Abstract
Triptolide (TP), the main active compound extracted from medicine—tripterygium wilfordii Hook f. (TWHF). It has anti-tumor and immunomodulatory properties. Our study aimed to investigate the mechanisms of hepatotoxicity treated with TP in vivo and in vitro, as well as their relationship with the NF-κB (p65) signal pathway; and to assess TP-induced hepatotoxicity after CYP2E1 modulation by the known inhibitor, clomethiazole, and the known inducer, pyrazole. Mice were given TP to cause liver injury and IHHA-1 cells were given TP to cause hepatocyte injury. The enzyme activity and hepatotoxicity changed dramatically when the CYP2E1 inhibitor and inducer were added. In comparison to the control group, the enzyme inducer increased the activity of CYP2E1, whereas the enzyme inhibitor had the opposite effect. Our findings suggest that TP is an inducer of CYP2E1 via a time-dependent activation mechanism. In addition, TP can promote oxidative stress, inflammatory and involving the NF-κB (p65) signal pathway. Therefore, we used triptolide to stimulate C57 mice and IHHA-1 cells to determine whether TP can promote oxidative stress and inflammation by activating CYP2E1 in response to exacerbated liver damage and participate in NF-κB (p65) signaling pathway.

Introduction
The roots are the traditional Chinese herb with anti-inflammatory and immunosuppression actions1,2 and are a herb of trypterygum wilfordii crook f. (TWHF). As a widely used Chinese medicine in clinical practice, there has been ample evidence indicated that TP has caused the clinic to be apparently hepatotoxic.3,4 As all we know, TP is one of the main ingredients of TWHF. It has similar effects to TWHF, and it is also the main toxic component. Published studies have shown that triptolide can induce hepatotoxicity.5,6 CYP2E1 has a significant role in hepatotoxicity. In vivo and in vitro experiments found that CYP2E1 can mediate oxidative stress and take part in liver disease pathogenesis such as hepatotoxicity.7,8 At the same time, CYP2E1 expression level may increase both trypterygium glycosides and TP. 9 Therefore, it is important to understand the toxicity of the TP mechanisms.
Oxidative stress is known to be a strong correlation to hepatotoxicity-related disease incidents and to the important mechanism of exogenous compounds for hepatotoxicity.10,11 In addition, accumulating studies have shown that the progression of hepatotoxicity can be beneficial for oxidative stress.12,13 The TP toxicity was considered to be the primary cause of TP-induced death due to a high incidence of the hepatotoxicity. Many researches suggest that hepatotoxicity plays an important role in oxidative stress and inflammation.14,15 The mediated metabolism of CYP2E1 in its substrates results in a stimulated oxidant stress and toxicity, resulting in reactive oxygen species and reactive metabolites (ROS). Previous investigations have shown that the occurrence of hepatotoxicity is etiologically influenced by CYP2E1. 16 Literature has indicated the abundance of ROS caused by the excitement of CYP2E1.17,18,19 Moreover, the release of pro-inflammatory cytokines (TNF-α, IL-6) is facilitated by oxidative stress. 20 Taken together, oxidative stress, CYP2E1, and NF-κB always interact with each other, ultimately leading to the development of hepatotoxicity. 21 However, the precise mechanism for the occurrence and development of hepatotoxicity among TP and CYP2E1, especially in oxidative stress and inflammatory hepatotoxicity responses, remains clearly defined. 22
Cytochrome P450 (CYP) is a superfamily of enzymes which can be linked by exogenous compounds and metabolism of endogenous, including steroids, a number of medicines, fatty acids, and a number of chemicals P450 cytochrome is a superfamily of enzymes which can be linked by exogenous compounds and endogenous metabolism, including steroids, a number of medicines, fatty acids, and a number of smaller chemical molecules. 23 Cytochrome P450 CYP2E1, which has a large influence on oxidative stress, metabolic transformation, and other reactions, is one of the major metabolistic enzymes in the liver. In addition, the metabolic excitation of toxins is mainly mediated by CYP2E1. CYP2E1 is a major factor in oxidative stress and toxicity. Clinicians should therefore closely monitor and adapt the medication to avoid toxic and adverse events in combination to monitor the patients’ liver-related indicators. NF-κB (p65) is considered a therapeutic target for overcoming oxidative stress-induced injury in a variety of diseases. 24 Interestingly, the NF-κB (p65) signaling pathway can be involved in hepatotoxic diseases by regulating oxidative stress-induced damage. Therefore, learning more about the regulatory mechanism of NF-κB (p65) signaling in TP induced hepatotoxicity will contribute to the treatment of liver damage.
Materials and methods
Reagents and materials
Triptolide (TP) (purity >98%) was obtained from Purcell biology (Chengdu, China). Clomethiazole (CMZ) (purity >98%) and pyrazole (PZ) (purity>98%) were purchased from Aladdin (Shanghai, China). 3-(4, 5-dimethyl-2-thiazolyl)-2, 5-diphenylterazolium bromide (MTT) was obtained from Beyotine Biotechnology Research Institute (Shanghai, China). Anti-CYP2E1 and anti-β-actin were purchased from Proteintech (Shanghai, China). Anti-P65 was obtained from Wanleibio (Shengyang, China). Anti-β-actin antibody and secondary antibodies for goat were obtained from ZSGBBIO (Beijing, China). Primary Antibody Dilution Buffer, RIPA Lysis, Penicillin-Streptomycin Solution and Extraction Buffer and PMSF were obtained from Beyotime (Hangzhou, China). Transcriptor First Strand cDNA Synthesis Kit, PrimeSTAR, and TB Green® Premix Ex Taq™ II were obtained from TaKaRa (Shiga, Japan). LipofectamineTM2000 and Trizol were purchased from Invitrogen (Carlsbad, CA, USA). CYP2E1, TNF-α, IL-6, (p-P65), and β-actin primers were obtained from Sangon Biotech (Shanghai, China). ROS assay kit and BCA protein assay kit were obtained from Beyotime (Hangzhou, China). The alanine transaminase (ALT), aspartate aminotransferase (AST), malondialdehyde (MDA), superoxide dismutase (SOD), Catalase (CAT) and glutathione (GSH) determination kits were obtained from Jiancheng Bioengineering Institute of Nanjing (Nanjing, China).
Animals and experimental design
Male C57 mice (about 18–22 g) aged 6–8 weeks were applied to a model of hepatogenic lesion. Mice have been taken from Anhui Medical University’s Animal Center. The experiment has been approved by the Ethics Committee in separately ventilated cages for adaptation to the laboratory environment (the animal ethics approval number: LLSC20200720). All procedures performed in studies on animals were in compliance with ethical standards of the institution in which the studies were conducted and with the approved legal acts of the Russian Federation and international organizations. The mice had been grouped by 4 groups, 10 in each group, after animals had been adapted for a week for the facilities. a. The mice were fed physiological saline by Gavage during 2 weeks of control (control group). b. Group TP (triptolide), 2 weeks gavage was used to feed the mice TP (500 μg/kg/day). The mice have been feeding TP (500 mg/kg/day) for 1 week by gavage before the week (75 mg/kg/day) in the CMZ (triptolide + clomethiazole) group. d. Group PZ (triptolide + pyazole), 1 week TP was supplied to the mice (500 μg/kg/day) by gavage prior to PZ (120 mg/kg/day). 24 h following the last gavage, the mice were anesthetized.
Cell culture and transfection
Human hepatocyte IHHA-1 cells have been obtained and kept in DMEM supported by 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin from Wuhan Procell Life Science&Technology Co., Ltd. In cell culture incubator (37°C) with 5% carbon dioxide. The TP (20 nM) stimulated IHHA-1 cells during 12 h. DMSO has dissolved the TP, CMZ (80 μM) and PZ (60 μM). Transfection was conducted using Lipofectamine® 2000. The DMSO content is not greater than 0.1% (V/V).
MTT Assay
IHHA-1 cells were cultivated at a density of 0.5 × 104/mL overnight on a 96-well plate. Then, cell pretreatment was performed with TP (0, 15, 20, 25, 30, 35, 40, 45, 50, and 55) nM, CMZ (0, 60, 80, 90, 100, 110, 120, 130, and 140) μM, PZ (0, 40, 60, 70, 80, 90, 100, 110, and 120) μM, and with 12 h PBS (control). All cells in all wells were co-incubated with TP for 12 h, excluding the control group. Following processing, every well had 20 μL MTT (5 mg/mL) and 4 h had to be cultivated at 37°C; 150 μL DMSO was removed and the formazan crystals were dissolved. The optical density (OD) with an Enzyme standard instrument (BioTek Company, USA) was recorded at a wavelength of 490 nm.
Flow cytometry
The detection kit for oxidative stress ROS of IHHA-1 was used in accordance with the instructions of the manufacturer in the FITC Annexin V ROS. The cells were placed on 6-well plating at a density of 2 x 105 mL and cultivated at 37°C for 12 h with CMZ and PZ. Afterward, the cells were collected and cultivated in a place outside of light for 20 min in 2′, 7′-dichlorofluoresce diacetate (DCFH-DA) and then measured by cytometry of the flow (Beckman Company, USA).
Plasmid construction
The expression plasmid for CYP2E1 (PG-CMV-CYP2E1) empty PG-CMV-control was synthesized by GenaPharma Corporation (Shanghai, China). Then, LipofectamineTM 2000 was used to transfect PG-CMV-CYP2E1 and PG-CMV as required.
RNA interference analysis
CYP2E1 small interfering RNA (CYP2E1 siRNA) and negative control (NC) were designed and synthesized by GenaPharma Corporation (Shanghai, China). Prior to the operation, IHHA-1 cells were incubated on 6-well plates for 24 h on a medium of antibiotic-free cultivation. Then, LipofectamineTM 2000 (Invitrogen, USA) was transfected with CYP2E1 siRNAs and Negative Control siRNA (NC) as needed. The sequences are as following: CYP2E1 siRNA: F: 5′-GCAACGUCAUAGCCGACAUTT-3′, R: 5′-AUGUCGGCUAUGACGUUGCTT-3′; NC siRNA: F:5′-UUCUCC GAAC GU GUC ACG UTT-3′; R: 5′-ACGUGA CACGUU CGG AGA ATT-3′.
Histopathological examinations
In short, the liver was soaked in the trachea with 4% paraformaldehyde phosphate buffer and incorporated into paraffin. After fixation, hematoxyline was stained (4 μm) for approximately 10 min. The sections of the tissue were duplicated water washed in and eosin-dyed for 5 min. Light microscopy (OLYMPUS Company, Japan) after dehydration was examined in tissue sections.
Serum biochemical assay
The blood examples were collected from mice and the activity ranges of alanine aminotransferase (ALT), aspartate aminotransferase (AST), malondialdehyde (MDA), superoxide dismutase (SOD), catalase (CAT), and glutathione (GSH) enzyme activity were detected with the kit.
Western blot
All samples were lysed in a protein extraction kit. BCA Protein Assay Kits (Biyuntian, China) were all extract and protein concentration. The total protein of the samples was electrophoresed by SDS-PAGE and blotted into PVDF membranes. After sealing with 5% fat free milk for 2 h, PVDF membranes and primary antibodies were incubated at 4°C overnight. The mixture was diluted 1:6000 with TBS/Tween-20 (TBST) and incubated for 1 h. After washing with TBST, finally, a band was formed using an ECL-chemiluminescence kit (Bole Company, USA). Concentration of primary antibody: CYP2E1, TNF-α, IL-6, (p-P65), (P65), and β-actin were used 1:500, 1:500, 1:500, 1:1000, 1:1000, and 1:1000, respectively. The blots were detected by chemiluminescence assay.
Quantitative real-time PCR
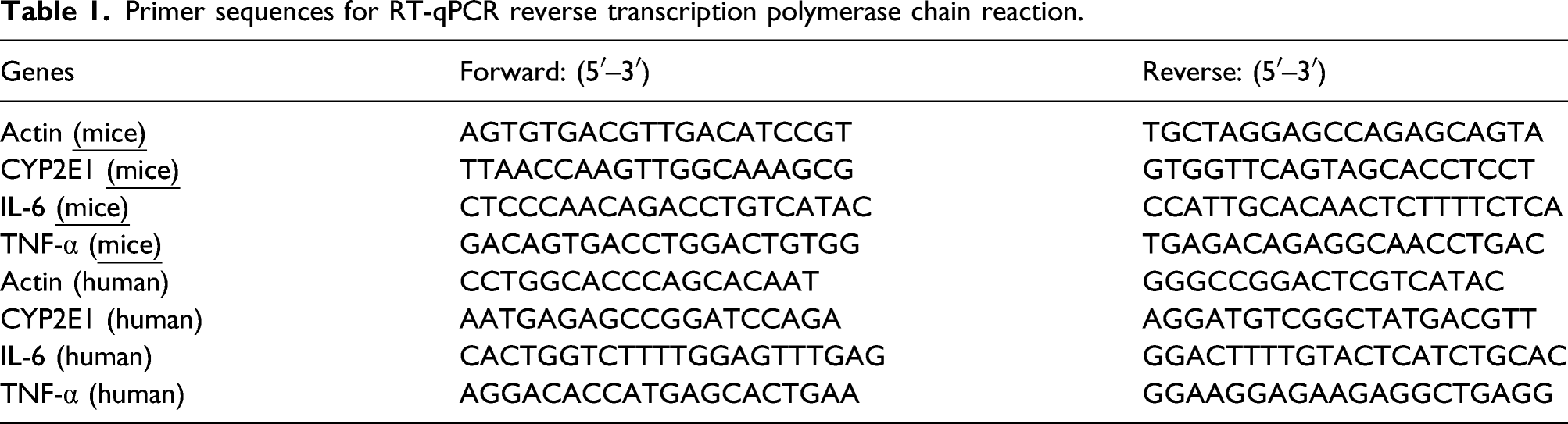
Primer sequences for RT-qPCR reverse transcription polymerase chain reaction.
Statistical analysis
Results were expressed as the mean ± SD analyzed by SPSS 16.0. One-way ANOVA was used to measure significant differences. In all cases, the results were from at least three experiments, meanwhile, when *p < 0.05, **p < 0.01, ***p < 0.001 and **p < 0.01, #p < 0.05, ##p < 0.01, ###p < 0.001. The data is considered statistically significant.
Results
TP induced liver injury in C57 mice
The liver tissue of C57 mice has been used for study to determine if TP causes liver injury to C57 mice. First of all, an ALT and AST test kit assessed the extent of the hepatic injury of the C57 mice treated with TP. In TP’s serum of C57, the expression of the ALT and AST was significantly more important than in the control group in TP’s ALT and AST groups (Figure 1(a) and (b)). In addition, hematoxylin-eosin (H&E) staining was assessed in the extent of the hepatic injury in TP-treated mice C57. The H&E staining data indicated that liver damage caused significant degeneration of hepatocytes, irregularly arrangement, and infiltration of inflammatory cells in the hepatic lobules and hillier areas in comparison to control tissues in vivo (Figure 1(c)). (a) The serum level of ALT content was measured. (b) The serum level of AST content was measured. (c) Vehicle and TP-treatment mice liver tissue sections in liver tissues were detected by hematoxylin eosin (H&E) staining. Representative views of each group are presented (×200) (scale bar = 50 μm). The results (mean ± SD) were evaluated relative expression against control values. *p <0.05, **p<0.01, ***p<0.001 compared to the control group, #p < 0.05, ##p < 0.01, ###p < 0.001 compared to the TP group.
Effects of TP on oxidative stress in C57 mice
We have measured the ROS, MDA, GSH, GSH, and CAT level for C57 mice in the liver in order to further investigate whether TP induced oxidative stress. The results demonstrated that in TP-fed C57 mice, ROS levels in comparison with the control group were significantly increased (Figure 2(a) and (b)). In contrast to TP-fed C57 mice, MDA levels were significantly increased in comparison to the control group, with significantly lower SOD, CAT, and GSH levels (Figure 2(c–f)). Note: MDA levels have fallen significantly in the CMZ group, compared to the TP group, while GSH, SOD, and CAT levels have significantly increased. The levels of MDA were considerably increased in PZ group compared with the TP group while those of GSH, SOD, and CAT were substantially reduced (Figure 2(c–f)). (a) and (b) ROS in the liver of C57 mice was measured by a slide scanner. (c) and (d) Effects of changes in the levels of MDA and SOD in C57 mice. (e) and (f) Effects of changes in the levels of CAT and GSH in C57 mice. The results were expressed as the mean ± SD of three different experiments. *p < 0.05, **p < 0.01, ***p < 0.001 compared to the control group, #p < 0.05, ##p < 0.01, ###p < 0.001 compared to the TP group.
CYP2E1 is upregulated induced by TP in C57 mice
We tested the expression level of the CYP2E1 control and TP treatment to determine if CYP2E1 has participated in TP-induced liver injury. Western blotting (Figure 3(a) and (b)) showed that CYP2E1 expression level has been updated after treatment with TP in comparison to the control group and has further been confirmed in mRNA levels by real-time q-PCR (Figure 3(c)). The results showed that TP may upgrade the CYP2E1 expression. (a) and (b) The expression of CYP2E1 in mice was detected by western blot analysis. (c) The expression of CYP2E1 in mice was detected by real-time q-PCR analyses. (d) and (e) TNF-α, IL-6, and P-NF-κB (p-P65) expression levels were by western blot analysis in mice. (f) IL-6 and TNF-α expression levels were by RT-qPCR in mice. (g) Effect of different concentrations of TP on the viability of IHHA-1 cells by MTT assay. (h) Effect of different concentrations of CMZ and PZ on the viability of IHHA-1 cells by MTT assay. (i) Effect of different concentrations of PZ on the viability of IHHA-1 cells by MTT assay. (j) and (k) TP time-dependent expression of CYP2E1 in IHHA-1 cells. By using western blotting, a significant upregulation of CYP2E1 protein was shown within 12 h after stimulation with TP. (L and M) TP dose-dependent expression of CYP2E1 in IHHA-1 cells. The results were expressed as the mean ± SD of three different experiments. *p < 0.05, **p < 0.01, ***p < 0.001 compared to the control group, #p < 0.05, ##p < 0.01, ###p < 0.001 compared to the TP group.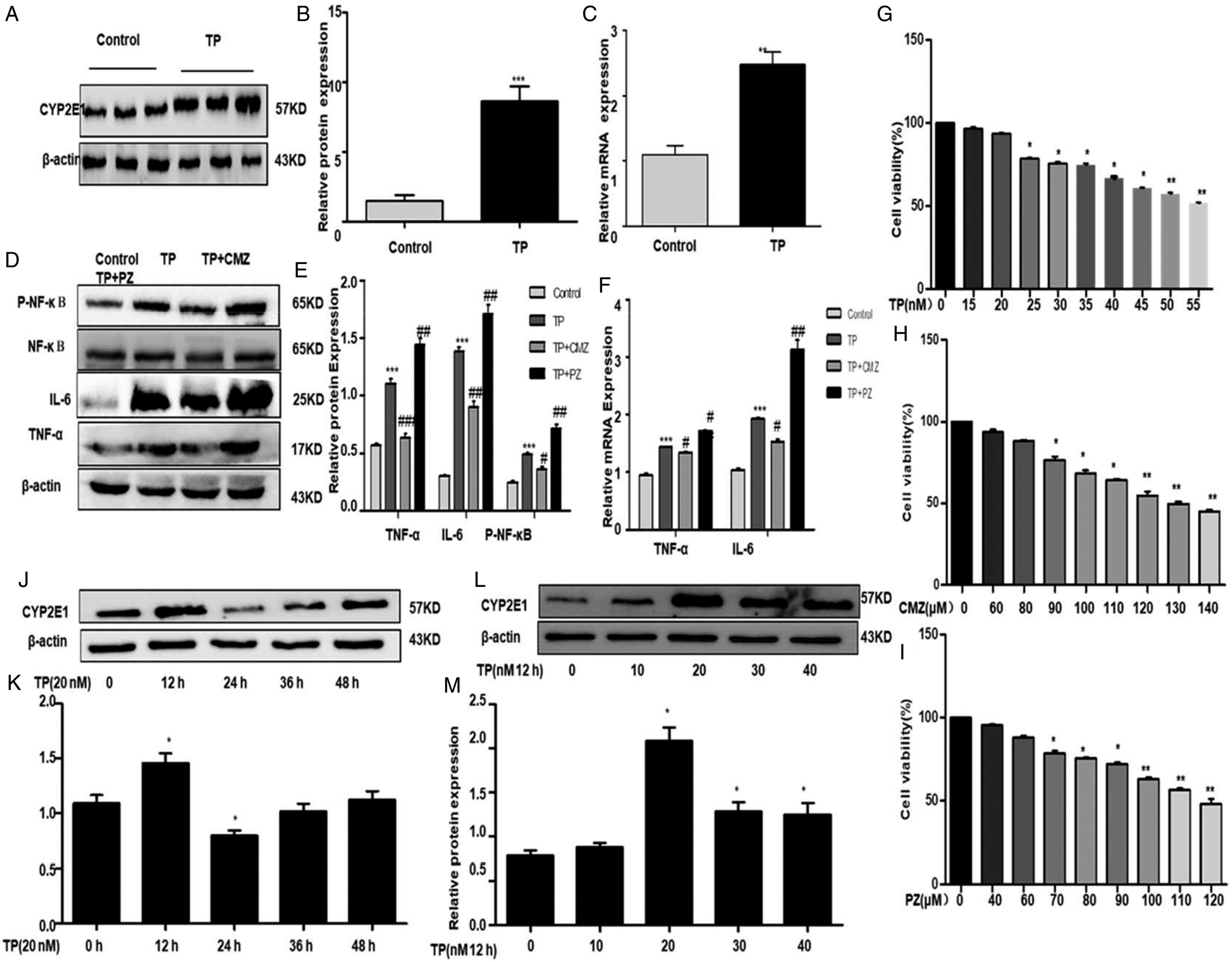
TP increased IL-6, TNF-α level by upregulating CYP2E1 in mice
The CYP2E1 was higher in C57 mice in the TP Group than the control group. CYP2E1, including pro-inflammatory cytokines (IL-6 and TNF-α), were fed to TP-treated C57 mice, respectively, to clarification on the involvement of CYP2E1 in TP-mediated inflammatory secretion. The IL-6, TNF-α, and (p-P65) expression grades in C57 mice were detected by western blot and real-time q-PCR. TP greatly promoted the IL-6, TNF-α, and (p-P65) expression levels (Figure 3(d–f)). Note that the CMZ group had anti-inflammatory and hepato-protective effects in relation to the TP group; notably, in the TP group, the CMZ group had hepato-protective and anti-inflammatory effects; and the PZ group had serious hepatotoxicity and inflammatory effects in the TP group (Figure 3(d–f)). We concluded that CYP2E1 activation may exacerbate injuries to the liver and inflammation.
TP induced IHHA-1cell hepatotoxicity and inflammation
Western blotting to detect hepatotoxicity was performed to further study the impact of CYP2E1 on TP-induced IHHA-1 cells. A figure showing the survival rates for medications TP, CMZ, and PZ in different IHHA-1 cells (Figure 3(g–i)), the survival rates are shown in the figure and an optimal concentration of the medicine in the cells can be selected. The concentration of TP in medicinal products in this research therefore amounts to 20 nM, CMZ is 80 μM, and PZ is 60 μM. When TP activated IHHA-1 in various times and concentrations, we checked the expression of CYP2E1. First, the treatment of IHHA-1 cells for 12 h with various TP levels (Figure 3(j–m)) was shown to increase dose-dependent cell viability. Following 20 nM of TP induction, protein levels were higher. The protein concentration of CYP2E1 reached the peak 12 h after TP induction. The western blotting results showed that the expression level of CYP2E1 was significantly higher than that of the control group, after 20 nM TP stimulation for 12 h. IHHA-1 cells were thus encouraged to detect CYP2E1 expression with 20 nM TP for 12 h.
TP induced IHHA-1 cell hepatotoxicity
The extent of hepatoxicity in TP-cultivated IHHA-1 cell was assessed by ALT and AST test kits to determine whether TP-induces hepatotoxicity in IHHA-1 cell. The results show that TP had a major impact on ALT and AST in IHHA-1 cell and that the ALT and AST expression levels were significantly higher in the TP group than those of the control group (Figure 4(a) and (b)). (a) Effects of TP on the activity of ALT in IHHA-1 cells. (b) Effects of TP on the activity of AST in IHHA-1 cells. (c) and (d) ROS of IHHA-1 cells were measured by flow cytometry analysis. (e) and (f) Effects of changes in the levels of MDA and SOD in IHHA-1 cells. (g) and (h) Effects of changes in the levels of CAT and GSH in IHHA-1 cells. (Date represents the mean ± standard deviation (SD). *p < 0.05, **p < 0.01, ***p < 0.001 vs control group; #p < 0.05, ##p < 0.01, ###p < 0.001 vs TP group).
Effects of TP on oxidative stress in IHHA-1cell
To further investigate whether TP caused oxidative stress, we measured the levels of ROS, MDA, GSH, GSH, and CAT in IHHA-1 cell to detect the level of oxidative stress. The results showed that in TP-cropped IHHA-1, the ROS levels were significantly higher than in the control group (Figure 4(c) and (d)). The MDA levels in TP cultured IHHA-1 were considerably higher compared to the control group, while the SOD, GSH, and CAT levels were significantly lower (Figure 4(e–h)). Of note, MDA levels have fallen significantly in the CMZ group, compared to the TP group, while GSH, SOD, and CAT levels have significantly increased. MDA was significantly higher than the TP group in the PZ group while GSH, SOD, and CAT levels were reduced significantly (Figure 4(e–h)).
CYP2E1 is upregulated induced by TP in IHHA-1 cells
We tested the CYP2E1 control and TP expression levels for determining whether CYP2E1 was involved in hepatotoxicity due to TP. Western blotting (Figure 5(a) and (b)) and q-PCR in real-time (Figure 5(c)) demonstrated that the expression level of CYP2E1 was upregulated after TP-culture compared with the control group. The results showed that TP may upgrade the CYP2E1 expression. (a) and (b) The expression of CYP2E1 in IHHA-1 cells was detected by western blot analysis. (c) The expression of CYP2E1 in IHHA-1 cells was detected by real-time q-PCR analyses. (d) and (e) TNF-α, IL-6 and (p-P65) expression levels were by western blot analysis in IHHA-1 cells. (f) IL-6 and TNF-α expression levels were by RT-qPCR in IHHA-1 cells. The results were expressed as the mean ± SD of three different experiments. *p < 0.05, **p < 0.01, ***p < 0.001 compared to the control group, #p < 0.05, ##p < 0.01, ###p < 0.001 compared to the TP group.
TP increased IL-6, TNF-α expression by upregulating CYP2E1 in IHHA-1 cells
The expression level of CYP2E1 in TP group in IHHA-1 cells was higher compared with the control group. CMZ and PZ were respectively cultivated with TP-induced IHHA-1 cells in order to clarify whether CYP 2E1 was associated with the inflammatory secretion by TP, including pro-inflammatory cytokines (IL-6 and TNF-α). The IL-6, TNF-α, and (p-P65) expression in IHHA-1 cell were measured using western blotting and q-PCR in real-time. The results demonstrate that TP greatly promoted IL-6, TNF-α, and NF-κB (p65) path levels (Figure 5(d–f)). It should be noted that in comparison with the TP group, hepatoprotective and anti-inflammatory effects were demonstrated by the CMZ; in comparison to the TP group, there was serious hepatotoxicity and inflammatory effects. We concluded that CYP2E1 activation may worsen inflammation and hepatotoxicity.
CYP2E1 siRNA downregulated expression of CYP2E1
The cultured IHHA-1 cells were transfected with CYP2E1 siRNA and NC siRNA by Lip 2000, respectively. RT-qPCR and western blot detected the CYP2E1 expression level after successful transfection. The expression of CYP2E1 at mRNA and protein levels was inhibited by CYP2E1 siRNA compared with the group of cells transfected by NC siRNA (Figure 6(a) and (b)). (a) and (b) The effects of different CYP2E1 siRNA on the expression of CYP2E1 in IHHA-1 cells. (c) and (d): Expression of CYP2E1, IL-6, TNF-α, and (p-P65) proteins. (e): The mRNA level of CYP2E1, IL-6 and TNF-α. *p < 0.05, **p < 0.01, ***p < 0.001 vs control group; #p < 0.05, ##p < 0.01, ###p < 0.001 vs TP group.
Knockdown of CYP2E1 suppressed inflammatory through the NF-κB (p65) pathways
Western blot and RT-qPCR have been detected for the effect of CYP2E1 silences on NF-κB (p65) mediated, inflammatory cytokines. It has been shown, that CYP2E1 and CYP2E1 silencing compared to the control group significantly reduces mRNA and protein expression values of IL-6, TNF-α and (p-P65) (Figure 6(c–e)). We concluded that CYP2E1 downregulation can reduce the inflammatory response caused by TP.
CYP2E1 plasmid upregulated expression of CYP2E1
The cultured IHHA-1 cells were transfected with PG-CMV-CYP2E1 and PG-CMV by Lip 2000, respectively. After successful transfection, the expression level of CYP2E1 was detected by western blot and RT-qPCR. This figure shows that PG-CMV-CYP2E1 improved protein and mRNA expression relative to the PG-CMV group of transfected cells CYP2E1 (Figure 7(a) and (b)). (a) and (b) The effects of different CYP2E1 plasmid on the expression of CYP2E1 in IHHA-1 cells. (c) and (d): Expression of CYP2E1, IL-6, TNF-α, and (p-P65) proteins. (e): The mRNA level of CYP2E1, IL-6, and TNF-α. *p < 0.05, **p < 0.01, ***p < 0.001 vs control group; #p < 0.05, ##p < 0.01, ###p < 0.001 vs TP group.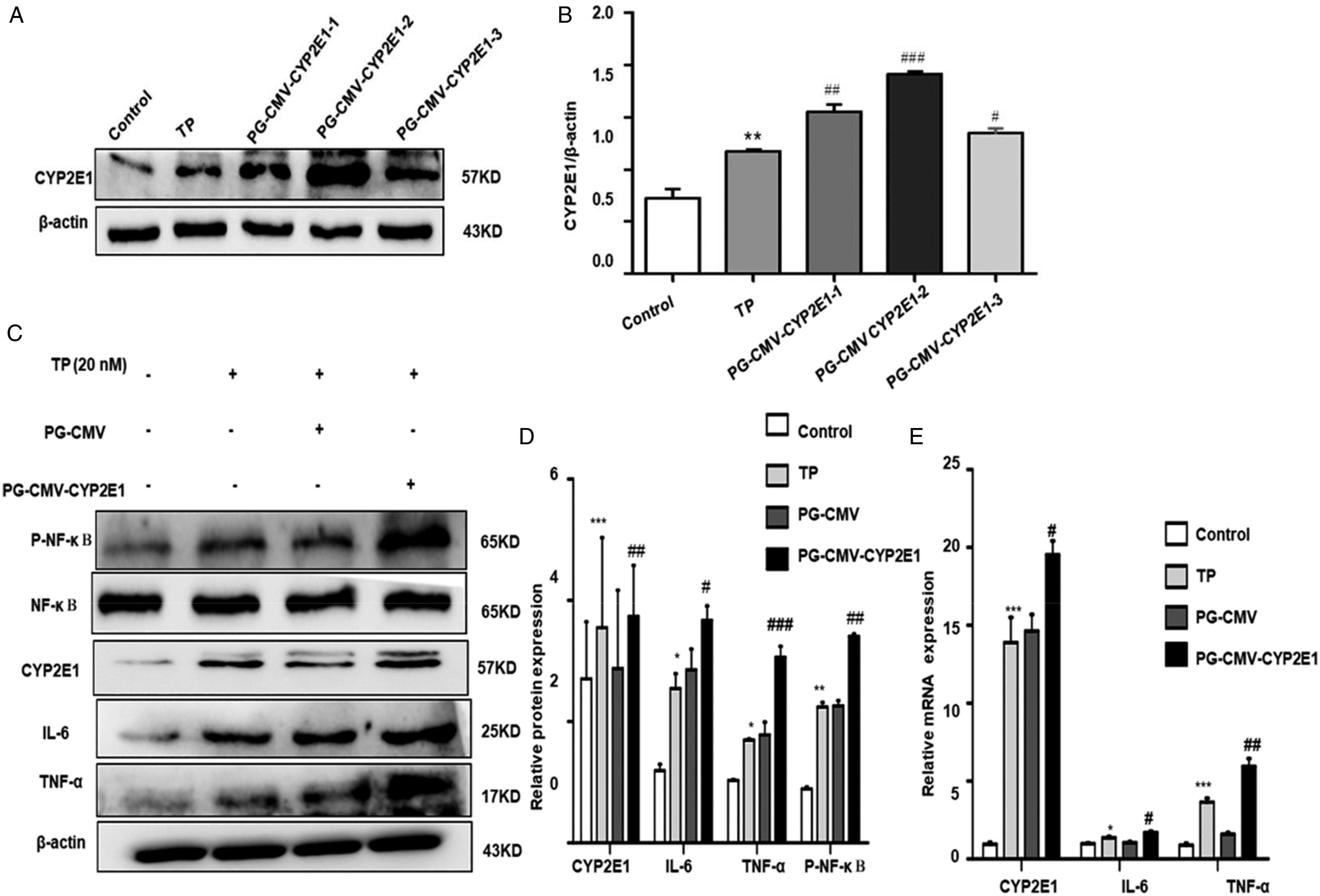
The activation of CYP2E1 inhibits inflammation through the NF-κB (p65) pathway
Western blot and RT-qPCR have been detected for the effect of the activation CYP2E1, IL-6, TNF-α, and (p-P65) from NF-κB (p65)–mediated inflammatory cytokines, mRNA levels, and protein expression. Results indicated that CYP2E1 and CYP2E1 have been significantly improved compared to the control group by activating mRNA and protein expression levels of IL-6, TNF-α, and (p-P65) (Figure 7(c–e)). We concluded that CYP2E1 upregulation can increase inflammatory response due to TP.
Discussion
At present, numerous Chinese herbs have attracted a lot of attention from researchers worldwide, which are identified to be a promising therapeutic method for hepatotoxicity and inflammatory sickness.24–26 Among them, triptolide is widely known to possess anti-inflammatory, anticancer, autoimmune, antitumor, and so on. As the major active ingredient of TWHF, TP has been proven to have a good therapeutic effect on rheumatoid arthritis and systemic lupus erythematosus.27,28 Recently, TP-induced hepatotoxicity has caused increasing safety concerns in the clinic. 29 Considerable evidence has indicated that TP induces CYP2E1 leading to hepatotoxicity. CYP2E1 is a momentous hepatic microsomal oxidase related to fatty acid oxidation and the formation of ROS. 30 CYP2E1 is a direct source of ROS and also reportedly has a greater ability to generate ROS compared to CYP3A4. 31 Excessive levels of ROS are derived from the imbalance between ROS production and the antioxidant system.32,33 Moreover, Excessive ROS can affect the production of lipid peroxides thus exacerbating oxidative stress and mitochondrial damage. To the best of our knowledge, oxidative stress can produce abundant ROS, which is deemed to be a momentous cause of hepatotoxicity. 34 Therefore, inhibiting oxidative stress is one of the crucial tactics to reduce liver damage. In this study, TP-induced CYP2E1 caused hepatotoxicity, which is deeded to be closely connected with its pathogenesis. Given these observations, the association between TP and hepatotoxicity is unclear and remains to be further studied, particularly in CYP2E1 activation.
In this research, we come to know that the level of CYP2E1 was promoted sharply in C57 mice and IHHA-1 cells stimulated by TP. Additionally, it was observed that mRNA levels were also significantly increased in TP-treated C57 mice and IHHA-1 cells.
There is numerous evidence that continued, and serious inflammation can create and sharpen hepatotoxicity.35,36 Of note, IL-6 and TNF-α are pro-inflammatory markers of the body.37,38 TP plays its role by effectively activating the expression of CYP2E1, IL-6, and TNF-α. Our experiments also showed that TP-induced elevated the level of IL-6 and TNF-α in mice and IHHA-1 cells. Studies have shown that CYP2E1 may be closely connected with inflammation-connected sickness. According to our studies, overexpression of CYP2E1 increased the expression of IL-6 and TNF-α in TP stimulated mice, both at the protein and mRNA levels. In contrast, CYP2E1 siRNA was found to decrease the protein and mRNA levels of IL-6 and TNF-α. To sum up, we can conclude that CYP2E1 has a significant regulatory effect on the cytokine level in TP -induced and can positively regulate the level of proinflammatory cytokines.
NF-κB (p65) signaling pathway is reacted to oxidative stress, and the NF-κB (p65) signaling pathway presents abnormalities in the process of hepatotoxicity in many sicknesses, including the liver. Although the function of CYP2E1 has been gradually understood, many sides of the effect still unknown, moreover, the relationship between CYP2E1 and NF-κB (p65) pathway remains unclear and further studies are needed, especially in the liver. Our results show that TP could promote the expression of (p-P65); the expression of (p-P65) was greatly increased in the PZ group. These conclusions demonstrated that CYP2E1 played a critical part in increasing inflammatory cytokines by negatively regulating the NF-κB (p65) signaling pathway.
In summary, our study demonstrates that after TP stimulation, CYP2E1 expression in C57 mice and IHHA-1 cells increased and induced oxidative stress and inflammatory response, which were closely related to NF-κB (p65) signaling pathway. In future studies, we are going to concentrate on knockout mice to detect the miRNA expression on TP and study its impact on inflammatory injury and hepatotoxicity and its further association with the NF-κB (p65) signaling pathway. Understanding the mechanisms of CYP2E1 and its relationship with the NF-κB (p65) signaling pathway is crucial and meaningful work; it provides a new way of thinking and effective means for the treatment of hepatotoxicity diseases.
Footnotes
Author contributions
The study was designed by Hai-yan Jiang; data was collected and analyzed by Yan-ni Bao and Feng-mei Lin; data interpretation and manuscript preparation was undertaken by Yong Jin. All authors approved the final version of the paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by Anhui Province Natural Science Foundation (No.1808085MH235) and Scientific Research Foundation of Institute of Anhui Province Transformational Medicine (No.2017zhyx32).
Abbreviations
TWHF, tripterygium wilfordii Hook F; TP, triptolide; CYP2E1, cytochrome P450 2E1; CMZ, clomethiazole; PZ, pyrazole; TNF-α, tumor necrosis factor-α; IL-6, interleukin- 6; NF-κB (p65), nuclear factor-κB; IHHA-1, human normal liver cell; ROS, reactive oxygen species; DMSO, dimethylsulfoxide; MTT, 3-45-dimethylthiazol-2-yl)-2,5 diphenyltetrazolium bromide; ALT, alanine aminotransferase; AST, aspartate aminotransferase; GSH, glutathione; MDA, malondialdehyde; SOD, superoxide dismutase; CAT, catalase.
