Abstract
Background
Due to the fact that methotrexate is widely used both as an immunosuppressive drug and as a chemotherapy agent, many studies are needed to reduce the side effects of this drug on non-target organs.
Purpose
This study was designed to investigate the effects of epicatechin (Epi) on MTX (methotrexate)-induced hepatotoxicity in mice.
Research Design
After 1 week for adaptation, we randomly divided 42 male Naval Medical Research Institute mice into six groups: (I) control; (II) Epi (100 mg/kg, po); (III) MTX (20 mg/kg, i.p.) on the fifth day; and (IV, V, and VI) Epi (25, 50, and 100 mg/kg, po) + MTX (20 mg/kg, i.p.) on the fifth day. At day 10, the mice were sacrificed and serum factors, oxidative stress markers, and inflammatory cytokines were measured.
Results
MTX increased activity level of serum enzymes (alanine aminotransferase and aspartate aminotransferase), lipid peroxidation marker (malondialdehyde), and inflammatory factors including interleukin-1 beta, tumor necrosis factor-alpha, and nitric oxide. Furthermore, MTX decreased glutathione level and activity level of catalase, superoxide dismutase, and glutathione peroxidase. Epi was able to reduce the destructive effects of oxidative/antioxidant system imbalance and inflammatory reactions and also histopathological damage in MTX intoxicated mice. Epi pretreatment reduced liver dysfunction by improving the antioxidant defense system, anti-inflammatory effects, and alleviation of histopathological damage in MTX hepatotoxicity.
Conclusions
Accordingly, Epi can be used as a therapeutic agent in hepatotoxicity associated with MTX chemotherapy.
Introduction
In 1949, methotrexate (MTX) was developed as a synthetic folic acid analogue and interfering with cell proliferation. 1 MTX is a chemotherapy drug used to treat some types of inflammatory diseases and cancer. 2 MTX is used at high doses for treatment of various malignancies such as acute lymphoblastic leukemia, lymphoma, medulloblastoma, and osteosarcoma. It also used in low doses for the treatment of non-malignant disorders such as rheumatoid arthritis, psoriasis, Crohn’s disease, and ectopic conception.3,4 MTX is one of the folic acid analogues that prevent the conversion of dihydrofolate (DHF) to tetrahydrofolate (THF) through competitive inhibition of DHF reductase with tendency 1000 times greater than folate. THF is necessary to make both purines and pyrimidines. The decrease in THF thymidylate synthase enzyme reduces the synthesis of thymidine nucleotide, which is an important component of deoxyribonucleic acid (DNA). MTX also inhibits the 5-aminoimidazole-4-carboxamide-1-β-d-ribofuranoside formyltransferase enzyme in the pathway of purine synthesis. Finally, by inhibiting these enzymes and reducing THF levels, MTX reduces the synthesis of DNA, RNA, and protein in the body.5–8
Many studies have reported MTX side effects, including gastrointestinal disorders, 9 intestinal mucositis, 10 myelosuppression, 11 kidney damage, 12 reproductive toxicity, 13 and life-threatening liver dysfunction. The process of MTX toxicity in the liver includes oxidative stress, inflammation, and destruction of liver tissue leading to fibrosis and cirrhosis.14,15
Flavonoids with low molecular weight are a broad group of polyphenolic agents that are found in fruits and vegetables. Flavonoids not only have oxidative stress suppressing activities but also have anti-tumor, anti-inflammatory, and immunomodulatory properties.16–18
Flavonoids as antioxidant encompass a wide range of activities from the removal of reactive oxygen species to the chelation of metals and the activation of antioxidant systems in the cells. 19 Epicatechin (Epi) is a flavonoid, which has many phenolic groups in its main structure. 20 Based on previous studies, epicatechin ameliorates doxorubicin-induced toxicity of brain and also prevents IL-6 and IL-8 and increases IL-10 production in the whole blood stimulated system.21,22 Therefore, in the present study, we have examined the effects of Epi as a natural antioxidant molecule against hepatotoxicity of MTX in mice.
Materials and methods
Chemicals and reagents
Epicatechin, MTX, thiobarbituric acid, 5,5-dithiobis (2-nitrobenzoic acid), trichloroacetic acid, phosphate-buffered saline (PBS), Bradford reagent, and other chemicals and reagents were purchased from Sigma–Aldrich Chemical Company (St Louis, MO, USA).
Animals
Forty-two male mice from the Naval Medical Research Institute (age 7 weeks; 25–30 g weights) were obtained from the Animals Center of Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran. The mice were kept under a 12 h light/dark cycles and standard conditions (temperature of 25 ± 3°C and relative humidity of 65 ± 4%). Animals were fed with standard rodent diet with free access to water and food.
Treatment schedule
After a week of adaptation, the mice were randomly divided into six groups. Each group consisted of seven mice.
Group I (control) was given distilled water (vehicle of Epi, po) 10 mL/kg for nine consecutive days and MTX vehicle (saline, 10 mL/kg, i.p.) on the fifth day.
Group II (Epi control) received 100 mg/kg/day of Epi (po) for nine consecutive days and vehicle of MTX (saline, 10 mL/kg, i.p.) on the fifth day.
Group III (MTX) was given distilled water for nine consecutive days and MTX (20 mg/kg, i.p.) on the fifth day, 2 hours before receiving distilled water.
Groups IV, V, and VI (MTX + Epi): these groups received doses of 25, 50, and 100 mg/kg/day of Epi (po) for nine consecutive days and MTX (20 mg/kg, i.p.) on the fifth day, 2 hours before taking Epi. The administration procedure of Epi and MTX is shown schematically in Figure 1. The experimental design to evaluate the effect of Epi on methotrexate hepatotoxicity in mice.
MTX dose was selected according to the study by Abdel-Daim et al. 23 and Epi doses were selected based on the research by Shariati et al. 24
After 24 h of the last Epi dose or vehicle, on the 10th day, the mice were anesthetized with ketamine HCl (100 mg/kg) and xylazine HCl (10 mg/kg), intraperitoneally. Samples of blood were obtained after decapitation and then centrifuged for 15 min at a speed of 3000 RPM. Collected sera were stored at −20 C to measure liver enzyme activity (ALT and AST). Next, the liver tissues were harvested. One portion of the liver was stored in solution of phosphate-buffered formalin (10%) to examine histological criteria, and the other portions of the liver were frozen at −80 C to evaluate subsequent hepatic markers.
Homogeneous tissue preparation
Liver samples were carefully homogenized in PBS (1:10 w/v) (0.1 M, pH 7.4) at 750 RPM for 2 min. Then, the homogenized samples were centrifuged (6000 RPM for 10 min) to collect supernatants and transferred into the microtubes. 25
Biochemical assays
Serum biomarkers
Serum AST and ALT activities were analyzed in serum, in accordance with the kit manufacturer’s protocols (Pars Azmoon Co, Tehran, Iran).
Hepatic oxidative stress assay
Catalase activity, 26 glutathione (GSH) content, 27 and malondialdehyde (MDA) levels as a lipid oxidation marker 28 were determined based on mentioned articles. The ZellBio kits were used to measure nitric oxide (NO) contents, activity of superoxide dismutase (SOD), and glutathione peroxidase (GPx) according to the kit protocols.
Liver cytokine assay
Hepatic contents of interleukin-1β (IL-1β) and tumor necrosis factor-alpha (TNF-α) were measured using the Karmania Pars Gene ELISA kits (Kerman, Iran) by the manufacturer’s procedures.
Histopathologic evaluation
The mice hepatic tissues were immediately harvested and placed in 10% formalin solution. The fixed hepatic tissues were then dehydrated in graded alcohol for embedding in paraffin. Sections of 4–6 µm were prepared and stained with hematoxylin and eosin (H&E). Six microscopy slides per mouse and six fields per slide were examined to assess histological alterations include vascular congestion, infiltration of inflammatory cells, fat deposit, and hepatocyte necrosis. The histological features were graded into four categories between 0 and 3 as normal, mild, moderate, and severe, respectively. The slides were read in a “blind” fashion and the average score was calculated for each mouse.
Statistical analysis
All data are presented as means ±SD for seven mice in all groups. The group differences were evaluated using a one-way analysis of variance followed by Tukey’s post hoc analysis by the PRISM 6 software package. p < .05 was regarded as statistically significant.
Results
Epicatechin effects on serum biomarkers
Mice exposed to MTX had a remarkable elevation in serum activity of ALT and AST compared to the control (Figure 2; p < .001). However, in Epi pretreated mice, the activity level of ALT and AST in serum was significantly reduced compared to the MTX intoxicated group, dose dependently. Effect of epicatechin (Epi) on serum parameters (alanine aminotransferase and aspartate aminotransferase) in mice hepatotoxicity of methotrexate (MTX). * (in comparison to the control group) and # (in comparison to the MTX group). *** p < .001, # p < .05, and ### p < .001.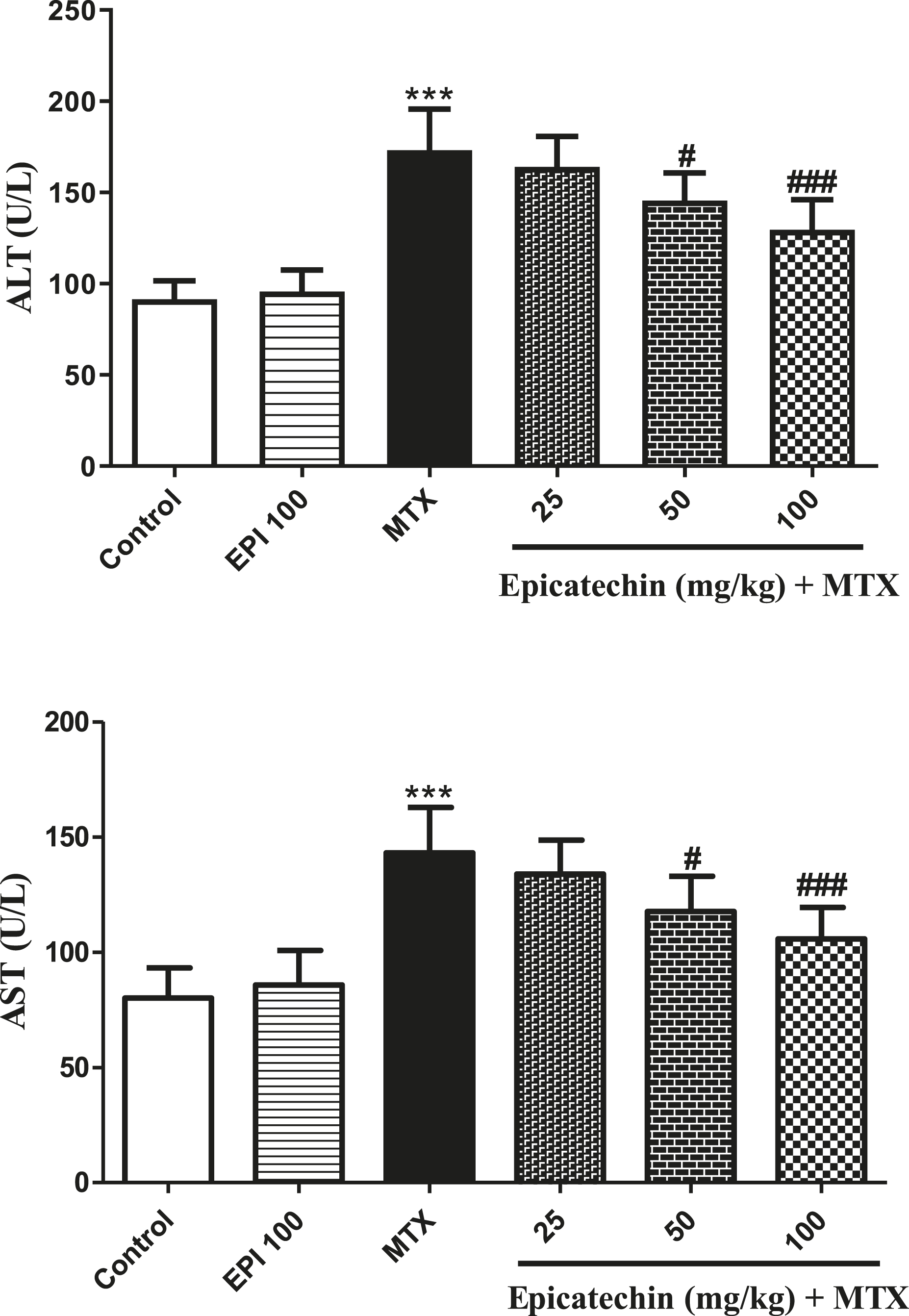
Epicatechin effects on oxidative stress and inflammatory factors
The effects of Epi on hepatotoxicity of MTX are illustrated in Figures 3–5. SOD, GPx, CAT (antioxidant enzymes), MDA (lipid peroxidation factor), interleukin-1 beta (IL-1β) and TNF-α (inflammatory cytokine factors), GSH and NO content were determined. Analysis of data represented that MTX injection significantly increased MDA, NO, and IL-1β and TNF-α formation in comparison with the control (p < .001). In contrast, pretreatment with Epi at the dose of 100 mg/kg significantly reduced the contents of MDA, NO, IL-1β, and TNF-α and enhanced the activity of antioxidant enzymes GPx, SOD, CAT, and GSH contents in comparison with the MTX administered group. The effect of epicatechin (Epi) on glutathione, nitric oxide, and MDA levels in mice hepatotoxicity of methotrexate (MTX). * (in comparison to the control group) or # (in comparison to the MTX group). *** p < .001, # p < .05, and ## p < .01. The effect of epicatechin (Epi) on activities of antioxidant enzymes (glutathione peroxidase, superoxide dismutase, and catalase) in mice hepatotoxicity of methotrexate (MTX). *(in comparison to the control group) or #(in comparison to the MTX group). *** p < .001, # p < .05, ## p < .01, and ### p < .001. The effect of epicatechin (Epi) on inflammatory cytokines (interleukin-1 beta and tumor necrosis factor-alpha) contents in mice hepatotoxicity of methotrexate (MTX). * (in comparison to the control group) or # (in comparison to the MTX group). *** p < .001, # p < .05, and ## p < .01.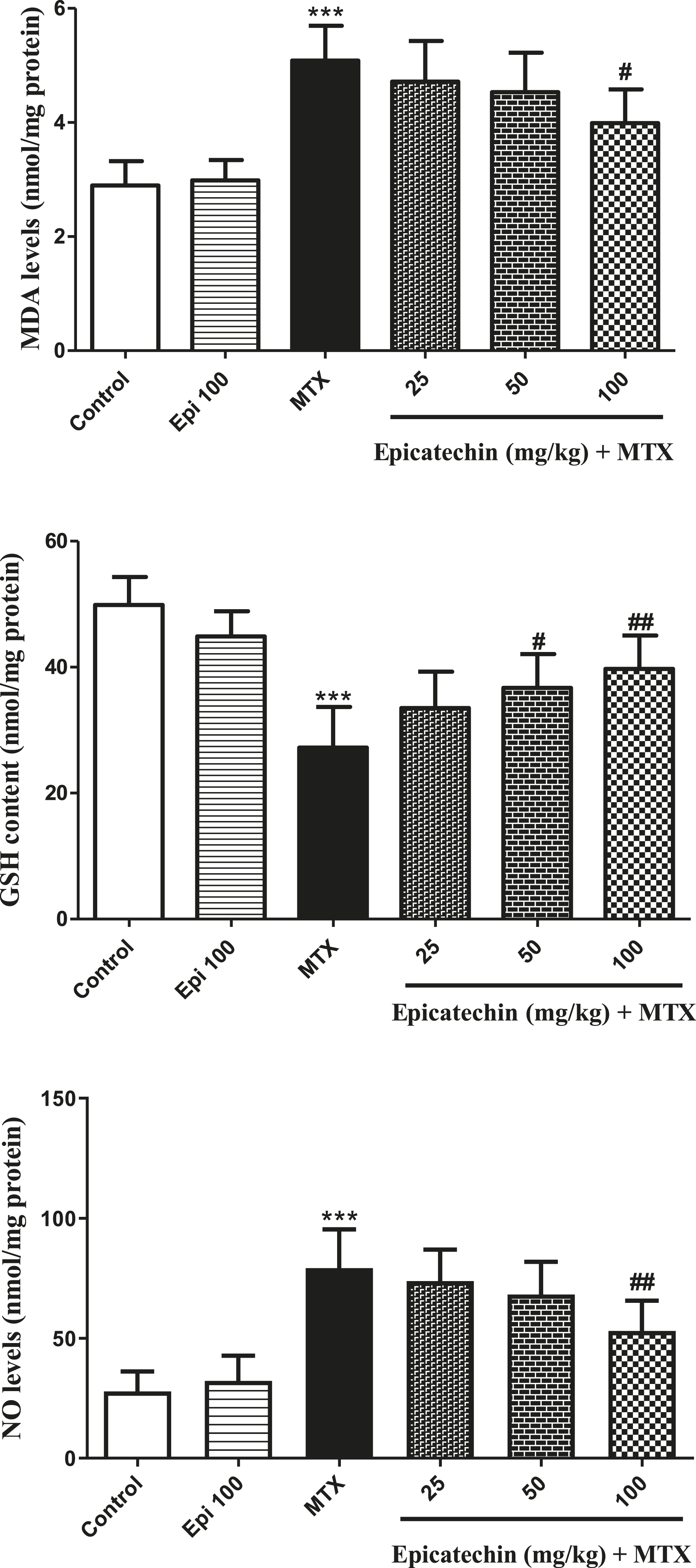


Histopathologic changes
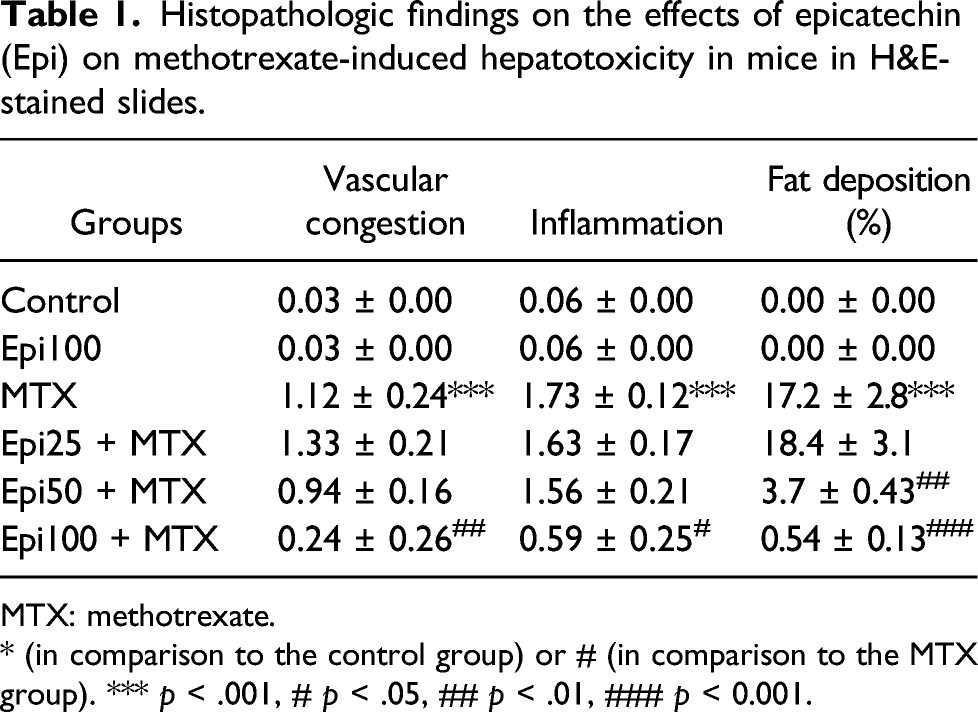
Histological parameters in the Epi group were similar to the control. MTX impaired normal histological patterns in mice hepatic tissue. Inflammatory cell infiltration, vascular congestion, and fat deposit were the most important findings in the MTX intoxicated group. Epi 100 mg/kg remarkably inhibited histological damage due to MTX (Figure 6 and Table 1). Histopathologic findings of liver sections stained with H&E under the light microscope in mice hepatotoxicity of methotrexate and the role of epicatechin (Epi). Magnification × 250. Black arrow: vascular congestion, white arrow: inflammation, yellow arrow: fat deposition. Histopathologic findings on the effects of epicatechin (Epi) on methotrexate-induced hepatotoxicity in mice in H&E-stained slides. MTX: methotrexate. * (in comparison to the control group) or # (in comparison to the MTX group). *** p < .001, # p < .05, ## p < .01, ### p < 0.001.
Discussion
According to the findings, MTX injection leads to major liver toxicity, which is associated with increased serum parameters, hepatic oxidative stress, and inflammatory biomarkers in mice. Analysis of histopathologic data confirmed these biochemical changes and indicated major liver damage in the MTX group. 28 However, pretreatment with Epi reduced liver enzyme activity levels and improved liver function. Epi significantly reduced biochemical and histopathologic changes, indicating the protective effect of this flavonoid against hepatotoxicity of MTX.
Nitric oxide is a molecule that is produced naturally in the body. Overproduction of NO contributes to overexpression of inducible NO synthase enzyme and tissue oxidative stress. NO acts as a potent cytotoxic agent in the cell through the interaction with superoxide to form peroxynitrite. 30 Our findings about NO pathogenesis in MTX toxicity are in line with previous studies.29,31
An increase in the level of MDA indicates lipid peroxidation, which ultimately leads to oxidative damage. 32 MDA is considered as a mutation agent because of its ability to change the structure of DNA and proteins.33,34 In the current study, MDA was remarkably elevated in the MTX group. However, pretreatment with Epi reduced the MDA marker in the treated groups. The findings of this experiment are consistent with previous reported study. 35
Glutathione, GPx, SOD, and CAT are important components to balance the oxidative/antioxidant system and play significant roles in preventing oxidative stress. The redox cycle of GSH is known as an important antioxidant system in the cells. MTX reduces cell GSH storage and stimulates oxidative stress by inhibiting NADPH synthesis. As an intracellular enzyme, SOD protects tissues against oxidative damage and converts accumulated superoxide radical anions (O2−) to hydrogen peroxide (H2O2). GPx and CAT convert H2O2 directly (CAT) or through the redox cycle (GPx).
36
MTX significantly reduced the levels of these antioxidants in our study. The results are consistent with previous published studies.37,38 Epi in pretreated groups had effective role against MTX hepatotoxicity. In confirmation, Epi has been reported to have antioxidant effects against doxorubicin,21,39 bleomycin,
24
nicotine,
40
and acetaminophen.
35
TNF-α and IL-1β as proinflammatory cytokines can trigger inflammatory responses and damage in mice hepatotoxicity of MTX, which is not far from the expectations of previous studies.29,41,42 The levels of IL-1β and TNF-α in MTX-intoxicated group were remarkably elevated in comparison to the control. Nevertheless, the level of these markers was significantly reduced in Epi-pretreated MTX groups. These findings are consistent with previous findings showing Epi anti-inflammatory responses in various cases.35,43,44 Histopathologic findings revealed that MTX causes histopathologic lesions including inflammatory cell infiltration, vascular congestion, and fat deposition in hepatocytes. Several other studies have confirmed these findings.45,46 As well as, histopathologic analyses indicated alleviative effects of Epi against MTX on liver. Other findings support the result of current study.47,48 The protective effects of Epi on hepatotoxicity of MTX are shown in Figure 7. Graphical abstract regarding the protective effects of epicatechin in prevention of methotrexate hepatotoxicity in mice.
Conclusion
Epi increases the body’s antioxidant capacity through enzymatic and non-enzymatic pathways. Accordingly, Epi could be a good candidate against the hepatotoxicity of MTX. Epi can also prevent elevated proinflammatory cytokines and destructive histopathological changes in MTX hepatotoxicity. Thus, Epi has promising therapeutic potential to attenuate the side effects associated with MTX chemotherapy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This paper is taken from the M.Sc. thesis of Reza Azadnasab and was financially supported by Toxicology Research Center (grant number: TRC-9802), funded by the Vice-Chancellor of Research, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran.
Ethical approval
All the experiments were done according to the Animal Ethics Committee Guidelines and approved by the Institutional Animal Care and Use Committee of the Ahvaz Jundishapur University of Medical Sciences (ethics code: IR.AJUMS.ABHC.REC.1398.002 on 15 April 2019).
