Abstract
Introduction
Nephrotoxicity and hepatotoxicity are substantial side effects triggered in individuals injected with 5-fluorouracil (5-FU), an anticancer drug. This study aimed to investigate the impact of the natural antioxidant and anti-inflammatory phenolic compound; protocatechuic acid (PCA) on 5-FU-provoked renal and hepatic injury in rats.
Methods
Rats were allocated to 4 groups: control, 5-FU, 5-FU + PCA (50 mg/kg), and 5-FU + PCA (100 mg/kg). Rats were intraperitoneally injected 5-FU (75 mg/kg; once a week for 21 days. Protocatechuic acid (50 and 100 mg/kg/day; orally) was administered for 3 weeks.
Results
Rats co-treated with PCA had lower serum kidney and liver function markers than those receiving 5-FU alone. Furthermore, co-treatment with PCA successfully modulated kidney and liver contents of TNF-α, NF-κB p65, active caspase-1, IL-1β, p-p38 MAPK, SOD, GSH, Nrf-2, HO-1 and MDA. Moreover, PCA improved histopathological alterations of both kidney and liver tissues.
Conclusion
PCA exerts its hepatoprotective and nephroprotective effects against 5-FU-triggered toxicity through modulation of oxidative stress and inflammatory pathways, particularly via Nrf-2 activation and NF-κB inhibition.
Introduction
5-Fluorouracil (5-FU) is a chemotherapeutic agent belonging to the pyrimidine analogue anti-metabolites group. It is used for the management of various types of cancer including colorectal, breast, esophageal, and head and neck cancers. 5-FU reported toxicities on many body organs sometimes restrict its use.1–3
Protocatechuic acid (PCA) is a phenolic compound (3,4-dihydroxybenzoic acid) found in many plants such as Hibiscus sabdariffa (roselle), Olea europaea (olives), Eucommia ulmoides (du-zhong) and Vitis vinifera (white wine grapes). The content of PCA varies significantly according to the plants. 4 PCA is a principal metabolite of anthocyanins which has been found useful for treatment and/or prophylaxis for a large number of various disorders associated with oxidative stress damage. 5 It possesses various pharmacological activities and these effects are due to their antioxidant activities, along with other possible mechanisms, such as anti-inflammatory, and antiapoptotic properties.6–9
In previous studies, PCA showed nephroprotective effects against lipopolysaccharide (LPS) induced acute kidney injury in mice via suppressing TLR-4/NF-κB and MAPK/COX-2 pathways. 10 It also was able to protect hepatocytes against hydrogen peroxide-triggered oxidative stress and apoptosis via enhancing the antioxidant enzymes (catalase and SOD) and HO-1/Nrf2 pathway. 11 Other various properties were also reported including antihyperglycemic, antimicrobial, anticancer, antiviral, antiaging, antifibrotic, antiatherosclerotic, antihypertensive, anti-Inflammatory, analgesic, and antiseptic effects. 7
This study aimed to investigate the possible hepatorenal-protective effect of PCA against 5-FU-triggered injury in rats and explicate its probable mechanisms.
Materials and methods
Experimental animals
Twenty-four male rats (average weight 190–210 g) were kept in cages for 2 weeks to acclimate to laboratory conditions. They were kept in an animal house at 25°C with 50% humidity and 12-h light/12-h dark cycle. Rats had free access to plenty of standard food and water. The experimental procedures applied in this work were approved by Mansoura University Animal Care and Use Committee (MU-ACUC) (PHARM. PhD.23.01.7) and compiled animal care guidelines and use of the National Institute of Health Guide and Laboratory Animals Use.
Drugs and chemicals
5-FU and PCA were obtained from Al-Hikma Co. (Egypt) and Sigma Aldrich Chemical Co.
Experimental design
Experimental protocol.

On day 22, rats were anesthetized by I.P injection of thiopental sodium at a dose of 50 mg/kg. 15 Blood samples were withdrawn and then centrifuged to obtain serum which kept at −80°C until the assays were performed. Kidney and liver were excised, parts of them were homogenized for ELISA and western blot analysis while the other parts were stored in 10% neutral buffered formalin for histopathological and immunohistochemical studies.
Assessment of kidney and liver weight indices
The kidney and liver weight indices were calculated by dividing the kidney and liver weight, respectively, by the total body weight of the rat.
Kidney function biomarkers
Urea and creatinine levels in serum were measured using commercial kits (SPINREACT Co., Spain).
Liver function biomarkers
Alkaline phosphatase, aspartate transaminase, alanine transaminase, γ-glutamyl transferase, albumin, lactate dehydrogenase and bilirubin levels in serum were measured using commercial kits (Biodiagnostic Co., Badr, Egypt).
Preparation of kidney and liver homogenates
Kidney and liver tissues were homogenized in phosphate buffer saline to yield a 10% w/v homogenate for the biochemical tests. After that, they were centrifuged at 1200 g at 4°C for 20 min and the supernatant was kept at −80°C until further assays.
Assessment of oxidative stress biomarkers
Malondialdehyde (MDA) (Cat. No: MD 2529), Superoxide dismutase (SOD) (Cat. No: SD 2521) & reduced glutathione (GSH) (Cat. No: GR 2511) were determined in kidney and liver homogenates spectrophotometrically (Biodiagnostic Co., Badr, Egypt).
ELISA measurement of renal and hepatic Nrf-2 and HO-1, TNF-α, IL-1β and NF-κB p65 contents
Renal and hepatic contents of Nrf-2 (Cat. No: RD-NFE2L2-Ra; Reddot Biotech Inc. Co., Canada), HO-1 (Cat. No: ER1041; FineTest Co., USA), TNF-α (Cat. No: 438204; BioLegend Co., USA), IL-1β (Cat. No: E0119Ra; BT LAB Co., China) and NF-κB p65 (Cat. No: E-EL-R0674; Elabscience Co., USA) were measured using ELISA technique according to the manufacturer’s instructions.
Western blot analysis of renal and hepatic p-p38 MAPK contents
The expression of renal and hepatic p-p38 MAPK protein was carried out using Western blot analysis as previously described. 13 The signal density was calculated by ImageJ software (version 1.8.0). The band intensity of the target proteins was normalized against the control sample beta-actin (β-actin), which acts as a housekeeping protein, by the ChemiDoc MP imager.
Immunohistochemical determination of renal and hepatic active caspase-1 contents
The expression of caspase-1 in kidney and liver tissues was assessed by immunohistochemistry technique using anti-caspase-1 rabbit monoclonal antibody (Cat. No: M00048; Boster Bio Co., USA) as previously described. 13
Histopathological analysis
Kidney and liver tissues were fixed in buffered formalin, processed, and stained with hematoxylin and eosin (H&E), as described previously.
13
Sections were then examined and grading score was carried out by the pathologist under light microscopy in a blinded manner as previously described
Statistics
Parametric data were represented as mean ± standard error of the mean (SEM) and analyzed using one-way ANOVA then Tukey–Kramer post-hoc test. Non-parametric data (histopathology scores) were expressed as median ± interquartile range (IQR) and analyzed using Kruskal-Wallis then Dunn’s multiple comparison test. Statistical analyses and graphical representations were performed using GraphPad Prism software (Version 9, San Diego, CA, USA).
Results
Effect of protocatechuic acid (PCA) on liver and kidney weight indices in 5-FU-injected rats
As depicted in (Figure 1(A) and (B), respectively), 5-FU group had significantly higher liver and kidney weight indices than the control group. Both PCA 50 and 100 mg/kg significantly decreased both liver and kidney weight indices compared to 5-FU group. PCA 100 Produced a significant decrease in liver weight index compared to PCA 50 (Figure 1(A)). Effect of protocatechuic acid (PCA) (50, 100 mg/kg) on liver (A) and kidney weight indices (B) in 5-FU-injected rats. N = 6 rats/group. The results were presented as a mean and SEM. The statistical analysis was done using one-way ANOVA and Tukey- Kramer post hoc test. * significant versus “control” # significant versus “5-FU” @ significant versus “PCA 50 mg/kg” at p < 0.05.
Effect of protocatechuic acid (PCA) on liver function markers in 5-FU- injected rats
In 5-FU-treated rats, serum levels of bilirubin (Figure 2(A)), alkaline phosphatase (ALP) (Figure 2(B)), alanine transaminase (ALT) (Figure 2(C)), aspartate transaminase (AST) (Figure 2(D)), lactate dehydrogenase (LDH) (Figure 2(E)) and gamma glutamyl transferase (GGT) (Figure 2(G)) were significantly higher than those of control group while serum albumin level (Figure 2(F)) was lower. Co-treatment with PCA 50 or 100 mg/kg did not significantly affect serum bilirubin, LDH, GGT and albumin levels while significantly decreased levels of ALP, AST, ALT compared to 5-FU group. Effect of protocatechuic acid (PCA) (50, 100 mg/kg) on liver function markers in 5-FU-injected rats. N = 6 rats/group. The results were presented as a mean and SEM. The statistical analysis was done using one-way ANOVA and Tukey- Kramer post hoc test. *significant versus “control” # significant versus “5-FU” at p < 0.05.
Effect of protocatechuic acid (PCA) on renal function markers in 5-FU-injected rats
In 5-FU-treated rats, serum levels of urea (Figure 3(A)) and creatinine (Figure 3(B)) were significantly higher than those of the control group. In contrast, PCA (50 mg/kg) + 5-FU and PCA (100 mg/kg) + 5-FU groups significantly decreased serum parameters compared to the 5-FU group. Effect of protocatechuic acid (PCA) (50, 100 mg/kg) on serum urea (A) and creatinine concentrations (B) in 5-FU-injected rats. N = 6 rats/group. The results were presented as a mean and SEM. The statistical analysis was done using one-way ANOVA and Tukey- Kramer post hoc test. *significant versus “control” # significant versus “5-FU” @ significant versus “PCA 50 mg/kg” at p < 0.05.
Effect of protocatechuic acid (PCA) on hepatic oxidative stress in 5-FU-injected rats
5-FU-injected rats had significantly higher contents of hepatic MDA (Figure 4(A)) whereas hepatic contents of GSH (Figure 4(C)), Nrf-2 (Figure 4(D)) and HO-1 (Figure 4(E)) as well as SOD activity (Figure 4(B)) were significantly lower than those of control rats. Co-treatment with PCA (100 mg/kg) significantly decreased hepatic MDA content compared to 5-FU-injected rats (Figure 4(A)). Effect of protocatechuic acid (PCA) (50, 100 mg/kg) on hepatic oxidative stress in 5-FU-injected rats. N = 6 rats/group. The results were presented as a mean and SEM. The statistical analysis was done using one-way ANOVA and Tukey- Kramer post hoc test. *significant versus “control” # significant versus “5-FU” @ significant versus “PCA 50 mg/kg” at p < 0.05.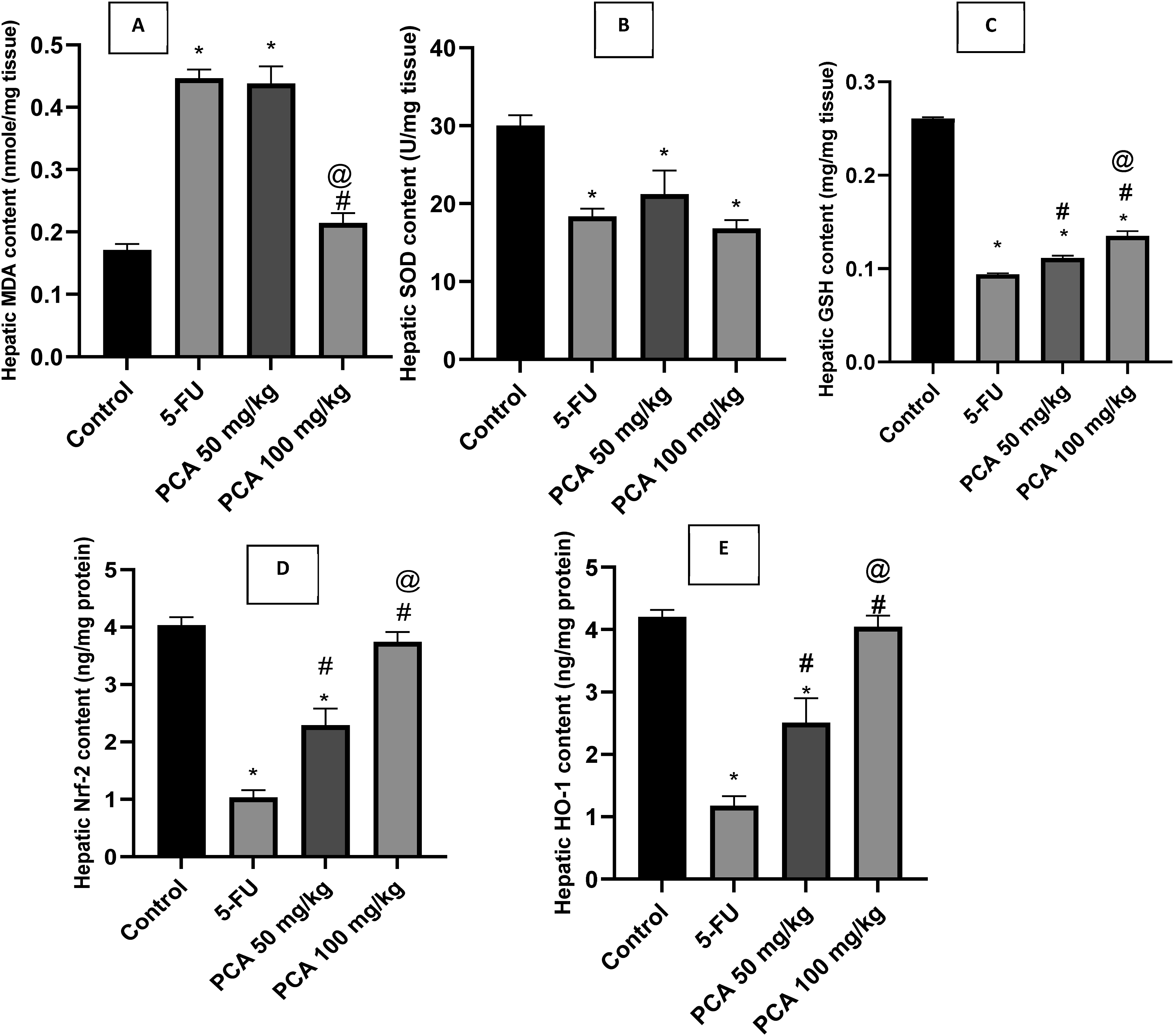
PCA (50 and 100 mg/kg) significantly increased hepatic GSH, Nrf-2 and HO-1 contents in a dose-dependent manner but failed to affect SOD activity compared to 5-FU (Figure 4(B)–(E)).
Effect of protocatechuic acid (PCA) on renal oxidative stress in 5-FU-injected rats
5-FU-injected rats had significantly higher contents of renal MDA (Figure 5(A)) whereas renal contents of GSH (Figure 5(C)), Nrf-2 (Figure 5(D)) and HO-1 (Figure 5(E)) as well as SOD activity (Figure 5(B)) were significantly lower than those of control rats. Effect of protocatechuic acid (PCA) (50, 100 mg/kg) on renal oxidative stress in 5-FU-injected rats. N = 6 rats/group. The results were presented as a mean and SEM. The statistical analysis was done using one-way ANOVA and Tukey- Kramer post hoc test. *significant versus “control” # significant versus “5-FU” @ significant versus “PCA 50 mg/kg” at p < 0.05.
PCA (50 and 100 mg/kg) significantly decreased renal MDA while increased renal GSH, Nrf-2 and HO-1 contents in a dose-dependent manner. Both doses of PCA failed to affect SOD activity compared to 5-FU.
Effect of protocatechuic acid (PCA) on hepatic inflammatory markers in 5-FU-injected rats
5-FU-injected rats had significantly higher contents of hepatic IL-1β (Figure 6(A)), TNF-α (Figure 6(B)) NF-κB p65 (Figure 6(C)) and p-p38 MAPK (Figure 6(D)) than those of control rats. Effect of protocatechuic acid (PCA) (50, 100 mg/kg) on hepatic inflammatory markers in 5-FU- injected rats. N = 6 rats/group. The results were presented as a mean and SEM. The statistical analysis was done using one-way ANOVA and Tukey- Kramer post hoc test. *significant versus “control” # significant versus “5-FU” @ significant versus “PCA 50 mg/kg” at p < 0.05.
PCA (50 and 100 mg/kg) significantly decreased hepatic contents of TNF-α, NF-κB p65 in a dose-dependent manner. Both doses of PCA were of equal efficacy on the level of decreasing hepatic IL-1β and p-p38 MAPK contents.
Effect of protocatechuic acid (PCA) on hepatic caspase-1 expression in 5-FU-injected rats
5-FU-injected rats had significantly higher hepatic caspase-1 expression than those of control rats (Figure 7(A)–(C) and (F)). PCA (50 and 100 mg/kg) significantly decreased caspase-1 expression with a superior effect for PCA50 than PCA100 (Figure 7(D)–(F)). Effect of protocatechuic acid (PCA) on hepatic caspase-1 expression in 5-FU-injected rats. (A): Control group with no staining. (B and C): 5-FU group with high intensity staining. (D): PCA 50 mg/kg group with very few faint immuopositivity in cytoplasm of hepatocytes. (E): PCA 100 mg/kg group with mild to moderate staining in portal area and hepatic parenchyma. Image magnification = 100×. (F): Effect of protocatechuic acid (PCA) (50, 100 mg/kg) on % positive area of hepatic caspase-1. N = 6 rats/group. The results were presented as a mean and SEM. The statistical analysis was done using one-way ANOVA and Tukey- Kramer post hoc test. *significant versus “control” # significant versus “5-FU” @ significant versus “PCA 50 mg/kg” at p < 0.05.
Effect of protocatechuic acid (PCA) on renal inflammatory markers in 5-FU-injected rats
5-FU-injected rats had significantly higher contents of renal IL-1β (Figure 8(A)), TNF-α (Figure 8(B)) NF-κB p65 (Figure 8(C)) and p-p38 MAPK (Figure 8(D)) than those of control rats. Effect of protocatechuic acid (PCA) (50, 100 mg/kg) on renal inflammatory markers in 5-FU- injected rats. N = 6 rats/group. The results were presented as a mean and SEM. The statistical analysis was done using one-way ANOVA and Tukey- Kramer post hoc test. *significant versus “control” # significant versus “5-FU” @ significant versus “PCA 50 mg/kg” at p < 0.05.
PCA (50 and 100 mg/kg) significantly decreased renal contents of TNF-α, NF-κB p65 and p-p38 MAPK in a dose-dependent manner. Both doses of PCA were of equal efficacy on the level of decreasing renal IL-1β contents.
Effect of protocatechuic acid (PCA) on renal caspase-1 expression in 5-FU-injected rats
As depicted in Figure (9(A)–(G), 5-FU-injected rats had significantly higher renal caspase-1 expression than those of control rats. PCA (50 and 100 mg/kg) significantly decreased caspase-1 expression compared to 5-FU group. Effect of protocatechuic acid (PCA) on renal caspase-1 expression in 5-FU-injected rats. (A): Control group with no staining. (B and C): 5-FU group showing diffuse and intense staining in glomerulus, cytoplasm of tubular epithelial cell and interstitial cells. (D): PCA 50 mg/kg group with few immunopositivity in glomerular bowman’s capsule. (E and F): PCA 100 mg/kg group showing moderate immunopositivity in interstitial tissue, glomerular tufts. Image magnification = 100×. (G): Effect of protocatechuic acid (PCA) (50, 100 mg/kg) on % positive area renal caspase-1. N = 6 rats/group. The results were presented as a mean and SEM. The statistical analysis was done using one-way ANOVA and Tukey- Kramer post hoc test. *significant versus “control” # significant versus “5-FU” at p < 0.05.
Effect of protocatechuic acid (PCA) on hepatic histopathology in 5-FU-injected rats
5-FU-injected rats had significantly higher hepatic inflammation (Figures 10(A) and 11), fibrosis (Figures 10(B) and 11) and necrosis scores (Figures 10(C) and 11) than those of control rats. Effect of protocatechuic acid (PCA) (50, 100 mg/kg) on hepatic different histopathological scores in 5-FU-injected rats. N = 4 rats/group. The results were presented as a median and IQR. The statistical analysis was done using Kruskall Wallis followed by Dunn’s multiple comparisons test. *significant versus “control” # significant versus “5-FU” at p < 0.05. Effect of protocatechuic acid (PCA) on hepatic histopathology in 5-FU-injected rats. (A): Control liver with normal appearance. (B and C): 5-FU group with necrosis and significant portal congestion (thick arrow), periportal fibrosis with edema (arrowhead) and inflammatory infiltrates (thin arrow). (D): PCA 50 mg/kg group showing few perivascular inflammatory cells (thin arrow) and occasional individual cell necrosis (thick arrow). (E): PCA 100 mg/kg group with minimal cellular aggregations of plumb fibroblast and macrophages (thin arrow). Image magnification = 400×.

PCA 50 or 100 mg/kg had no significant effect on hepatic inflammation score whereas both doses significantly reduced hepatic fibrosis and necrosis scores compared to 5-FU group.
Effect of protocatechuic acid (PCA) on renal histopathology in 5-FU-injected rats
5-FU-injected rats had significantly higher renal inflammation (Figure 12(A) and 13), necrosis (Figure 12(B) and 13) and glomerular damage scores (Figure 12(C) and 13) than those of control rats. PCA 50 or 100 mg/kg significantly reduced all scores compared to 5-FU group. Effect of protocatechuic acid (PCA) (50, 100 mg/kg) on renal different histopathological scores in 5-FU-injected rats. N = 4 rats/group. The results were presented as a median and IQR. The statistical analysis was done using Kruskall Wallis followed by Dunn’s multiple comparisons test. *significant versus “control” # significant versus “5-FU” at p < 0.05. Effect of protocatechuic acid (PCA) on renal histopathology in 5-FU-injected rats. (A): control group with normal appearance. (B–D) 5-FU group with diffuse tubular necrosis with intraluminal cellular cast, focal to coalescing inflammation and perivascular edema and hemorrhage together with glomerular damage. Tubular damage with intraluminal cast (thin arrow), glomerular damage with loss or shrunken glomerular tufts (thick arrow), hemorrhage (star), inflammation (arrowhead). (E): PCA 50 mg/kg group with normal renal parenchyma with focal few periglomerular inflammatory cells (arrowhead), inset few aggregations of lymphocytes (thin arrow). (F): PCA 100 mg/kg group with mild periglomerular and interstitial aggregation of inflammatory cells (arrowhead) beside mild to moderate tubular damage (thin arrow), inset periglomerular aggregation of lymphocytes, macrophages and fibroblast (arrowhead), intraluminal necrotic aggregates (thin arrow). Image magnification = 100×, inset = 400×.

Discussion
The current study aimed to test the probable protective effects of PCA in combating 5-FU-triggered hepatorenal injury in rats. PCA has been reported to have anti-inflammatory antioxidant effects in previous studies.6,17,18
A dose of 75 mg/kg 5-FU (intraperitoneally-injected once a week for 21 days was previously reported to produce hepatorenal injury. 12 The proposed underlying mechanisms of its nephro- and hepatotoxicity were related to augmenting inflammation and generation of reactive oxygen species (ROS).12,13 To study the possible protective effects of PCA in improving hepatorenal damage, two doses of PCA (50 or 100 mg/kg) were orally-administered daily for 21 consecutive days. PCA doses were extrapolated from previous studies where they investigated the PCA effects on dexamethasone-induced hepatotoxicity1 19 and cisplatin-induced hepatotoxicity. 20
Upon incidence of hepatocyte injury, AST and ALT leak extracellularly with subsequent elevation in their serum activity.21,22 In our study, 5-FU increased serum AST and ALT levels, a finding that is in agreement with a previous study. 13 PCA was able to reduce their elevated levels which is in line with the study in which the hepatoprotective effect of PCA administered by a dose of 100 mg/kg against liver insult associated with monosodium glutamate intoxication was proven. 23
Lactate dehydrogenase has been related to hepatic injury besides other hepatic injury biomarkers. 24 5-FU significantly increased LDH levels while PCA failed to reduce its elevated levels.
Bilirubin, gamma-glutamyltransferase (GGT), and alkaline phosphatase (ALP) are indicators of biliary function. 25 Their elevation may reflect cholestatic liver injury, while the elevation of bilirubin may also result from high production due to the destruction of erythrocytes or disrupted bilirubin metabolism caused by attenuated liver function. 26
5-FU was able to significantly increase serum ALP, GGT, and bilirubin levels while both PCA 50 and PCA 100 were able to reduce elevated ALP levels matching with an earlier study. 27 PCA was not able to reduce elevated GGT and bilirubin levels. These conflicting observations make the drug’s impact on bile excretion doubtful and warrant more studies.
To further explore the effects of PCA on 5-FU-induced hepatic damage, histopathological examination of hepatic tissues was carried out. The results of the present study were parallel with other studies13,28 in which the hepatotoxic effects of 5-FU were elucidated. Although PCA 50 and PCA 100 were not able to decrease hepatic inflammation score, they were able to decrease the fibrosis and necrosis scores versus elevated inflammation, necrosis, and fibrosis scores with 5-FU. Our results were consistent with previous studies14,29,30 which have demonstrated that PCA was able to preserve the hepatic architecture against different models of experimentally-induced hepatic injury.
Serum creatinine and urea levels were estimated for evaluating renal function. In our study, PCA was able to reduce the elevated serum creatinine and urea induced by 5-FU. This agrees with a study in which PCA showed a protective effect against monosodium glutamate-induced renal damage evidenced by the reduction of these biomarkers. 23
Supporting these findings, upon histopathological study on kidneys, 5-FU increased scores of renal and glomerular inflammation and necrosis, a finding which is in line with other studies.13,28 Surprisingly, only PCA 50 was able to decrease renal inflammation and necrosis scores, a finding that needs further investigation. This is parallel with a previous study 14 in which PCA was effective in mitigating hepatorenal injury induced by LPS. These results emphasize the advantage of PCA as a significant agent for treating hepatorenal injury and open a new approach for additional investigations to prove potential mechanisms.
The reno and hepatoprotective effects of PCA against 5-FU-induced distortions in kidney and liver functions have been established in our study at the biochemical and histopathological studies levels. In an attempt to explore the mechanism of PCA therapeutic effects, different molecular pathways were measured as shown below.
Regarding the inflammatory pathway, NF-κB, TNF-α, IL-1β, caspase-1 and p-p38 MAPK were measured to demonstrate further mechanisms mediating the hepato and nephroprotective effects of PCA. Our results showed elevation in IL-1β, TNF-α, NF-κB p65, caspase-1 and p-p38 MAPK levels upon 5-FU administration. These elevations are consistent with other studies in which the inflammatory effects of 5-FU were evidenced.13,28
Nuclear factor kappa B (NF-κB) is a protein transcription factor regulating the innate type of immunity. It is responsible for complex cellular signaling of inflammation. 31 PCA 50 and PCA 100 reduced the elevated renal and hepatic NF-κB content which was induced by 5-FU injection. Our results are along with an earlier study in which authors concluded that PCA was successful in suppressing NF-κB among other inflammatory biomarkers in hepatic and renal tissues of monosodium glutamate-intoxicated rats. 23
Activated NF-κB increases the gene expressions of pro-inflammatory cytokines such as IL-1β and TNF-α, causing inflammation and subsequent tissue damage. Tumor necrosis factor (TNF)-α is a principal controller of inflammation. 32 PCA had the ability to reduce the raised renal and hepatic TNF-α content which was induced by 5-FU injection confirming the involvement of inflammation in the pathogenesis of the model. This observation agreed with another where PCA was effective in inhibiting TNF-α in renal damage in rats. 23
Caspase-1 induces activation of IL-1β via mediating the cleavage of pro IL-1β into active IL-1β, a powerful pro-inflammatory cytokine in response to cellular injury. 33 Our study showed that PCA 50 and PCA 100 were able to reduce 5-FU-induced capsase-1 expression resulting in the reduction of renal and hepatic IL-1β contents, these observations are in line with previous studies.14,23
IL-1β release can activate the MyD88 inflammatory cascade dependent on the Toll/IL-1R domain, inducing phosphorylation of the MAPKs, ERK1/2, p38, and JNK, resulting in modulation of many transcription factors. 34
Mitogen activated protein kinases (MAPKs) are serine/threonine protein kinase enzymes. The expression of hepatic and renal p-p38 MAPK was detected in our study since it was previously reported to be elevated in 5-FU-induced intestinal mucosal inflammation through activating Toll-like/MyD88/NF-κB/MAPK pathway. 35 PCA 50 and PCA 100 reduced the raised contents of renal and hepatic p-p36 MAPK induced by 5-FU injection. Our results are matched with a study in which PCA was able to reduce p-p38 MAPK content in injured tissues. 36
Taken together, 5-FU possessed the ability to increase renal and hepatic p-p38 MAPK, NF-κB p65, TNF-α, and IL-1β content and capsase-1 expression indicating the participation of inflammation in the pathogenicity of the model. PCA was able to reduce all these measurements. These findings reflect the reno and hepatoprotective effects of PCA via anti-inflammatory effects. The anti-inflammatory effects of PCA were also previously proven in other studies.20,23
In many pathological states, the inflammatory infiltrate into tissue is highly related to reactive oxygen species formation and hence a state of oxidative stress. 37 This is why, the possible contribution of the anti-oxidant mechanism of PCA in its hepato and renoprotective effects against 5-FU-induced toxicity was also explored in our study. In this context, hepatic and renal contents of GSH, MDA, SOD, Nrf2, and HO-1 were evaluated.
The current study showed that 5-FU injection induced a state of oxidative stress; evidenced by elevated malondialdehyde (MDA) contents, diminished superoxide dismutase (SOD) activities, and reduced glutathione (GSH) contents as well as decreased expression of hepatic and renal Nrf2, and its downstream HO-1.
PCA was able to regenerate GSH contents, a result that is consistent with a previous study 23 in which PCA proved to have anti-oxidant activities evidenced by restoration of anti-oxidant/oxidant balance. PCA 100 was able to decrease the elevated renal and hepatic MDA content which was induced under 5-FU injection, a finding that agrees with a previous study. 23 PCA 50 was able to decrease the elevated renal MDA content only with no effect on hepatic MDA content.
Nuclear factor erythroid 2-related factor (Nrf)-2 is a principal anti-oxidant providing cellular resistance against oxidative stress. 38 PCA was able to regenerate the depleted renal and hepatic Nrf-2 content and the downstream anti-oxidant enzyme, heme oxygenase (HO-1) due to 5-FU injection. Our results are consistent with a previous study in which PCA was able to induce HO-1 levels. 39
Collectively, 5-FU could increase renal and hepatic MDA content while decrease GSH, SOD, HO-1, and Nrf-2 contents. These findings may reflect its ability to hinder the endogenous anti-oxidant defense mechanisms and increase lipid peroxidation leading to detrimental effects on renal and hepatic functions. PCA was able to reverse the deleterious effects of 5-FU confirming its anti-oxidative properties. Further clinical studies are required to prove the beneficial effect of PCA in clinical situations.
Conclusion
PCA by the two doses 50 and 100 mg/kg was efficacious in enhancing the pathological findings in 5-FU-induced renal and hepatic injury at the functional, molecular, and structural studies but the better response was shown at the higher dose (PCA 100) compared to corresponding lower dose (PCA 50). This was evidenced through hepatic oxidative stress biomarker; Nrf-2 and renal oxidative stress biomarkers; SOD, GSH, MDA, HO-1, and Nrf-2 as well as hepatic and renal inflammatory biomarkers; NF-κB p65, TNF-α and p-p38 MAPK
Finally, the present study revealed that PCA with antioxidant/antiinflammatory effects could have reduced 5-FU-induced oxidative stress, inflammation, and oxidative tissue damage by suppressing NF-KB activation and stimulating Nrf-2. The results obtained from the study may be used as a guide for further research on the interplay between inflammation, oxidative stress, and hepatorenal injury.
Study limitation
One study limitation might have been the lack of a comparison between the effects of PCA with other well-known antioxidants. In addition, despite the absence of bias in the experiments performed, the use of a rat model makes it difficult to quantitatively extrapolate to a clinical scenario in humans.
Footnotes
Acknowledgements
The authors appreciate the help of Dr. Eman Abdelwahab, Faculty of Veterinary Medicine, Mansoura University, for assistance with histopathological and immunohistochemical experiments.
Statements and declarations
Author contributions
A.M.A. Methodology, Software, Writing original draft, Data curation, Investigation, H.E.K. Supervision, Methodology, Software, Revision of original draft, R.R.A. Supervision, Methodology, Software, Revision of original draft, G.M.S. Conceptualization, Software, Supervision, Revision of original draft. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data presented in this study are original, and no part of this manuscript has been published or submitted elsewhere. Figures and Western blot images were obtained without any digital alteration, and datasets used or analyzed during the current study are available from the corresponding author upon reasonable request.
