Abstract
Methotrexate (MTX) is frequently used drug in treatment of cancer and autoimmune diseases. Unfortunately, MTX has many side effects including the hepato-renal toxicity. In this study, we hypothesized that Luteolin (Lut) exhibits protective effect against the MTX-induced hepato-renal toxicity. In order to investigate our hypothesis, the experiment was designed to examine the effect of exposure of male rats to MTX (20 mg/kg, i.p., at day 9) alone or together with Lut (50 mg/kg, oral for 14 days) compared to the control rats (received saline). The findings demonstrated that MTX treatment induced significant increases in the liver and kidney functions markers in serum samples including Aspartate transaminase (AST), Alanine transaminase (ALT), creatinine, urea and uric acid. MTX also mediated an oxidative stress expressed by elevated malondialdehyde (MDA) level and decreased level of reduced glutathione (GSH), antioxidant enzyme activities, and downregulation of the Nrf2 gene expression as an antioxidant trigger. Moreover, the inflammatory markers (NF-κB, TNF-α, and IL-1β) were significantly elevated upon MTX treatment. In addition, MTX showed an apoptotic response mediated by elevating the pro-apoptotic (Bax) and lowering the anti-apoptotic (Bcl-2) proteins. All of these changes were confirmed by the observed alterations in the histopathological examination of the hepatic and renal tissues. Lut exposure significantly reversed all the MTX-induced changes in the measured parameters suggesting its potential protective role against the MTX-induced toxicity. Finally, our findings concluded the antioxidative, anti-inflammatory and anti-apoptotic effects of Lut as a mechanism of its protective role against the MTX-induced hepato-renal toxicity in rats.
Introduction
Methotrexate (MTX)—a folic acid antagonist—is an effective anticancer drug, used to treat many cases such as lymphoma, leukemia, breast cancer and osteosarcoma. 1,2 Also, MTX is widely used to treat autoimmune diseases including rheumatoid arthritis and psoriasis. 3 MTX has been proved to induce hepatotoxicity showing tissue damage, liver dysfunction, fibrosis and cirrhosis with remarkable alterations in the hepatic function biochemical markers and the histological structure. 4 MTX metabolites are excreted through the kidney and precipitate in the renal tubules, this induces nephrotoxicity and the high level of creatinine and blood urea nitrogen lead to renal failure. 5 However, the mechanisms of the MTX-mediated hepato-renal toxicity have been studied by many researchers, 6,7 the protection against this toxicity is still obscured. Although many drugs have been tested to decrease the MTX-induced side effects, nothing has shown a high efficiency when used in the clinical trials. Therefore, finding a potential protective therapy against the MTX-induced hepato-renal toxicity becomes an important research topic recently.
Herbal therapy, plant-derived products, has been widely used to treat several diseases. 8,9 This is due to their higher safety and lower side effects compared to the synthetic therapy. 10 Luteolin (Lut) - 3,4,5,7 tetrahydroxyflavone - is one of the plant-derived flavonoids, which found in many different plants including peppers, thyme, carrot, rosemary, oregano, parsley, onion leaves and celery. 11 Many reports mentioned the beneficial effects of Lut in treatment of many diseases such as diabetes, liver and cardiovascular defects. 12,13 Lut exhibits its protective role via different known biological effects including the antioxidative, anti-inflammatory, anti-mutagenic and anti-tumorigenic effects. 14,15 A recent study has concluded that the antagonistic effect of Lut against the lead acetate-mediated hepatotoxicity is occurred via the antioxidant, anti-apoptotic and anti-inflammatory properties. 14 Moreover, Lut has protected against the diabetic nephropathy by inducing changes in the oxidant/antioxidant capacity. 16 Therefore, this research aimed to explore the potential protective role of Lut against the MTX-induced hepato-renal toxicity and its molecular mechanism in rats.
Material and methods
Chemicals
Methotrexate (vial 50 mg) was purchased from ACDIMA International (Cairo, Egypt), and luteolin (C19H18O6; CAS number 855-97-0) was purchased from Alfa Aesar (Kandel, Germany). Firstly, Lut was dissolved in DMSO (dimethyl sulfoxide) and then diluted with normal saline (0.9% NaCl); the final solution to be administered contained 2.5% DMSO, which corresponded to 10 µl per rat. Control rats received the same amount of DMSO (2.5% DMSO in saline).
Experimental animals and ethics approval
Thirty-two male Wistar Albino rats were used in this experiment. Rats were obtained from EgyVac (Giza, Egypt) at age of 8-weeks old in a clinical healthy condition. Polypropylene cages were used for housing in a clean room and controlled conditions (23 ± 2°C temperature, 55–60% humidity and 12 h light then 12 h dark cycles). Rats were fed on standard pellets of food and water ad libitum. Ethics committee for laboratory animals care and experimentation has approved this experimental animal study at the Zoology department, Faculty of Science, Helwan University (Approval number: HU2020/Z/AEG0220-07). The protocol followed the guidelines (8th edition) of the National Institute of Health (NIH) regarding to the care and use of experimental animals.
Experimental protocol
After two weeks of acclimatization, rats were randomly distributed into four groups (n = 8). The first one is the control group which received 3 ml/kg B.wt normal saline solution containing 2.5% DMSO orally for successive 14 days and a single intraperitoneal injection of 1 ml/kg B.wt normal saline solution on the day 9 of the experiment. The second group is the Lut group which orally received 50 mg/kg B.wt/day of Lut for successive 14 days according to Al-Megrin et al. method 17 ; and a single intraperitoneal injection of 1 ml/kg B.wt normal saline solution on the day 9. The third group is the positive MTX-intoxicated group (MTX) which received a single intraperitoneal injection of 20 mg/kg B.wt of MTX on day 9 as an optimal dose to induce the toxicity 18 ; and orally received 3 ml/kg B.wt normal saline solution containing 2.5% DMSO for successive 14 days. The fourth group is the Lut+MTX group which orally received 50 mg/kg B.wt/day of Lut for successive 14 days; and a single intraperitoneal injection of 20 mg/kg B.wt of MTX on day 9 of the experiment. The health condition of all experimental animals was observed daily till the end of the study.
Samples collection
After 24 h of the last treatment, all rats were euthanized by intraperitoneal injection of 300 mg/kg sodium pentobarbital (Sigma Aldrich) to minimize the stress in the animals and then sacrificed by cervical dislocation. All samples were collected at the terminal stage of the study, so the blood samples were obtained from the abdominal aorta immediately after sacrificing and serum samples were separated and kept at -80°C for biochemical analysis. Samples of liver and kidney tissues were collected and washed twice in 50 mM Tris–HCl of neutral pH and each sample was divided into three parts. First part of samples was minced and mixed with 10 mM phosphate buffer to get the tissue homogenate. Then, homogenates were centrifuged at 3000x g for 10 min. at 4°C and supernatants were then used in the biochemical analysis. Second part of tissues were kept in 10% buffered formalin for fixation to be used in the histopathological examination. The last part of the tissue samples was kept at -80°C for the real-time PCR analysis.
Assessment of liver and kidney function biomarkers
The enzymatic activities of liver function biomarkers; aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were calorimetrically estimated in serum samples. Also, serum samples were analyzed for kidney function biomarkers including uric acid, urea and creatinine levels. All biomarkers were analyzed using commercially available standard kits (Randox Laboratories Ltd., Crumlin, UK) following the manufacturer’s protocol.
Assessment of hepatic and renal oxidative stress markers
Levels of lipid peroxidation in liver and kidney tissues were assessed in the tissues homogenates and expressed by malondialdehyde (MDA), a secondary product of lipid peroxidation, based on method of Ohkawa et al. 19 Also, the reduced glutathione (GSH) levels were analyzed by Ellman’s method 20 that relied on the ability of glutathione to convert the 5,5-dithiobis (2-nitrobenzoic acid) into yellow-colored 5-thionitrobenzoic acid.
Assessment of the antioxidant capacity
Superoxide dismutase (SOD) activity was measured spectrophotometrically at 560 nm using method of Nishikimi et al. 21 This method based on the inhibition of phenazine methosulphate-mediated reduction of the nitroblue tetrazolium dye by SOD enzyme. Also, catalase (CAT) activity was determined following Aebi’s method. 22 The method depends on the decomposition of H2O2 by catalase enzyme and is measured at 510 nm. In addition, the activity of glutathione peroxidase (GPx) was assessed spectrophotometrically by Paglia and Valentine’s method 23 and glutathione reductase (GR) was assessed according to De Vega et al. 24 method.
Analysis of inflammatory biomarkers
The levels of NF-κB p65, a subunit of the NF-kappa-B (NF-κB) transcription complex, tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β) in tissues were analyzed by ELISA kit purchased from Thermo Fisher Scientific depending on the manufacturer’s protocol.
Determination of apoptotic biomarkers
The protein levels of Bcl-2 (B-cell lymphoma 2) and Bax (bcl-2-like protein 4) in hepatic and renal tissues were measured by ELISA kits (CSB-E13604r and CSB-EL002573RA, respectively) obtained from (CUSABIO Life Sciences, Wuhan, China). The procedure was done according to the manufacturer’s instructions.
Gene expression analysis
Nuclaer factor erythroid 2–related factor 2 (Nrf2) is an essential transcription factor which controls the antioxidant signals in liver. Reverse transcription-quantitative polymerase chain reaction (RT-qPCR) analysis was used to analyze the Nrf2 gene expression. Total RNA was extracted from liver and kidney using TRIzol® reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. An aliquot of the extracted total RNA was reverse transcripted into cDNA. The employed primer sequences of Nrf2 gene were 5′-GGTTGCCCACATTCCCAAAC-3′ (forward) and 5′-GGCTGGGAATATCCAGGGC-3′ (reverse). β-actin was used as a reference housekeeping gene to normalize the data. The employed primer sequences for β-actin gene were 5′-GCAGGAGTACGATGAGTCCG-3′ (forward) and 5′-ACGCAGCTCAGTAACAGTCC-3′ (reverse). Real-time PCR reactions were conducted using Power SYBR® Green PCR Master Mix, (Applied Biosystems, Life Technologies, Foster, CA, USA) and then the analysis was performed using a 7500 Real-Time PCR System (Applied Biosystems, Foster City, CA, USA). The thermocyclic condition for the PCR analysis was 95°C for 4 min, 40 cycles at 95°C for 10 s, 60°C for 30 s, and 72°C for 10 s. mRNA expression was presented as a relative fold change in the expression compared to the control expression.
Histopathological examination
Specimens of liver and kidney tissues were fixed in 10% buffered formalin for 24 h, dehydrated, and paraffinized in wax. Then, sections of 5-µm thickness were prepared and stained with hematoxylin and eosin (H&E) for light microscopy examination. Slides were examined using a Nikon microscope (Eclipse E200-LED, Tokyo, Japan) by a magnification power 400×.
Immunohistochemical examination
The paraffin sections of hepatic and renal tissues were mounted on glass slides. To reveal the antigen sites, sections were washed using boiling water after treatment with 0.03% H2O2 in methanol for 10 min to prevent the activity of the endogenous peroxidase. After that, Sections were exposed to the anti-caspases 3 antibody (Santa Cruz, CA, USA) according to the manual instructions. After washing the sections using phosphate buffer saline (PBS), they were incubated with secondary anti-rabbit antibody conjugated to horseradish peroxidase for 30 min. Finally, sections were incubated with chromogen 3,3′-diaminobenzidine tetrachloride (DAB-H2O2) for 10 min at room temperature. Hepatic and renal sections were examined using 400 x magnification lens (Nikon Eclipse E200-LED, Tokyo, Japan).
Statistical analysis
All data were subjected to statistical analysis using SBSS software program. The analyzed values were presented as the mean ± standard deviation (SD). One-way ANOVA with Duncan multiple comparison tests were used to determine the difference between the mean values of the different groups. P value less than 0.05 was considered statistically significant.
Results
Liver and kidney functions biomarkers
Liver transaminases; aspartate aminotransferase (AST) and alanine aminotransferase (ALT) are useful biomarkers in case of liver injury which are associated with hepatic cellular integrity. In case of kidney function tests, creatinine, urea and uric acid levels are the most related biomarkers to the renal dysfunction. Therefore, the mentioned liver and kidney function biomarkers were determined in serum samples to reveal the hepatic and renal function condition.
As shown in Table 1, all the liver and kidney functions biomarkers were significantly increased (p < 0.05) in MTX-treated group compared to the control (CNT) group. While, the group treated with luteolin before and after the methotrexate injection (Lut+MTX group) showed significant decreases in all the liver and kidney functions biomarkers compared to the MTX-treated group. However, luteolin alone didn’t show any significant difference compared to the control group. These results demonstrate that the luteolin can improve the hepatic and renal functions against the MTX-induced hepato-renal toxicity.
Levels of liver and kidney functions biomarkers in serum samples after exposure of male rats to luteolin (Lut, 50 mg/kg, oral for 14 days) and/or methotrexate (MTX, 20 mg/kg, i.p., at day 9).

All values represent the mean ± SD (n = 8). #, $ represent the statistical significance compared to the control group and the MTX-treated group respectively, at p-values < 0.05.
Hepatic and renal oxidative stress
Oxidative stress is enhanced by the over production of the reactive oxygen species (ROS) along with the alteration in the antioxidant system. Oxidative stress leads to lipid peroxidation with a higher level of malondialdehyde (MDA) as a secondary product. Also, it induces an alteration in the level of the GSH as an important scavenger of ROS. Therefore, the protein levels of MDA and GSH were measured in hepatic and renal tissues homogenates.
As shown in Figure 1(a), in both liver and kidney tissues, MDA levels were significantly increased (p < 0.05) in MTX-treated group compared to the control (CNT) group. While, the exposure of rats to luteolin before and after the methotrexate injection (Lut+MTX group) showed significant decreases in MDA levels compared to the MTX-treated group. In kidney tissue only, luteolin-treated group showed a significant decrease in MDA level compared to the control group. In Figure 1(b), GSH levels revealed significant decreases (p < 0.05) in MTX-treated group compared to the control (CNT) group in both liver and kidney tissues. Reversely, Lut+MTX-treated group showed significant increases of GSH levels compared to the MTX-treated group. The data of the oxidative stress markers demonstrate that the luteolin reverses the MTX-induced changes in MDA and GSH levels.
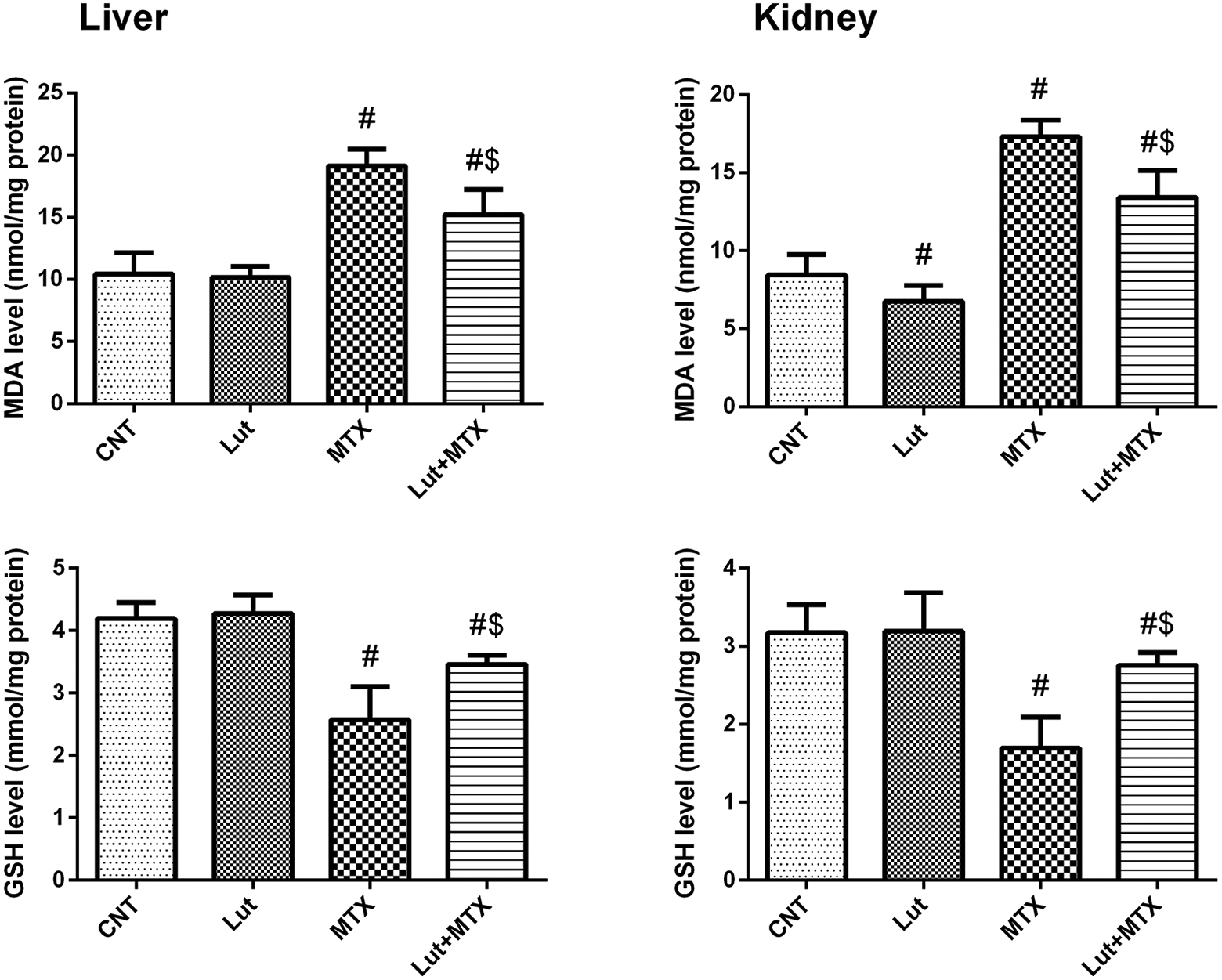
Hepatic and renal protein levels of oxidative stress markers; malondialdehyde (MDA) and reduced glutathione (GSH) after exposure of male rats to luteolin (Lut, 50 mg/kg, oral for 14 days) and/or methotrexate (MTX, 20 mg/kg, i.p., at day 9). All values represent the mean ± SD (n = 8). #, $ represent the significant difference compared to the control group and the MTX-treated group, respectively, at p-values < 0.05.
The antioxidant capacity in hepatic and renal tissues
To evaluate the antioxidant role of luteolin against the MTX-induced oxidative stress, the activities of SOD, CAT, GPx and GR were measured in the homogenates of hepatic and renal tissues and expressed by protein level/mg. As shown in Figure 2, in both hepatic and renal tissues, the activities of SOD (A), CAT (B), GPx (C) and GR (D) were significantly decreased in the MTX-treated group compared to the control (CNT) group. While, the treatment of rats with luteolin before and after the methotrexate injection (Lut+MTX group) showed significant increases in all the inhibited activities compared to the MTX-treated group. These data demonstrate that the luteolin has an antioxidant effect against the MTX-induced oxidative stress by reversing the antioxidants enzymes alterations.

The hepatic and renal antioxidant capacity expressed by the activities of superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), and glutathione reductase (GR). Rats were exposed to luteolin (Lut, 50 mg/kg, oral for 14 days) and/or methotrexate (MTX, 20 mg/kg, i.p., at day 9). All values represent the mean ± SD (n = 8). #, $ represent the significant difference compared to the control group and the MTX-treated group, respectively, at p-values < 0.05.
Inflammatory markers
In this study, methotrexate (MTX) exposure mediated an inflammatory response in liver and kidney through the significant elevation (p < 0.05) in the inflammatory markers levels; A) NF-κB p65, B) TNF-α, and C) IL-1β, compared to the control group as shown in Figure 3. Conversely, Lut treatment before and after the methotrexate injection reduced the hepatic and renal inflammation as evidenced by the significant decrease in the inflammatory markers in the Lut+MTX-treated group. It is notable in Figure 3 that the Lut+MTX-treated group still showed significant increases in the inflammatory markers levels when compared to the control group. The inflammatory markers data demonstrate the anti-inflammatory role of luteolin against the MTX-induced inflammatory response.

The inflammatory markers in both hepatic and renal tissues. Nuclear factor Kappa B subunit P65 (NF-κB), tumor necrosis factor alpha (TNF-α), and interleukin 1β (IL-1β) were investigated in male rats exposed to luteolin (Lut, 50 mg/kg, oral for 14 days) and/or methotrexate (MTX, 20 mg/kg, i.p., at day 9). All values represent the mean ± SD (n = 8). #, $ represent the significant difference compared to the control group and the MTX-treated group, respectively, at p-values < 0.05.
Apoptotic markers
To evaluate the apoptotic response in liver and kidney, the protein levels of the anti-apoptotic (Bcl-2) and the pro-apoptotic (Bax) proteins were assessed by ELISA in the tissues. In comparison with the control group in both liver and kidney tissues, the MTX-treated group showed the apoptotic effect as revealed by the significant decrease in the anti-apoptotic Bcl-2 level (Figure 4(a)), along with the significant increase in the pro-apoptotic Bax level (Figure 4(b)). In contrast, luteolin treatment before and after the methotrexate injection (Lut+MTX) showed an anti-apoptotic effect as revealed by the significant increase in the anti-apoptotic Bcl-2 level (Figure 4(a)), along with the significant decreased in the pro-apoptotic Bax level (Figure 4(b)) compared to the MTX-treated group. Interestingly, in the Lut+MTX-treated group, the pro-apoptotic Bax level in both liver and kidney tissues, also the anti-apoptotic Bcl-2 level in liver tissue showed no significant difference compared to the control group.
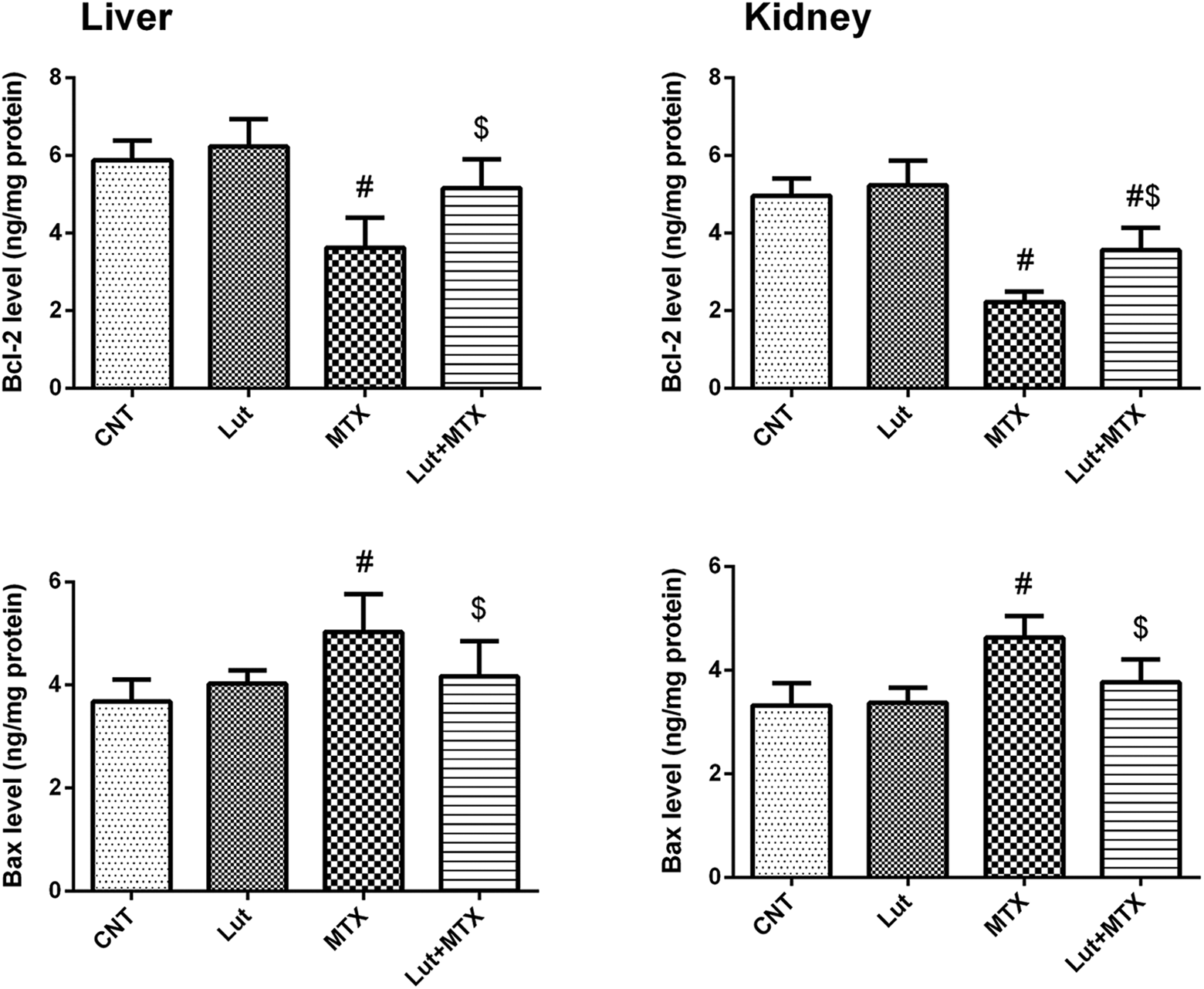
The apoptotic markers in both hepatic and renal tissues. The protein levels of the anti-apoptotic marker (Bcl-2), and pro-apoptotic marker (Bax) were measured by ELISA after exposure of male rats to luteolin (Lut, 50 mg/kg, oral for 14 days) and/or methotrexate (MTX, 20 mg/kg, i.p., at day 9). All values represent the mean ± SD (n = 8). #, $ represent the significant difference compared to the control group and the MTX-treated group, respectively, at p-values < 0.05.
These data were confirmed using the immunohistochemical examination of both liver and kidney tissues for the signaling of caspase 3 as an apoptotic protein. As shown in Figure 5, The MTX-treated group potentiated the apoptotic effect by increasing the immunostaining intensity signal of caspase 3 in both liver and kidney tissues. The caspase 3 intensity signals were reduced in the Lut+MTX-treated group in both tissues. These data demonstrate that the luteolin has an anti-apoptotic effect against the MTX-induced apoptosis by reversing the changes in the apoptotic markers to the normal levels.
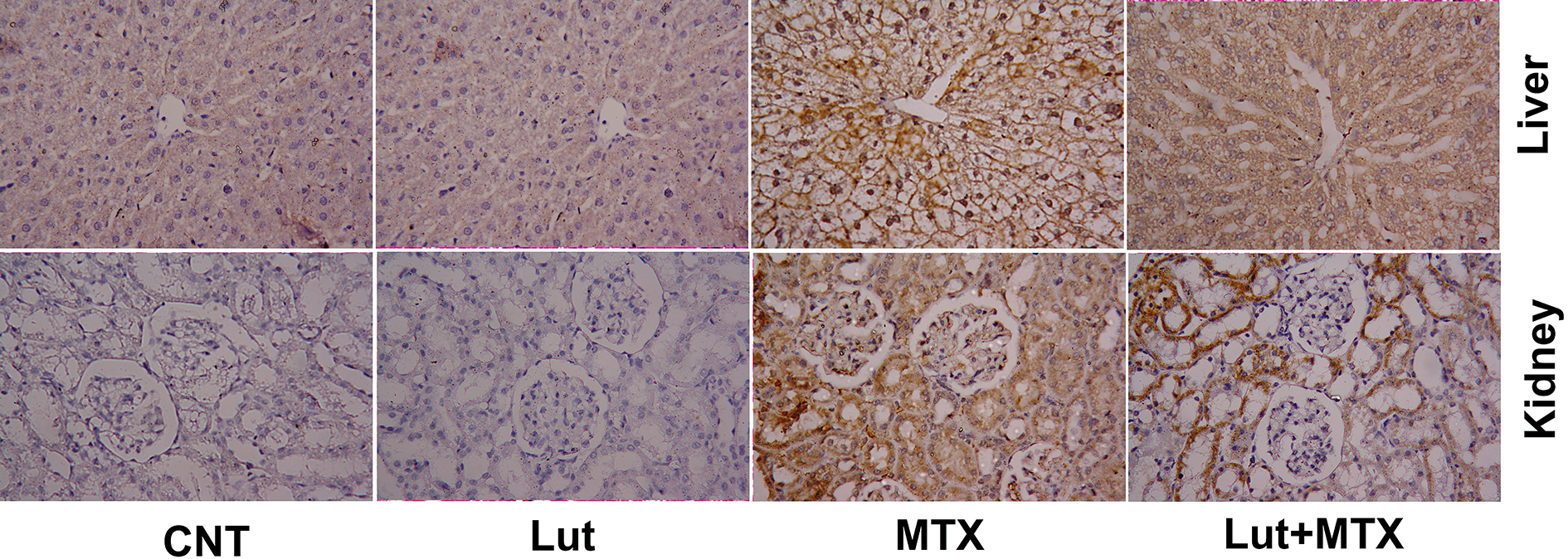
Photomicrographs showing changes in caspase-3 expression in both liver and kidney tissues by the immunohistochemical examination. Male rats were exposed to luteolin (Lut, 50 mg/kg, oral for 14 days) and/or methotrexate (MTX, 20 mg/kg, i.p., at day 9). The used magnification was 400×.
Nrf2 gene expression
The transcription factor Nrf2 plays an essential role in both the antioxidant and the anti-inflammatory cellular responses. Therefore, to elucidate if the Nrf2 has a role in the molecular mechanism of the luteolin’s effect against the MTX-induced oxidative stress and inflammatory response or not, Nrf2 gene expression was determined in both liver and kidney. As shown in Figure 6, MTX-treated group exhibited significant downregulation in Nrf2 gene expression compared to the control group. In contrast, Lut+MTX-treated group showed significant upregulation of Nrf2 expression compared to the MTX-treated group. Also, the downregulation of Nrf2 gene expression was still notable in the Lut+MTX-treated group compared to the control one. The gene expression analysis data demonstrate the effect of the transcription factor Nrf2 in the molecular antioxidant and anti-inflammatory mechanisms of the luteolin against the MTX-induced oxidative and inflammatory responses.

Expression of the nuclear factor erythroid 2-related factor 2 (Nrf2) mRNA in both hepatic and renal tissues of rats exposed to luteolin (Lut, 50 mg/kg, oral for 14 days) and/or methotrexate (MTX, 20 mg/kg, i.p., at day 9). mRNA expression was measured using RT-PCR. All values represent the mean ± SD (n = 8). #, $ represent the significant difference compared to the control group and the MTX-treated group, respectively, at p-values < 0.05.
Histopathological changes in liver and kidney
Liver
The histological microphotograph of the liver tissue stained with H&E methods of control and Lut-treated alone rats showing central veins with normal intact hepatocytes surrounding it (Figure 7). However in the MTX-treated rats, the hepatic strands showed disarrangement, and sinusoids dilatation with many hepatocyte vacuolation. Massive inflammatory cells infiltrated in the liver in the MTX-treated group were also observed. However, Lut pre-treatment efficiently attenuated the hepatic lesions, which were healthier than those observed in the MTX-treated groups.

Microphotographs of histological changes in the liver and kidney following treatment with luteolin (Lut, 50 mg/kg, oral for 14 days) and/or methotrexate (MTX, 20 mg/kg, i.p., at day 9). Scale bar = 50 µm (400×).
Kidney
Furthermore, the representative microphotograph of the kidney tissues showed the normal morphological structures of the glomerulus and renal tubules in the control and Lut-treated rats. However, in the MTX-treated rats, kidney tissue showed congested glomerular capillaries, edema within Bowman’s corpuscles, vacuolar degeneration, infiltration of inflammatory cells in the interstitial area and debris in the lumen of some renal tubules. Lut pre-treatment markedly improved the renal lesions in MTX-intoxicated rats.
Discussion
MTX is one of the most common drugs which treats cancer and many autoimmune diseases. 25 The MTX-cytotoxicity can also affect the normal healthy cells of vital organs, along with the tumor cells. Liver and kidney are the most affected organs showing the signs of hepato-renal toxicity. 26 The mechanisms of the MTX-induced hepato-renal toxicity have been explained using different pathways including the inflammatory, oxidative stress and the apoptotic pathways. 27,28 In the current study, we used Lut as an herbal therapy to investigate its protective role against the MTX-induced toxicity. Therefore, we chose a safe dose of Lut (50 mg/kg, orally) which provided a protective role without any side effect. 29 Moreover, a high dose of Lut (100 mg/kg) was proved to protect diabetic rats from ischemia/reperfusion injury with no cytotoxic effect 30 which provide a higher safe area to the chosen dose in our study. Our findings proved that the luteolin confers a protective effect against the MTX-induced hepato-renal toxicity. Also, we suggested the mechanism of such protection through the antioxidant, anti-inflammatory and anti-apoptotic activities.
In the current study, MTX-induced disturbances in the hepatic and renal functions, evidenced by the significant increases in the hepatic and renal function biomarkers including AST, ALT, creatinine, urea and uric acid as shown in Table 1. The elevated levels of the biochemical markers are usually related to the leakage of the enzymes into the circulation because of changes in the cell membrane permeability and the loss of the structural integrity. 31 This was confirmed by the changes in the histological architecture of liver and kidney tissues as shown in Figure 7 and agreed with many previous studies. 32,33 To elucidate the protective role of Lut as an herbal therapy, group of rats were orally administered luteolin for 14 successive days with a single i.p injection of MTX at day 9 to induce the toxicity. This group (Lut+MTX-treated rats) showed an improvement in the liver and kidney functions, evidenced by the reduction in the MTX-induced increases in AST, ALT, creatinine, urea and uric acid levels. This could be explained that the luteolin can improve the cell membrane permeability and increase the clearance of creatinine and urea by the kidney. The protective effect of Lut was also confirmed by the less damage which observed in the histopathological examination of both liver and kidney tissues. Therefore, Lut treatment has the ability to improve the hepatic and renal functions after induction of toxicity by MTX.
To investigate the molecular mechanisms involved in the MTX-induced hepato-renal toxicity and the protective effect of the luteolin, three different pathways were investigated including the oxidative stress, inflammation and apoptosis. In both hepatic and renal tissues (Figures 1 and 2), MTX mediated an oxidative stress, imbalance between the oxidants and antioxidants, through increasing the lipid peroxidation expressed by elevated MDA as a secondary product. Also, MTX reduced the GSH level- the most important cellular antioxidant- and the activities of the antioxidant enzymes SOD, CAT, GPx and GR. The production of the antioxidant enzymes is controlled by the Nuclear factor erythroid 2-related factor 2 (Nrf2) that has an essential role in the antioxidative mechanism. When the oxidative stress occurred, Nrf2 liberates from Kaap1, transcripted in the nucleus and then stimulates the production of the antioxidant enzymes such as SOD, CAT, GPx and GR. 34 MTX-induced significant downregulation in the Nrf2 gene expression (Figure 6) in consistence with the reduction in the activities of the antioxidant enzymes. The oxidative stress lead to many pathological and cellular damages as confirmed by the histopathological study of both liver and kidney tissues. These results were in agreement with previous reported data about the MTX-oxidative stress. 35,36 Luteolin itself is known to have antioxidant activity. 12 Our results showed that Lut exhibited a significant upregulation of Nrf2 gene expression and thus reversed the MTX-induced lipid peroxidation (MDA), changes in the GSH level and in the SOD, CAT, GPx and GR activities in both hepatic and renal tissues. Consequently, Lut enhanced the scavenging of the free radicals with decrease in reactive oxygen species (ROS) level. These data suggested that Lut has an antioxidant effect against the MTX-induced oxidative stress via the Nrf2 signal transduction (Figure 8).

Illustration explains the molecular mechanism of the Lut effect against the MTX-induced hepato-renal toxicity via its antioxidant, anti-inflammatory, and anti-apoptotic activities. MTX mediates the ROS production and the oxidative stress which then stimulate the NF-κB-inflammatory response and the apoptosis. In contrast, Lut stimulates the Nrf2 signal transduction which triggers the antioxidant activity and reveals the anti-inflammatory and the anti-apoptotic response. The red arrows (→) indicate the stimulatory effects of MTX. The green arrows (→) indicate the stimulatory effects of Lut, while the green blockers (⊥) indicate the Lut inhibitory effect.
The inflammatory pathway depending on the NF- κB transcription is an important mechanism which previously reported to be involved in the MTX-induced inflammatory response by increasing the interleukins expression. 37 In our study, MTX mediated the inflammatory response in liver and kidney through elevation of the protein levels of NF- κB, TNF-α, and IL-1β. While, the Lut+MTX-treated group revealed significant decreases in the MTX-induced elevated inflammatory markers as shown in Figure 3. These findings agreed with previous studies which concluded that the Lut treatment reduced the inflammatory response via lowering the pro-inflammatory cytokines in hepatic tissue. 12,38 Moreover, Lut antagonized the mercury-induced inflammation by reducing the NF-κB expression. 38 Hence, our data clarified that the Lut shows an anti-inflammatory activity against the MTX-induced inflammatory response.
Apoptosis is a programed cell death and a properly regulated process. It could be initiated either by the intrinsic pathway, cell kills itself responding to the internal stress, or the extrinsic pathway, cell kills itself depending on signals from another cells. 39 The relationship between Bax as a pro-apoptotic protein and Bcl-2 as an anti-apoptotic protein can regulate the exposure of cells to the apoptosis. 40 Therefore, the effect of MTX and Lut on these proteins and the apoptotic pathway was investigated in this study. Our results, in accordance with previous report, 41 showed that MTX mediated an apoptotic effect via elevated Bax and decreased Bcl-2 levels revealing the change in the pro-apoptotic/anti-apoptotic markers quantification. Also, the immunohistochemical intensity signal of the apoptotic caspase 3 was increased in both hepatic and renal tissues revealing the apoptotic effect of the MTX (Figure 5). These data demonstrate the mechanism underlying the MTX-induced apoptosis; the MTX-induced mitochondrial stress which affects the mitochondrial membrane depolarization, thus lead to caspase 3 activation and Bax/Bcl-2 release which then trigger the mitochondrial apoptotic pathway. To clarify the protective role of Lut against the MTX-induced apoptosis, the apoptotic markers (caspase 3, Bax and Bcl-2) were investigated after Lut treatment with MTX exposure. Lut treatment significantly reversed the MTX-induced changes in caspase 3, Bax and Bcl-2 levels showing its anti-apoptotic activity. The mechanism of the anti-apoptotic activity of luteolin could be explained by the antioxidant effect of luteolin which decreases the oxidative stress, consequently reduce the mitochondrial stress, decrease both caspase 3, Bax levels and increase Bcl-2 level as an anti-apoptotic protein, thereby trigger its anti-apoptotic activity (Figure 8). Our data agreed with a reported study which concluded the anti-apoptotic effect of Lut against the lead acetate-induced toxicity. 12 The current findings demonstrate the anti-apoptotic activity of Lut against the MTX-induced apoptosis and its molecular mechanism.
Conclusion
Altogether, this study concluded that luteolin could reduce the MTX-induced hepato-renal toxicity in male Wistar albino rats. Also, a mechanism of this antagonistic effect was proposed including the antioxidant, anti-inflammatory and anti-apoptotic activities of luteolin (Figure 8). Finally, this study recommends the use of luteolin as a dietary supplement during the disease’s treatment by MTX to protect against the MTX-cytotoxicity. Also, further laboratory and clinical trials are needed to ensure the potential protective effects of luteolin.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by King Saud University, Riyadh, Saudi Arabia (Research Supporting Project number: RSP-2020/95).
