Abstract
MicroRNAs plays important role in cerebral ischemia-reperfusion (CIR). However, the role of miR-26b-5p in CIR injury remains unclear. PC12 cells were treated with oxygen-glucose deprivation (OGD) for 0 h, 2 h, 4 h, 6 h, and then reoxygenated for 24 h to construct an in vitro I/R model. Then, miR-26b-5p mimic, small interfering RNA of KLF10 and KLF10 overexpression plasmid were transfected into cells respectively for mechanism study. Our results showed that miR-26b-5p was downregulated in OGD/R-induced PC12 cells. After overexpression of miR-26b-5p, cell proliferation ability was enhanced, apoptosis, ROS and inflammatory mediators were inhibited. Bioinformatics analysis indicated that miR-26b-5p was directly bound to the 3’ UTR of KLF10, and downregulated the expression of KLF10. KLF10 was upregulated in OGD/R cells, and transfection with si-KLF10 promoted cell proliferation and reduced apoptosis, NO concentration and inflammatory factor secretion. Moreover, pcDNA-KLF10 reversed the inhibitory effects of miR-26b-5p mimic on apoptosis, NO content and inflammatory factor secretion, as well as the downregulation of N-myc and PTEN expression. Meanwhile, I/R rat models were constructed and divided into sham operation group (femoral artery isolation only), model group (middle cerebral artery occlusion model of rats was prepared by thread embolization), treatment group (200 µL of miR-26b-5p mimic was injected into the brain of model rats). We observed that the infarct size of brain tissue was reduced, KLF10 expression was downregulated, and apoptosis and inflammatory response were reduced. These results suggest that miR-26b-5p had protective effects on CIRI and it may be a potential treatment target.
Introduction
Cerebral ischemia-reperfusion injury (CIRI) refers to the recanalization of blood flow after ischemia in brain tissue in some cases, leading to further tissue damage and dysfunction. 1 Restoring the blood supply to the brain in time is a fundamental measure for the treatment of global cerebral ischemia. However, due to complex pathophysiological changes during reperfusion, the recovery of blood supply to the brain is reversed. It may cause a wide range of microvascular dysfunction and changes in tissue barrier function, aggravating brain dysfunction and structural damage. 2 In addition, the inflammatory response caused by CIRI can trigger systemic inflammatory response or multiple organ dysfunction syndrome, accounting for 30–40% of severe mortality. Clinical studies have shown that the pathophysiological mechanism of CIRI is related to ROS production and the secretion of pro-inflammatory factors, including IL-6 and TNF-α. 2
MiRNAs are small noncoding nucleotides and specifically expressed in various types of cells and tissues. As an important post-transcriptional regulatory molecule, microRNA can regulate the expression of target genes by binding to multiple target mRNAs. CIRI is a complex pathophysiological process involving multiple factors. It has been found that many miRNAs are upregulated or downregulated during cerebral ischemia-reperfusion. 3,4 This part of aberrantly expressed miRNAs are mainly involved in the regulation of the occurrence and development of CIRI by affecting life activities such as energy metabolism, apoptosis, ROS production and secretion of inflammatory cytokines IL-6 and TNF-α. 5,6 Our previous gene analysis found that the expression of miR-26b-5p was downregulated in the infarcted brain tissue of cerebral ischemia-reperfusion rats, which was consistent with the results of another gene chip analysis. 7 Recently, microarray analysis of mouse cerebral artery ischemia model showed that miR-26b-5p was downregulated in CIR brain tissue. Other studies have shown that downregulation of miR-26b-5p expression accompanies the progression of myocardial infarction and that it can play a central role by controlling targets related to endothelial cell growth, angiogenesis and left ventricular function. 8 It is well known that miRNAs usually act through their target genes. We predicted and screened KLF10 as a target gene by online bioinformatics. Uniquely and occasionally, some studies have reported that KLFs family members are closely related to ischemic brain injury, however, the mechanism of action of miR-26b-5p and KLF10 in cerebral ischemia-reperfusion brain still deserves further study.
Krüppel-like factor 10 (KLF10), originally named TGF-β (Transforming growth factor β) inducible early gene 1 (TIEG1), is a DNA-binding transcriptional regulator containing a triple C2H2 zinc-finger domain. 9 By binding to Sp1 (specificity protein 1) sites on the DNA and interactions with other regulatory transcription factors, KLF10 encourages and suppresses the expression of multiple genes in many cell types. 10 KLF family plays a role in proliferation, differentiation as well as apoptosis, just like other members of the SP (specificity proteins)/KLF. 11,12 At present, it has been found that KLF10-dependent TGF-β1/Smad signaling pathway is involved in the ischemia-reperfusion process of the lower limbs of rats. 13
Existing studies have shown that N-myc and PTEN are widely involved in the occurrence and development of cerebral ischemia. In OGD/R-induced astrocytes, the expression of N-myc downstream regulatory gene-2 and the secretion of pro-inflammatory cytokines increased, and inhibition of N-myc downstream regulatory gene-2 expression with pyrrolidine dithiocarbamate inhibited the inflammatory response, thereby alleviating ischemia-induced neuronal injury and hippocampal-dependent cognitive impairment. 14 Other studies found that miR-26a-5p was downregulated in myocardial I/R injury, and overexpression of miR-26a-5p activated the PI3K/AKT signal transduction pathway by targeting inhibition of PTEN expression, effectively improving the viability of cardiomyocytes and inhibiting apoptosis after I/R injury. 15
In this study, miR-26b-5p was taken as the research object, and its regulation mechanism on the occurrence and development of cerebral ischemia-reperfusion injury was explored in vivo and in vitro, respectively, to provide new ideas for the targeted treatment of this disease.
Materials and method
Cell culture, transfection and interference
PC12 cells were purchased from American Type Culture Collection (ATCC, Manassas, VA, USA) and maintained in Dulbecco’s modified Eagle’s medium (DMEM), supplemented with 10% fetal bovine serum (FBS; Gibco BRL), 100 U/mL penicillin and 100 μg/mL streptomycin in a 5% CO2 humidified atmosphere at 37°C. MiR-26b-5p mimic, negative control oligonucleotides (NC-mimic) were purchased from RiboBio Co., Ltd (Guangzhou, China) and transfected by using RiboBio Transfection Kits (RiboBio Co., Ltd). Small interfering RNA of KLF10 (siKLF10) and scramble siRNA of KLF10 (Scramble) were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). The overexpression plasmid of MAT2A (pcDNA-KLF10) and the negative control (pcDNA3.1) were purchased from RiboBio Co., Ltd (Guangzhou, China). All transfection reagent transfected into cells by using Lipofectamine 3000 Transfection Reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions.
Oxygen-glucose deprivation and reperfusion (OGD/R)
To mimic ischemic-like conditions in vitro, PC12 cells were incubated in an anaerobic chamber containing a gas mixture composed of 5% CO2 and 95% N2. The culture medium was replaced with deoxygenated glucose-free Hanks’ Balanced Salt Solution (Invitrogen). After 0 h, 2 h, 4 h, 6 h of OGD treatment, the cells were removed from the anoxia incubator and cultured under normal conditions for 24 h. Cells that were not exposed to OGD were used as controls and incubated under normal conditions.
Animals
Specific pathogen free (SPF) Sprague-Dawley rats (8-week, male) were obtained from the experimental animal center of the Fourth Military Medical University. Rats (n = 30) were housed at room where temperature and humidity were controlled (25–27°C and 45–50%, respectively), with each 12 hours light/dark cycle. Foods and water were provided ad libitum. Thirty rats were divided into two groups, of which 10 were sham operation group (femoral artery isolation only), and the remaining 20 were prepared by silk suture method and randomly divided into two groups, named model group (middle cerebral artery occlusion model of rats was prepared by thread embolization) and treatment group (200 µL of miR-26b-5p mimic was injected into the brain of model rats), with 10 rats in each group. The specific operation was to remove the thread plug and suture the wound after 90 min ischemia (survival rats as the experimental group), reperfusion for 24 h, anesthetize 2 groups of mice, take blood from the eyelids (500 µL each), and take the core tissue of cerebral infarction after cardiac perfusion with phosphate buffer saline. And then take the brain infarcted tissue for TTC staining. 16 All procedures were performed in accordance with Health Guidelines for the Care and Use of Animals and were approved by the Institutional Animal Care and Use Committee of The First Affiliated Hospital of Xi’an Jiaotong University (Xi’an, China).
RNA extraction and qPCR
Total RNA was isolated from tissues and cell lines by using the TRIzol (Invitrogen, USA). Single-stranded cDNA was synthesized with the PrimeScript Reagent Kit (Promega, USA). Real-time PCR was conducted by using SYBR Premix Ex TaqTM Kit (TaKaRa). The reaction was run in ABI7500 Real-time PCR system (Applied Biosystems, Carlsbad, CA). U6 was used as an endogenous control. The RT-qPCR cycling conditions consisted of: 95°C for 10 min; then 35 cycle amplification for 20 s at 95°C, 30 s at 55°C, 15 s at 72°C; followed by 1 min at 72°C. The primers used in this study were synthesized from Sangon Biotech (Wuhai, China). Data were analyzed by using the 2−ΔΔCT method. First, for all test samples and calibration samples, the CT values of target genes were normalized with the CT values of internal reference genes: ΔCT (test) = CT (target, test) − CT (ref, test), ΔCT (calibrator) = CT (target, calibrator) − CT (ref, Calibrator); secondly, normalize the ΔCT value of the test sample with the ΔCT value of the calibration sample: ΔΔCT = ΔCT (test) − ΔCT (calibrator), and finally, calculate the expression level ratio: 2−ΔΔCT = the ratio of expression amount. 17 The following primers were used: miR-26b-5p (forward 5’–3’: ATCTCAGTCCTGTACAGTAC, reverse 5’-3’: CGT ACA TGC ACA TAC AGT C); KLF10 (forward 5’–3’: ACG GTG CCT CGG GTT GTG; reverse 5’–3’: TCG ACC AAT CAG CGG TAA AGG).
Western blotting
Cells were harvested and lysed in RIPA lysis buffer (Beyotime, Haimen, China). Protein concentration was assessed by using a BCA protein assay kit (Beyotime, China). The equal amount (10 μg/lane) of protein samples was separated by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) at 70 V for 30 min and 120 V for 90 min, and then transferred onto polyvinylidenedifluoride (PVDF) membranes at 250 mA for 80 min (Bio-Rad, Hercules, CA, USA). The membranes were blocked in 5% (w/v) nonfat dry milk in TBST (Tris-buffered saline–0.1% Tween) at 25°C for 3 h and then incubated with the following primary antibodies: rabbit polyclonal anti-GAPDH antibody (1:800, Abcam, ab8227), rabbit monoclonal anti-Bax-antibody (1:300, Abcam, ab32503), rabbit polyclonal anti-KLF10 antibody (1:500, Abcam, ab73537), rabbit polyclonal anti-Cleaved Casepase3 antibody (1:700, Abcam, ab2302), rabbit monoclonal anti-PTEN anti-body (1:400, Abcam, ab32199). The membranes were all incubated overnight at 4°C, washed with 1×TBST (6 washes, 6 min per wash). Then, the bands were incubated by using horseradish peroxidase (HRP) conjugated goat anti-rabbit IgG (1:12000, Boster) for 2 h incubation at room temperature. Changes in protein expression were determined after normalizing the band intensity of each lane to that of GAPDH. The imaging was performed with electron-chemiluminescence (ECL) emitting solution. The proteins were visualized with an Anmobilon Western Chemiluminescent HRP Substrate system (Millipore Corp., Billerica, MA, USA).
ELISA
The secretion of IL-6 and TNF-α in cerebrospinal fluid and cell culture supernatant were detected by ELISA kits (Sigma, St Louis, MO, USA) following the manufacturer’s instructions.
Luciferase reporter gene assay
The putative relationship between miR-26b-5p and KLF10 were predicted by using TargetScan (http://www.targetscan.org/). The wild type and mutant 3’-UTR sequences of KLF10 were cloned into the pcDNA3.1 (+) vector (Invitrogen, Carlsbad, CA, USA) containing the luciferase reporter gene, respectively. HEK293 cells were seeded in 24-well plates, and when grown to approximately 70% confluence, co-transfected with luciferase plasmid and miR-26b-5p mimic or NC-mimic by using Lipofectamine 2000. After 24 hours of transfection, the luciferase reporter activity was measured by Dual-Luciferase Reporter Assay System (Promega, Madison, WI) under the manufacturer’s instructions.
Cell proliferation
MTT kit (Glenview, USA) was used to measure cell proliferation by comparing optical density. OGD/R cells were seeded at a density of 104 cells/well in 96-well plates. After 24 h, 10 µL of MTT at a concentration of 5 mg/mL was added to each well and incubated at 37°C for 4 h in the dark. Finally, the reduction of MTT was quantified by a microplate reader by measuring the absorbance at 450 nm.
Cell apoptosis
The treated cells (100 μL) of 106 cells/mL were incubated with Annexin-VAPC (allophycocyanin, BD Biosciences Pharmingen, San Diego, CA, USA) and PI (BD Biosciences Pharmingen) in the dark for 15 min. Then, 400 µL of binding buffer was added to the sample. Finally, the samples were analyzed by flow cytometry, and the data were analyzed with the CELLQUEST software.
ROS measurement
To detect intracellular ROS, cells were incubated with 20 μM CM-H2DCFDA [5-(and-6)-chloromethyl-2, 7’-dichlorodihydrofluorescein diacetate, acetyl ester] for 30 min at 37°C, followed by fixing with 4% paraformaldehyde at room temperature for 10 min, and then mounted with DAPI. The fluorescence intensity was measured by flow cytometry at the excitation wavelength of 485 nm and emission wavelength of 538 nm.
Statistical analysis
All statistical analyses were performed by using the SPSS software (ver. 13.0; SPSS, Chicago, IL). The quantitative data derived from three independent experiments are expressed as mean ±SD. Statistical comparisons were performed by using one-way analysis of variance followed by the post-hoc test. P < 0.05 was considered statistically significant.
Results
MiR-26b-5p was downregulated in OGD/R cells and promoted apoptosis and secretion of inflammatory factors
In order to establish an in vitro ischemia-reperfusion injury model, PC12 cells were treated with hypoxic and sugar-free conditions for 0 h, 2 h, 4 h and 6 h, respectively and then reoxygenated for 24 h. RT-qPCR results showed that the expression of miR-26b-5p was significantly decreased with the prolongation of OGD treatment time (Figure 1(a)). MTT assay results showed that the longer OGD treatment significantly inhibited PC12 cells proliferation (Figure 1(b)). Flow cytometry were used to detect apoptosis of PC12 cells, and the results showed that apoptosis was significantly increased after OGD treatment for 6 h. Similarly, the expression of apoptosis-related proteins such as cleaved casepase3 and BaX were significantly increased (Figure 1(c)). In addition, we found that the levels of ROS, IL-6 and TNF-α were significantly increased after OGD treatment for 6 h compared with 0 h (Figure 1(d) and (e)).
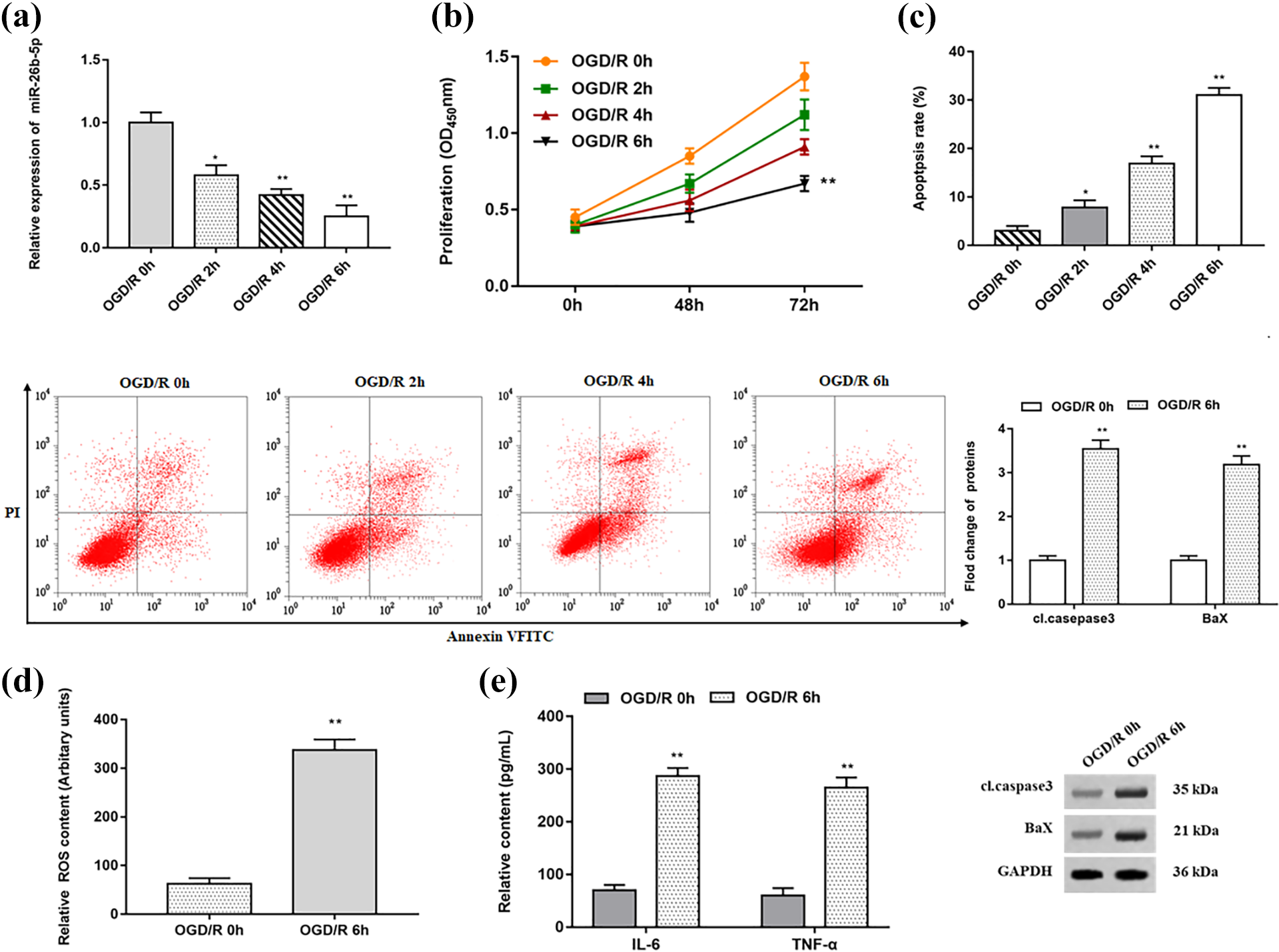
MiR-26b-5p was downregulated in OGD/R cells. PC12 cells were reconstituted for 48 h after oxygen deprivation treatment for 0 h, 2 h, 4 h and 6 h to establish an I/R model in vitro. (a) Relative expression of miR-26b-5p in OGD/R cells were analyzed by RT-qPCR assay. (b) OGD/R cell proliferation were analyzed by MTT assay. (c) Apoptosis of OGD/R cells were detected by flow cytometry and Western blotting was used to detect the expression changes of apoptosis-related proteins. (d) Relative content of ROS in OGD/R cells were analyzed by DCFH-DA stain. (e) ELISA assay was used to detect the secretion levels of IL-6 and TNF-α. β-actin was used as an internal reference. N = 3, * P < 0.05, ** P < 0.01.
MiR-26b-5p mimic promoted proliferation and inhibited apoptosis of OGD/R cells
PC12 cells treated with OGD/R were transfected with NC mimic and miR-26b-5p mimic, respectively. RT-qPCR results showed that miR-26b-5p mimic significantly upregulated miR-26b-5p, but with the prolongation of OGD treatment time, the expression of miR-26b-5p was gradually inhibited (Figure 2(a)). In addition, the proliferation ability of PC12 cells transfected with miR-26b-5p mimic were significantly enhanced compared with the control group (Figure 2(b)). Similarly, miR-26b-5p mimic significantly reduced the percentage of cells in the G1 phase and increased the percentage of cells in the S phase (Figure 2(c)). Flow cytometry results showed that miR-26b-5p mimic significantly inhibited apoptosis after OGD treatment for 6 h. Furthermore, the expression of apoptosis-related proteins such as cleaved casepase3 and BaX were significantly decreased (Figure 2(d)). In addition, we found that miR-26b-5p mimic significantly reduced ROS, IL-6 and TNF-α levels after 6 h of OGD treatment (Figure 2(e) and (f)).

The role of miR-26b-5p in the model of ischemia-reperfusion in vitro. (a) OGD/R cells were transfected with miR-26b-5p mimic or NC mimic, and the expression of miR-26b-5p was detected by RT-qPCR assay. (b) Cell proliferation was analyzed by MTT assay. (c) Flow cytometry was used to detect the changes in cell cycle. (d) Apoptosis of OGD/R cells after transfected with miR-26b-5p mimic were detected by flow cytometry and Western blotting was used to detect the expression changes of apoptosis-related proteins. (e) ROS content was detected by DCFH-DA stain. (f) The secretion levels of IL-6 and TNF-α were detected by ELISA assay. β-actin was used as the loading control. N = 3, * P < 0.05, ** P < 0.01.
MiR-26b-5p protected against damage caused by OGD/R by targeting KLF10
We predicted possible target genes for miR-26b-5p through TargetScan 7.1, and we found that miR-26b-5p directly targets the 3’UTR of KLF10. The wild type and the mutant sequence of KLF10 were shown in Figure 3(a). Next, the luciferase reporter gene analysis showed that miR-26b-5p mimic significantly reduced the luciferase activity of wild-type KLF10, but had no significant effect on the luciferase activity of mutant-type KLF10 (Figure 3(b)). In addition, RT-qPCR and Western blotting results showed that miR-26b-5p mimic significantly reduced the mRNA and protein levels of KLF10 after 6 h of OGD treatment compared with the control group (Figure 3(c)).

KLF10 is a target gene of miR-26b-5p. (a) TargetScan 7.1 was used to predict the targeting site of miR-26b-5p in KLF10 mRNA 3’UTR. (b) The relative luciferase activities in wild-type KLF10 group and mutant-type KLF10 group were tested. (c) The mRNA and protein expression of KLF10 in OGD/R cells transfected with miR-26b-5p mimic were analyzed by RT-qPCR assay and Western blotting. β-actin was used as an invariant internal control for calculating protein-fold changes. N = 3, * P < 0.05, ** P < 0.01.
Furthermore, RT-qPCR and Western blotting results confirmed that KLF10 expression was significantly upregulated with the prolongation of OGD time (Figure 4(a)). Next, the KLF10 siRNA was transfected into OGD-treated cells at 0 h and 6 h, respectively. The results showed that transfection of KLF10 siRNA significantly decreased the expression of KLF10 at mRNA and protein levels after 6 h of OGD treatment (Figure 4(b)). Similarly, MTT assay results showed that KLF10 siRNA transfection after OGD treatment for 6 h significantly improved cell proliferation (Figure 4(c)), while apoptosis was inhibited, and the expression of apoptosis-related proteins such as cleaved casepase3 and BaX were also significantly reduced (Figure 4(d)). What’s more, compared with the control group treated with OGD for 6 h, the content of ROS and NO in the KLF10 siRNA transfection group was significantly decreased (Figure 4(e)), and the secretion of IL-6 and TNF-α were also significantly decreased (Figure 4(f)).

KLF10 promoted CIRI in OGD/R cells. PC12 cells were reconstituted for 48 h after oxygen deprivation treatment for 0 h, 2 h, 4 h and 6 h to establish OGD/R model in vitro. (a) The mRNA and protein expression of KLF10 in OGD/R cells were analyzed by RT-qPCR assay and Western blotting. (b) KLF10 mRNA and protein levels were detected after the transfection of scramble or KLF10 siRNA. (c) Cell proliferation was measured by the MTT assay. (d) Cell apoptosis was measured by flow cytometry and Western blotting was used to detect the expression changes of apoptosis-related proteins. (e) ROS content and NO concentration were detected by DCFH-DA stain and ELUSA kits, respectively. (f) The secretion levels of IL-6 and TNF-α were detected by ELISA. β-actin was used as the loading control. N = 3, * P < 0.05, ** P < 0.01.
MiR-26b-5p post-transcriptionally suppressing the N-myc/PTEN axis by inhibiting KLF10 expression to reduce the OGD/R injury of PC12 cells
To further explore the regulatory effect of KLF10 on OGD/R in in vitro model, miR-26b-5p mimic were transfected alone or together with pcDNA-KLF10 into PC12 cells treated with OGD for 6 h, respectively. Western blotting results showed that pcDNA-KLF10 significantly reversed the expression of apoptosis-related proteins such as cleaved casepase3 and BaX compared with miR-26b-5p mimic transfected alone (Figure 5(a)). N-myc and PTEN are downstream genes of KLF10, and their expression is positively regulated by KLF10. As expected, the protein expression of N-myc and PTEN was significantly upregulated when miR-26b-5p mimic and pcDNA-KLF10 transfected together (Figure 5(a)). What’s more, pcDNA-KLF10 significantly reversed the reduction of ROS, NO, IL-6 and TNF-α secretion by miR-26b-5p mimic (Figure 5(b) and 5(c)).

MiR-26b-5p regulated the N-myc/PTEN axis by inhibiting KLF10 to reduce the injury in IRI. OGD/R cells were transfected with miR-26b-5p mimic alone or together with pcDNA-KLF10. (a) Cleaved casepase3, Bax, N-myc and PTEN protein levels were detected by Western blot assay. (b) ROS content and NO concentration were detected by DCFH-DA stain and ELISA kits, respectively. (c) The secretion levels of IL-6 and TNF-α were detected by ELISA. β-actin was used as an internal reference. N = 3, * P < 0.05, ** P < 0.01.
MiR-26b-5p had a protective effect on cerebral ischemia reperfusion in vivo
To further explore the protective effect of miR-26b-5p in vivo, we set up sham groups and I/R groups, in which I/R groups was further divided into NC mimic groups (control groups) and miR-26b-5p mimic groups (experimental groups). TTC staining results showed that the gray and white infarct tissue areas in experimental groups were significantly smaller than that of control groups (Figure 6(a)). In addition, RT-qPCR results showed that miR-26b-5p was upregulated in cerebral infarction tissues and peripheral blood in experimental groups (Figure 6(b) and 6(c)). In contrast, mRNA and protein expression of KLF10 were significantly inhibited in experimental groups (Figure 6(d)). Next, Western blotting results revealed that miR-26b-5p mimic downregulated the protein expression of cleaved casepase3, BaX, N-myc and PTEN (Figure 7(a)). Finally, we found that the levels of oxidative stress factor ROS, or inflammatory factors IL-6 and TNF-α were significantly reduced in experimental groups (Figure 7(b) and 7(c)).
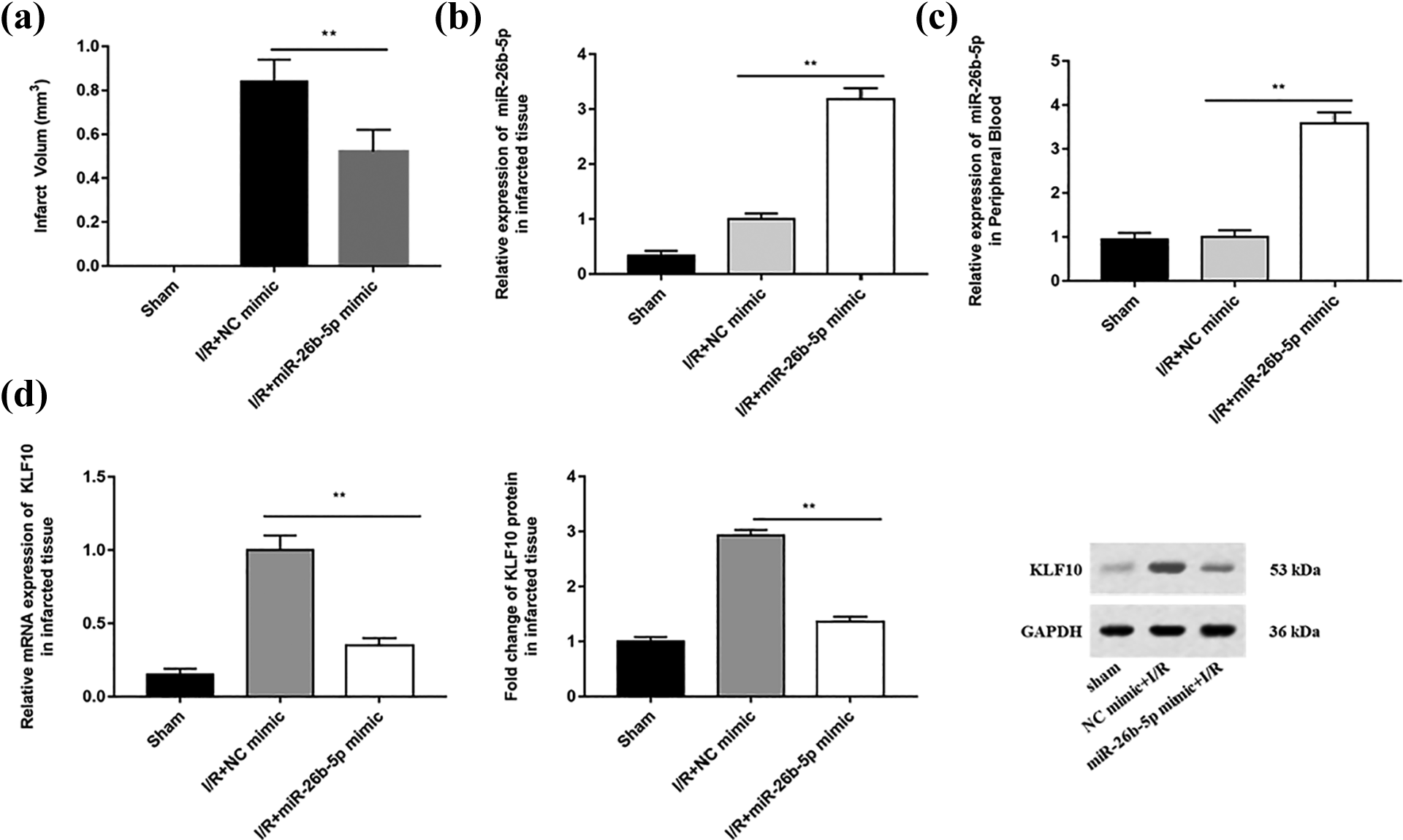
Downregulation of miR-26b-5p and upregulation of KLF10 in MCAO rat model. We divided 30 adult Sprague-Dawley rats into two groups, 10 rats were only used for femoral artery separation (sham), and the remaining 20 rats were used to construct MCAO model (experimental). (a) The cerebral infarction tissue sections of MCAO rats were stained with TTC staining. (b–c) Relative expression of miR-26b-5p in cerebral infarction tissue and peripheral blood of MCAO rats were analyzed by RT-qPCR. (d) The mRNA and protein expression of KLF10 in MCAO rats were analyzed by RT-qPCR assay and Western blotting. β-actin was used as an invariant internal control for calculating protein-fold changes. N = 3, * P < 0.05, ** P < 0.01.

Regulation of miR-26b-5p in MCAO rat model. (a) Cleaved asepase3, Bax, N-myc and PTEN protein levels were detected by Western blot assay. (b) ROS content was detected by DCFH-DA stain. (c) The secretion levels of IL-6 and TNF-α were detected by ELISA. β-actin was used as an internal reference. N = 3, * P < 0.05, ** P < 0.01.
Discussion
Cerebral ischemia-reperfusion injury can cause extensive microvascular dysfunction and changes in tissue barrier function, which is an important pathogenesis of secondary cerebral ischemia injury. 18 At present, there is no very effective treatment for brain IRI. Therefore, elucidating the pathogenesis of brain IRI is essential to discover new and effective treatment modalities.
In recent years, there has been increasing evidence that many miRNAs regulate the survival and apoptosis of neurons in vitro OGD/R and in vivo IRI models. 19 –21 In addition, 118 differentially expressed miRNAs were identified in the infarcted area of mice in the middle cerebral artery occlusion mouse model, of which 12 had an upregulation fold change of more than 2 and 18 had a downregulation fold change of less than 0.5. 22 Overexpression of miR-22 can lead to increased expression of anti-apoptotic gene Bcl-2 and decreased expression of apoptotic gene Bax, so as to inhibit inflammatory response and apoptosis after IRI. 23 New evidence shows that the expression of GAS5 is increased in CI/R rats, while the expression of miR-26b-5p is decreased. Knocking down GAS5 can reverse the apoptosis and inflammation induced by CI/R. Similarly, the overexpression of miR-26b-5p reverses the apoptosis and inflammation induced by CI/R. 24 Other studies have reported that exosomes from human umbilical cord mesenchymal stem cells can upregulate the expression of miR-26b-5p in microglia induced by OGD/R, and inhibit the M1 polarization of microglia by inactivating the TLR pathway by targeting CH25 H, thereby alleviating neurological injury after cerebral I/R. 25 This provided a new idea for us to further explore the role and regulatory mechanism of miR-26b-5p in ischemia reperfusion injury.
Different expression disorders of miRNAs have been found in various pathological processes, involving many biological processes such as cell proliferation, migration, invasion and apoptosis. 26,27 In this study, we found that the expression of miR-26b-5p was significantly downregulated in both in vitro OGD/R and rat CIRI models. In addition, miR-26b-5p simulators overcome OGD/R-induced apoptosis and injury by enhancing cell proliferation, and reduce the secretion levels of ROS, IL-6 and TNF-α, indicating that miR-26b-5p plays an important protective role in CIRI.
MiRNAs usually function through their target genes. Through bioinformatics analysis, we found that the 3’UTR of KLF10 has the binding site of miR-26b-5p, and luciferase reporter gene analysis confirmed the target relationship between KLF10 and miR-26b-5p. Krüppel-like family is a group of zinc-finger transcription factors which play key regulatory roles in cellular growth, development, differentiation and vascularization. 28 –30 A study of transcriptome changes after oxygen-glucose deprivation showed that KLF10 was upregulated in rat aortic smooth muscle cells cultured under ischemic conditions, and transfection of hypoxia-inducible factor-1α siRNA reduced the hypoxia-induced increase in KLF10 mRNA. 31 Although there is not much research on the regulation mechanism of KLF10 in ischemia reperfusion, there are many evidences that Krüppel-like family members play an important biological role in the process of ischemia-reperfusion. For example, studies have shown that KLF2 regulates eNOS uncoupling via Nrf2/HO-1 in endothelial cells under hypoxia and reoxygenation. 32 Another study found that Simvastatin ameliorates total liver ischemia-reperfusion injury via KLF2-mediated mechanism in rats. 33 Similarly, some studies have shown that KLF5 overexpression attempts cardiomyocyte information induced by oxygen glucose innovation / reconstruction through the PPAR γ/PGC-1 α/TNF-α signaling pathway. 34
In this study, we found that transfection of pcDNA-KLF10 significantly upregulated the expressions of N-myc and PTEN, as well as the secretion levels of ROS, IL-6 and TNF-α. This suggests that KLF10 aggravates ischemia-reperfusion injury by upregulating the protein expression of N-myc and PTEN. To some extent, these reports support our existing research results, in addition to investigating the role of miR-26b-5p targeting KLF10 in OGD/R in cell model, we also constructed a rat brain IRI model, and the results showed that miR-26b-5p expression was decreased and KLF10 was highly expressed in the rat cerebral infarction core tissues. However, we only confirmed the role of miR-26b-5p in PC12 cells, which still needs to be verified in other cells. In addition, this study only confirmed one of several miRNAs with abnormal expression in brain IRI, and further validation and in-depth analysis of other miRNAs with altered expression are needed. In the follow-up work, we will validate the protective effect of miR-26b-5p on brain IRI in a variety of brain tissue cells, explore the effectiveness of targeting the target gene of miR-26b-5p, KFL10, in the treatment of brain IRI in rats, and find more miRNAs that play a protective role in brain IRI, providing more effective targets for clinical treatment.
In conclusion, our study demonstrated that miR-26b-5p blocked the N-myc/PTEN axis by inhibiting KLF10 expression, thereby alleviating cerebral ischemia-reperfusion injury in rats, and provided a reference for the development of new targeted therapies.
Footnotes
Author contributions
Jian Wen: investigation, conceptualization, project administration; Ying Xiao: methodology, writing-original draft; Shao-hua Zheng: formal analysis, data curation; Na Duan: interpretation of data; Xin Li: resources. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets used during the present study are available from the corresponding author upon reasonable request.
Ethics approval
All procedures were performed in accordance with Health Guidelines for the Care and Use of Animals and were approved by the Institutional Animal Care and Use Committee of The First Affiliated Hospital of Xi’an Jiaotong University (Approval No. XJ2018-236).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (Grant No. 81801388) and Natural Science Basic Research Plan in Shaanxi Province of China (Program No. 2020JM-388).
