Abstract
Doxorubicin is an effective anti-neoplastic agent; the reported toxicities of DOX limit its use. Luteolin is a polyphenolic phytochemical that exhibits beneficial biological effects via several mechanisms. We investigate luteolin protective effects on hepatorenal toxicity associated with doxorubicin treatment in rats. For 2 weeks, randomly assigned rat cohorts were treated as follows: control, luteolin (100 mg/kg; per os), doxorubicin alone (2mg/kg; by intraperitoneal injection), co-treated cohorts received luteolin (50 and 100 mg/kg) in addition to doxorubicin. Treatment with doxorubicin alone significantly (p < 0.05) increased biomarkers of hepatorenal toxicities in the serum. Doxorubicin also reduced relative organ weights, antioxidant capacity, and anti-inflammatory cytokine interleukine-10. Doxorubicin also increased reactive oxygen and nitrogen species, lipid peroxidation, pro-inflammatory-interleukin-1β and tumour necrosis factor-α-cytokine, and apoptotic caspases-3 and -9). Morphological damage accompanied these biochemical alterations in the rat’s liver and kidney treated with doxorubicin alone. Luteolin co-treatment dose-dependently abated doxorubicin-mediated toxic responses, improved antioxidant capacity and interleukine-10 level. Luteolin reduced (p < 0.05) lipid peroxidation, caspases-3 and -9 activities and marginally improved rats’ survivability. Similarly, luteolin co-treated rats exhibited improvement in hepatorenal pathological lesions observed in rats treated with doxorubicin alone. In summary, luteolin co-treatment blocked doxorubicin-mediated hepatorenal injuries linked with pro-oxidative, inflammatory, and apoptotic mechanisms. Therefore, luteolin can act as a chemoprotective agent in abating toxicities associated with doxorubicin usage and improve its therapeutic efficacy.
Keywords
Introduction
Doxorubicin (DOX), is an anthracycline glycoside antibiotic, 1 commonly prescribed against several tumours.2,3 DOX exerts its anti-neoplastic activity by intercalating with the DNA double-helix, thus inhibiting nucleic acid and protein synthesis. DOX covalently bind proteins necessary for DNA replication and transcription, thereby disrupting DNA replication and transcription,4,5 and harming neoplastic cells. Cytochrome P450 reductase metabolizes DOX to a semi-quinone radical that generates reactive oxygen species (ROS),6,7 and increases its toxicity, inducing calcium overload mitochondrial dysfunction.8–10 Toxicity of DOX to non-target tissues includes hepatic, cardiac, haematologic, and reproductive organs,11–14 and this often limits its therapeutic dosages. 15 DOX has been observed to induce hepatocyte damage by cell cycle arrest, 16 thus inhibiting the self-regeneration potential of the liver. 17 RONS produced from DOX metabolism increases the propensity for lipid peroxidation, DNA damage, and decreases cellular thiols and vitamin E levels.18,19 DOX reported side effects to include hepatoxicity and nephrotoxicity.20,21 The presence of severe proteinuria, 22 and glomerular capillary permeability 23 has also been reported. Furthermore, DOX interferes with mitochondrial function, leading to decreased mitochondrial complex I–IV activities and increased superoxide (O2•−) levels, 24 thus triggering lipid peroxidation and depletion of cellular antioxidant. 25
Luteolin (LUT) – 2-[3,4-dihydroxyphenyl]-5,7-dihydroxy-4-chromenone – is a member of the flavonoid family present in fruits and vegetables.26,27 LUT is reported to exhibit multiple pharmacological effects: cytoprotective, 28 neuroprotective, 29 antioxidative, 30 anti-inflammatory, 27 anti-apoptotic and anti-neoplastic activities26,31 in several disease models. The efficacy of LUT to induce apoptosis in breast cancer cells 32 and sensitize carcinoma cells to a chemotherapeutic agent by LUT-loaded phytosomes 33 has been reported. These prospects open up new avenues for research to improve treatment efficacy by mitigating drug-related toxicities. Therefore, we investigated the beneficial effect of LUT as a chemoprotective agent for preventing DOX-mediated toxicity in an in vivo model. We assessed oxidative stress biomarkers’, antioxidant enzyme activities, and inflammatory mediators’ levels alongside the liver and kidney histopathological examination to achieve these goals. To gain mechanistic insight on how LUT modulates DOX-induce hepatorenal dysfunction in rats. Finally, we further report LUT’s protective mechanisms in the rats’ hepatorenal system treated with DOX.
Materials and methods
Chemicals
Doxorubicin, epinephrine, glutathione (GSH), thiobarbituric acid (TBA), hydrogen peroxide (H2O2), 5,5-dithio-bis-2-nitrobenzoic acid (DTNB), Griess reagent, 1-chloro-2, 4-dinitrobenzene (CDNB), xanthine, trichloroacetic acid, bovine serum albumin (BSA), hydrogen peroxide (H2O2) was purchased from Sigma (St Louis, Missouri, USA). Monosodium dihydrogen phosphate, disodium hydrogen phosphate, sodium carbonate, sodium hydroxide, copper sulphate, potassium iodide, sodium–potassium tartrate, and sodium chloride were obtained from BDH Ltd. (Poole, Dorset, UK) and William Hopkins Ltd. (Birmingham, UK). Alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), γ-glutamyl transferase (γ-GT), lactate dehydrogenase (LDH), urea and creatinine kits were obtained from Randox™ Laboratories Ltd. (Ardmore, Crumlin, Co., Antrim, UK). Enzyme-Linked Immunosorbent Assay (ELISA) kits for the assessment of Interleukin-1β (IL-1β), interleukin-10 (IL-10), tumour necrosis factor-alpha (TNF-α), caspase-3 and caspase-9 were obtained from Elabscience Biotechnology Company (Beijing, China). All other chemicals used for these experiments are of analytical grade.
Animal model and experimental design
Sexually mature, healthy male Wistar rats (160 ± 5 g, n = 50) were obtained from the Experimental Laboratory animal house, Faculty of Basic Medical Sciences, University of Ibadan. Experimental rats were housed under natural photoperiod conditions (12/12-hour light-dark) in a well-ventilated rodent facility of the Department of Biochemistry, provided with rat pellets (Ladokun™ Feeds, Ibadan, Nigeria) and allowed free access to water. Rats were allowed to adapt (7 days) to their new environment preceding experimentation and adequately cared for as specified by ‘Guide for the Care and Use of Laboratory Animals’ published by the National Institute of Health. Also, testing was performed following the University of Ibadan Ethical Committee’s approval (UI-ACUREC/20/035) and following the United States National Academy of Sciences guidelines.
Experimental protocol
The doses of Doxorubicin (2 mg/kg) and Luteolin (50 and 100 mg/kg dissolved in corn oil) used in the current study were selected based on previously published data.34–36 New stock solutions of DOX (2 mg/mL) and LUT (50 mg/mL) were prepared every other day for dosing experimental rats. Rats were randomly divided into five treatment groups (n = 10), and specific groups were treated with a single dose of DOX, every other day for 6 days by intraperitoneal injection (i.p.),37,38 and with LUT per os (p.o.)30,39 for 14 consecutive days
30
as follows: Group I: Control – administered corn oil-vehicle – (2 ml/kg; p. o) alone. Group II: treated with LUT dissolved in corn oil (100 mg/kg; p.o) alone. Group III: treated with DOX (2mg/kg; i.p.). Group IV: DOX + LUT1: treated with (DOX: 2 mg/kg) and (LUT: 50 mg/kg; p.o.). Group V: (DOX + LUT2): treated with (DOX: 2 mg/kg) and (LUT: 100 mg/kg; p.o.).
Following the last treatment, on day 15, the rats were weighed. Blood was collected from the retro-orbital venous plexus into both plain and ethylenediaminetetraacetic acid (EDTA)-containing pre-labelled sample bottles, and the experimental rats humanely sacrificed by carbon dioxide (CO2) asphyxiation and confirmed by cervical dislocation.40,41 Subsequently, the clotted blood was centrifuged at 3000g for 10 minutes, 4°C to obtain the serum. The liver and kidney were immediately excised, weighed, and processed for biochemical and histological analyses. The serum samples were stored (−20°C) until required for specific biochemical analysis.
Assessment of biomarkers of liver and kidney function
Analysis of serum activities of aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), γ-glutamyl transferase (γ-GT), and lactate dehydrogenase (LDH), as well as creatinine and urea levels, were performed using available commercial kits from Randox™ Laboratories Limited (UK).
Preparation of tissue homogenate and assessment of biomarkers of liver and kidney oxidative and nitrative stress
The experimental rats’ liver and kidney samples were homogenized in phosphate buffer (0.05 M; pH 7.4). The tissue homogenates were centrifuged at 12,000g for 15 min at 4°C to obtain the supernatant used to assess oxidative stress, inflammatory and apoptotic biomarkers. According to Lowry’s method, 42 liver and kidney protein concentrations were evaluated, and total sulfhydryl group were estimated by Ellman’s method. 43 Reduced glutathione (GSH) was determined by Jollow protocol 44 while glutathione-S-transferase (GST), glutathione peroxidase (GPx) and superoxide dismutase (SOD) activities were determined as previously described and reported.45–48 Catalase (CAT) activity was determined using H2O2 as substrate as previously described by Claiborne, 49 while xanthine oxidase (XO) was quantified following Bergmeyer method. 50 According to the previously reported method by Buege and Aust51,52 lipid peroxidation was assessed and expressed as μmol MDA/mg protein.
Reactive oxygen and nitrogen species (RONS) detection
Liver and kidney RONS production was evaluated according to an established protocol based on the RONS-dependent oxidation of 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA) to dichlorofluorescein (DCF). 53 Briefly, the reaction mixture (150 µL 0.1 M potassium phosphate buffer; 35 µL distilled water, 10 µL sample and 5 µL freshly prepared DCFH-DA was constituted with minimal exposure to air. Fluorescence emission of DCF produced from DCFH-DA oxidation was spectrophotometrically analysed (wavelengths: 488 nm excitation; 525 nm emission) for 10 min at 30 s intervals using a Spectra Max 384 multi-modal plate reader (Molecular Devices, San Jose, CA, USA). DCF produced was expressed as a percentage over control.
Assay for pro-inflammatory biomarkers
The liver and kidney nitric oxide (NO) level was assessed using Griess reagent according to the established protocol. 54 Briefly, the reaction mixture consisting of an equal volume of sample and Griess reagent was incubated for 15 min before the absorbance was evaluated at 540 nm. The level of NO was extrapolated from the standard curve and then expressed as Units/mg protein. Moreover, myeloperoxidase (MPO) activity was evaluated according to the method described by Granell et al. 55 Additionally, IL-1β, IL-10, and TNF-α levels were determined using commercially available ELISA Kits and a SpectraMax™ plate reader (Molecular Devices, CA, USA), as stated in the manufacturer’s manual.
Assay for biomarkers of apoptosis caspase 3 and 9 activities
Liver and kidney caspase-3 and 9 activities were evaluated using Enzyme-Linked Immunosorbent Assay (ELISA) Kits (Elabscience; Beijing, China) following the manufacturer’s instructions. The results were obtained using a SpectraMax™ M384 Multi-modal plate reader (Molecular Devices, San Jose, CA, USA). Before the start of each assay, all reagents were calibrated to room temperature of 25°C. Briefly, 100 µL of standard and specimen were pipetted into designated wells in the specific caspase 3 and 9 ELISA and incubated for 90 min at 37°C. After that, standards and samples were discarded, and 100 µL of biotinylated detection antibody working solution was added to probe specific well and incubated for 60 min at 37°C. The reagent was aspirated off and each well washed thrice with approximately 300 µL of the 1X wash buffer. 100 µL of HRP conjugated working solution was added to each well, incubated for 30 min at 37°C and each well washed five times with the wash buffer. Following the substrate reagent (90 μL) was added to each well, and incubated for another 15 min at 37°C. The reaction was quenched with 50 µL of the stop solution and gently mixed by swirling for 20 seconds. Subsequently, the absorbance was immediately read using a Molecular Devices multi-modal plate reader at 450 nm (Caspase-3 and -9). The results were extrapolated from the standard curve generated and expressed in ng/mL.
Histopathological examination of liver and kidney
Liver and kidney samples were fixed using 10% phosphate-buffered formalin for 3 days. The samples were embedded in paraffin after dehydration procedures. Microtome cut tissue sections (4–5μm), fixed on charged microscopic glass slides, were subsequently stained with haematoxylin and eosin (H&E). 56 The tissue histology slides were blinded and examined under a light microscope (Leica DM 500, Germany) by a pathologist. The histopathological aberrations were scored and reported accordingly. Representative images were captured with a digital camera (Leica ICC50 E, Germany) attached to the microscope. Table 1 depicts the findings of semi-quantitative evaluation of rats’ liver and kidney scored for pathological aberration as previously reported,57–60 following DOX and LUT treatment. The scoring criteria are denoted as none (0); mild (1); mild-moderate (2); moderate (3); moderate to severe (4) and severe (5). 61
The incidence of the pathological lesions in the rat liver and kidney following treatment with LUT and DOX for 14 consecutive days.
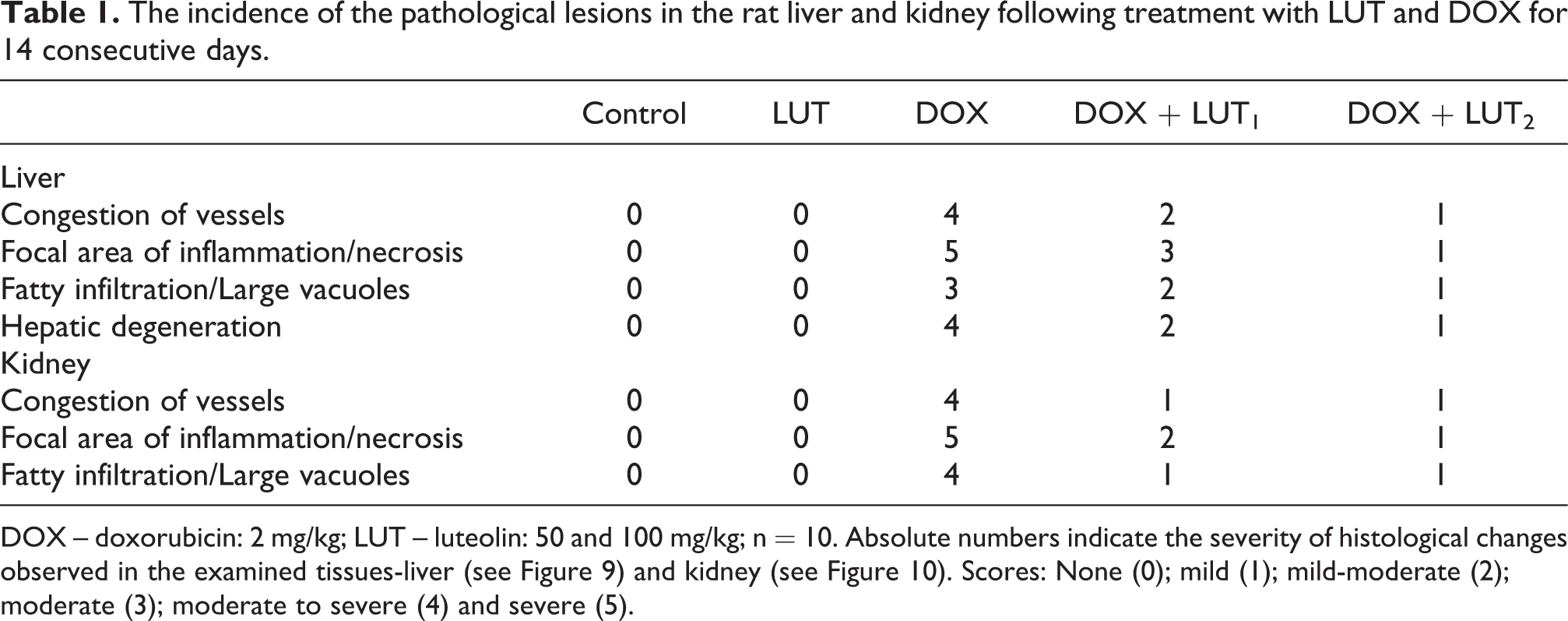
DOX – doxorubicin: 2 mg/kg; LUT – luteolin: 50 and 100 mg/kg; n = 10. Absolute numbers indicate the severity of histological changes observed in the examined tissues-liver (see Figure 9) and kidney (see Figure 10). Scores: None (0); mild (1); mild-moderate (2); moderate (3); moderate to severe (4) and severe (5).
Statistical analysis
Data were analysed using the one-way analysis of variance (ANOVA) and the post hoc Tukey test (GraphPad Prism 5 Software, La Jolla, California, USA, www.graphpad.com) ascertain significant differences in the treatment groups. P values <0.05 were considered significant. The results were subsequently expressed as mean ± standard deviation (SD).
Results
Effect of luteolin on rat survival and relative organ weight of doxorubicin-treated rats
The impact of LUT on rat survival (Kaplan-Meyers) and percentage weight change are presented in Figures 1(a) and (b). LUT (50 and 100 mg/kg) improved survival of rats co-treated with DOX, while DOX only reduced rat survivability (Figure 1(a)). DOX alone significantly (p < 0.05) decreased the body weight gain of rats compared to other treatment groups (Figure 1(b)). In comparison to DOX alone, groups of rats that received both DOX and LUT (50 and 100 mg/kg) showed improvement in body weight change, with respective percentage weight loss of 8.34% (LUT 50 mg/kg) and 4.30% (LUT 100 mg/Kg). The cohort of rats treated with LUT 100 mg/kg weight change was increased (p < 0.05) compared to the DOX alone and the DOX+LUT 50 and 100 mg/kg groups. DOX alone resulted in decreases in liver and kidney weight compared to the untreated control and LUT alone groups (Table 2)

Effect of luteolin on percentage survival (a), and percentage weight change (b) of doxorubicin-treated rats. DOX: 2 mg/kg, i.p.; LUT1 (50 mg/kg, p.o.); LUT2 (100 mg/kg). DOX: Doxorubicin; LUT: Luteolin; i.p.: interperitoneally: p.o: per os.
Body weight gain and relative organ – liver and kidney – weights of rats following treatment with LUT and DOX for 14 consecutive days.

Values are expressed as mean ± SD of 10 rats, *p < 0.05 versus untreated control; **p < 0.05 versus DOX alone. SD: standard deviation; LUT, luteolin; DOX, doxorubicin.
Luteolin improves liver and kidney toxicities in doxorubicin-treated rats
The effect of DOX on the serum liver and kidney function biomarkers in rats is presented in Figure 2. DOX treatment alone caused increases (p < 0.05) in liver transaminases (ALT, AST, ALP, GGT), lactate dehydrogenase (LDH), as well as markers of kidney function, urea and creatinine, in rat serum compared to the control. Alterations in liver and kidney functional biomarkers were reduced (p < 0.05) by LUT at 50 and 100 mg/kg. These observed LUT-mediated improvements are dose-dependent.

Effect of luteolin on hepatic and renal function markers of doxorubicin-treated rats. DOX: 2 mg/kg, i.p.; LUT: 100 mg/kg, per os; LUT1 (50 mg/kg); LUT2 (100 mg/kg). Each bar represents the mean ± SD of 10 rats. Connecting lines indicate groups compared to one another, the significance level was set at (p < 0.05); * to ****: indicates the level of significance; ns: not significant. DOX: doxorubicin; LUT: luteolin; SD: standard deviation. ALP: alkaline phosphatase; AST: aspartate amino transferase; ALT: alanine amino transferase; GGT: gamma-glutamyl transferase; LDH: lactate dehydrogenase.
Luteolin abates doxorubicin-induced hepatic and renal oxidative damage in rats
The protective effect of LUT against DOX-induced toxicities in rats’ liver and kidney is presented in Figures 3–5. DOX alone increased (p < 0.05) liver and kidney activities of GPx and GST, and reduced (p < 0.05) GSH level compared to control (Figure 3). Conversely, LUT (50 and 100 mg/kg) co-treatment reversed the observed changes. Relative to the control, DOX treatment caused a similar increase in liver and kidney CAT and SOD activities and reduced TSH levels (Figure 4) reversed by LUT co-treatment at 50 and 100 mg/kg. Additionally, DOX treatment alone increased (p < 0.05) the levels of MDA and RONS, and XO activity in the liver and kidney compared to the control (Figure 5). These increase in pro-oxidant levels of RONS, MDA, and the free radical generating enzyme activity, XO reduced (p < 0.05) following co-treatment with LUT at both 50 and 100 mg/kg. LUT alone did not alter the balance between antioxidants and RONS species in rat liver and kidney.
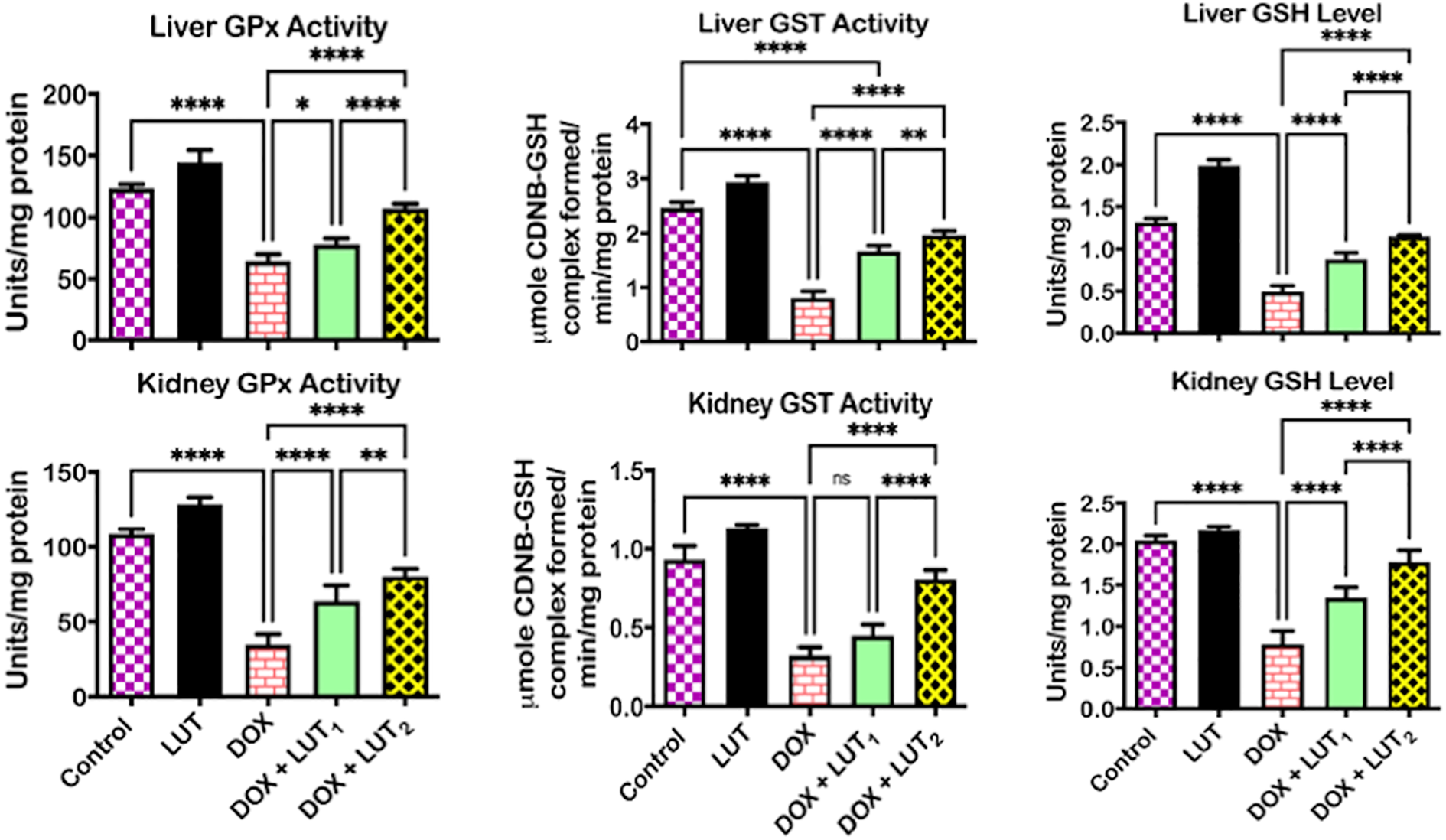
Effect of luteolin on GSH-dependent enzyme activities and GSH level in the liver and kidney of doxorubicin-treated rats. DOX: 2 mg/kg, i.p.; LUT: 100 mg/kg, per os; LUT1 (50 mg/kg); LUT2 (100 mg/kg). Each bar represents the mean ± SD of 10 rats. Connecting lines indicate groups compared to one another, the significance level was set at (p < 0.05); * to ****: indicates the level of significance; ns: not significant. DOX: doxorubicin; LUT: luteolin; SD: standard deviation. GPx: glutathione peroxidase; GST: glutathione-S-transferase; GSH: glutathione.
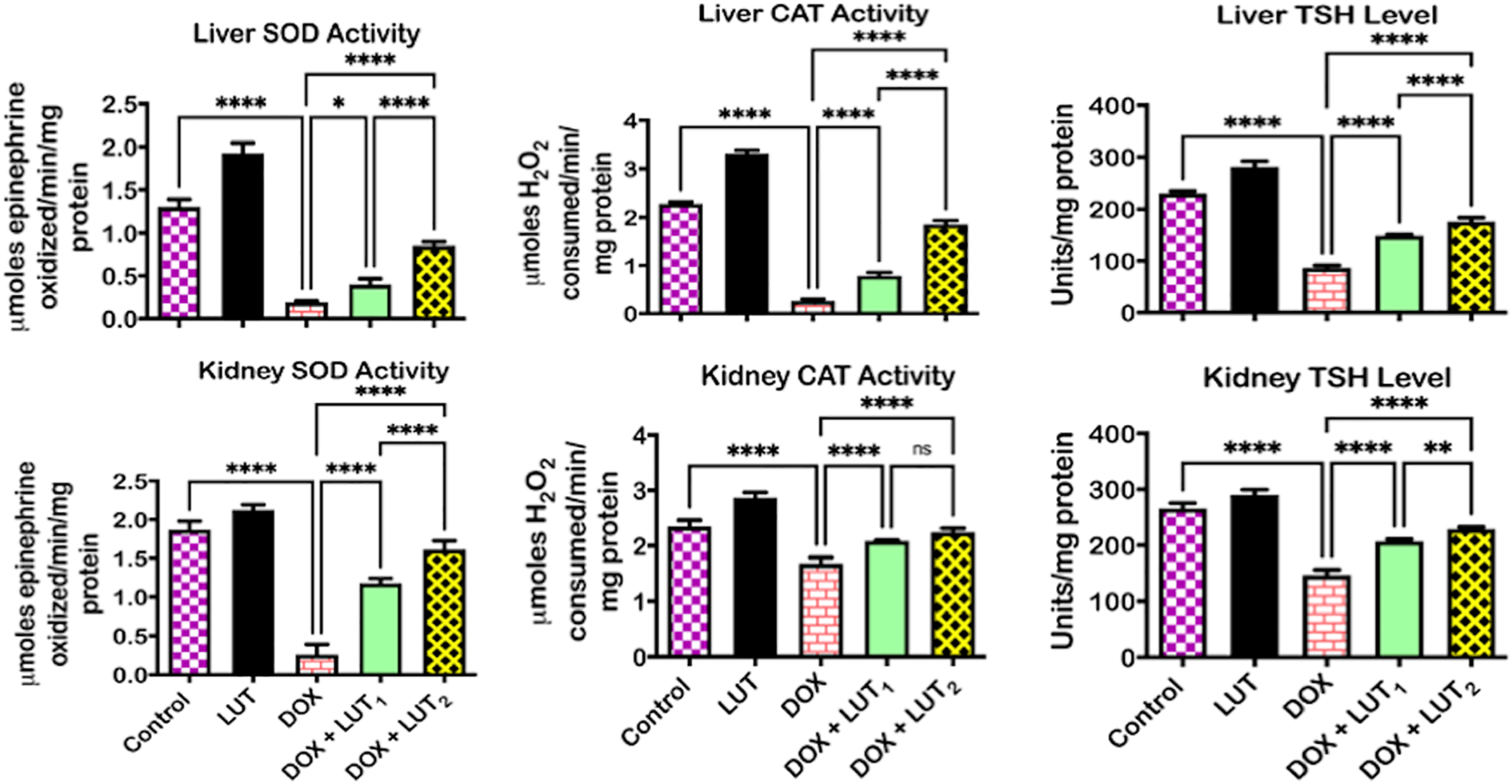
Effect of Luteolin on SOD and CAT activities and TSH level in the liver and kidney of doxorubicin-treated rats. DOX: 2 mg/kg, i.p.; LUT: 100 mg/kg, per os; LUT1 (50 mg/kg); LUT2 (100 mg/kg). Each bar represents the mean ± SD of 10 rats. Connecting lines indicate groups compared to one another, the significance level was set at (p < 0.05); * to ****: indicates the level of significance; ns: not significant. DOX: doxorubicin; LUT: luteolin; SD: standard deviation. SOD: superoxide dismutase; CAT: catalase; TSH: total sulfhydryl group.

Effect of luteolin on levels of RONS and LPO and activity of XO in the liver and kidney of Doxorubicin-treated rats. DOX: 2 mg/kg, i.p.; LUT: 100 mg/kg, per os; LUT1 (50 mg/kg); LUT2 (100 mg/kg). Each bar represents the mean ± SD of 10 rats. Connecting lines indicate groups compared to one another, the significance level was set at (p < 0.05); * to ****: indicates the level of significance; ns: not significant. DOX: doxorubicin; LUT: luteolin; SD: standard deviation. RONS: reactive oxygen/nitrogen species; LPO: lipid peroxidation; XO: xanthine oxidase.
Luteolin reduces the doxorubicin-mediated increase in biomarkers of inflammation in the liver and kidney of rats
The effects of LUT against DOX-mediated increase in inflammatory biomarkers are presented in Figures 6 and 7. DOX treatment alone increased (p < 0.05) MPO activity and NO level in rat liver and kidney compared to control (Figure 6). These were reduced (p < 0.05) in the presence of LUT at 50 and 100 mg/kg. Furthermore, DOX alone caused increases (p < 0.05) in pro-inflammatory: TNF-α, and IL-1β, and decreased (p < 0.05) anti-inflammatory IL-10, in rat’s liver and kidney compared to control (Figure 7). The observed changes were reversed in the presence of LUT, and more so at LUT 100mg/kg; by significantly decreasing TNF-α, and IL-1β and increasing IL-10.

Effect of luteolin on NO level and MPO activity in the liver and kidney of doxorubicin-treated rats. DOX: 2 mg/kg, i.p.; LUT: 100 mg/kg, per os; LUT1 (50 mg/kg); LUT2 (100 mg/kg). Each bar represents the mean ± SD of 10 rats. Connecting lines indicate groups compared to one another, the significance level was set at (p < 0.05); * to ****: indicates the level of significance; ns: not significant. DOX: doxorubicin; LUT: luteolin; SD: standard deviation; NO: nitric oxide; MPO: myeloperoxidase.

Effect of luteolin on TNF-α, IL-1β and IL-10 levels in the liver and kidney of doxorubicin-treated rats. DOX: 2 mg/kg, i.p.; LUT: 100 mg/kg, per os; LUT1 (50 mg/kg); LUT2 (100 mg/kg). Each bar represents the mean ± SD of 10 rats. Connecting lines indicate groups compared to one another, the significance level was set at (p < 0.05); * to ****: indicates the level of significance; ns: not significant. DOX: doxorubicin; LUT: luteolin; SD: standard deviation; TNF-α: tumour necrosis factor-alpha; IL1β: interleukin-1beta; IL-10: interleukin-10.
Luteolin attenuates the doxorubicin-mediated increase in apoptosis in rat liver and kidney
DOX treatment alone caused increases (p < 0.05) in the activities of initiator Caspase-9 (Casp-9) and executioner Caspase-3 (Casp-3) in treated rats’ liver and kidney compared to control (Figure 8). However, groups co-treated with LUT (50 and 100 mg/kg) caused reduction of DOX-mediated increase in Casp-9 and -3 activities in rats’ liver and kidney.

Effect of luteolin on Caspase-3 and Caspase-9 activities in the liver and kidney of doxorubicin-treated rats. DOX: 2 mg/kg, i.p.; LUT: 100 mg/kg, per os; LUT1 (50mg/kg); LUT2 (100mg/kg). Each bar represents the mean ± SD of 10 rats. Connecting lines indicate groups compared to one another, the significance level was set at (p < 0.05); * to ****: indicates the level of significance; ns: not significant. DOX: doxorubicin; LUT: luteolin; SD: standard deviation.
Luteolin abated histopathological lesions in rat’s liver and kidney following treatment with doxorubicin
Photomicrographs of liver and kidney from rats treated with DOX and LUT were combined from microscopic examination and digital camera capture. Liver: Control and LUT treated rats consistently displayed typical liver morphology and histological-architecture. DOX alone showed marked disseminated congestion of veins and sinusoids infiltrated by inflammatory cells (black arrow), and disseminated microvascular steatosis. Sections from the DOX + LUT50 mg/kg group showed mild congestion of veins and sinusoids, and mild disseminated microvascular steatosis. In contrast, those from the DOX + LUT100 mg/kg group revealed mild disseminated congestion of veins and sinusoids and mild disseminated infiltration (Figure 9). Kidney: Control and LUT alone rats showed typical kidney morphology with no significant lesions. Treatment with DOX alone resulted in moderate disseminated congestion and glomerular hypercellularity/congestion of the kidney. DOX and LUT (50 and 100 mg/kg) decreased the observed changes and improved kidney cyto-architecture similar to typical histology features. The kidney sections from the DOX + LUT 50 mg/kg group showed mild glomerular hypercellularity/congestion. In contrast, those from rats in the DOX + LUT 100 mg/kg group are relatively more similar to the control, and LUT alone treated groups (Figure 10).

The liver’s representative photomicrographs from control, LUT alone, DOX alone, and co-exposed (DOX+LUT) experimental rats. Control and LUT alone show no significant lesion, characterized by structural and functionally normal hepatocytes. Hepatic structure of rats treated with DOX alone presented with congested veins and sinusoids (black arrows), and the incidence of small-sized cytoplasmic vacuoles. The morphological attributes of the liver treated with DOX + LUT appeared to be improving dependent on the doses of LUT (LUT2 > LUT1), with lesser congestion (black arrow), mild disseminated congestion of sinusoids and infiltration by inflammatory cells. H and E stained, Magnification ×400: top panel; ×100: lower panel. DOX: doxorubicin; LUT: luteolin; LUT1 (50 mg/kg); LUT2 (100 mg/kg).

Representative photomicrographs of the kidney from control, LUT alone, DOX alone, and co-exposed (DOX + LUT) experimental rats. Control and LUT groups showed no significant lesion. DOX showed mild to moderate disseminated congestion and glomerular hypercellularity/congestion (black arrows). DOX + LUT1 showed mild glomerular hypercellularity/congestion (black arrows). The histology of DOX + LUT2 appears similar to that of the control and LUT alone treated groups. H and E stained, Magnification ×400: top panel; ×100: lower panel. DOX: doxorubicin; LUT: luteolin; LUT1 (50 mg/kg); LUT2 (100 mg/kg).
Discussion
DOX is an active anti-neoplastic agent, limited in use by its associated hepatorenal toxicity.7,17 We explore opportunities to improve DOX usage by devising strategies to mitigate DOX toxicity. These include compromised antioxidant defences due to oxidative stress, RONS generation, inflammatory, and apoptotic responses. 62 Strategies focused on preventing DOX-induced toxicity include the use of chemopreventive agents,63,64 such as plant-derived flavonoids. We hypothesized that LUT could be beneficial in attenuating drug-induced oxidative, inflammatory, and apoptotic responses in the rat hepatorenal system following DOX treatment. Our findings demonstrate that LUT lessened DOX-mediated hepatorenal dysfunction by abating tissue injury, oxido-inflammatory perturbation, and easing apoptotic cell death in rats. Our results show that DOX treatment resulted in a significant reduction of relative organ weight and rat survivability compared to control, consistent with a previous report. 65
The decrease in organ weight may be due in part to gastrointestinal toxicity, and reduced food consumption. The direct impact of DOX on renal tubules may prompt improper sodium excretion, and decrease water reabsorption, leading to polyuria, dehydration, and reduced body weight.66,67 In contrast, improved relative organ weight and survivability were observed in LUT and DOX co-treated rats in our study. DOX alone increased hepatic transaminases in the serum, useful in diagnosing hepatic toxicity and necrotic damage,68,69 supporting previous reports.6,63 Co-administration of LUT dose-dependently decreased serum transaminase (ALT, ALP, LDH, GGT) levels indicates the protective role of LUT. Kidney function can be diagnosed from the byproducts of protein metabolism (urea) and a product from creatinine phosphate breakdown (creatinine). However, creatinine is a more accurate predictor of kidney injury than urea. Both the liver and kidney must be functioning correctly for the body to maintain a healthy level of urea.70,71 DOX alters renal functions, and can significantly increase serum urea and creatinine levels.7,62 LUT co-administration reduced serum urea and creatinine levels, indicative of the chemoprotective effect of LUT, a deduction supported by previous reports.72,73
Oxidative stress results from an imbalance among antioxidants and pro-oxidants favouring pro-oxidants that can harm cells. 74 Oxidative stress-derived free radicals can overwhelm cellular enzymatic and non-enzymatic antioxidant defences. Consequently, damage macromolecules, worsen lipid peroxidation and increase further RONS production. 53 Increased RONS production further contributes to the initiation and progression of injuries and various hepatorenal system disorders.9,65 Increase in RONS, LPO levels, and XO activity typically occurs with waning enzymatic antioxidant (SOD, CAT, GPx, GST) activities and GSH and TSH levels. 75 Data from our study shows that DOX alone caused an apparent decrease in hepatorenal antioxidant enzyme activities, GSH and TSH levels, as well as increases in LPO, RONS levels, and XO activity. DOX-induced oxidative stress can result in injuries to the hepatorenal system. LUT co-treated rats showed significant improvement in enzymatic antioxidant activities (SOD, CAT, GPx, GST) and GSH and TSH levels.
Similarly, LPO, and RONS levels and XO activity were reduced in rat’s cohort co-treated with LUT. These findings further emphasize the chemoprotective role of LUT on the rat hepatorenal system treated with DOX, consistent with earlier reports demonstrating the LUT protective effect in mice with acute renal injury 72 and rats with liver injury. 76 The observed effects of LUT can be attributed to its inherent antioxidant activities.77,78 MPO, NO, IL-1β, IL-10, and TNF-α play important inflammatory/pathological roles in several disorders and DOX-induced hepatorenal injuries. 70 LUT is reported to suppress oxido-inflammatory stress via the upregulation of the Nrf2/ARE/HO-1 signalling pathways. 79 The controlled release of MPO at the site of infection is of prime importance for its efficient use. However, any uncontrolled degranulation worsens inflammation and can cause tissue damage. 80 The combined roles of IL-1β, IL-10, and TNF-α in inflammatory processes have long been reported as they are known to recruit cells at the site of tissue injuries. 81 The increases in liver and kidney levels of IL-1β and TNF-α, as well as a decrease in IL-10 in DOX treated rats, indicate an inflammatory response. Increases in IL-1β and TNF-α and the associated reduction of IL-10 levels can trigger inducible nitric oxide synthase (iNOS) to produce nitric oxide.
Subsequently, nitric oxide can combine with superoxide anion to form peroxynitrite, a harmful reactive nitrogen species. 82 DOX administration triggers inflammatory-mediated actions by markedly increasing MPO activity and NO, IL-1β, TNF-α levels, and decreasing IL-10 levels in our study’s rat hepatorenal system. Our findings agree with previously published data on the DOX-mediated alteration of inflammation biomarkers.83,84 The observed increases in hepatorenal MPO activity, NO, IL-1β and TNF-α levels, together with decreases in the level of IL-10 in DOX treated rats, may contribute to hepatorenal damage by inducing inflammatory and nitrosative stress. Conversely, decreases in MPO activity, NO, IL-1β, and TNF-α levels accompanying an increase in IL-10 level in rats co-treated with LUT and DOX demonstrated the alleviating effects of LUT on DOX-mediated inflammatory responses. As earlier previously reported, LUT exhibits anti-inflammatory properties.27,85
Several apoptotic responses, including the intrinsic mitochondrial-dependent and extrinsic signalling pathways, have been implicated in the induction of programmed cell death. Signal transduction either through the intrinsic apoptotic molecules like Bax or extrinsic apoptotic molecules like Fas activate caspases, cysteine-dependent aspartate-specific proteases86,87 including Casp-9, Casp-3, the initiator and executioner caspases respectively. A marked increase in the hepatorenal Casp-9 and Casp-3 activities in DOX alone treated rats connotes apoptosis activation in the present study. This observation is in line with earlier findings on the ability of DOX to induce apoptosis in mice and rats.10,13 The observed reduction of Casp-9 and Casp-3 activities in rats co-treated with LUT signifies the suppression of apoptosis, due to the mitigating effect of LUT. Our findings support the previous report that LUT delayed apoptosis in an H2O2-induced ischaemic cerebrovascular disease model. 88 The histopathological examination appraises tissue integrity and disease progression and corroborates biochemical assays’ findings in serum and tissue enzyme assays. In this study, photomicrographs from the liver of rats administered with DOX alone showed marked disseminated congestion of veins and sinusoids compared to the liver sections from the control. LUT alone treated rats were found to be without significant lesions, hence corroborating our biochemical findings. Co-administration of LUT and DOX revealed mild congestion of veins and sinusoids, mild disseminated microvascular steatosis compared with the DOX alone treated group. These findings show that co-administration of LUT can protect the liver against DOX-mediated toxicity. The kidney photomicrographs revealed a similar pattern of DOX-induced toxicity, as previously reported on the nephrotoxic potentials of DOX in a rat model. 7 Similarly, co-administration of LUT in the presence of DOX showed non-significant lesions compared with the DOX alone treated group. The nephroprotective effect of LUT has been reported in bisphenol A (BPA)-induced renal toxicity in rats, 79 owing to its additional antioxidant activity. 89
Conclusion
DOX elicits hepatorenal toxicity via oxidative stress, inflammation, and apoptotic cell damage, thereby limiting the therapeutic uses of DOX. Co-administration of LUT, dose-dependently reduced hepatorenal toxicities observed in rats treated with DOX alone. LUT co-treatment improved the antioxidant status and anti-inflammatory cytokine levels in experimental rats. LUT also suppressed pro-inflammatory cytokines production and the actions of Casp-3 and -9 involved in apoptosis as depicted in Figure 11. The combined administration of DOX and LUT may help improve the therapeutic efficacies of DOX by reducing the intolerable side effects observed in the patient when well-coordinated.

Proposed mechanism of LUT in abating doxorubicin-induced hepatorenal toxicities in rats. Note that Cytochrome P450 reductase (CYP), carbonyl reductase (CBR) and aldo/keto reductase (A/KR) mediated the production of doxorubicinol. This toxic intermediate mediates oxidative stress, inflammation and apoptosis via the generation of ROS, RNS, IL-1β; and TNF-α; caspase-8 and -3. LUT abrogated these biochemical perturbations.
Footnotes
Acknowledgement
The authors would like to acknowledge Mr Eric Sabo’s technical assistance of the Department of Biochemistry, University of Ibadan.
Author contributions
All authors partook in the design, interpretation, analysis of the data generated from the study. SEO, UOA, and DOL conceptualized the experiments; carried out the research and preliminary data analysis. SEO and UOA supervised the investigation. SEO checked the generated data for error. The manuscript was written and revised by SEO, UOA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
