–21 miR-122, composing about 70% of the total miRNAs found in hepatocytes, is one of the most abundant miRNAs in the liver and plays a critical role in liver fibrosis by negatively regulating the proliferation and transactivation of HSCs.22–24 miR-122-5p overexpression remarkably suppressed hepatocellular carcinoma cell proliferation, migration, and invasion. 25 Furthermore, miR-122-5p was found to be poorly expressed in skeletal muscle fibrosis. 26 In addition, miR-203 regulates smooth muscle cell proliferation by modulating cellular-Abelsongene (c-Abl) expression, and miR-489 inhibits cardiac fibrosis by down-regulating histone deacetylases 2 (HDAC2).27,28 However, the action of miR-122-5p on HSCs in liver fibrosis is still unknown to us. And whether miR-122-5p can affect HSC proliferation and apoptosis by regulating the c-Abl/HDAC2 pathway requires further research. This study was conducted to identify whether miR-122-5p affects HSC proliferation and apoptosis by modulating the c-Abl/HDAC2 pathway and thus theoretically to provide a new treatment method for patients with liver fibrosis.
Material and methods
Ethical statement
Ethical approval of this study was obtained from the Ethical Committee of the Affiliated People’s Hospital of Ningbo University.
Cell culture and transfection
Human HSCs (LX-2 cell line) were from Shanghai Institute of Biochemistry and Cell Biology (Chinese Academy of Sciences, Shanghai, China). Cells were cultured with RPMI 1640 culture medium containing 10% fetal bovine serum in an incubator containing 5% CO2 at 37°C. Cells at the logarithmic phase were harvested and detached using 0.25% trypsin. Cell density was modified at 6 × 104 cells/mL. Cells were seeded into 96-well plates at 0.2 mL/well (4 wells per group) and incubated overnight. Transforming growth factor-β1 (TGF-β1, R&D Systems, Shanghai, China) was used as a stimulating factor to activate HSCs, and each well was added with 5 ng/mL TGF-β1 and treated for 24 h. 29 Cells were assigned into two groups: TGF-β1 group (treated with 5 ng/mL TGF-β1 for 24 h) and blank group (treated with the same amount of PBS for 24 h). Alterations of cellular morphology were observed using an inverted microscope (Olympus, Tokyo, Japan).
Cell transfection was performed using Lipofectamine 2000 (Invitrogen, Carlsbad, California, USA). After transfection, cells were incubated in an incubator for following experiments. Cells transfected with miR-122-5p mimic for 24 h were treated with c-Abl/HDAC2 activator DPH (HY-12070, MCE, Monmouth Junction, New Jersey, USA) for another 24 h. Cells in the control group were treated with the same amount of DSMO. miR-NC, miR-122-5p mimic, miR-122-5p inhibitor, and inhibitor NC were purchased from Shanghai Sangon Biotech Co. Ltd. (Shanghai, China).
Reverse transcription-quantitative polymerase chain reaction
Primer sequences.

miR: microRNA; F: forward; R: reverse; α-SMA: α-smooth muscle actin.
Western blot
The bicinchoninic acid kit (Qiagen, Wiessberg, Germany) was used to assess concentrations of proteins extracted from LX-2 cells. Proteins were separated by SDS-PAGE with voltage changing from 80 v to 120 v, and then transferred onto polyvinylidene fluoride membranes by semi-dry method at the voltage of 80 mv for 30–45 min. After blocking with 5% bovine serum albumin for 1 h, the membranes were probed with primary antibodies collagen I (1:1,000, ab260043, Abcam, Cambridge, Massachusetts, USA), α-smooth muscle actin (α-SMA, 1:1,000, ab108424, Abcam), c-Abl (1:1,000, ab254341, Abcam), p-c-Abl (1:1,000, ab4717, Abcam), HDAC2 (1:1,000, ab219053, Abcam), p-HDAC2 (1:1,000, ab75602, Abcam), and β-actin (1:5,000, ab8227, Abcam) at 4°C overnight. After three TBST washes (each time for 5 min), corresponding secondary antibody (Beyotime, Shanghai, China) was added and incubated for 1 h. Then, the membranes were rinsed 3 times (each time for 5 min). Chemiluminescence reagent was added for color development. Protein bands were analyzed using Bio-Rad Gel Dol EZ Imager (Hercules, California, USA). Gray value analysis was conducted using Image J software (NIH, Bethesda, Maryland, USA).
Cell counting kit-8 assay
LX-2 cells were seeded at 5000 cells/well into 96-well plates and placed into an incubator containing 5% CO2 at 37°C. A total of 20 μL CCK-8 solution (Nanjing JianCheng Bioengineering, Nanjing, China) was added to each well and incubated for 24, 48, 72, and 96 h. The optical density (OD) value at 450 nm was detected.
Flow cytometry
LX-2 cells were seeded into 6-well plates and adjusted to 1 × 105 cells/mL. After centrifugation, cells were resuspended in 50 μL PBS. As instructed by the protocols of the Annexin V-FITC apoptosis assay kit (YEASEN Biotech, Shanghai, China), 5 μL Alexa Fluor 488 Annexin V-FITC and 10 μL 100 mg/mL PI staining solution were added to 100 μL 1 × binding buffer resuspended cells and incubated for 15 min without light exposure. Following thorough mixture, cell apoptosis was determined by flow cytometry (MoFloAstrios EQ, Fullerton, California, USA). About 100,000 cells were detected by flow cytometry. The percentage of cell AV+ (apoptotic cells) was calculated.
Dual-luciferase reporter assay
The binding sites of miR-122-5p and c-Abl were predicted using bioinformatics website (https://starbase.sysu.edu.cn/index.php). LX-2 cells at the logarithmic phase were harvested and lysed using TRIzol lysis buffer. Then, 5 μL lysis buffer, luciferase buffer, and 5 μL substrate were mixed thoroughly and the fluorescence intensity was determined. Subsequently, the c-Abl 3’UTR sequence containing miR-122-5p binding sites were synthesized to construct c-Abl 3’UTR wild-type (WT) plasmid (c-Abl-WT). The binding sites were mutated on the basis of the plasmid to construct c-Abl 3’UTR mutant-type (MUT) plasmid (c-Abl-MUT). The plasmids of c-Abl-WT and c-Abl-MUT were mixed with NC and miR-122-5p, respectively, and transferred into 293T cells (ATCC, Manassas, Virginia, USA). Cells were collected and lysed 48 h post transfection. Luciferase activity was determined by dual-luciferase reporter assay kit (BioVision, SanFrancisco, California, USA) and Glomax20/20 lUminometer fluoroscope (Promega, Madison, Wisconsin, USA).
Statistical analysis
All data were processed using SPSS21.0 statistics software (IBM Corp., Armonk, New York, USA) and GraphPad Prism 8.0.1. Measurement data were expressed as mean ± standard deviation (SD). Normality test and homogeneity of variance test were performed at first. The result showed that data were in accordance with normal distribution and homoscedasticity. The comparison between the two groups was carried out by t test. Data were analyzed by one-way analysis of variance (ANOVA) or two-way ANOVA, followed by Tukey’s multiple comparisons test. The rank sum test was performed if data were not in accordance with normal distribution and homoscedasticity. p < 0.05 showed statistical significance.
Results
MicroRNA-122-5p was poorly expressed in TGF-β1-stimulated LX-2 cells
miR-122-5p are small regulatory molecules which affect liver fibrosis progression in some ways.
30
Meanwhile, TGF-β1 is the most potent fibrogenic cytokines so far.
29
To investigate the effect of miR-122-5p on LX-2 HSCs, normal human HSC line LX-2 and LX-2 cells stimulated by TGF-β1 for 24 h were cultured and assigned into the blank group and the TGF-β1 group. miR-122-5p expression was evaluated by RT-qPCR. The result showed that miR-122-5p expression was decreased in TGF-β1-stimulated cells (Figure 1(a), p < 0.01). Observation under inverted microscope showed that cells in the TGF-β1 group changed from fibroblasts in large star or fusiform shape to fibrocytes in spindle shape with flat fibrous structure and multiple processes, displaying significant differentiation and obvious morphological variation (Figure 1(c)). The miR-122-5p inhibitor and its NC were transfected in blank group cells, and miR-122-5p mimic and its NC were transfected in TGF-β1 group cells. RT-qPCR result showed successful transfection (Figure 1(b), p < 0.01). Finally, the inverted microscope showed that miR-122-5p overexpression impeded LX-2 cell differentiation, while downregulation of miR-122-5p facilitated LX-2 cell differentiation (Figure 1(c)). miR-122-5p was poorly expressed in transforming growth factor-β1-stimulated LX-2 cells. (a) miR-122-5p expression in LX-2 cells was detected by RT-qPCR, (b) miR-122-5p expression in transfected cells was detected by RT-qPCR, and (c) cell morphology was observed under an inverted microscope. Cell experiment was repeated 3 times independently. Data were expressed as mean ± SD. Data in panels (a)(b) were analyzed using the t test, followed by Tukey’s multiple comparisons test. miR: microRNA; RT-qPCR: reverse transcription-quantitative polymerase chain reaction. **p < 0.01.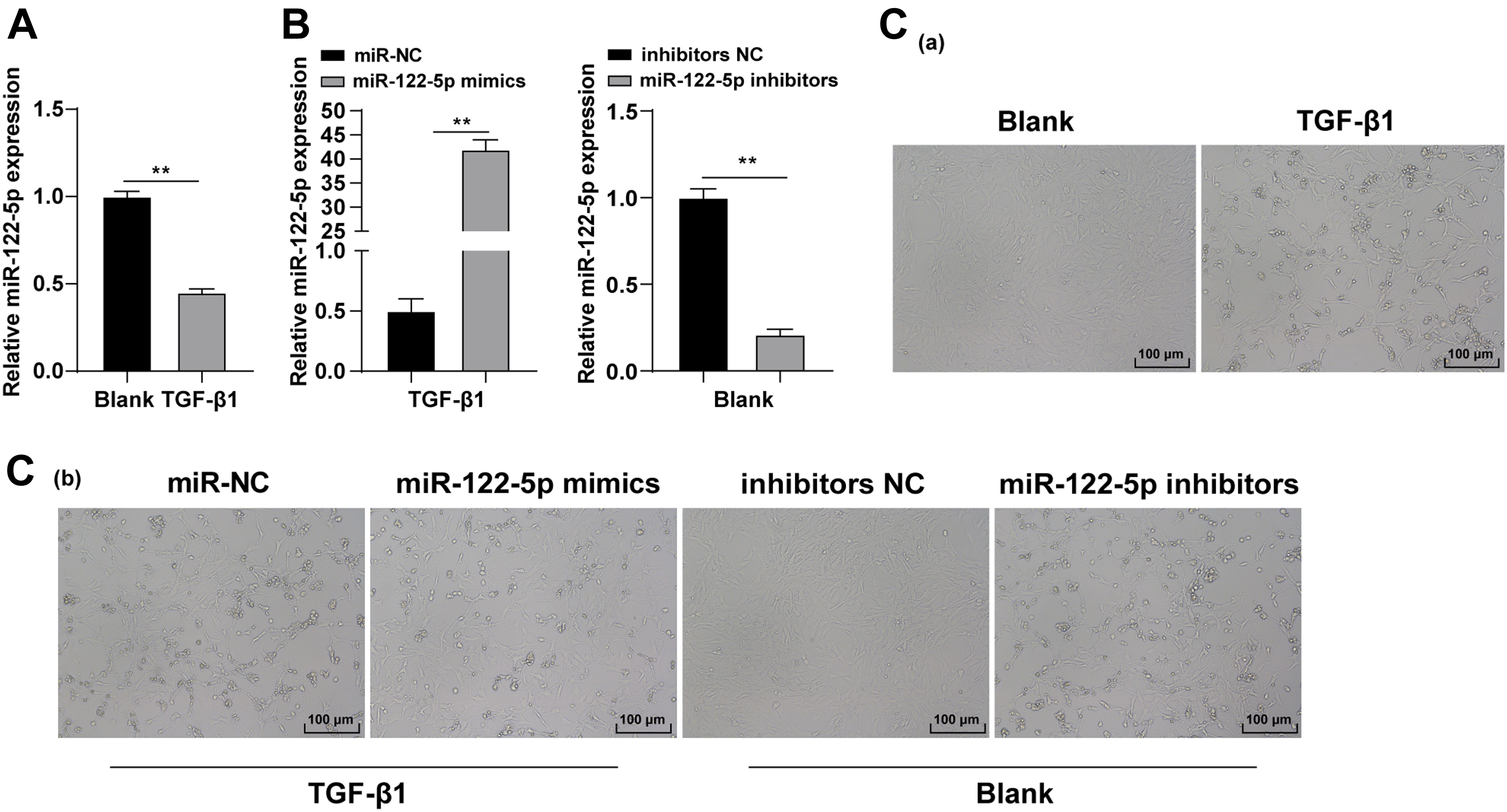
MicroRNA-122-5p overexpression inhibited expressions of collagen I and α-smooth muscle actin
Collagen I and α-SMA are common fibrosis markers.
31
The expressions of collagen I and α-SMA were determined by RT-qPCR. The result showed that mRNA expressions of collagen I and α-SMA were obviously lower in the miR-122-5p mimic group than that in the miR-NC group (Figure 2(a), p < 0.01). However, mRNA expressions of collagen I and α-SMA were remarkably higher in the miR-122-5p inhibitor group than that in the inhibitor NC group (Figure 2(a), p < 0.01). Western blot also showed that miR-122-5p overexpression reduced protein levels of collagen I and α-SMA while silencing miR-122-5p upregulated protein levels of collagen I and α-SMA (Figure 2(b), p < 0.01). miR-122-5p overexpression inhibited expressions of collagen I and α-SMA. miR-122-5p mimic and miR-NC were transfected into transforming growth factor-β1-stimulated LX-2 cells. miR-122-5p inhibitor and inhibitor NC were transfected into LX-2 cells. (a) mRNA expressions of collagen I and α-SMA were detected by Reverse transcription-quantitative polymerase chain reaction, and (b) protein levels of collagen I and α-SMA were detected by Western blot. Cell experiment was repeated 3 times independently. Data were expressed as mean ± SD. Data were analyzed using two-way analysis of variance, followed by Tukey’s multiple comparisons test. miR: microRNA; α-SMA: α-smooth muscle actin. **p < 0.01.
MicroRNA-122-5p overexpression inhibited LX-2 cell proliferation and promoted apoptosis
The inhibition of proliferation and the induction of apoptosis of stellate cells are effective measures of liver fibrosis management. Increasing evidence showed that miRNA participates in the regulation of HSC proliferation and apoptosis.32,33 Therefore, the proliferative and apoptotic ability of LX-2 cells in each group were determined by the CCK-8 method and flow cytometry. The CCK-8 result showed that the OD value and proliferative capacity of LX-2 cells were decreased at 72 h and 96 h after transfection in the miR-122-5p mimic group, while OD value and proliferative capacity of LX-2 cells in the miR-122-5p inhibitor group were increased at 72 h and 96 h after transfection (Figure 3(a), p < 0.01). Also, flow cytometry showed that the apoptotic rate of LX-2 cells was enhanced in the miR-122-5p mimic group relative to that in the miR-NC group, and the apoptotic rate of LX-2 cells was lowered in the miR-122-5p inhibitor group relative to that in the inhibitor NC group (Figure 3(b), p < 0.01). To sum up, miR-122-5p overexpression inhibited the proliferation of LX-2 cells and promoted cell apoptosis. MicroRNA-122-5p overexpression inhibited LX-2 cell proliferation and promoted apoptosis. (a) Cell proliferative capacity was evaluated by cell counting kit-8 method, (b) cell apoptosis was determined by flow cytometry. In images of flow cytometry, cells in the UL region represented necrotic cells, those in the UR region representing apoptotic cells at late stage, those in the LR region representing apoptotic cells at early stage, and those in the LL region representing living cells. Cell experiment was repeated 3 times independently. Data were expressed as mean ± SD. Data were analyzed using one-way ANOVA or two-way ANOVA, followed by Tukey’s multiple comparisons test. ANOVA: analysis of variance. UL: upper left; UR: upper right; LR: lower right; LL: lower left. **p < 0.01.
MicroRNA-122-5p targeted cellular-Abelsongene
Target relations between miR-122-5p and multiple genes were predicted using the Starbase database (Figure 4(a)). c-Abl is closely associated with liver fibrosis among the target genes.
34
Subsequently, based on the binding sites of miR-122-5p and c-Abl 3’UTR, the binding relation of miR-122-5p and c-Abl was verified by dual-luciferase reporter assay (Figure 4(b), p < 0.01). expression of c-Abl was detected by RT-qPCR. The result showed that miR-122-5p targeted c-Abl (Figure 4(c), p < 0.01). miR-122-5p targeted c-Abl. (a) The binding sites of miR-122-5p and c-Abl were predicted by bioinformatics website, (b) binding relationship between miR-122-5p and c-Abl was validated by dual-luciferase reporter assay, (c) mRNA expression of c-Abl was determined by reverse transcription-quantitative polymerase chain reaction. Experiment was repeated 3 times. Data were expressed as mean ± SD. Data were analyzed using one-way ANOVA or two-way ANOVA, followed by Tukey’s multiple comparisons test. **p < 0.01. miR: microRNA; ANOVA: analysis of variance; c-Abl: cellular-Abelsongene.
MicroRNA-122-5p inhibited hepatic stellate cell activation by suppressing the cellular-Abelsongene/histone deacetylases 2 pathway
It has been reported that c-Abl is associated with HSC activation.
35
HDAC2 is highly expressed in human liver fibrotic tissues.
36
Also, the c-Abl/HDAC2 pathway regulates cell malignant growth.
37
Therefore, we speculated that miR-122-5p might inhibit HSC activation by suppressing the c-Abl/HDAC2 pathway. The phosphorylation levels of p-c-Abl and p-HDAC2 were determined by Western blot. The result showed that the phosphorylation levels of p-c-Abl/c-Abl and p-HDAC2/HDAC2 were decreased after overexpression of miR-122-5p (Figure 5(a), p < 0.01). Then, cells treated with miR-122-5p overexpression were treated with the c-Abl/HDAC2 pathway activator DPH. Cell proliferation was detected by the CCK-8 method. The result showed that cell proliferative capacity was elevated in the miR-122-5p mimic + DPH group (Figure 5(b), p < 0.01). Flow cytometry result showed that cell apoptosis was attenuated in the miR-122-5p mimic + DPH group (Figure 5(c), p < 0.01). Finally, expressions of collagen I and α-SMA in each group were detected by RT-qPCR (Figure 5(d), p < 0.01), and the result suggested that the fibrosis degree of cells in the miR-122-5p mimic + DPH group was significantly increased. miR-122-5p inhibited HSC activation by suppressing the c-Abl/HDAC2 pathway. (a) Levels of p-c-Abl and p-HDAC2 were detected by Western blot, (b) cell proliferative capacity was evaluated by cell counting kit-8 method, (c) cell apoptosis was determined by flow cytometry, (d) mRNA expressions of collagen I and α-smooth muscle actin were determined by reverse transcription-quantitative polymerase chain reaction. In images of flow cytometry, cells in the UL region represented necrotic cells, those in the UR region representing apoptotic cells at late stage, those in the LR region representing apoptotic cells at early stage, and those in the LL region representing living cells. Cell experiment was repeated 3 times independently. Data were expressed as mean ± SD. Data were analyzed using one-way ANOVA or two-way ANOVA, followed by Tukey’s multiple comparisons test. c-Abl: cellular-Abelsongene; HDAC: histone deacetylases; ANOVA: analysis of variance. UL, upper left; UR, upper right; LR, lower right; LL, lower left. **p < 0.01.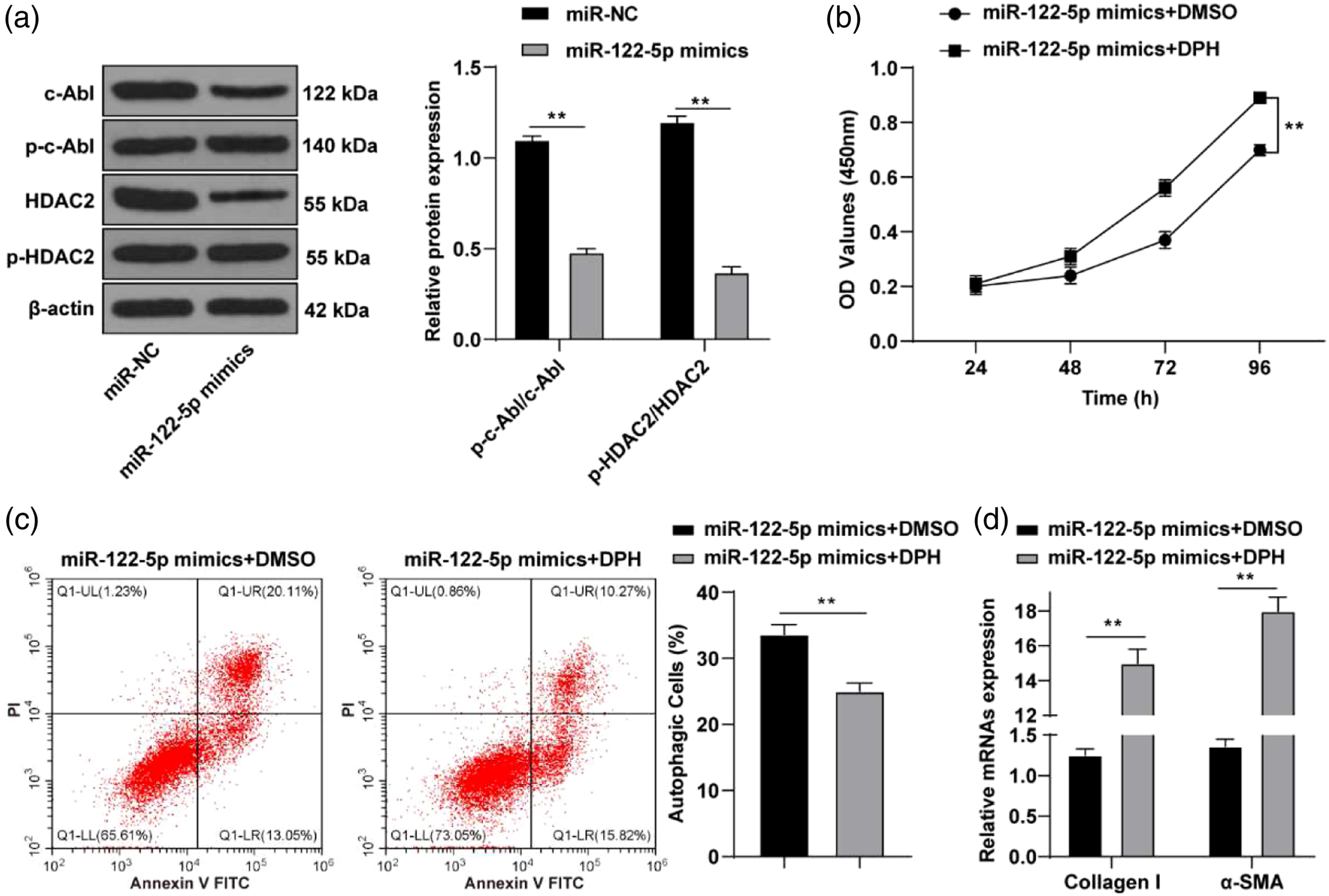
Discussion
Liver fibrosis is a chronic liver disease that constitutes a major health problem.
38
Some miRNAs have been suggested to correlate with liver fibrosis.
33
Therefore, investigating the role of miRNAs in liver fibrosis may expedite the discovery of efficacious treatment for liver fibrosis. This study found that miR-122-5p overexpression exerted an anti-fibrosis effect and suppressed HSC activation by inhibiting the c-Abl/HDAC2 pathway (Figure 6). miR-122-5p impeded HSC activation by inhibiting the c-Abl/HDAC2 pathway. miR-122-5p abated proliferation and strengthened apoptosis of HSCs by targeting c-Abl, and then repressing the activation of the c-Abl/HDAC2 pathway, which contributed to inhibition of HSC activation and ultimately led to counteraction of liver fibrosis. miR: microRNA; c-Abl: cellular-Abelsongene; HSC: hepatic stellate cell; HDAC: histone deacetylases.
Some miRNAs have been demonstrated to correlate with a variety of diseases by regulating the various biological processes of cells. 33 miR-122 is capable of manipulating the course of disease in gastric cancer and liver cancer by jeopardizing tumor cell multiplication and inducing tumor cell apoptosis.39,40 Moreover, miR-122 has significant functions in liver diseases. 41 In our study, the result showed that miR-122-5p was down-regulated in TGF-β1-stimulated HSCs. The structure of HSCs changes from star-shaped to that of fibroblast-like cells in pathological conditions like liver cirrhosis. 42 In our study, TGF-β1-stimulated cells changed from fibroblasts in a large star or fusiform shape to fibrocytes in spindle shape with flat fibrous structure and multiple processes. The excessive deposition of α-SMA and collagen I are typical characteristics of liver fibrosis. 31 The reintroduction of miR-122 was reported to suppress expressions of α-SMA and collagen I in hepatic fibrogenesis. 43 Our result showed that collagen I and α-SMA were remarkably lowered in the miR-122-5p mimic group, but significantly enhanced in the miR-122-5p inhibitor group. miR-122 was demonstrated to negatively regulate the production of collagen in HSCs. 44 Collectively, miR-122-5p overexpression inhibited the expressions of collagen I and α-SMA in LX-2 cells.
HSC activation participates in the progression of liver fibrosis and apoptosis plays a pivotal role in HSC activation.45,46 In addition to its ability to regulate collagen production, miR-122 inhibits HSC proliferation. 24 In our study, proliferative capacity of cells in the miR-122-5p mimic group was decreased at 72 h and 96 h after transfection, while OD value and proliferative capacity of cells in the miR-122-5p inhibitor group were increased at 72 h and 96 h after transfection. Also, the apoptotic rate of cells was elevated in the miR-122-5p mimic group and the apoptotic rate of cells was lowered in the miR-122-5p inhibitor group. The transportation of extracellular miR-122 to HSCs led to the inhibition of HSC proliferation. 47 Overexpression of miR-122 induced the apoptosis of hepatocellular carcinoma. 48 Taken together, miR-122-5p overexpression inhibited LX-2 cell proliferation and promoted LX-2 cell apoptosis.
In order to explore the downstream mechanism of miR-122-5p on liver fibrosis, target genes of miR-122-5p were predicted using the Starbase database. Among the target genes, the inhibition of c-Abl attenuates liver fibrosis. 49 Therefore, we predicted the binding sits of miR-122-5p and c-Abl and verified their binding relation using dual-luciferase reporter assay. The result showed that c-Abl mRNA expression was inhibited by miR-122-5p. Increasing evidence indicated that c-Abl could stabilize HDAC2 which plays a key part in HSC activation and liver fibrosis.36,50 Therefore, it was speculated that miR-122-5p inhibited HSC activation by suppressing the c-Abl/HDAC2 pathway. In our study, the result showed that the phosphorylation levels of p-c-Abl/c-Abl and p-HDAC2/HDAC2 were decreased after overexpression of miR-122-5p. The proliferative ability was increased while apoptosis was decreased in cells treated with the combination of miR-122-5p overexpression and the c-Abl/HDAC2 pathway activator DPH. The fibrosis degree was also increased. The c-Abl/HDAC2 signaling pathway participates in the malignant bovine mammary epithelial cell growth. 37 HDAC2 knockdown contributed to the downregulation of collagen I and α-SMA in cardiac fibrosis. 28 Similarly, our study demonstrated that miR-122-5p inhibited HSC activation by suppressing the c-Abl/HDAC2 pathway.
In conclusion, this study suggested that miR-122-5p overexpression exerted an anti-fibrosis effect by inhibiting HSC activation by suppressing the c-Abl/HDAC2 pathway. However, this study only simply revealed the effect of miR-122-5p on HSC proliferation and apoptosis by modulating the c-Abl/HDAC2 pathway. The specific functions of miR-122-5p in the management of liver fibrosis in clinic were left undiscovered. Further study is necessary to accurately identify the curative effect of miR-122-5p overexpression on patients with liver fibrosis.
Footnotes
Author contributions
ZYW is the guarantor of integrity of the entire study; ZYW contributed to the study concepts, study design, and definition of intellectual content; ZYW contributed to the literature research; LPY contributed to the manuscript preparation, and LPY contributed to the manuscript editing and review; ZYW and JBW contributed to the clinical studies; ZYW and JBW contributed to the experimental studies and data acquisition; JBW and JYF contributed to the data analysis and statistical analysis. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability
All the data generated or analyzed during this study are included in this published article.
