Abstract
Luteolin (LUT) is a glycosylated flavonoid compound that has multiple beneficial pharmacological and biological impacts. The current investigation was undertaken to evaluate the putative neuroprotective potency of LUT against neuronal damage induced by lead acetate (PbAc). Twenty-eight rats were placed into four equal groups. Group 1: served as the control group, group 2: rats were supplemented orally with LUT (50 mg kg−1), group 3: rats were intraperitoneally injected with PbAc (20 mg kg−1), and group 4: rats were pretreated with LUT before PbAc injection with the same doses. All animals were treated for 7 days. The exposure to PbAc increased the concentration of lead in the cortical tissue, neuronal lipid peroxidation, and nitric oxide (NO) production and decreased the antioxidant enzymes. Additionally, PbAc enhanced a neuroinflammatory response in the cortical tissue through increasing the pro-inflammatory cytokines secretion and inducible NO synthase expression. Moreover, cortical cell death was recorded following PbAc intoxication as evidenced by the enhancement of the proapoptotic and inhibiting the antiapoptotic markers. Interestingly, LUT supplementation reversed the cortical adverse reactions induced by PbAc. Taken together, these findings may suggest that LUT may be useful for attenuating neuronal damage induced by PbAc through inhibiting the oxidative damage, neuroinflammation, and the cortical cell death.
Introduction
Lead (Pb) is a toxic heavy metal, ubiquitous worldwide, and its exposure is associated with the development of severe health problems including neuronal damage. 1 Occupational exposure represents the main source of Pb intoxication. Human may also expose to Pb from industrial products including cosmetics, pipes, pigments, and paints. 2 Pb is known to affect all the body organs, and its accumulation is associated with pathological changes. 2,3 Cumulative evidences confirm that the exposure to Pb is associated with adverse reactions on the central nervous system in animals and humans. Pb penetrates the blood–brain barrier (BBB) and substitutes calcium and zinc ions causing neurological deficits. 4 Among other brain areas, cerebral cortex has been identified as a target for Pb toxicity leading to learning, memory, and behavioral impairments. 5 Although the pathophysiology of neurotoxicity produced following heavy metals exposure remains unclear, it is well identified that Pb-induced neuronal injury is correlated with oxidative challenge, neuroinflammation, and the enhancement of programmed cell death, which may play an important role in the development of the neurodegenerative diseases. 1,6,7
Therefore, searching for natural agents with a wide spectrum of health-promoting capacities including antioxidant and anti-inflammatory activities may provide a neuroprotective effect against Pb-induced neuronal damage. Luteolin (LUT; 3′,4′,5,7-tetrahydroxy flavones) is a glycosylated flavonoid compound found in different fruits and vegetables such as celery, broccoli, perilla leaf, pepper, honeysuckle bloom, and chamomile tea. 8,9 Recent studies have shown the beneficial effects of LUT in different experimental models. LUT protected the renal tissue against bisphenol A-induced nephrotoxicity via inhibiting the oxidative insults, inflammation, and activating antioxidants signaling in rats. 9 Additionally, LUT was found to provide a neuroprotective effect in a spinal cord ischemia–reperfusion (I/R) model through decreasing the oxidative damage, neuroinflammation, and neuronal cell death in rats. 10 Moreover, LUT protected the cortical tissue through upregulating the antioxidant kinases, inhibiting the production of the pro-inflammatory cytokines, and inhibiting the apoptotic events in cerebral I/R in rats. 11 Furthermore, LUT alleviated the lung injury in response to cadmium exposure through suppressing nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and activating the antioxidant defense system. 12
Based on these previous reports, the current investigation was undertaken to evaluate the putative neuroprotective potency of LUT versus lead acetate (PbAc)-induced neuronal damage through addressing the oxidative challenge, neuroinflammation, and neuronal cell death and seeking for a potential link between the molecular and biochemical alterations with the histopathological deformations in the cortical tissue in rats.
Materials and methods
Chemicals and experimental animals
Lead (II) acetate trihydrate (Pb(CH3CO2)2·3H2O; CAS number 6080-56-4) was purchased from Sigma-Aldrich (St Louis, Missouri, USA) and LUT (C19H18O6; CAS number 855-97-0) with 97% purity sourced from Alfa Aesar (Kandel, Germany). LUT was first dissolved in dimethyl sulfoxide (DMSO) and then diluted with normal saline (0.9% sodium chloride (NaCl)); the final administered solution contained 5% DMSO, corresponding to 20 µl per rat. Control rats received the same dose of DMSO (5% DMSO in saline). Although DMSO could represent free radical scavenger effect, its use was necessary because LUT is hydrophobic. It was, therefore, used at the lowest level possible. All other chemicals and reagents used in this study were of analytical grade. Double-distilled water was used as the solvent.
Ten-week-old male Wistar albino rats weighing 180–220 g were purchased from VACSERA (Giza, Egypt) and housed in wire polypropylene cages in a room under controlled laboratory conditions (12 h light–dark cycle; 25 ± 2°C). The rats were supplied with a rodent chewing standard diet and water ad libitum and allowed to acclimatize 2 weeks prior to the experiment.
Experimental design
To examine the ameliorative effect of LUT on PbAc-induced neurotoxicity, the rats were randomly allocated into four groups: the control group (n = 7), LUT group (n = 7), PbAc group (n = 7), and LUT + PbAc group (n = 7). The control group was administered 3 ml kg−1 day−1 of normal saline solution (0.9% NaCl) containing 5% DMSO orally, followed by intraperitoneal (i.p.) injection with 1 ml kg−1 day−1 of saline after 3 h. LUT was administered orally daily at 10:00 a.m. at a dose of 50 mg kg−1 day−1 according to Kalbolandi et al., 13 and PbAc was i.p. injected at a dose of 20 mg kg−1 day−1 according to Abdel Moneim 3 to induce acute neurotoxicity in rats. The LUT was pre-administrated 3 h before the PbAc injection. The animals were observed daily for any sign of toxicity throughout the experimental period.
The animals were euthanized 24 h after the last treatment using sodium pentobarbital (300 mg kg−1).
The rat’s skull was quickly opened, and the brain was carefully removed and washed twice in ice-cold 50-mM Tris–hydrochloride (HCl) (pH 7.4). The cortex was excised and divided into three parts. The first part was weighed and immediately homogenized in ice-cold medium containing 50 mM Tris-HCl (pH 7.4) to give a 10% (w/v) homogenate. The homogenates were centrifuged at 3000 × g for 10 min at 4°C. The supernatants were used for the various biochemical determinations. All experimental protocols were conducted according to the European Community Directive (86/609/EEC) and the national rules on animal care that was carried out in accordance with the NIH Guidelines for the Care and Use of Laboratory Animals 8th edition and were approved by the Institutional Animal Ethics Committee guidelines for animal care and use at Helwan University (approval number: HU/Z/012-19). The cortical total protein content of the homogenized was determined by the method of Lowry et al. 14 using bovine serum albumin as a standard. However, the second part of cortex was stored at −80°C for molecular analysis, and the third part was placed in 10% neutral-buffered formalin for the pathological examination.
Pb concentration in the cortical tissue
Cortical Pb concentration was analyzed by utilizing a flame atomic absorption spectrophotometry method according to Szkoda and Zmudzki. 15 Cortical samples were digested in a hot solution of 1 M nitric acid. Pb concentration in the digested samples was analyzed at 283.3 nm using an atomic absorption spectrophotometer (Perkin-Elmer, 3100, Perkin-Elmer Corporation, Norwalk, CT, USA). Results were expressed as micrograms per gram wet tissue weight.
Oxidative stress markers in the cortex
Lipid peroxidation (LPO) was spectrophotometrically measured in terms of malondialdehyde (MDA) formation according to Ohkawa et al. 16 Nitric oxide (NO) was estimated by the colorimetric method of Green et al. 17 Glutathione (GSH) was assayed according to the method of Ellman 18 utilizing the ability of GSH to reduce 5,5′-dithiobis(2-nitrobenzoic acid) forming a yellow compound spectrophotometrically measured at 405 nm. The activities of superoxide dismutase (SOD) and catalase (CAT) were determined according to the described methods of Sun et al. 19 and Aebi, 20 respectively. Glutathione peroxidase (GPx) and glutathione reductase (GR) activities were measured using the methods of Paglia and Valentine 21 and Carlberg and Mannervik, 22 respectively.
Cortical inflammatory and apoptotic markers
Cortical levels of tumor necrosis factor-alpha (TNF-α; catalog number: MBS175904) and interleukin-1 beta (IL-1β; catalog number: MBS764668) were detected using enzyme-linked immunosorbent assay (ELISA) kits obtained from MyBioSource (Southern California, San Diego, USA). Cortical apoptotic proteins including Bax (code: CSB-EL002573RA), caspase-3 (code: CSB-E08857r), and Bcl2 (code: CSB-E08854r) were measured using ELISA kits purchased from Cusabio (Wuhan, China) according to the manufacturer’s manual instructions.
Quantitative real-time polymerase chain reaction
Total RNA was isolated with the TRIzol reagent (Life Technologies, Gaithersburg, Maryland, USA) according to the manufacturer’s instructions. Complementary DNA (cDNA) was immediately prepared using the MultiScribe RT enzyme kit (Applied Biosystems, Foster City, California, USA). The resulting cDNA was subjected in triplicate for real-time PCR (RT-PCR) analysis. RT-PCR reactions were performed using Power SYBR Green PCR Master Mix (Applied Biosystems and Life Technologies) on a 7500 Real-Time PCR Systems (Applied Biosystems). Relative values of gene expression of superoxide dismutase (Sod2), catalase (Cat), glutathione peroxidase (Gpx1), glutathione reductase (Gsr), nuclear factor erythroid 2-related factor 2 (Nfe2l2), heme oxygenase-1 (Hmox1), tumor necrosis factor-alpha (Tnf), interleukin-1 beta (Il1β), inducible nitric oxide synthase (Nos2), caspase-3, Bcl-2-associated X protein (Bax), and B-cell lymphoma 2(Bcl2) genes were normalized to the endogenous glyceraldehyde-3-phosphate dehydrogenase (Gapdh). Primer sequences and accession numbers of the genes are presented in Table 1.
Primer sequences of genes analyzed in RT-PCR.

RT-PCR: real-time polymerase chain reaction; Gapdh: glyceraldehyde-3-phosphate dehydrogenase; Sod2: superoxide dismutase 2 mitochondrial (MnSOD); Cat: catalase; Gpx1: glutathione peroxidase 1; Gsr: glutathione reductase; Nfe2l2: nuclear factor erythroid 2-related factor 2; Hmox1: heme oxygenase 1; Nos2: inducible nitric oxide synthase; Il-1β: interleukin-1 beta; Tnf: tumor necrosis factor; Bcl2: B-cell lymphoma 2; Bax: Bcl-2-like protein 4; Casp3: caspase-3;
Histological changes
The cortical tissue was fixed in 10% neutral buffered formalin for 24 h, dehydrated in ethyl alcohol through increasing concentrations of ethyl alcohol (70%, 80%, 90%, and 100%), cleared in xylene, and mounted in molten paraplast. Sections of 5 µm were obtained from the prepared blocks and stained with hematoxylin–eosin (H&E). Sections were observed under a light microscope (Eclipse E200-LED; Nikon, Tokyo, Japan).
Statistical analysis
Results were expressed as the mean ± standard deviation. All data were analyzed by one-way analysis of variance. For the comparison of significance between groups, Duncan’s test was used as a post hoc test and p values less than 0.05 was considered statistically significant.
Results
In the current experiment, there was no incidence of death and both PbAc and LUT administration did not lead to any abnormal clinical signs, with no marked change in food and water consumption in the LUT-treated group. However, food consumption in PbAc-treated rats was decreased. As a result, the body weight of PbAc-treated rats was decreased significantly (data not shown).
The level of Pb in the cortical tissue following the treatment with LUT and/or PbAc
The data recorded in Figure 1 exhibited that the exposure to PbAc at a dose 20 mg kg−1 for 7 days increased significantly Pb level in the cortical tissue greater than its level in the control rats. Interestingly, LUT administration prior to PbAc at a dose 50 mg kg−1 for 7 days decreased significantly Pb deposition relative to PbAc-exposed rats; however, its level is still higher than the control group.

Represents the protective effect of LUT against Pb accumulation in the cortical tissue following PbAc exposure. Each column represents the mean ± SD (n = 7). “a” represents the statistical significance against the control group at p < 0.05 and “b” represents the statistical significance against the PbAc-injected group at p < 0.05. LUT: luteolin; Pb: lead; PbAc: lead acetate; SD: standard deviation.
LUT suppresses the cortical oxidative insult produced by PbAc in the cortical tissue
Rats intoxicated with PbAc showed a significant elevation in the oxidants including LPO and NO levels and decreased the cortical antioxidants including GSH content along with GR, GPx, SOD, and CAT activity as compared with their control values. The effect of PbAc is extended also to the molecular level through downregulating the messenger RNA (mRNA) expression of Gsr, Gpx1, Sod2, and Cat. LUT-treated rats showed a nonsignificant change in the investigated oxidative stress indices. Meanwhile, the pretreatment with LUT improved significantly the redox status in the cortical tissue in response to PbAc exposure through inhibiting the oxidants and enhancing the antioxidant molecules and their mRNA expression (Figures 2 and 3).
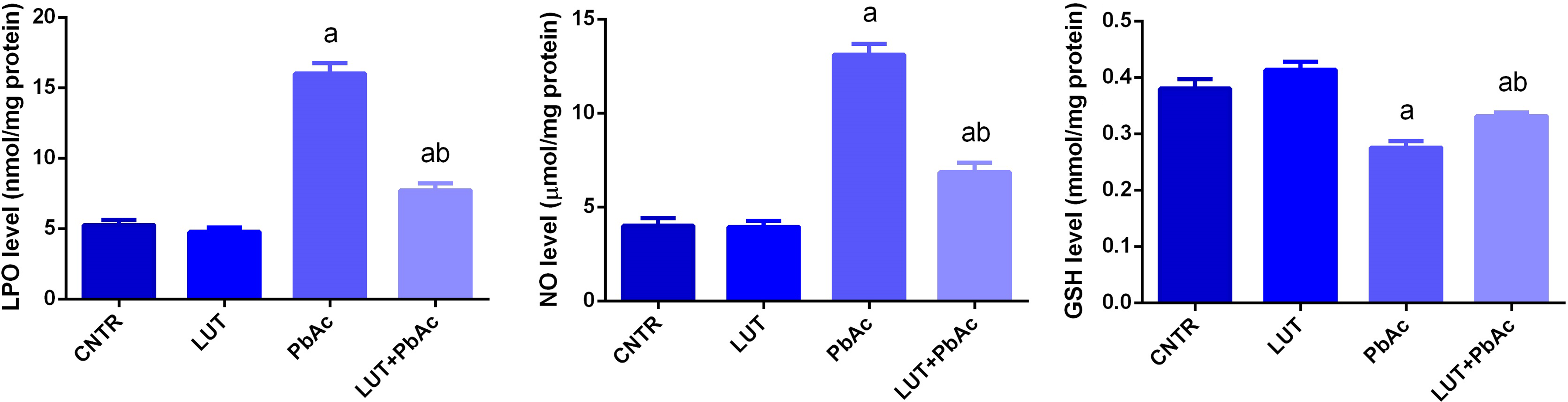
The ameliorative effect of LUT on the cortical levels of LPO, NO, and GSH content following PbAc exposure. Each column represents the mean ± SD (n = 7). “a” represents the statistical significance against the control group at p < 0.05 and “b” represents the statistical significance against the PbAc-injected group at p < 0.05. LUT: luteolin; LPO: lipid peroxidation; NO: nitric oxide; GSH: glutathione; PbAc: lead acetate; SD: standard deviation.

LUT enhances the activity of GR, GPx, SOD, and CAT and upregulates their mRNA expression in the cortical tissue following PbAc exposure. Each column represents the mean ± SD (n = 7). “a” represents the statistical significance against the control group at p < 0.05 and “b” represents the statistical significance against the PbAc-injected group at p < 0.05. mRNA expression was compared with the Gapdh. LUT: luteolin; GR: glutathione reductase; GPx: glutathione peroxidase; SOD: superoxide dismutase; CAT: catalase; mRNA: messenger RNA; PbAc: lead acetate; SD: standard deviation; Gapdh: glyceraldehyde-3-phosphate dehydrogenase.
To further assess the molecular mechanism involved in oxidative stress-mediated neurotoxicity following PbAc exposure, the mRNA expression of Nfe2l2 and Homx1 was evaluated in the cortical tissue. The quantitative RT-PCR (qRT-PCR) findings (Figure 4) that showed a significant downregulation in Nfe2l2 and Homx1 mRNA expression was detected in the PbAc-injected group relative to their expression in the control group. LUT administration was found to enhance significantly Nfe2l2 expression, while Homx1 did not change as compared to the control group. Meanwhile, the pretreated group with LUT showed a significant overexpression in Nfe2l2 and Homx1 when compared against the PbAc-exposed group; reflecting the neuroprotective role of LUT against oxidative stress-induced cortical damage following PbAc intoxication.

LUT activates the upregulation of the cortical mRNA expression of Nfe2l2 and Hmox1 following PbAc exposure. mRNA expression was compared with the Gapdh. “a” represents the statistical significance against the control group at p < 0.05 and “b” represents the statistical significance against the PbAc-injected group at p < 0.05. LUT: luteolin; mRNA: messenger RNA; PbAc: lead acetate; Gapdh: glyceraldehyde-3-phosphate dehydrogenase; Nfe2l2: nuclear factor erythroid 2-related factor 2; Hmox1: heme oxygenase 1.
LUT protects the cortical tissue against PbAc-induced neuroinflammation
To evaluate the inflammatory response following PbAc and the potential anti-inflammatory effect of LUT, the concentration of the pro-inflammatory cytokines (TNF-α and IL-1β) and Nos2 mRNA expression were determined in the cortical tissue. Our findings showed a significant increase in TNF-α and IL-1β and upregulation of Nos2 expression in PbAc-treated rats as compared with their levels in the control group. Rats treated with LUT alone showed a nonsignificant change in the levels of these inflammatory messengers. Meanwhile, rats pretreated with LUT showed a marked decrease in the inflammatory response as indicated by the decreased TNF-α and IL-1β in the cortical tissue as compared with PbAc-injected rats (Figure 5).

The protective role of LUT on the levels of TNF-α and IL-1β and Nos2 mRNA expression in the cortex tissue following PbAc exposure. mRNA expression was compared with the Gapdh. “a” represents the statistical significance against the control group at p < 0.05 and “b” represents the statistical significance against the PbAc-injected group at p < 0.05. LUT: luteolin; TNF-α: tumor necrosis factor-alpha; IL-1β: interleukin-1 beta; Nos2: inducible NO synthase; mRNA: messenger RNA; PbAc: lead acetate; Gapdh: glyceraldehyde-3-phosphate dehydrogenase.
LUT suppresses the cortical apoptosis in response to PbAc intoxication
Neuronal apoptosis has been suggested to play a role in Pb-induced neurotoxicity. In the current study, PbAc exposure enhanced the mRNA expression of Bax and caspase-3, the proapoptotic markers and suppressed the mRNA expression of Bcl2, the antiapoptotic marker in the cortical tissue as compared with the control group. The proapoptotic proteins were found to be downregulated and the antiapoptotic protein was upregulated in LUT pretreated group (Figure 6).

LUT inhibits the cortical proapoptotic protein expression and enhances the antiapoptotic protein expression following PbAc exposure. mRNA expression was compared with the Gapdh. “a” represents the statistical significance against the control group at p < 0.05 and “b” represents the statistical significance against the PbAc-injected group at p < 0.05. LUT: luteolin; PbAc: lead acetate; mRNA: messenger RNA; Gapdh: glyceraldehyde-3-phosphate dehydrogenase
LUT protects the cortical tissue in response to PbAc-induced histological alterations
Histological sections of brain stained with H&E are illustrated in Figure 7. Control and LUT-treated rats showed normal cortical morphology with normal distribution of neurons (Figure 7(a) and (b), respectively). By Contrary, PbAc-treated rats revealed deleterious histopathological changes evidenced by degenerative changes in neurons and severe vacuolation with perineural edema; some neurons were shrunken while others were shrunken and showed pyknotic nuclei (Figure 7(c)). However, LUT pre-administration repaired these abnormalities and largely preserved the neuronal structures (Figure 7(d)).

Photomicrographs of the control and LUT-treated rats showed normal cortical morphology with normal distribution of neurons ((a) and (b), respectively). By Contrary, PbAc-treated rats revealed deleterious histopathological changes evidenced by degenerative changes in neurons (blue arrow), severe vacuolation with perineural edema (black arrow), some neurons were shrunken while others were shrunken and showed pyknotic nuclei (red arrow) (c). However, LUT pre-administration repaired these abnormalities and largely preserved the neuronal structures (d). LUT: luteolin; PbAc: lead acetate;
Discussion
Heavy metals are ubiquitous environmental toxicants, originated naturally and from human activities, and caused adverse impacts in the biological system. 23 Due to its abundance and widespread applications, the exposure to Pb is associated with the progression of irreversible health problems including neuronal disturbances in animals and humans. 4 The current investigation was designed to explore the protective potency of LUT against PbAc-induced neurotoxicity through addressing the oxidative challenge, neuroinflammation, neuronal cell death, and histopathological deformations in the cortical tissue in rats. Rats treated with PbAc showed a significant increase in Pb concentration in the cortical tissue. The deposition of Pb was coupled with the development of oxidative alterations as indicated by the increased cortical LPO and NO levels along with GSH depletion. The accumulation of Pb in the cortical tissue is due to its ability to penetrate the BBB and concentrate in the brain tissue, resulting in cognitive and behavioral deficits. 24 LUT administration prior to PbAc significantly decreased the deposition of Pb in the cortical tissue. A recent study speculated that LUT may eliminate Pb through its effective chelating properties. 25 Accumulative evidences confirmed the development of oxidative stress in the brain tissue following Pb intoxication. 23,26 Brain is characterized by poor antioxidant capacity, enrich lipid content, and high oxygen consumption rate. 27 The increased neuronal lipids peroxidation in response to PbAc has been previously reported. 26 Pb is thought to induce neurotoxicity through disturbing the mitochondrial structure and function, and enhancing reactive oxygen species (ROS) generation, 28 which may trigger LPO in the cortical tissue. Oxidation of the neuronal membrane lipids upon Pb exposure impairs the membrane biophysics properties, integrity, fluidity, and function. 26 The elevated NO level may be due to the overexpression of inducible NO synthase, the rate-limiting enzyme in NO production which has been recorded in our study. These findings are in agreement with previous reports. 26,29 The overproduced NO interacts with superoxide anions, forming peroxynitrite radicals. These reactive nitrogen species have been suggested to play an essential role in Pb-mediated neuronal damage. 28 GSH is a powerful nonenzymatic antioxidant protein that protects the cell through scavenging ROS and detoxifying the toxicants. The decline in GSH following PbAc injection has been correlated with the overproduction of ROS, development of oxidative reactions, and further cellular damages. 30 The consumption of GSH in Pb biotransformation and its excretion through bile may also explain the GSH depletion. 31
PbAc injection deactivated significantly the antioxidant enzymes, namely, GR, GPx, SOD, and CAT in the cortical tissue. This decrease might be attributed to the downregulation of their mRNA expressions in the cortical tissue, which has been recorded in the current investigation. The disturbance in the cortical antioxidant enzymes following Pb intoxication has been demonstrated previously. 30,32 Pb is known to interact with sulfhydryl-containing enzymes leading to their deactivation. Additionally, Pb interferes with the enzyme activity and replaces Cu2+, Zn2+, and Fe2+ which are necessary for functioning of antioxidant enzymes leading to their deactivation. 30,33 Nrf2 signaling provides cellular protection through restoring the balance between oxidants and antioxidants and detoxifying toxicants including heavy metals. 34 Under stress conditions, Nrf2 translocates into the nucleus to bind with antioxidant response element; activating several cytoprotective genes including HO-1. 35 Our findings showed that the cortical Nfe2l2 and Hmox1 mRNA expression were downregulated in PbAc-treated rats. The downregulation of Nrf2/HO-1 signaling in the current study may explain the deactivation of the cortical antioxidant molecules.
Interestingly, LUT supplementation was able to suppress the oxidative stress produced in response to PbAc as indicated by decreasing the oxidant molecules along with increasing the enzymatic and nonenzymatic proteins in the cortical tissue. LUT has been suggested to provide neuroprotective activity through suppressing the oxidative load including the neuronal LPO. 36,37 This effect may be attributed to its ability to quench ROS. 38 Additionally, LUT was found to enhance the activity of the enzymatic (GSH) and nonenzymatic (SOD, CAT, GPx, and GR), this may be due to the upregulation of Gsr, Gpx1, Sod2, and Cat in the cortical tissue. Previous reports stated that LUT activated the cellular antioxidant defense system in different experimental models. 11,39 Additionally, LUT activated the mRNA expression of Nfe2l2 and Hmox1. Collectively, our findings indicated that LUT is able to promote Nrf2/HO-1 signaling pathway which subsequently activates the antioxidant molecules against oxidative insult-mediated neurotoxicity developed following PbAc exposure.
In the present study, levels and expression of pro-inflammatory cytokines, namely, TNF-α and IL-1β in addition to Nos2 expression, were upregulated significantly in cortical tissue of rats treated with PbAc. Previous studies indicated that Pb exposure activated microglia and enhanced the release of pro-inflammatory cytokines in these cells. 40 Our results are consistent with the data of Khalil et al. 7 and Shvachiy et al. 41 Oxidative stress-induced premature senescence in the astrocytes that may be accountable for their production of pro-inflammatory cytokines. 42 Exposure to Pb stimulates phosphorylation of NF-κB that transactivated from cytoplasm into nucleus, stimulating the activation of downstream pro-inflammatory genes. 43 Furthermore, many studies linked between oxidative stress and inflammation, and overproduction of ROS enhances cytokines release, which in turn, potentiates oxidative stress and progression of Pb-induced neurotoxicity. Therefore, restraining oxidative stress and inflammatory cytokines release is a good strategy to attenuate PbAc-induced neurotoxicity. Our results revealed that LUT markedly suppressed the levels and expression of inflammatory cytokines and Nos2 expression in cortical tissue. This is in agreement with previous reports which demonstrated that LUT inhibited secretion of high mobility group B-1 (HMGB1), and HMGB1 promotes the production of pro-inflammatory factors that involved in the aggravation of inflammatory cascades. 44 Similarly, LUT inhibited NO production and ROS generation in lipopolysaccharide-activated macrophages. 45 Further, LUT suppressed TNF-α and IL-6 production and shortened the half-lives of TNF-α and IL-6 mRNA. 46 Therefore, our results signify that the protective effect of LUT may be attributed to its ability to decrease the production of pro-inflammatory cytokines in the cortical tissue.
The biochemical results together with histopathological observations indicated that PbAc induced neuronal damage; however, the histological structure of the cortex was restored with LUT pretreatment, suggesting the efficiency of LUT to rescue cortical tissue from PbAc-induced damage. These histopathological findings suggest that PbAc induces cell death, which was confirmed by an elevation in the oxidative stress markers in the PbAc-intoxicated rats. Khalil et al. 7 reported that Pb readily crosses the BBB and directly alerted BBB structure, which further leads to morphological and functional changes in the brain architecture. However, LUT preventing the pathological changes in cortical tissue was reliable even in terms of biochemical findings. This implies that LUT effectively prevented the PbAc-induced neurotoxic damage in rats. Our findings are in accordance with Qiao et al. 40 who found that LUT administrations at 25 and 10 mg kg−1 effectively preserved the brain structure from pathological changes in cerebral ischemia.
Exposure to Pb mediates type I cell death (apoptosis) causing neuronal hyperphosphorylation. Furthermore, Pb-induced oxidative stress mediated by overproduction of ROS causes oxidative damage to cellular macromolecules such as DNA, proteins, and membrane phospholipids, which affects membrane integrity and induces apoptosis. Moreover, increased MDA and NO could lead to structural damage dysfunction to mitochondria and stimulating cytochrome c release into the cytosol, upregulating the expression levels of Bax/Bcl-2, and enhancing the activity of caspase-3. In the resent work, we found that PbAc-induced neuronal programmed cell death (apoptosis) and LUT significantly mitigated PbAc upregulated the levels and expression of Bax and caspase-3 and downregulated Bcl-2. Our findings are in agreement with the previous studies. Galal et al. 6 reported that Pb-induced apoptosis in neuronal tissue of rats by increasing intercellular calcium that distrusting mitochondrial membrane depolarization resulting in the release of cytochrome c that binds to apoptotic protease activating factor 1 (Apaf-1) which activates caspase-9 and -3. Thangarajan et al.’s 47 results indicated that PbAc administration altered Bax/Bcl-2 expression thereby increased cytochrome c release from the mitochondria. Abdel Moneim et al. 48 and Al-Quraishy et al. 26 found that Pb-induced neurotoxicity by inducing DNA fragmentation and alerting the Bcl-2/Bax ratio. However, pre-administration of LUT to PbAc protected the cortical tissue from Pb-induced apoptosis. In accordance with previous studies, Zhang et al. 49 found that flavonoid riched with LUT has neuroprotection effect against I/R injury by suppressing p53 expression and downregulating of Apaf-1 expression. Qiao et al. 40 found that the neuroprotective effects of LUT likely occurred through multiple mechanisms in experimental stroke, including suppression of oxidative stress and apoptosis.
Conclusion
In summary, LUT supplementation improved markedly the cortical injury induced following PbAc intoxication through restoring the redox homeostasis, inhibiting the inflammatory, and apoptotic cascades, along with protecting the neuronal tissue. The mechanism by which LUT exerts antioxidant, anti-inflammatory, and antiapoptotic effects is to stimulate the Nrf2/HO-1 pathway.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
