Abstract
The anti-neoplastic use of Doxorubicin (DOX) is hampered by several limitations, including reproductive toxicity. Luteolin (LUT)–a phytochemical-biological benefits include antioxidative and anti-inflammatory actions. Here we examined the protective effect of LUT against DOX-induced reproductive toxicity in an in vivo model—male albino Wistar rats—randomly assigned to five groups and treated as follows: Control (corn oil 2 mL/kg; per os), LUT (100 mg/kg; per os), DOX (2 mg/kg) by intraperitoneal injections, co-treated groups received LUT (50 and 100 mg/kg) with DOX. Treatment with DOX alone, significantly (p > 0.05), reduced biomarkers of testicular function, reproductive hormone levels, testicular and epididymal antioxidant, and anti-inflammatory cytokine. DOX increased (p > 0.05) sperm morphological abnormalities, as well as reactive oxygen and nitrogen species, lipid peroxidation, xanthine oxidase, a pro-inflammatory cytokine, and apoptotic biomarkers. Furthermore, testicular and epididymal histological lesion complemented the observed biochemical changes in treated rats. LUT co-treatment resulted in a dosage-dependent improvement in rats’ survivability, antioxidants capacity, reduction in biomarkers of oxidative stress, pro-inflammatory cytokines, and apoptosis in rat’s testis and epididymis. Also, LUT treatment resulted in improved histological features in the testis and epididymis, relative to DOX alone treated rats. LUT co-treatment abated DOX-mediated reproductive organ injuries associated with pro-oxidative, inflammatory, and apoptotic mechanisms. LUT supplementation may serve as a phyto-protective agent in alleviating male reproductive organ toxic injuries associated with Doxorubicin therapy.
Introduction
Doxorubicin (DOX) is a widely used anthracycline in the management of cancers including; breast, bladder, ovarian, esophageal, liver, and leukemia and lymphoma. 1,2 Primarily, DOX interacts with the DNA by intercalation and binds to the α-isoform of topoisomerase II, thus inhibiting DNA replication. 3,4 DOX generated reactive oxygen and nitrogen species (RONS) is another mechanism for the cytotoxic activity of DOX. 5 Consequently, DOX can cause a marked alteration in DNA structure through histone separation from the active chromatin. 6 Nevertheless, ascertaining the mechanisms involved in DOX-mediated testicular and epididymal toxicity are yet to be established. However, recent studies have shown DOX treatment mediated cell death, DNA breaks, altered genes involved in the cell cycle, oxidative stress, release of inflammatory mediators, and cellular apoptosis. 2,5,7,8 These effects linked to DOX may contribute to gonadal damage and perturbation of the spermatogenesis process, and male infertility. 9 –11 Besides gonadal toxicity, the administration of chemotherapeutic agents, including DOX, correlates with several adverse clinical manifestations ranging from organ dysfunction, decreased fertility, loss of a pregnancy, impaired cognitive function, an overall reduction in life’s quality, and early death. 12 ,–15
Various studies have shown that the use of chemoprotective agents can contribute to attenuating the unwanted side effects of anticancer drugs aimed at promoting a better quality of survivors. 2,16,17 As the risk of DOX-induced toxicity in cancer patients increases, 9,18 –21 there is a need to explore better strategies at reducing DOX toxicity. Luteolin (LUT, 2-[3,4-dihydroxyphenyl]-5,7-dihydroxy-4-chromenone) is a naturally occurring flavonoid found in fruits, leaves, vegetables, and natural herbs including parsley, thyme, peppermint, and celery. 22 –24 LUT exhibits antioxidative, anti-inflammatory, autophagic regulatory, apoptotic, and anti-neoplastic effects in several disease models from different studies. 25 –28 However, there is a lack of information on the protective effect of LUT against DOX-induced oxido-inflammatory responses in testicular and epidermal tissues of male Wistar rats. Accordingly, this study aimed to investigate the protective role of oral LUT co-treatment on oxidative and inflammatory reactions in DOX-treated rats. Here we focus on the role of LUT in modulating survivability, organ-body weight indices, testicular function biomarkers, and reproductive hormones in DOX-treated rats. Additionally, we quantified markers of antioxidant enzymes, oxidative stress, inflammatory and apoptotic responses in the rats’ testicle and epididymis. These results should provide critical insights into the effect of LUT on the links between DOX-mediated oxido-inflammatory stress and apoptotic responses in rat’s testicular and epididymal functions.
Materials and methods
Chemicals
Doxorubicin, epinephrine, glutathione (GSH), thiobarbituric acid (TBA), hydrogen peroxide (H2O2), 5,5-dithio-bis-2-nitrobenzoic acid (DTNB), Griess reagent, 1-chloro-2, 4-dinitrobenzene (CDNB), xanthine, trichloroacetic acid, bovine serum albumin (BSA), hydrogen peroxide (H2O2) were purchased from Sigma (St Louis, Missouri, USA). Monosodium dihydrogen phosphate, disodium hydrogen phosphate, sodium carbonate, sodium hydroxide, copper sulfate, potassium iodide, sodium–potassium tartrate, and sodium chloride were obtained from BDH Ltd. (Poole, Dorset, UK) and William Hopkins Ltd. (Birmingham, UK). Alkaline phosphatase (ALP), acid phosphatase (ACP), and glucose 6-phosphate dehydrogenase (G6PD) kits were obtained from Randox™ Laboratories Ltd. (Ardmore, Crumlin, Co., Antrim, UK). Enzyme-Linked Immunosorbent Assay (ELISA) kits for the assessment of Interleukin-1β (IL-1β), interleukin-10 (IL-10), tumor necrosis factor-alpha (TNF-α), and caspase-3, as well as the evaluation of serum hormones including luteinizing hormone, LH (E-EL-R0026), follicle-stimulating hormone, FSH (E-EL-R0391), prolactin (E-ELR0052) and testosterone (E-EL-R0033), were obtained from Elabscience Biotechnology Company, (Beijing, China). All other chemicals used for these experiments are of analytical grade.
Animal model and experimental design
Sexually matured, healthy male albino Wistar rats (9 weeks old, 160 ± 5 g, n = 50) were obtained from the Experimental Laboratory animal house, Faculty of Basic Medical Sciences, University of Ibadan. Experimental rats were housed in natural photoperiod (12/12-hour light–dark) conditions in a well-ventilated rodent facility, Department of Biochemistry, provided with rat pellets (Ladokun™ Feeds, Ibadan, Nigeria) and allowed free access to water. Rats were allowed to adapt (7-days) to their new environment preceding experimentation and adequately cared for as specified by “Guide for the Care and Use of Laboratory Animals” published by the National Institute of Health. Also, testing was performed following the University of Ibadan Ethical Committee’s approval (UI-ACUREC/20/035) and following the guidelines of the United States National Academy of Sciences.
Experimental protocol
The doses of Doxorubicin (2 mg/kg) and Luteolin (50 mg/kg) used in the current study were selected based on previously published data.
5,29
–31
New stock solutions of DOX (2 mg/mL) and LUT (50 mg/mL) were prepared every other day for dosing experimental rats. Rats were randomly divided to five treatment groups (n = 10), and specific groups were treated with DOX single dose, every other day for 6 days by intraperitoneal injection (i.p.), and with LUT per os (p.o.) for 14 consecutive days as follows:
Following the last treatment, on day 15, the rats were weighed. Blood was collected from the retro-orbital venous plexus into both plain and ethylenediaminetetraacetic acid (EDTA)-containing pre-labeled sample bottles under light ether anesthesia, before sacrificing the rats by cervical dislocation. Subsequently, the clotted blood was centrifuged at 3000 g for 10 minutes, 4°C to obtain the serum. The liver and kidney were immediately excised, weighed, and processed for biochemical and histological analyses. The serum samples were stored (−20°C) until required for specific biochemical analysis.
Sperm progressive motility assay
Sperm motility from the control, DOX, and LUT-treated rats was evaluated using the method of Zemjanis. 32 Epididymal sperm were obtained by cutting the cauda epididymis with surgical blades and released onto a sterile clean glass slide. The sperm was subsequently diluted with a 2.9% sodium citrate dehydrate solution, which had been pre-warmed to 37°C, mixed thoroughly, and covered with a 24 × 24 mm coverslip. At least 10 microscopic fields were observed under a phase-contrast microscope at ×200 magnification to evaluate the sperm motility. Sperm motility was calculated by scoring the number of all progressive sperm, followed by the non-progressive and then the immotile sperm in the same field. The data were expressed as a percentage of sperm progressive motility.
Epididymal sperm concentration
The epididymal sperm number (ESN) from the control, DOX, and LUT-treated rats was evaluated by the method outlined in the World Health Organization manual 33 for semen analysis. Briefly, the sperm was obtained by mincing the caudal epididymis in normal saline and filtering through a nylon mesh. An aliquot of 5 µL of the sperm was mixed with 95 µL of diluent (0.35% formalin containing 5% NaHCO3 and 0.25% trypan blue). Subsequently, 10 µL of the diluted sperm was transferred to the hemocytometer, allowed to sediment by standing for 5 minutes in a humid chamber to prevent drying before they were counted using the improved Neubauer (Deep 1/10 m; LAB ART, Munich, Germany) chamber with a light microscope at ×400.
Sperm viability and morphological abnormalities
A portion of the sperm suspension from the control, DOX, and LUT-treated rats were placed on glass slides, smeared out with another slide, and stained with a reagent containing 0.2 g eosin and 0.6 g fast green dissolved in distilled water and ethanol in a ratio of two to one (2:1) for morphological examination. In contrast, sperm viability was determined by staining with 1% eosin and 5% nigrosine in 3% sodium citrate dehydrate solution. 34 A total of 400 sperm cells from each rat were used for morphological examination.
Evaluation of serum hormones
Serum levels of LH (E-EL-R0026), FSH (E-EL-R0391), prolactin (E-ELR0052), and testosterone (E-EL-R0033) from the control, DOX, and LUT-treated rats were evaluated using ELISA plates from Elabscience Biotechnology, according to manufacturer’s protocol. The sensitivities of FSH were 0.28 ng, LH was 0.54 ng, and prolactin was 0.39 ng, whereas that of testosterone was 0.58 ng/ml. Assessment of the hormones concurrently minimized variation in inter-assay, with a minimal intra-assay coefficient of variations for FSH (2.9%), LH (3.3%), prolactin (2.4%), and testosterone (3.8%).
Evaluation of marker enzymes of testicular function
The testicular activities of acid phosphatase (ACP) and alkaline phosphatase (ALP) from the control, DOX, and LUT-treated rats were evaluated in line with the established method, which is based on the hydrolysis of p-nitrophenyl-phosphate in acid and alkaline medium respectively. 35,36 Glucose-6-phosphate dehydrogenase (G6PD) activity was determined using NADP+ and glucose-6-phosphate as substrates in line with the modified method of Wolf et al. 37
Evaluation of testicular and epididymal oxidative stress level
After the rat was sacrificed, each right caudal epididymis was harvested and placed in cold phosphate-buffered saline and cut with sterile surgical blades into pieces to collect the sperm. The solution was pipetted several times to obtain the sperm suspension and then filtered through a nylon mesh. The sperm suspensions were subsequently homogenized at 4°C with a glass Teflon homogenizer for 10 seconds and centrifuged at 2000×g for 10 minutes to obtain the supernatant, which was used for biochemical assays. The right testes were homogenized in four volumes of phosphate buffer (pH 7.4), and the resulting homogenate was centrifuged at 10 000×g for 15 minutes at 4°C. The supernatant was after that used for the oxidative stress assays. Total protein concentration was determined at 595 nm by the method of Lowry et al. 38 Total sulfhydryl group was determined at 412 nm by Ellman’s method 39 ; glutathione (GSH) levels were determined at 412 nm as previously described 40 ; glutathione-S-transferase (GST) was assayed at 340 nm by the method of Habig et al., 41 glutathione peroxidase (GPx) activity was determined at 412 nm according to the method of Rotruck 42 ; superoxide dismutase (SOD) activity was determined at 480 nm by the method of Misra and Fridovich 43 ; catalase (CAT) activity was determined at 240 nm using H2O2 as a substrate according to the method of Clairborne. 44 Furthermore, Xanthine oxidase was quantified at 290 nm by the method of Bergmeyer, 45 and Lipid peroxidation marker was quantified as malondialdehyde (MDA) at 532 nm according to the method of Buege and Aust 46 and expressed as µmol MDA/mg protein.
Quantification of reactive oxygen and nitrogen species (RONS) level
Epididymal and testicular RONS levels from the control, DOX, and LUT-treated rats were determined by the RONS-dependent oxidation of 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA) to dichlorofluorescein (DCF) as previously described. 47 The reaction mixture (150 µL 0.1 M potassium phosphate buffer; 35 µL distilled water, 10 µL sample, and 5 µL freshly prepared DCFH-DA was constituted with minimal exposure to air. Fluorescence emission of DCF produced from DCFH-DA oxidation was spectrophotometrically analyzed (wavelengths: 488 nm excitation; 525 nm emission) for 10 min at 30 s intervals using a Spectra Max 384 multimodal plate reader (Molecular Devices). DCF produced was expressed as a percentage over control.
Assay for pro-inflammatory and anti-inflammatory biomarkers
Testicular and epididymal nitric oxide (NO) level from the control, DOX, and LUT-treated rats were estimated according to the method of Green et al. 48 Briefly, an equal volume of Griess reagent and samples was incubated for 15 min; subsequently, the absorbance of the mixture was read (540 nm) and NO level in the sample calculated by extrapolation using a standard curve. Testicular and epididymal Myeloperoxidase (MPO) activities were analyzed, as previously described by Granell et al. 49 Furthermore, testicular and epididymal IL-1β, IL-10, TNF-α levels were assayed using commercially available ELISA Kits with the aid of a SpectraMax™ plate reader (Molecular Devices, CA, USA) stated in the manufacturer’s manual.
Assay for biomarkers of apoptosis
Testicular and epididymal caspase-3 activity from the control, DOX, and LUT-treated rats were evaluated using ELISA Kits Elabscience (Beijing, China) following the manufacturer’s manual. The results were obtained using a SpectraMax™ M384 Multimodal plate reader (Molecular Devices, San Jose, CA, USA)
Histopathology
Testis and epididymis samples were fixed using Bouin’s solution for 3 days. The samples were embedded in paraffin after dehydration procedures. Microtome cut tissue sections (4–5µm) and fixed on charged microscopic glass slides, were subsequently stained with hematoxylin and eosin (H & E). 50 The tissue histology slides were blinded and examined under a light microscope (Leica DM 500, Germany) by a pathologist. The histopathological aberrations were scored and reported accordingly. Representative images were captured with the aid of a digital camera (Leica ICC50 E, Germany) attached to the microscope.
Statistical analysis
Data were analyzed by the one-way analysis of variance (ANOVA) and post hoc Tukey test (GraphPad Prism 5 Software, La Jolla, California, USA, www.graphpad.com) was used to ascertain significant differences in the treatment groups. P values <0.05 were considered significant. The results were subsequently expressed as mean ± standard deviation (SD).
Results
Administration of LUT enhanced survivability, mean body weight and organo-somatic indices in DOX-treated rats
The results of survivability (Kaplan-Meyers), mean body weight, and relative testis and epididymis weights are presented in Figure 1A and B and Table 1. In comparison to the control, rats treated with DOX alone reduced rats’ survivability while LUT (50 and 100 mg/kg) enhanced the survival of rats co-treated with DOX (Figure 1a). DOX alone significantly (p < 0.05) reduced the final body weight of rats compared to other treatment groups (Figure 1b; Table 1). LUT alone resulted in weight gain. In comparison to DOX alone, groups that received DOX and LUT (50 and 100 mg/kg) revealed a slight reduction in final body weight, with percentage weight loss of 8.34% (LUT 50 mg/kg) and 4.30% (LUT 100 mg/kg). The weight change in (LUT 100 mg/kg) group is different (p < 0.05) compared to DOX alone and DOX+ (LUT 50 mg/kg). DOX alone decreased the weight of testis (p < 0.05) and epididymis (p > 0.05) compared to the control and LUT alone.
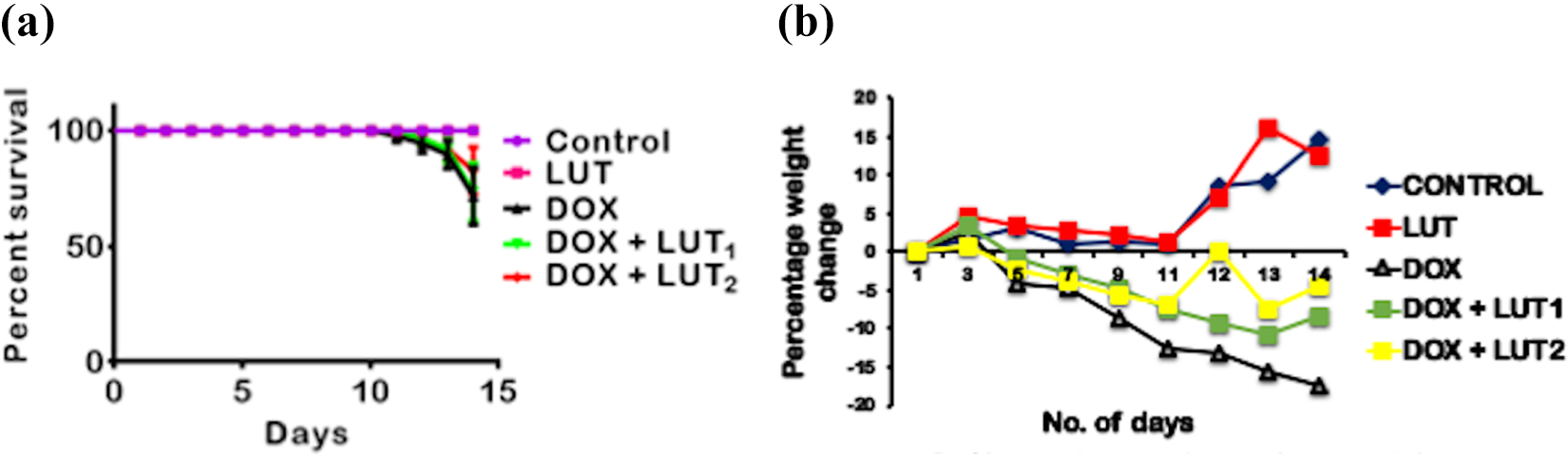
Effect of LUT on percentage survival proportion, body and relative body weight of Doxorubicin-treated rats. Doxorubicin: (DOX: 2 mg/kg); Luteolin: (LUT: 100 mg/kg); DOX+LUT1, (2 + 50) mg/kg; DOX+LUT2, (2 + 100) mg/kg. Each bar represents mean ± SD of 10 rats.
Bodyweight gain and relative testes and epididymis weights of rats following exposure to Dox for 14 days.
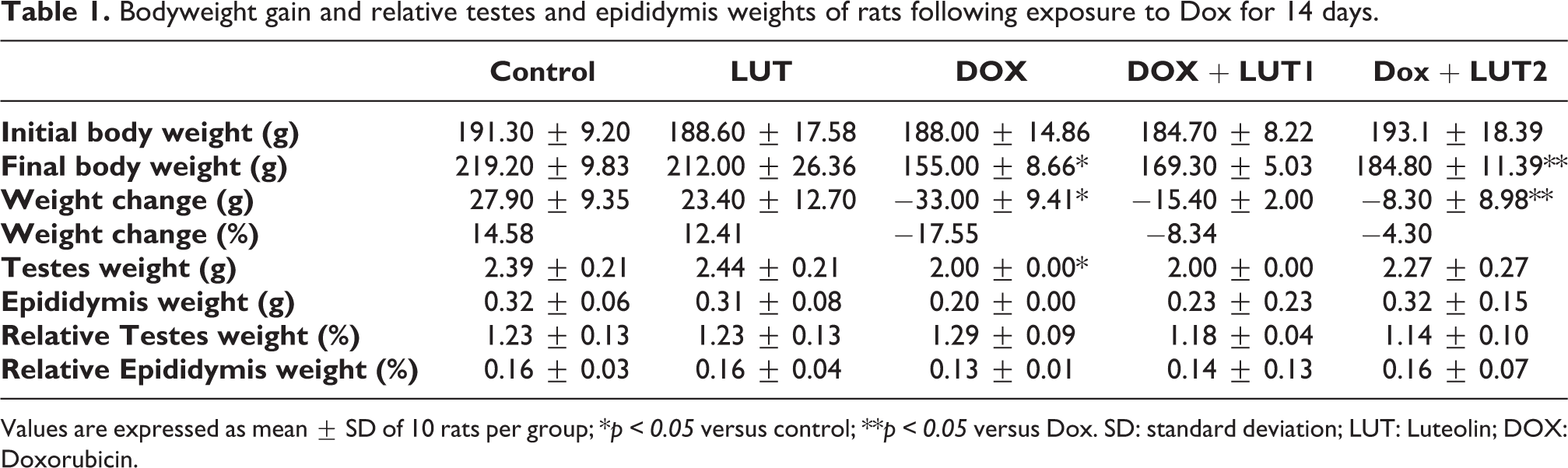
Values are expressed as mean ± SD of 10 rats per group; *p < 0.05 versus control; **p < 0.05 versus Dox. SD: standard deviation; LUT: Luteolin; DOX: Doxorubicin.
Administration of LUT ameliorated DOX-induced decreases in hormonal levels and impaired testicular function in rats
The impact of LUT on the serum concentrations of testosterone, prolactin, FSH, and LH, as well as on the activities of ALP, ACP, and G6PD in DOX-treated rats, is shown in Figure 2. Rats treated with DOX alone manifested significant (p < 0.05) variations in serum levels of testosterone, prolactin, FSH, and LH; testicular activities of ALP, ACP, and G6PD compared to the control. A negative correlation was found between serum levels of prolactin and serum levels of FSH, LH, and testosterone. However, the administration of LUT (50 and 100 mg/kg) markedly reversed DOX-induced alterations in the hormonal concentrations. Enzymatic indices of testicular functions of DOX-treated rats by significantly (p < 0.05) elevating the serum levels and testicular activities of testosterone, FSH, LH, ALP, ACP, G6PD as well as by significantly (p < 0.05) diminishing the serum level of prolactin.
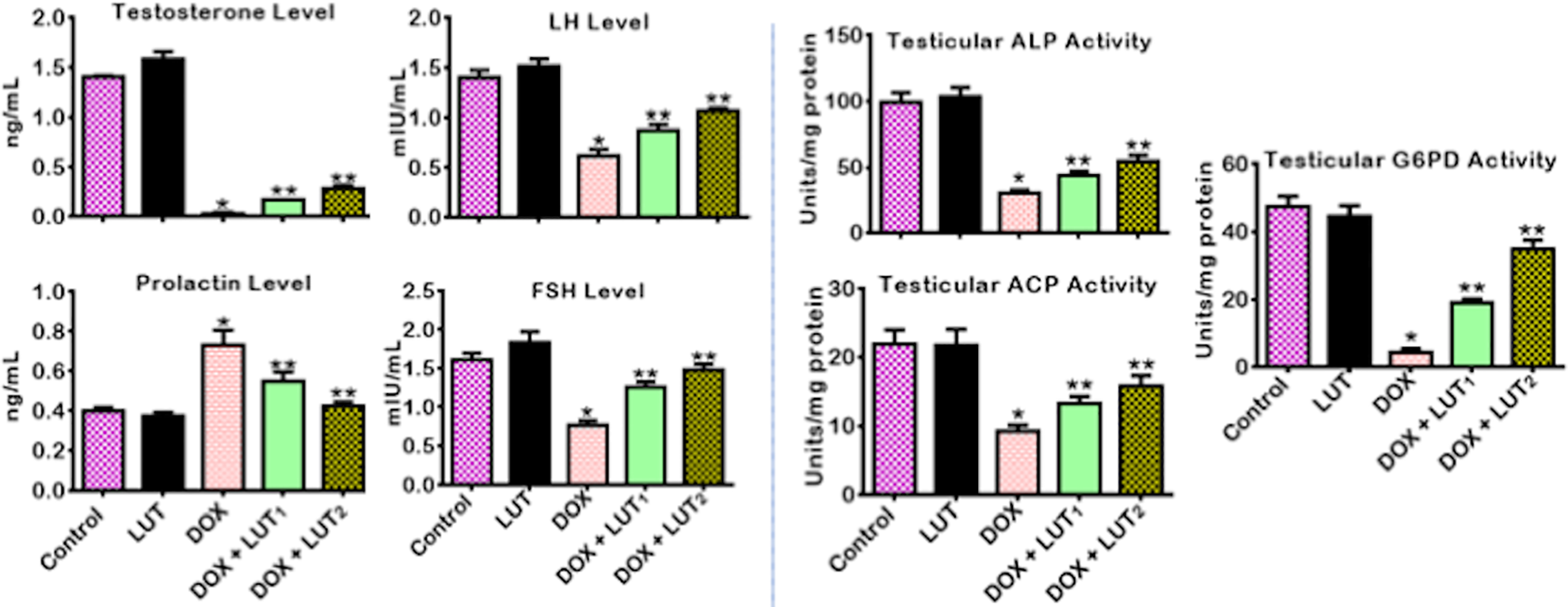
Effect of Luteolin on serum reproductive hormones and testicular biomarkers of Doxorubicin-treated rats. Doxorubicin: (DOX: 2 mg/kg); Luteolin: (LUT: 100 mg/kg); DOX+LUT1, (2 + 50) mg/kg; DOX+LUT2, (2 + 100) mg/kg. Each bar represents mean ± SD of 10 rats. *p < 0.05 versus control and **p < 0.05. Acid phosphatase: ACP; alkaline phosphatase: ALP; glucose 6-phosphate dehydrogenase: G6PD; Follicle-stimulating hormone: FSH; Luteinizing hormone: LH; standard deviation: SD.
Administration of LUT improved sperm functional parameters in DOX-treated rats
The influence of LUT on sperm motility, epididymal sperm number, sperm viability, and total sperm abnormality in DOX-treated rats is presented in Figure 3. In comparison to the control, the concentrations of epididymal sperm number, sperm motility, and sperm viability of rats exposed to DOX were diminutively (p < 0.05) decreased with a simultaneous increase in total sperm abnormality. Conversely, the administration of LUT (50 and 100 mg/kg) reduced DOX-induced deficits in sperm functional parameters and epididymal sperm number.
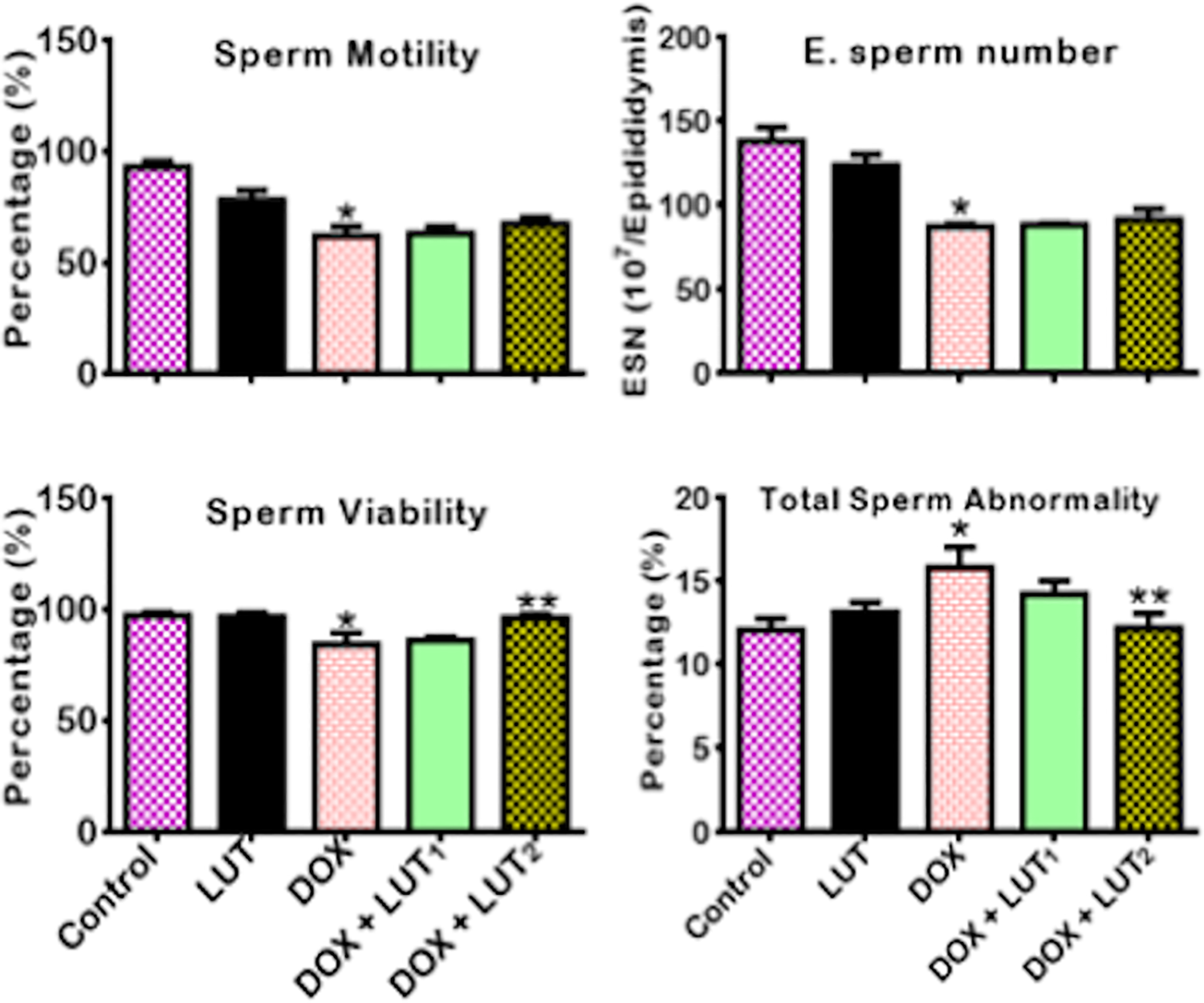
Effect of Luteolin on sperm morphological characteristics of Doxorubicin-treated rats. Doxorubicin: (DOX: 2 mg/kg); Luteolin: (LUT: 100 mg/kg); DOX+LUT1, (2 + 50) mg/kg; DOX+LUT2, (2 + 100) mg/kg. Each bar represents mean ± SD of 10 rats. *p < 0.05 versus control and **p < 0.05. Standard deviation: SD.
Administration of LUT refurbished the antioxidant status and oxidative damage in DOX-treated rats
The effects of LUT treatment on antioxidant status and biomarkers of oxidative stress in the testis and epididymis of DOX-treated rats are illustrated in Figures 4 –6. Rats treated with DOX alone manifested significant (p < 0.05) diminutions in specific antioxidant activities of GPx, GST, and level of GSH (Figure 4) together with the activities of CAT, SOD, and status of TSH (Figure 5). In contrast, the quantities of LPO, RONS, and XO (Figure 6) produced were significantly (p < 0.05) elevated in the testis and epididymis of rats. On the contrary, the administration of LUT (50 and 100 mg/kg) significantly restored the activities and levels of these antioxidants. Besides, the levels of LPO, RONS, and the activity of XO in the testis and epididymis of LUT and DOX-treated rats were significantly (p < 0.05) lowered in comparison with rats exposed to DOX alone.
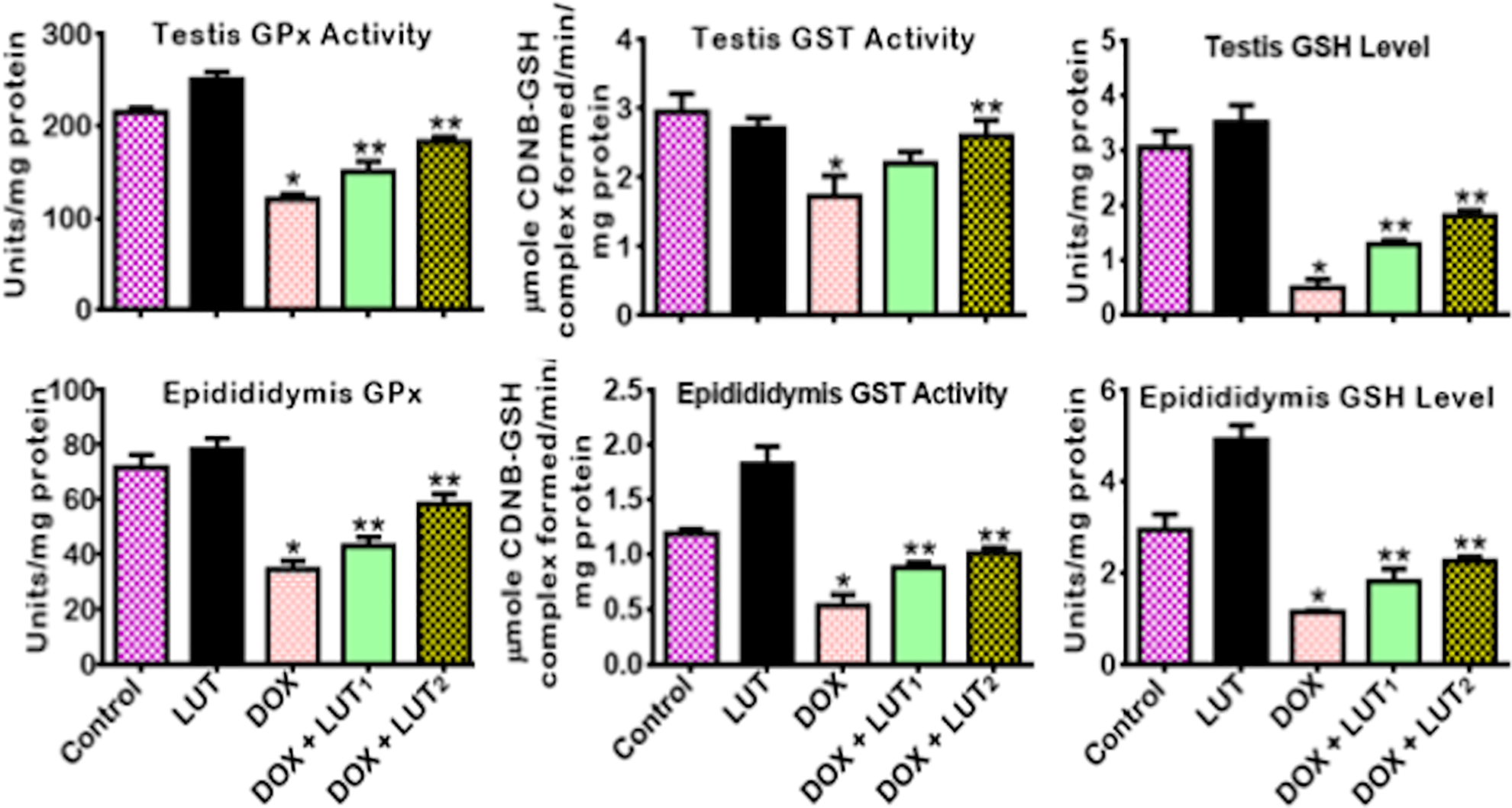
Effect of Luteolin on GPx, GST activity and GSH level in the testes and epididymis of Doxorubicin-treated rats. Doxorubicin: (Dox: 2 mg/kg); Luteolin: (Lut: 100 mg/kg); Dox+Lut1, (2 + 50) mg/kg; Dox+Lut2, (2 + 100) mg/kg. Each bar represents mean ± SD of 10 rats. *p < 0.05 versus control and **p < 0.05. Glutathione peroxidase: GPx; glutathione-S-transferase: GST; glutathione: GSH; standard deviation: SD.
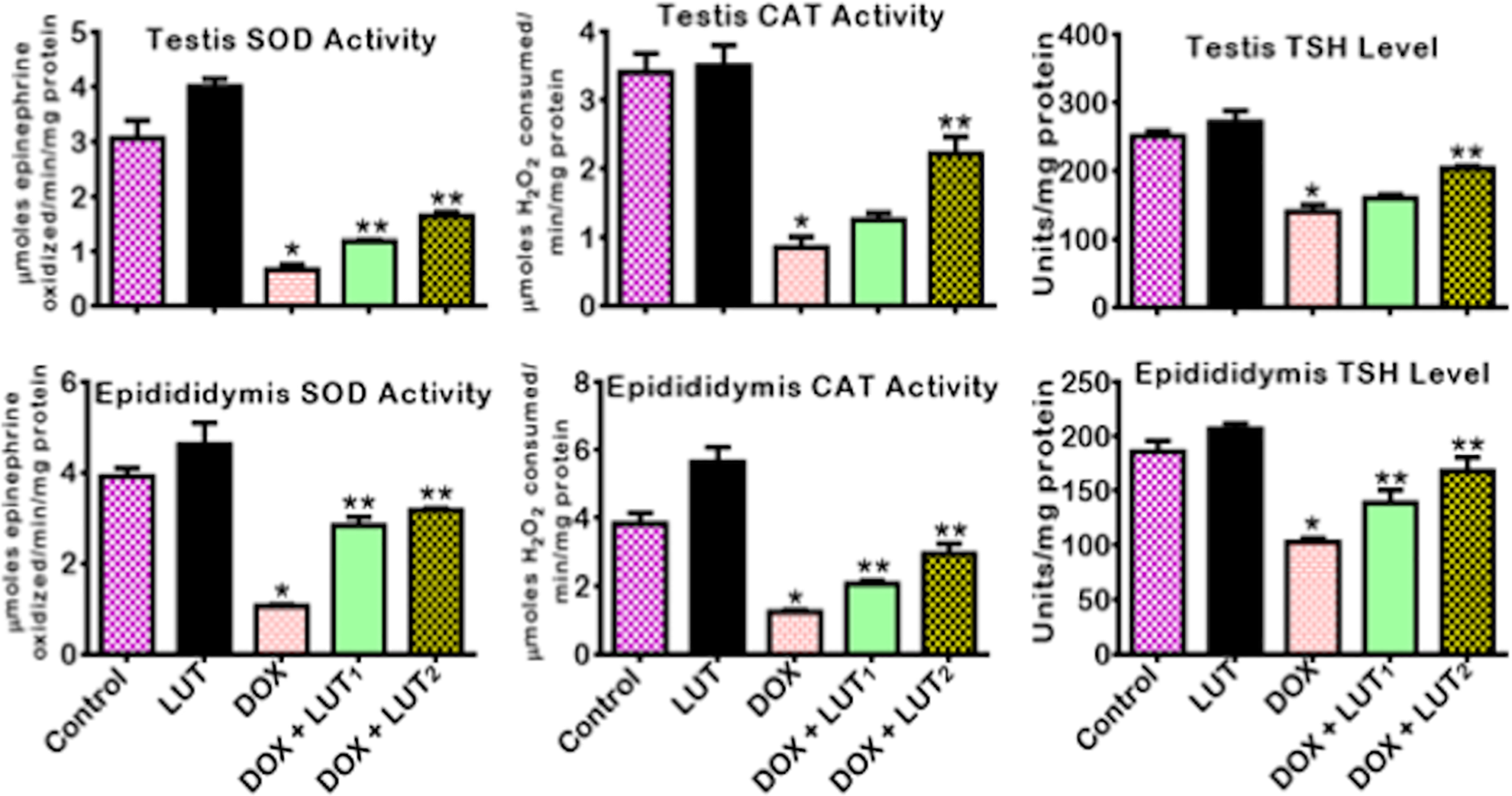
Effect of Luteolin on SOD, CAT activities and total TSH levels in the testes and epididymis of Doxorubicin-treated rats. Doxorubicin: (Dox: 2 mg/kg); Luteolin: (Lut: 100 mg/kg); Dox+Lut1, (2 + 50) mg/kg; Dox+Lut2, (2 + 100) mg/kg. Each bar represents mean ± SD of 10 rats. *p < 0.05 versus control and **p < 0.05. Superoxide dismutase: SOD; catalase: CAT; total sulfhydryl group: TSH; standard deviation: SD.
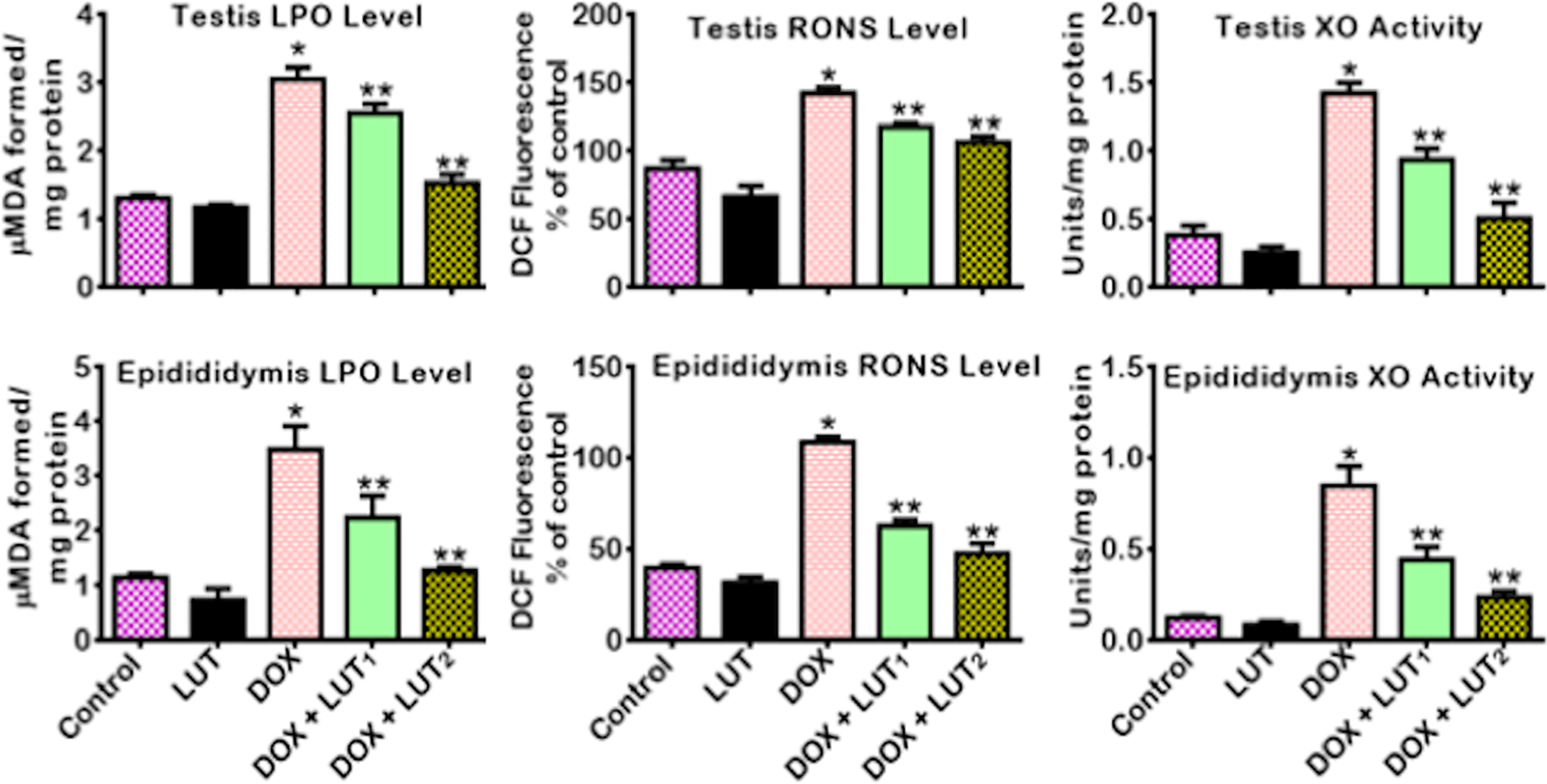
Effect of Luteolin on LPO level, and RONS and XO activities in the testes and epididymis of Doxorubicin-treated rats. Doxorubicin: (Dox: 2 mg/kg); Luteolin: (Lut: 100 mg/kg); Dox+Lut1, (2 + 50) mg/kg; Dox+Lut2, (2 + 100) mg/kg. Each bar represents mean ± SD of 10 rats. *p < 0.05 versus control and **p < 0.05. Lipid peroxidation: LPO; RONS, reactive oxygen and nitrogen species: RONS; xanthine oxidase: XO; standard deviation: SD.
Administration of LUT suppressed inflammatory responses in the testis and epididymis of DOX-treated rats
The effects of LUT treatment on pro- and anti-inflammatory cytokines in DOX-treated rats are presented in Figures 7 and 8. In comparison with the control, rats treated with DOX alone significantly (p < 0.05) increased MPO activity, NO level (Figure 7), IL-1β, and TNF-α levels with a concomitant decrease in the level of the anti-inflammatory cytokine, IL-10 (Figure 8) in the testis and epididymis of rats. Conversely, the administration of LUT (50 and 100 mg/kg) occasioned a significant (p < 0.05) decrease in the activity of MPO as well as in the levels of NO, IL-1β and TNF-α. At the same time, LUT increased IL-10 level in the testis and epididymis of DOX-treated rats compared to rats exposed to DOX alone.
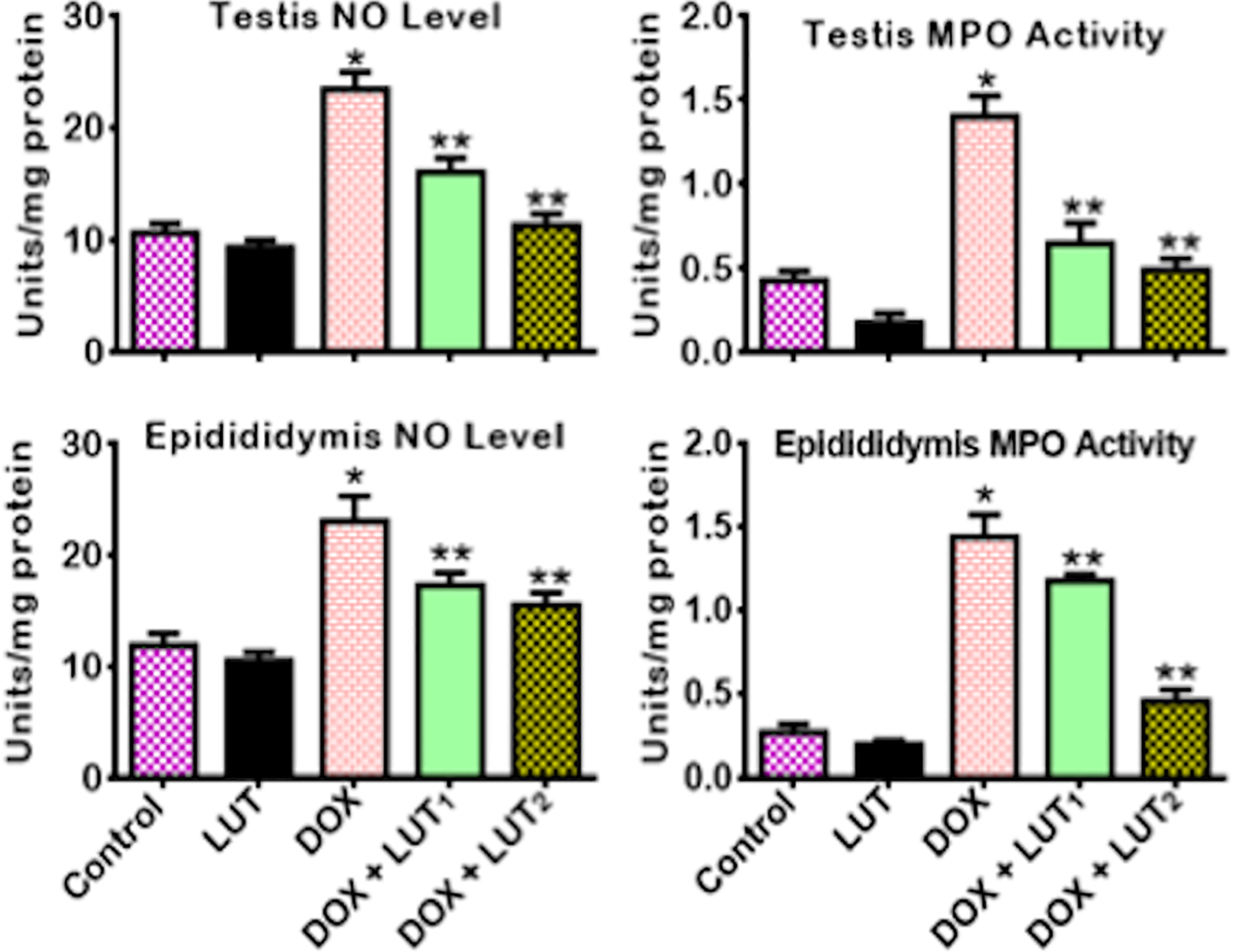
Effect of Luteolin on the NO level and MPO activity in the testes and epididymis of Doxorubicin-treated rats. Doxorubicin: (Dox: 2 mg/kg); Luteolin: (Lut: 100 mg/kg); Dox+Lut1, (2 + 50) mg/kg; Dox+Lut2, (2 + 100) mg/kg. Each bar represents mean ± SD of 10 rats. *p < 0.05 versus control and **p < 0.05. Nitric oxide: NO; myeloperoxidase: MPO; standard deviation.
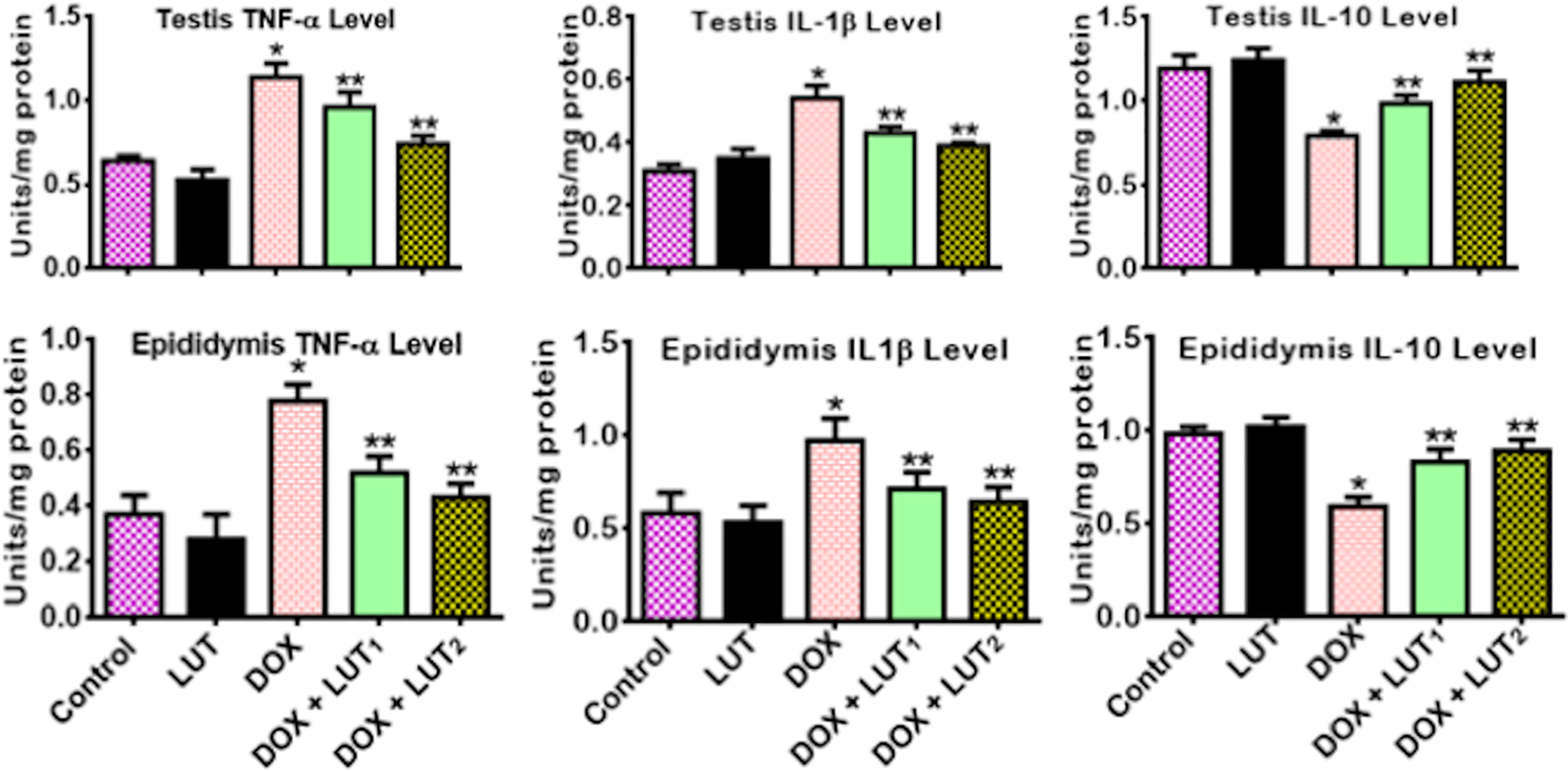
Effect of Luteolin on the level of TNF-α, IL-1β and IL-10 in the testes and epididymis of Doxorubicin-treated rats. Doxorubicin: (Dox: 2 mg/kg); Luteolin: (Lut: 100 mg/kg); Dox+Lut1, (2 + 50) mg/kg; Dox+Lut2, (2 + 100) mg/kg. Each bar represents mean ± SD of 10 rats. *p < 0.05 versus control and **p < 0.05. Tumor necrosis factor-alpha: TNF-α; interleukin-1 beta: IL-1β; interleukin-10: IL-10; standard deviation: SD.
Administration of LUT inhibited the activities of caspase-3 in the testis and epididymis of DOX-treated rats
The impact of LUT on caspase-3 activity in rats treated with DOX is presented in Figure 9. Rats exposed to DOX alone manifested a marked (p < 0.05) increase in the activities of caspase-3 in testis and epididymis in comparison with the control. Conversely, the administration of LUT to DOX exposed rats caused a significant (p < 0.05) reduction in the activity of caspases-3 in the testis and epididymis when compared to the group treated with DOX alone.
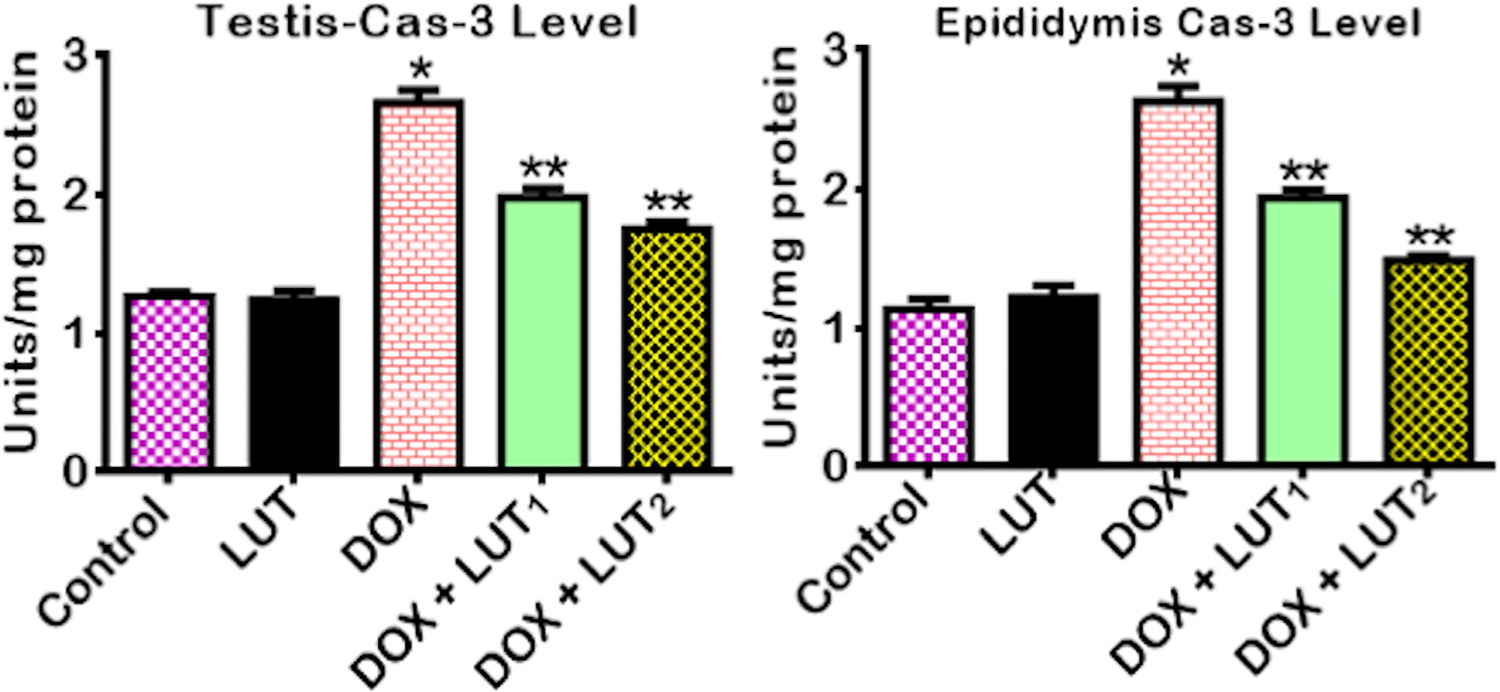
Effect of Luteolin on the activity of Caspase-3 in the testes and epididymis of Doxorubicin-treated rats. Doxorubicin: (Dox: 2 mg/kg); Luteolin: (Lut: 100 mg/kg); Dox+Lut1, (2 + 50) mg/kg; Dox+Lut2, (2 + 100) mg/kg. Each bar represents mean ± SD of 10 rats. *p < 0.05 versus control and **p < 0.05. Caspase-3: Cas-3; Standard deviation: S.D.
Administration of LUT abrogated histological alterations induced by DOX in rats
The representative histological characteristics of the testis and epididymis of treated rats are illustrated in Figures 10 and 11. Epididymal sections from the control and LUT alone revealed normal morphology of the epididymis, tubules as well as abundant spermatozoa. Rats treated with DOX alone exhibited vesicular nuclei of the epithelial lining of the epididymis with reduced spermatozoa. Conversely, the epididymis of rats administered with LUT at 50 and 100 mg/kg demonstrated histological characteristics similar to the control (Figures 10a and b). Testicular sections of the control and LUT alone exhibited the typical morphology of the testis. DOX-treated rats showed the detachment of the seminiferous tubules and vacuolization of the testicular tubules. On the contrary, testes of rats co-treated with DOX and LUT at 50 and 100 mg/kg displayed improved morphological characteristics and normal tubules (Figures 11a and b).
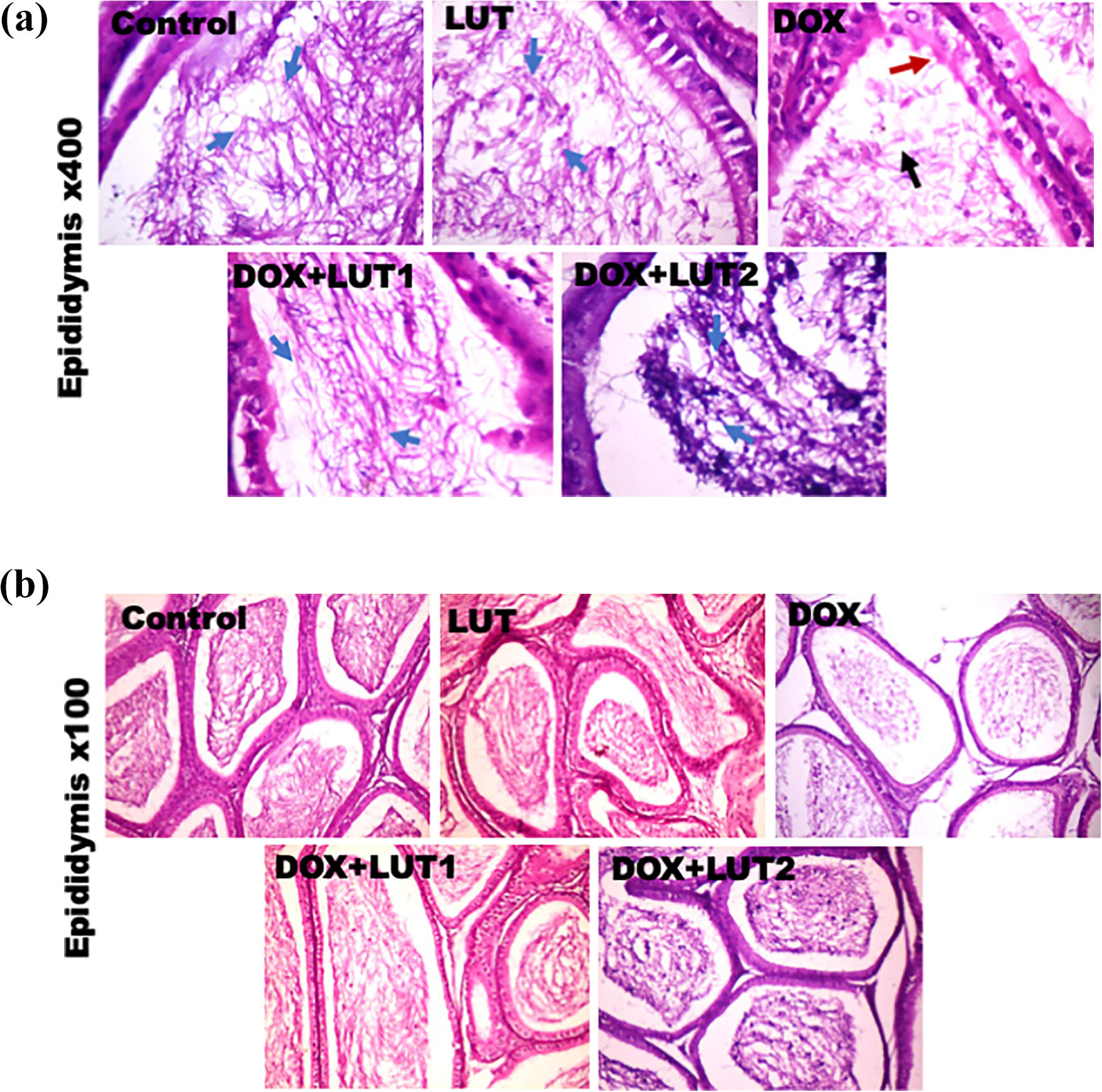
a–b: Representative photomicrographs of the epididymis from control, Lut alone, Dox alone, and co-treated groups. Control and Lut alone plates show normal morphology of the epididymis, and the tubules are essentially normal with abundant spermatozoa (short blue arrows). Dox shows vesicular nuclei of the epithelial lining of the epididymis (red arrows) with reduced spermatozoa (black arrows). Dox + Lut1 shows fundamentally improved morphology of the epididymis, with abundant spermatozoa (blue arrows). Dox + Lut2 shows normal morphology of the epididymis, and the tubules are normal with abundant spermatozoa (blue arrows). H and E stained Magnification ×400 (Figure 10a); ×100 (Figure 10b). Dox: Doxorubicin; Lut: Luteolin.
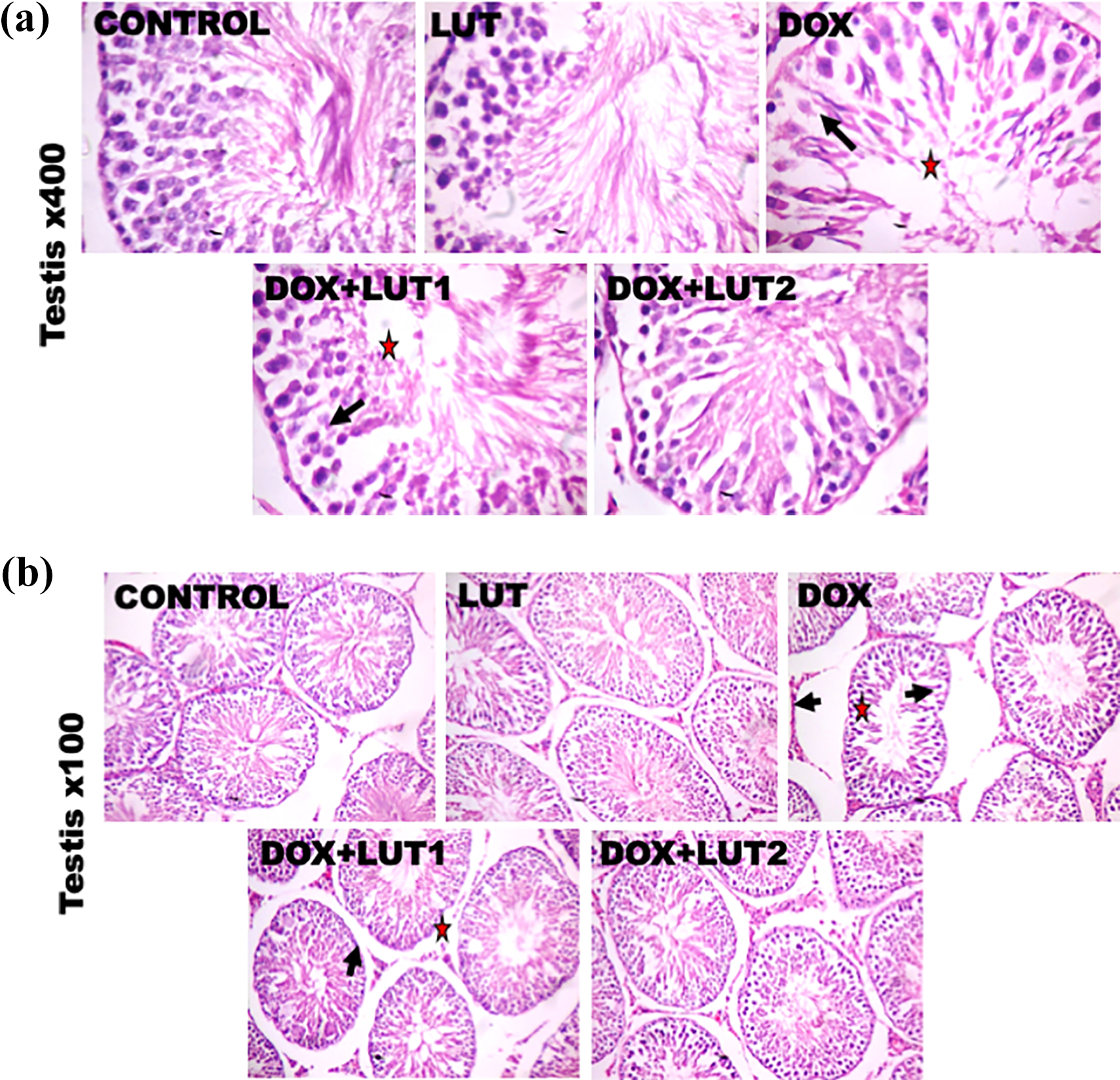
Histological representative photomicrographs of experimental rat testis treated with Luteolin and Doxorubicin. Control and Lut plates exhibit typical morphology of the testis. Dox alone treated rats showed seminiferous tubules detachment (black arrow), and vacuolization of the testicular tubules (red star). Equally, testes of rats co-treated with Dox and Lut (1 and 2) displays improved morphological features, and the tubules are essentially normal with the absence of vacuolization in testicular tubules dose-dependently. H and E stained; Magnification ×400 (Figure 11b); ×100 (Figure 11b). Dox: Doxorubicin; Lut: Luteolin.
Discussion
Luteolin is a biologically active compound found predominantly in the fruits, leaves, and vegetables of some medicinal plants. In vitro and in vivo investigations have shown few ethnopharmaceutical activities, including antioxidative, anti-inflammatory, and anti-neoplastic effects. 23,25,51 –53 Therefore, the present study revealed, for the first time, the beneficial health effects of LUT against DOX-induced oxido-inflammatory responses in the reproductive organs of male Wistar albino rats. Rats exposed to DOX exhibited marked (p < 0.05) diminutions in the mean body weight, thus indicating general toxicity and alterations of metabolic functions. The significant decrease in mean body weight in rats treated with DOX could be due to the upregulation of IL-6 and suppression of growth hormone in the experimental rats, 54,55 thus triggering anorexia, lethargy, diarrhea, depletion of nitrogen, and breakdown of vital body cell mass. Again, the reduction in the relative testis and epididymis weights following DOX treatment could be due to the degeneration of the structural morphology of the seminiferous tubules and loss of germline cells in rats. 56 Conversely, the administration of LUT abrogated the observed alterations in mean body weight and relative testis and epididymis weights of the rats, and this could be due to the direct effect of Luteolin on genes controlling IL-6 gene, 52 growth hormones well as germ cells.
In the present study, DOX caused a marked (p < 0.05) decline in serum levels of testosterone, LH, and FSH with a concomitant increase in prolactin level of rats. In this study, DOX may have elicited a gonadotoxic effect on the integrity and functionality of the Leydig and Sertoli cells, which express functional receptors for LH and FSH. 57 These hormones are responsible for the development and maintenance of the male reproductive tract and spermatogenesis, including the production of androgen, testosterone, and sperm. 58,59 The reduction (p < 0.05) in serum testosterone level in DOX alone treated rats could be as a result of germ cell apoptosis and degeneration of Leydig cell. 5,60 The increased serum level of prolactin following DOX treatment may have disturbed the hypothalamic-pituitary-gonadal-axis, inhibit gonadotropin-releasing hormone (GnRH) secretion, reduced the pulsatile release of LH, FSH, and testosterone, which are the primary cause of dysgonadogenesis, abnormal sperm motility and quality. 61,62 The balance between serum prolactin level and levels of LH, FSH, and testosterone following treatment with both doses of LUT showed the protective effect of LUT against reproductive hormone imbalance mediated by DOX in the experimental rats.
The activities of ALP, LDH, and G6PD in the testes of rats were evaluated to ascertain the protective effect of LUT in DOX-induced toxicity. The participation of these marker enzymes in the energy metabolism and stabilization of the testes is vital to sperm generation and maturation. The marked diminutions in the activities of testicular ACP and ALP in DOX-treated rats signify the release of these nonspecific phosphatases from lysosomes of the degenerating cells and rapid catabolism of the damaged germ cells, thereby leading to the interference with the division of spermatogenic cells, denaturation of the epithelium of the seminiferous tubules, phagocytosis of Sertoli cells, loss of germinal elements, reduction of the number of spermatids and subsequently decrease in the daily sperm production in the testes of rats. 63 Activities of free lysosomal enzymes have been shown to increase when testicular steroidogenesis is elevated. 64 However, a reduction in the ACP activity, as observed in DOX-treated rats, indicates decreased testicular steroidogenesis, and this may be correlated with reduced secretion of gonadotrophins. ALP is involved in the biosynthesis of nucleic acids, nuclear proteins, phospholipids, cleavage of phosphate esters, mobilizing carbohydrates and lipid metabolites for spermatozoa utilization. 65,66 Reduction in ALP activity observed in the DOX-treated rat may signify shortages in the supply of essential biosynthetic components to the Sertoli and germ cells. 59,67
Similarly, G6PD activity through the hexose monophosphate shunt in rat testes is required for the generation of reducing equivalent, nicotinamide adenine dinucleotide (NADPH) in the testes of rats. NADPH is used for the hydroxylation of steroids necessary for spermatogenesis. 68,69 In this study, the marked (p < 0.05) wane in the activity of G6PD in DOX-treated rats specified its inhibitory effect on the hexose monophosphate shunt in the testicular system of rats, which may have affected the availability of NADPH and consequently disrupted the hydroxylation of steroids in our present study. Other consequences of reduced G6PD activity may include diminution in the biosynthesis of GSH due to reduced NADPH generation as well as the production of ribulose-5-phosphate necessary for DNA biosynthesis, thus resulting to impaired DNA replication and perturbed spermatogenesis. Conversely, the administration of LUT significantly (p < 0.05) restored the activities of these marker enzymes of testicular function in DOX-treated rats similar to control. These findings indicate the beneficial properties of LUT on spermatogenesis by maintaining the activities of ALP, ALP, and G6PD, thereby mitigating DOX-induced testicular injury.
Furthermore, rats exposed to DOX alone exhibited a marked (p < 0.05) reduction in sperm functional parameters, as evidenced by diminutions in sperm motility, sperm viability, and epididymal sperm count with the concomitant elevation in total sperm abnormality. This observation delineates that DOX elicited harmful effects in the internal milieu of the epididymis, which is responsible for the transportation, storage, and maturity of sperm cells produced by the testes. Besides, wane in sperm motility, quality, and quantity could trigger male infertility and dysfunction. 59 The restoration of spermatogenic function and sperm characteristics in rats treated with LUT further validated the beneficial effect of LUT on DOX-induced testicular and epididymal toxicity.
The maintenance of normal testicular steroidogenesis, division of spermatogenic cells, and the efficiency of the epididymal epithelium is protected from oxidative stress by an antioxidant defense system comprising of SOD, CAT, GPx, GST, GSH, and TSH. The mechanism of antioxidant enzymes involves the rapid dismutation of superoxide anions (.O2 −) into hydrogen peroxide (H2O2) by SOD and the conversion of H2O2 into the water by either CAT or GPx, thereby preventing O2 − and H2O2 from participating in Haber-Weiss and Fenton reactions respectively to produce the highly hydroxyl radical (.OH). 70,71 However, inadequacy in antioxidant systems to scavenge ROS has been implicated in the onset of dysgonadogenesis. 7,59 In this study, we observed that the animals treated with DOX exhibited a marked (p < 0.05) diminution in the activities of antioxidant enzymes such as SOD, CAT, GPx, GST, as well as non-enzymatic antioxidants including GSH and TSH, similar to other reports. 5,67 Also, DOX-treated rats manifested an increase (p < 0.05) in the level of LPO and RONS, as well as the activity as XO. Increase oxidative stress may cause ROS-mediated damage to macromolecules and critical enzymes involved in testicular steroidogenesis and spermatogenesis, besides lipid peroxidation of the germ cell membrane that is rich in polyunsaturated fatty acids. 72 –74
Furthermore, the testis and epididymis of rats treated with DOX exhibited marked (p < 0.05) increased in XO activity. The activity of XO in this study may have increased following an increase in the concentration of Ca2+ since excess Ca2+ activates the protease Calpain, which converts xanthine dehydrogenase (XDH) into XO. XO and XDH are components of the xanthine oxidoreductase (XOR) system. While XDH accounts for 90% of the total activity of XOR and has no role in the initiation of oxidative stress in germ cells, it has been shown that in some pathological conditions, XDH is converted to XO, thus leading to increased production of .O2 − radicals. 75 –77 The decrease in LPO, RONS levels, and XO activity coupled with increases in antioxidant status following LUT co-treatment may be ascribed to the antioxidative and peroxidation properties of LUT as previously documented, 25,78,79 thereby suppressing oxidative injury in the testis and epididymis of treated rats.
Mediators of inflammation play a crucial role during spermatogenesis by regulating the processes of mitosis and meiosis of the spermatogenic cells, as well as by organizing the Sertoli cell activities throughout the cycle of the somniferous epithelium. 80 However, the presence of inflammatory mediators produced within the testes can interrupt the normal functions of the Sertoli cells and spermatogenic cells, thereby leading to altered spermatogenesis. In the present study, DOX-treated rats exhibited marked (p < 0.05) increases in pro-inflammatory markers such as NO, MPO, IL-1β, and TNF-α compared to the control and LUT alone. Increases in pro-inflammatory markers, denote the possibility of DOX to trigger the recognition of specific motifs, notably the Toll-like receptor (TLR), localized in the Sertoli cells. The stimulation of Sertoli cells by TLR ligand following DOX-induced inflammatory response will activate inflammatory signaling pathways leading to the mitogen-activating protein kinases (MAPK), inflammatory transcription factors, nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and interferon regulatory factors 3 (IRF3). 81 –83 MAPK, NF-κB and IRF3 then activate subsets of genes, particularly inducible nitric oxide synthase (iNOS), which catalyzes the NADPH- and O2-dependent oxidation of L-arginine to L-citrulline producing NO. 84 Excessive production of NO in the testis and epididymis of rats induces nitrosative stress, which reportedly modifies macromolecules such as DNA, protein, and lipids after exhaustion of antioxidant defense systems. Also, the activation of MPO, a heme protein synthesized during myeloid differentiation, produces hypochlorous acid, which causes further injuries to the testis and epididymis of rats. 85,86
Additionally, the role of IL-10 in the process of inflammation has been documented well. IL-10, otherwise known as an anti-inflammatory cytokine, is a potent negative feedback regulator that regulates inflammation through autocrine and paracrine mechanisms. 87 –89 Therefore, a marked decreased in IL-10, as shown in our studies, is an indication of inflammation, which could alter tissue homeostasis and trigger tissue injury. On the other hand, the significant diminutions in the levels of pro-inflammatory mediators with a concomitant increase in anti-inflammatory cytokine following LUT treatment revealed the involvement of the anti-inflammatory mechanism of LUT in abating testicular and epididymal toxicity associated with DOX.
A biological shift in the prooxidant-antioxidant cycle favoring prooxidants, as demonstrated in this study, can result in the activation of Calpain, and subsequently, p38. p38 phosphorylates and activates p53, which targets different proapoptotic genes such as Bax and Fas. Translocation of Bax into germ cells mitochondria induces its permeability resulting in the release of Cytochrome C (Cyt. C). The assemblage of Cyt. C with APaf-1 (apoptotic protease activating factor 1) and procaspase-9 to form the apoptosome will lead to the activation of procaspase-9 into active caspase 9 (initiator caspase), which in turn activates procaspase-3 into active caspase-3 (executioner caspase). Caspase-3 then cleaves apoptotic substrates containing the recognition motif DxxD leading to the proteolysis of multiple substrates, and eventually, the germ cells are committed to death. 90 –92 In this study, DOX-treated rats exhibited marked (p < 0.05) increased in the activities of caspase-9 and caspase-3, and elevated level of apoptosis in this study may have resulted in degenerations of the Sertoli and Leydig cells, thus reducing sperm production, viability, and motility. The activation of apoptosis by DOX, as observed in this study, is similar to earlier works. 93 –95 Reduction in Casp-9 and -3 activities following treatment with LUT at 50 and 100 mg/kg demonstrates the anti-apoptotic role of LUT by inhibiting the initiator and downstream executioner proteases in the testis and epididymis in treated rats. Earlier studies have shown the anti-apoptotic action of LUT. 96,97 The outcomes of the histopathology of the testis and epididymis validated the biochemical findings on the ameliorative effect of LUT against DOX-induced testicular and epididymal injuries.
In summary, the present study indicates that DOX causes impairment of testicular functions and affects the androgenicity in male rats. Concurrent treatment with LUT can ameliorate DOX-induced testicular and epididymal oxidative damage and ensure normal steroidogenic activities in experimental rats by mechanisms relating to the suppression of inflammation, oxidative stress, and apoptosis. The ethnopharmaceutical benefits elicited by LUT at the molecular levels are due to disturbance of intracellular signaling pathways. Earlier reports in the pharmacopeia delineated that LUT derives its antioxidant, anti-inflammatory, and anti-apoptotic effects via repression of JNK, p38, ERK1, NF-κB, p38 MAPK, Bax, as well as upregulation of BCl-2, Keap1, NrF2, HO-1, NQO1, KLF9, mTOR and Sirt1. 78,96 –99 It is, therefore, possible that the ameliorative impact of LUT against DOX-induced damage to the hypothalamic-pituitary-gonadal axis could be a combination of LUT scavenging ability of ROS/RNS and inhibition of proapoptotic proteins and markers of inflammations. Therefore, LUT supplementation is highly recommended to guard against male infertility among patients undergoing chemotherapy. Also, more research is needed to elucidate the molecular mechanisms by which natural supplements, including LUT, exert their testicular and epididymal ameliorative effect on the male reproductive system.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest for the research, authorship, and publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
